Abstract
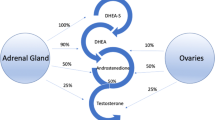
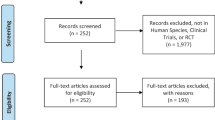
Selective androgen receptor modulators (SARMs) are a class of androgen receptor ligands that bind androgen receptors and display tissue selective activation of androgenic signaling. SARMs have selective anabolic effects on muscle and bone, and were originally synthesized for treatment of muscle wasting conditions, osteoporosis, breast cancer. To date, no SARM has been clinically approved and little is known about the beneficial effects and other adverse effects on users. We examined the adverse effects and potential benefits of SARMs amongst users. We performed an internet survey assessing the demographics of users via a 32-question survey. Using reddit as a platform, we distributed the survey through various subreddits that included potential SARMs users. Out of the 520 responses, 343 participants admitted having used SARMs. Most were males (98.5%), between the ages of 18–29 (72.3%). More than 90% of users acquired SARMs via the internet and did not consult with a physician. More than half of SARMs users experienced side effects including mood swings, decreased testicular size, and acne. More than 90% of men reported increased muscle mass and were satisfied with their SARMs usage. Despite having seemingly positive effects, more than 50% of SARMs users report significant adverse effects. Chi square was the main method of statistical analysis. Future studies should focus on comprehensive reproductive evaluation of men using SARMs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bhasin S, Jasuja R. Selective androgen receptor modulators as function promoting therapies. Curr Opin Clin Nutr Metab Care. 2009;12:232–40.
Narayanan R, Mohler ML, Bohl CE, Miller DD, Dalton JT. Selective androgen receptor modulators in preclinical and clinical development. Nucl Recept Signal. 2008;6:e010.
Rambhatla A, Mills JN, Rajfer J. The role of estrogen modulators in male hypogonadism and infertility. Rev Urol. 2016;18:66–72.
Narayanan R, Coss CC, Dalton JT. Development of selective androgen receptor modulators (SARMs). Mol Cell Endocrinol. 2018;465:134–42.
Barbara M, Dhingra S, Mindikoglu AL. Ligandrol (LGD-4033)-induced liver injury. ACG Case Rep. J 2020;7:e00370.
Van Wagoner RM, Eichner A, Bhasin S, Deuster PA, Eichner D. Chemical composition and labeling of substances marketed as selective androgen receptor modulators and sold via the internet. JAMA 2017;318:2004–10.
Basaria S, Collins L, Dillon EL, Orwoll K, Storer TW, Miciek R, et al. The safety, pharmacokinetics, and effects of LGD-4033, a novel nonsteroidal oral, selective androgen receptor modulator, in healthy young men. J Gerontol A Biol Sci Med Sci. 2013;68:87–95.
Session tCs. S.2895 - SARMs Control Act of 2019 [Control Act]. United States Congress; 2019 [updated 1/19/2019; cited 2020 9/15/2020]. 1]. Available at https://www.congress.gov/bill/116th-congress/senate-bill/2895/text.
Christiansen AR, Lipshultz LI, Hotaling JM, Pastuszak AW. Selective androgen receptor modulators: the future of androgen therapy? Transl Androl Urol 2020;9:S135–S48.
Curry D Reddit Revenue and Usage Statistics (2021) [Webpage]. Businessofapps.com: Business of Apps; 2021 [cited 2020 11/13/2020]. Available at https://www.businessofapps.com/data/reddit-statistics/.
Agency WA-D THE 2008 PROHIBITED LIST INTERNATIONAL STANDARD [Website]. WADA2008 [cited 2020. WADA Prohibited List (2008)]. Available at https://www.wada-ama.org/sites/default/files/resources/files/WADA_Prohibited_List_2008_EN.pdf.
Cohen J, Collins R, Darkes J, Gwartney D. A league of their own: demographics, motivations and patterns of use of 1,955 male adult non-medical anabolic steroid users in the United States. J Int Soc Sports Nutr. 2007;4:12.
Sagoe D, Molde H, Andreassen CS, Torsheim T, Pallesen S. The global epidemiology of anabolic-androgenic steroid use: a meta-analysis and meta-regression analysis. Ann Epidemiol. 2014;24:383–98.
Crawford J, Prado CM, Johnston MA, Gralla RJ, Taylor RP, Hancock ML, et al. Study design and rationale for the phase 3 clinical development program of enobosarm, a selective androgen receptor modulator, for the prevention and treatment of muscle wasting in cancer patients (POWER Trials). Curr Oncol Rep. 2016;18:37.
GTx I GTx Announces Top-Line Results from Placebo-Controlled ASTRID Trial of Enobosarm in Women with Stress Urinary Incontinence [Webpage]. Bloomberg.com: Bloomberg; 2018 [cited 2020 11/15/2020]. Available at https://www.bloomberg.com/press-releases/2018-09-21/gtx-announces-top-line-results-from-placebo-controlled-astrid-trial-of-enobosarm-in-women-with-stress-urinary-incontinence.
Ponnusamy S, Sullivan RD, Thiyagarajan T, Tillmann H, Getzenberg RH, Narayanan R. Tissue selective androgen receptor modulators (SARMs) increase pelvic floor muscle mass in ovariectomized mice. J Cell Biochem. 2017;118:640–6.
Osterberg EC, Bernie AM, Ramasamy R. Risks of testosterone replacement therapy in men. Indian J Urol. 2014;30:2–7.
Smit DL, de Ronde W. Outpatient clinic for users of anabolic androgenic steroids: an overview. Neth J Med. 2018;76:167.
Patel AS, Leong JY, Ramos L, Ramasamy R. Testosterone is a contraceptive and should not be used in men who desire fertility. World J Mens Health. 2019;37:45–54.
Catalani V, Arillotta D, Corkery JM, Guirguis A, Vento A, Schifano F. Identifying new/emerging psychoactive substances at the time of COVID-19; a web-based approach. Front Psychiatry. 2020;11:632405.
Leas EC, Hendrickson EM, Nobles AL, Todd R, Smith DM, Dredze M, et al. Self-reported cannabidiol (CBD) use for conditions with proven therapies. JAMA Netw Open. 2020;3:e2020977.
Ibarra JL, Agas JM, Lee M, Pan JL, Buttenheim AM. Comparison of online survey recruitment platforms for hard-to-reach pregnant smoking populations: feasibility study. JMIR Res Protoc. 2018;7:e101.
Wang M, Wu G, Gunasekaran V, Chen V, Vani A, Kim D. Internet survey: health screening in sports. J Atr Fibrillation. 2016;9:1471.
Acknowledgements
The authors have no financial disclosures. No funding was provided for this study.
Author information
Authors and Affiliations
Contributions
All listed authors contributed equally to writing of this manuscript.
Corresponding author
Ethics declarations
Competing interests
There are no competing financial interests in relation to the work described. Dr. Ranjith Ramasamy has the following disclosures: Acerus Pharmaceuticals (Consultant); Boston Scientific (Consultant, Grant recipient); Coloplast (Consultant, Grant recipient); Endo Pharmaceuticals (Consultant, Grant Recipient); Metuchen (Consultant); Nestle Health (Consultant).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Efimenko, I.V., Valancy, D., Dubin, J.M. et al. Adverse effects and potential benefits among selective androgen receptor modulators users: a cross-sectional survey. Int J Impot Res 34, 757–761 (2022). https://doi.org/10.1038/s41443-021-00465-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41443-021-00465-0
This article is cited by
-
Selective androgen receptor modulator use and related adverse events including drug-induced liver injury: Analysis of suspected cases
European Journal of Clinical Pharmacology (2024)
-
Ostarine blunts the effect of endurance training on submaximal endurance in rats
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
Emergence and occurrence of performance-enhancing substance use in Australia determined by wastewater analysis
Nature Water (2023)
-
Advancements in the evaluation and treatment for Testosterone Deficiency
International Journal of Impotence Research (2022)