Abstract
Erectile dysfunction (ED) is caused by microvascular or macrovascular insufficiency in the majority of patients. Recent studies have shown that hyperbaric oxygen therapy (HBOT) can induce angiogenesis in different body organs. The effect of HBOT on the non-surgery-related ED has not been investigated yet. The aim of the current study was to evaluate the effects of HBOT on sexual function and penile vascular bed in non-surgical ED patients. A prospective analysis of patients suffering from chronic ED treated with 40 daily HBOT sessions. Clinical efficacy was assessed using the International Index of Erectile Function questionnaire (IIEF) and a global efficacy question (GEQ). The effect on the penile vascular bed was evaluated by perfusion MRI. Thirty men (mean age of 59.2 ± 1.4) suffering from ED for 4.2 ± 0.6 years completed the protocol. HBOT significantly improved all IIEF domains by 15–88% (p < 0.01). Erectile function improved by 88% (p < 0.0001) and 80% of the patients reported positive outcome according to the GEQ. Angiogenesis was indicated by perfusion MRI that showed a significant increase by 153.3 ± 43.2% of K-trans values in the corpous cavernous (p < 0.0001). HBOT can induce penile angiogenesis and improve erectile function in men suffering from EcD. HBOT reverses the basic common pathophysiology, atherosclerosis and decreased penile perfusion, responsible for most cases of ED.
Similar content being viewed by others
Introduction
The normal erection is a complex event resulting from the coordinated function of psychological, neurological, hormonal, and vascular systems [1]. Penile blood flow disruption due to inadequate vascular perfusion is present in at least 60% of the erectile dysfunction (ED) patients [2]. The first-line therapy used for ED relies on the vasodilatation effect of phosphodiesterase-5 inhibitors (PDE5Is) [3]. However, since the vasodilatation effect of PDE5Is is transient and dependent on the presence of adequate blood vessels within the corpora cavernosa (CC), there is a need for a treatment modality that intervenes with the baseline pathology and induces generation of new blood vessels (angiogenesis). Hyperbaric oxygen therapy (HBOT) has been shown to induce angiogenesis in different tissues experiencing compromised blood perfusion such as the brain and non-healing wounds [4, 5]. The effect of HBOT on penile angiogenesis has not been investigated yet.
HBOT incorporates the inhalation of 100% oxygen at pressures exceeding 1 atmosphere absolute (ATA), thus increasing the amount of oxygen dissolved in the body tissues [6]. One of the most interesting mechanisms induced by HBOT is angiogenesis mediated by release of omnipotent stem cells capable of differentiating into endothelial cells [7,8,9]. HBOT also boosts the release of vascular endothelial growth factor (VEGF) and hypoxia-inducible factor-1alpha (HIF-1alpha), crucial mediators for the angiogenesis process [9]. In addition, the improved oxygenation by HBOT creates the necessary environment needed for stem cells proliferation. Pre-clinical and clinical studies have demonstrated that HBOT can induce angiogenesis even in tissues with low regenerative potential such as the brain [10,11,12].
Two studies, on both humans and animals, suggested a possible therapeutic effect of HBOT on ED related to surgical injuries [13, 14]. In the clinical study, which included a cohort of 12 men suffering from ED after posterior urethral reconstruction surgery, HBOT significantly improved erectile function [14].
Aims
The aim of the current study was to evaluate the effects of HBOT on sexual function and penile microvasculature in non-surgical ED patients.
Materials and methods
A prospective analysis of men, age 18 years or older, with a clinical diagnosis of erectile dysfunction,of more than six months duration. The study was approved by the institutional review board, and all participants gave written informed consent prior to their inclusion. The study was registered in the US National Institute of Health Clinical Trails registry (NCT02619383).
Exclusion criteria included penile anatomical defects, any active or history of malignancy including prostate cancer, spinal cord injury, any major psychiatric disorder uncontrolled with treatment, claustrophobia, chronic lung disease, or chronic middle ear or sinus diseases. Participants were allowed to continue with PDE5I medication if it was chronically used for more than 6 months prior to their inclusion.
Hyperbaric oxygen therapy
The treatment comprised 40 daily hyperbaric sessions, 5 days a week, in a multiplace hyperbaric chamber (HAUX-Life-Support GmbH, Germany). Each session consisted of 90 min of exposure to 100% oxygen at 2 ATA with 5 min air breaks every 30 min.
Sexual function assessment
Efficacy of the treatment was assessed at baseline (2 weeks after signing informed consent) and in 2 weeks post their last HBOT session, using the International Index of Erectile Function (IIEF) questionnaire [15]. The IIEF is a validated, multidimensional, self-administered questionnaire consisting of 15 items used for the clinical assessment of erectile dysfunction. Each item is rated on a five-point ordinal scale. Zero is scored when responders did not attempt intercourse.
Primary outcome was measured by questions 3 and 4. Question 3 asks ‘Over the past four weeks, when you have attempted sexual intercourse, how often were you able to penetrate (enter) your partner?’. Question 4 asks ‘Over the past four weeks, during sexual intercourse, how often were you able to maintain your erection after you have penetrated your partner?’.
Efficacy was also assessed on the basis of the scores for the five separate domains of male sexual function of the IIEF [15]. The domain scores were computed by adding the scores for the individual questions in each domain. In addition, efficacy was also assessed using the global efficacy question (GEQ) (“Did the treatment improve your erections?”), with a yes or no response.
Perfusion MRI protocol
Perfusion imaging technique called dynamic contrast-enhanced MRI (DCE-MRI) was used to evaluate the change in capillary leakage/flow as indicated by K-trans parameter values, serving as indicator of angiogenesis [16].
Seven patients underwent two MRI scans, baseline and after HBOT. Imaging was conducted using a 3 Tesla system (MAGNETOM Skyra, Siemens, Germany). The MRI protocol included anatomic T1 and T2 sequences and DCE (detailed in supplementary section S1).
K-trans calculations were performed in four specific axial sections located in the CC at the level of the penis base for better localization and reduced variability (Fig. 1a). Another control K-trans measurement was performed at the psoas muscle as it passes through the groin. The relative change in penile K-trans values (in %) was calculated as (post-pre HBOT values)/(pre HBOT value)*100.
DCE-MRI was performed using injection of contrast media (Gd-DTPA) and aquring serial images in order to measure blood perfusion.DCE-MRI Ktrans map is calculated from the serial images (see S-1 for further details). a Penile MRI coordinates of measurement. T2 MRI sequence demonstrates the 4 locations in each CC which were used to extract K-trans values. The base of the penis area was chosen for better localization in each patient and variability reduction. b Average MRI dynamic contrast enhancement (DCE) maps pre and post HBOT. Black to blue and green (in this order) reflect low K-trans values = low perfusion in the corporoa cavernousa. Yellow and red (in this order) reflect high K-trans values = increased perfusion in the corpora cavernousa. Top row: K-trans values at baseline. Middle row: K-trans values after HBOT. Bottom row: delta map showing increase in K-trans after HBOT
Statistical analysis
The mean frequency of responses to questions 3 and 4 of the IIEF was calculated. An analysis-of-covariance (ANCOVA) model was fitted for each question, which included main-effect terms for base-line score, patient age, smoking, diabetes mellitus, hypertension, ischemic heart disease, use of PDE5Is and duration as covariates. Mean domain scores from the IIEF were calculated, and the treatment effect was analyzed by the described ANCOVA model. The answers to the GEQ question (yes or no) were analyzed with the use of logistic-regression analysis, accounting for the same covariates as those listed for the ANCOVA models. Using IIEF and erectile function domain scores means and standard deviations, pairwise analysis was performed to calculate the sample size. In order to detect a two-sided change of one standard deviation in Q3, Q4 and erectile function domain, the calculated sample size was 20-23.
Two-sided paired t-test was performed to compare K-trans values before and after HBOT.
Continuous data were expressed as means ± standard errors. The normal distribution for all variables was tested using the Kolmogorov-Smirnov test. The alpha level was set to 0.05. Data were statistically analyzed using SPSS software (version 22.0).
Results
Patients
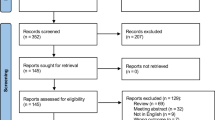
Between June 2013 to May 2015, 42 men suffering from ED were screened. Two did not fulfill inclusion criteria and 3 were excluded due to contraindications (2 middle ear diseases, 1 active malignancy). HBOT was applied for 37 patients. Seven patients did not complete 40 hyperbaric sessions (6 due to barotrauma, 1 claustrophobia). Accordingly, 30 men completed the study and were included in the final analysis (Fig. 2). The mean age was 59.2 ± 1.4 years and the mean duration of symptoms prior to inclusion was 4.2 ± 0.6 years. All 30 patients reported having previous experience with PDE5Is; 46.7% continued using PDE5Is while in the study. Baseline characteristics are summarized in Table 1.
Efficacy
HBOT improved the mean scores of IIEF questions assessing the frequency of penetration (3.0 ± 0.2 post-HBOT vs. 1.9 ± 0.2 pre-HBOT, p < 0.001) and maintenance of erections after penetration (3.4 ± 0.2 post-HBOT vs. 1.7 ± 0.2 pre-HBOT, p < 0.001). The percent increase from base line was 58% for question 3 and 100% for question 4 (Table 2, Fig. 3).
Erectile dysfunction measures before and after hyperbaric oxygen therapy (HBOT). a Penetration erections, Erections maintenance, intercourse satisfaction and overall satisfaction scores significantly increased after HBOT. b Erectile function and IIEF total score improved significantly after HBOT. b IIEF total and erectile function scores significantly improved after HBOT
Penetration and maintenance of erections improved to a response level of 4 or 5 (“success most times”) in 50% of men who were at level 0 or 1 (“no attempts or almost never successful”) before treatment, and in 50% and 70% of men who were at level 2 and 3 (“success at half or less than half the times”), respectively, prior to the treatment.
HBOT increased the mean scores for the erectile-function domain by 10.4 ± 1.2 (90%) (p < 0.001) (Table 2). After HBOT, 63% of the men had none to mild erectile dysfunction (total of questions 1, 2, 3, 4, 5, and 15 was within 19-30) (Fig. 4). The mean scores for the orgasmic-function, intercourse-satisfaction, and overall-satisfaction domains were significantly improved by 29–59% after HBOT (p < 0.001) (Table 2, Fig. 3).
Erectile function severity at baseline and after hyperbaric oxygen therapy (HBOT). After HBOT, 63% of the men had none to mild erectile dysfunction. Erectile function is calculated as the total of questions 1, 2, 3, 4, 5, and 15. Erectile dysfunction severity is graded as: 0–6 severe dysfunction, 7–12 moderate dysfunction, 13–18 mild to moderate dysfunction, 19–24 mild dysfunction, 25–30 normal function
After HBOT, 24 of the 30 men (80%) reported improved erections (global efficacy question).
Age, duration of symptoms, BMI, the presence of diabetes mellitus, hypertension, dyslipidemia, ischemic heart disease, history of smoking, and concurrent use of PDE5Is did not affect any of the above-mentioned outcome measures (p > 0.1).
Penile perfusion
A significant increase in the renal blood flow was indicated in the seven participants who agreed to undergo penile perfusion MRI pre and post HBOT. After HBOT, the relative change in K-trans values increased by 153.3 ± 43.3 in the CC (p < 0.0001). The average K-trans values are depicted in Fig. 1b.
Adverse events
Six from the 37 patients who included in the study discontinued the treatment due to barotrauma (16%) and one due to claustrophobia. Barotraumas were mild and fully recovered in all patients after 3 days. Five out of 30 men who completed the study protocol reported minor pain in the ear during recompression, recovered completely and resumed HBOT sessions without any need for additional treatment.
Discussion
The present study shows for the first time that penile angiogenesis and improved erectile function can be induced by HBOT in men suffering from chronic non-surgical ED. The improvement in sexual performance was significant in all IIEF domains and was most noticeable in both erectile function and the global efficacy question responses. Penile angiogenesis was demonstrated by perfusion MRI analysis.
Vascular integrity, including appropriate vascular dilatation and increased blood flow to the cavernous sinusoidal system, is a key component of the physiology of penile erections [17, 18]. Endothelial dysfunction, atherosclerosis and microvascular diseases disrupt the penile blood flow and accordingly vascular etiology is the leading cause of ED [2]. Because of the small diameter of penile arteries (1–2 mm), compared with other arteries (such as coronary arteries), the same level of endothelial dysfunction and atherosclerosis may lead to a clinically more significant reduction of blood flow to the penis culminating in ED [19]. Consequently, ED can be the earliest symptom of cardiovascular disease.
Angiogenesis is the sprouting of new blood vessels using vasculogenic stem cells which differentiate into endothelial cells, as well as other supporting structures. The use of stem cells and/or angiogenic growth factors for erectile dysfunction has been suggested previously. Several pre-clinical studies have shown beneficial effects using intracavernosal injections of bone marrow stem cells, as well as VEGF in different rat models [20, 21]. Both mobilization of vasculogenic stem cells and induction of angiogenic factors release (such as VEGF and HIF-1alpha) can be induced by repetitive HBOT sessions [5, 9]. Recent studies in patients suffering chronic neurological impairments stemming from stroke, traumatic brain injury and anoxic brain injury have shown that HBOT neuro-therapeutic effects are mediated by brain angiogenesis [22,23,24].
One of the known differentiation characteristics of newly formed blood vessels is their relatively high leakage compared to normal blood vessels [25]. The high leakage of these newly formed blood vessels has been exploited to demonstrate angiogenesis using DCE-MRI [25]. In the present study, by use of DCE-MRI and demonstration of higher K-trans values after HBOT, it was clearly demonstrated that angiogenesis was induced in the penis CC. The demonstrated penile angiogenesis may be one of the underlying physiological mechanisms responsible for the beneficial clinical effect of HBOT on erectile and sexual functions.
When compared to PDE5I or other ED treatments, HBOT holds the promise of unique new intervention modality with several advantages and disadvantages. Starting with the advantages, the induction of angiogenesis by HBOT is aiming for resolution of the basic vascular pathology instead of short-term symptomatic relief. PDE5I or other penile injections/interventions require pre-planning and preparation prior to sexual intercourse. When compared to PDE5I, HBOT enables regaining the ability to have spontaneous or unplanned intercourse. Second, diabetic patients had similar efficacy with HBOT, unlike the lower efficacy seen with PDE5I [26]. The third and most important advantage relates to the high potency of HBOT even in men in whom PDE5Is have no beneficial effect, compared to at least 30% of patients who do not regain erectile function using PDE5Is [27]. In the current study population, ED persisted 4.2 ± 0.6 years prior to inclusion and all men used PDE5Is prior to inclusion without gaining satisfactory erectile function. HBOT carries two main disadvantages. First, while PDE5Is or intracavernosal injections have immediate effect, in HBOT, as in other regenerative therapies, the beneficial effect can be recognized only after a series of sessions. Second, HBOT sessions require considerable time and dedication (40 daily treatments, 90 min each).
HBOT is considered to be a safe method of treatment in general [28], in other vascular related pathologies [29, 30] and was found to be safe also for ED patients. In general, the main adverse effects include middle ear or sinus barotraumas which are usually mild and temporary, with complete resolution after several days [28]. In the current study, the six patients who stopped HBOT due to barotrauma could have continued the treatment with no additional risk if they so wanted.
Our study has several limitations. The first is related to the relatively small number of patients. However, the clear statistical significant effect (a two-way pairwise method analysis) has considerable power (100%) even in a small group of patients. Second, our study lacks an appropriate control/placebo group. Nevertheless, one should not expect any significant improvement in IIEF questionnaires within 3 months in men suffering from ED for several years. In addition, the improvement in penile perfusion was clearly noticeable in objective analysis of perfusion MRI. The correlation between the clinical and the objective penile perfusion MRI findings give further strength to study results. Third, HBOT is a rather costly therapy compared to the accepted medications.
This is the first study evaluating the effect of HBOT on this population suffering from chronic non-surgical ED. Additional, studies are necessary in order to evaluate which subgroups of patients can benefit the most from this treatment, and the optimal HBOT protocol needed for those patients.
HBOT can induce penile angiogenesis and improve erectile function in men suffering from ED. HBOT reverses the basic common pathophysiology, atherosclerosis and decreased penile perfusion, responsible for most of ED cases. The treatment can be considered even years after the onset of erectile dysfunction and in men with unsatisfactory response to PDE5Is. Further studies are needed to evaluate the subgroups of men who can benefit the most from this treatment.
Appendix
The following is a case example, including the clinical data and the MR imaging results.
A 52-year-old male with history of hypertension, dyslipidemia, and ischemic heart disease who suffered from erectile dysfunction during the last 4 years prior to his inclusion. During the first year experienced partial improvement using PDE5I, but the effect waned off. After 40 sessions of HBOT, erectile function improved from 9 to 19, penetration erections (Q3) increased from 1 to 3 and maintenance of erections (Q4) increased from 1 to 3. Intercourse satisfaction and overall satisfaction improved from 8 to 11. K-trans increased by 225% in the right CC and by 61% in the left CC (Fig. 5).
Case #1 DCE-MRI. After HBOT there was a significant increase in K-trans values in both the right and left corpus cavernosum. K-trans values map was overlaid on the T2 sequence. Black to blue and green (in this order) reflect low K-trans values = low perfusion in the corporoa cavernousa. Yellow and red (in this order) reflect high K-trans values = increased perfusion in the corpora cavernousa
Change history
09 July 2018
This has been corrected in both the PDF and HTML versions of the Article
References
Burls A, Gold L, Clark W. Systematic review of randomised controlled trials of sildenafil (Viagra) in the treatment of male erectile dysfunction. Br J Gen Pract. 2001;51:1004–12.
Virag R, Bouilly P, Frydman D. Is impotence an arterial disorder? A study of arterial risk factors in 440 impotent men. Lancet. 1985;1:181–4.
Bella AJ, Lee JC, Carrier S, Benard F, Brock GB. 2015 CUA Practice guidelines for erectile dysfunction. Can Urol Assoc J. 2015;9:23–9.
Duan S, Shao G, Yu L, Ren C. Angiogenesis contributes to the neuroprotection induced by hyperbaric oxygen preconditioning against focal cerebral ischemia in rats. Int J Neurosci. 2015;125:625–34.
Lin KC, Niu KC, Tsai KJ, Kuo JR, Wang LC, et al. Attenuating inflammation but stimulating both angiogenesis and neurogenesis using hyperbaric oxygen in rats with traumatic brain injury. J Trauma Acute Care Surg. 2012;72:650–9.
Fosen KM, Thom SR. Hyperbaric oxygen, vasculogenic stem cells, and wound healing. Antioxid Redox Signal. 2014;21:1634–47.
Thom SR, Bhopale VM, Velazquez OC, Goldstein LJ, Thom LH, et al. Stem cell mobilization by hyperbaric oxygen. Am J Physiol Heart Circ Physiol. 2006;290:H1378–86.
Hopf HW, Gibson JJ, Angeles AP, Constant JS, Feng JJ, et al. Hyperoxia and angiogenesis. Wound Repair Regen. 2005;13:558–64.
Peng ZR, Yang AL, Yang QD. The effect of hyperbaric oxygen on intracephalic angiogenesis in rats with intracerebral hemorrhage. J Neurol Sci. 2014;342:114–23.
Chen J, Zhang ZG, Li Y, Wang L, Xu YX, et al. Intravenous administration of human bone marrow stromal cells induces angiogenesis in the ischemic boundary zone after stroke in rats. Circ Res. 2003;92:692–9.
Jiang Q, Zhang ZG, Ding GL, Zhang L, Ewing JR, et al. Investigation of neural progenitor cell induced angiogenesis after embolic stroke in rat using MRI. Neuroimage. 2005;28:698–707.
Tal S, Hadanny A, Berkovitz N, Sasson E, Ben-Jacob E, et al. Hyperbaric oxygen may induce angiogenesis in patients suffering from prolonged post-concussion syndrome due to traumatic brain injury. Restor Neurol Neurosci. 2015;33:943–51.
Muller A, Tal R, Donohue JF, Akin-Olugbade Y, Kobylarz K, et al. The effect of hyperbaric oxygen therapy on erectile function recovery in a rat cavernous nerve injury model. J Sex Med. 2008;5:562–70.
Yuan JB, Yang LY, Wang YH, Ding T, Chen TD, et al. Hyperbaric oxygen therapy for recovery of erectile function after posterior urethral reconstruction. Int Urol Nephrol. 2011;43:755–61.
Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, et al. The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology. 1997;49:822–30.
Li L, Wang K, Sun X, Wang K, Sun Y, et al. Parameters of dynamic contrast-enhanced MRI as imaging markers for angiogenesis and proliferation in human breast cancer. Med Sci Monit. 2015;21:376–82.
Fournier GR Jr., Juenemann KP, Lue TF, Tanagho EA. Mechanisms of venous occlusion during canine penile erection: an anatomic demonstration. J Urol. 1987;137:163–7.
Costa C, Virag R. The endothelial-erectile dysfunction connection: an essential update. J Sex Med. 2009;6:2390–404.
Montorsi P, Ravagnani PM, Galli S, Rotatori F, Veglia F, et al. Association between erectile dysfunction and coronary artery disease. Role of coronary clinical presentation and extent of coronary vessels involvement: the COBRA trial. Eur Heart J. 2006;27:2632–9.
Qiu X, Lin H, Wang Y, Yu W, Chen Y, et al. Intracavernous transplantation of bone marrow-derived mesenchymal stem cells restores erectile function of streptozocin-induced diabetic rats. J Sex Med. 2011;8:427–36.
Qiu X, Sun C, Yu W, Lin H, Sun Z, et al. Combined strategy of mesenchymal stem cell injection with vascular endothelial growth factor gene therapy for the treatment of diabetes-associated erectile dysfunction. J Androl. 2012;33:37–44.
Boussi-Gross R, Golan H, Fishlev G, Bechor Y, Volkov O, et al. Hyperbaric oxygen therapy can improve post concussion syndrome years after mild traumatic brain injury - randomized prospective trial. PLoS One. 2013;8:e79995.
Boussi-Gross R, Golan H, Volkov O, Bechor Y, Hoofien D, et al. Improvement of memory impairments in poststroke patients by hyperbaric oxygen therapy. Neuropsychology. 2015;29:610–21.
Hadanny A, Golan H, Fishlev G, Bechor Y, Volkov O, et al. Hyperbaric oxygen can induce neuroplasticity and improve cognitive functions of patients suffering from anoxic brain damage. Restor Neurol Neurosci. 2015;33:471–86.
Nagy JA, Benjamin L, Zeng H, Dvorak AM, Dvorak HF. Vascular permeability, vascular hyperpermeability and angiogenesis. Angiogenesis. 2008;11:109–19.
Salonia A, Rigatti P, Montorsi F. Sildenafil in erectile dysfunction: a critical review. Curr Med Res Opin. 2003;19:241–62.
McMahon CN, Smith CJ, Shabsigh R. Treating erectile dysfunction when PDE5 inhibitors fail. BMJ. 2006;332:589–92.
hadanny A, Meir O, Bechor Y, Fishlev G, Bergan J, et al. The safety of hyperbaric oxygen treatment--retrospective analysis in 2,334 patients. Undersea Hyperb Med. 2016;43:113–22.
Kalani M, Jorneskog G, Naderi N, Lind F, Brismar K. Hyperbaric oxygen (HBO) therapy in treatment of diabetic foot ulcers. Long-term follow-up. J Diabetes Complicat. 2002;16:153–8.
Mathews R, Rajan N, Josefson L, Camporesi E, Makhuli Z. Hyperbaric oxygen therapy for radiation induced hemorrhagic cystitis. J Urol. 1999;161:435–7.
Acknowledgements
The authors thank Ms. Michal Ben-Jacob and Mr. Raz Tuval for reviewing and proofing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hadanny, A., Lang, E., Copel, L. et al. Hyperbaric oxygen can induce angiogenesis and recover erectile function. Int J Impot Res 30, 292–299 (2018). https://doi.org/10.1038/s41443-018-0023-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41443-018-0023-9
This article is cited by
-
Investigating self-reported efficacy of lifestyle medicine approaches to tackle erectile dysfunction: a cross-sectional eSurvey based study
BMC Urology (2023)
-
Restorative therapy clinical trials for erectile dysfunction: a scoping review of endpoint measures
International Journal of Impotence Research (2023)
-
Conservative Non-surgical Options for Erectile Dysfunction
Current Urology Reports (2023)
-
Hyperbaric Oxygen Treatment Ameliorates the Decline in Oocyte Quality and Improves the Fertility of Aged Female Mice
Reproductive Sciences (2023)
-
Hyperbaric Oxygen Therapy Attenuated the Motor Coordination and Cognitive Impairment of Polyglutamine Spinocerebellar Ataxia SCA17 Mice
The Cerebellum (2023)