Abstract
The switch from vegetative growth to reproductive growth is a key event in the development of a plant. Here, the product of the chrysanthemum gene CmMYB2, an R2R3 MYB transcription factor that is localized in the nucleus, was shown to be a component of the switching mechanism. Plants engineered to overexpress CmMYB2 flowered earlier than did wild-type plants, while those in which CmMYB2 was suppressed flowered later. In both the overexpression and RNAi knockdown plants, a number of genes encoding proteins involved in gibberellin synthesis or signaling, as well as in the response to photoperiod, were transcribed at a level that differed from that in the wild type. Both yeast two-hybrid and bimolecular fluorescence complementation assays revealed that CmMYB2 interacts with CmBBX24, a zinc-finger transcription factor known to regulate flowering by its influence on gibberellin synthesis.
Similar content being viewed by others
Introduction
The switch from vegetative growth to reproductive growth is a major developmental event in the life cycle of a flowering plant. This switch is coordinated by the products of a suite of genes that differentially respond to the photoperiod, temperature, and the tissue content of several phytohormones1. The successful isolation of a number of such genes has led to the recognition that flowering can be triggered by proteins active in one or more of the photoperiod, gibberellin (GA), vernalization, autonomous, and senescence pathways2,3,4,5,6. These pathways come together to form a complex regulatory network that ultimately induces the key floral meristem genes APETALA1 (AP1) and LEAFY (LFY)4,5,6.
A number of MYB transcription factors participate in the regulation of flowering time. The following examples all relate to the model angiosperm species Arabidopsis thaliana. Plants engineered to overexpress the MYB-related gene CIRCADIAN CLOCK ASSOCIATED 1 (CCA1) are compromised with respect to their recognition of the circadian clock and consequently suffer from a delay in flowering7. Overexpression of the gene EARLY-PHYTOCHROME-RESPONSIVE 1 (EPR1) enhances responsiveness to far-red light, which also leads to delayed flowering8. Overexpression of CAPRICE 3 (CPL3) downregulates several key flowering time genes, including FLOWERING LOCUS T (FT), SUPPRESSOR OF OVEREXPRESSION OF CO1 (SOC1) and CONSTANS (CO)9. The product of WEREWOLF (WER) is known to act in the photoperiod pathway, since flowering is delayed in the wer mutant when exposed to long-day conditions10. MYB30 interacts with the FT promoter to promote flowering11. EARLY FLOWERING MYB PROTEIN (EFM) is involved in both the photoperiod and the temperature flowering pathways12, while PHLOEM DEVELOPMENT (APL) (syn. PE) activates FT13. The flowering of plants overexpressing MYB44 is delayed by 6–7 days14. MYB transcription factors that affect flowering are also present in other species. For example, the product of the poplar gene PtrMYB192 negatively affects flowering time, and its constitutive expression in A. thaliana also induces a delay in flowering15. Constitutive expression of the wheat gene TaMYB72 in rice delays flowering time by 12 days16. In alfalfa, MsSPL13 downregulates MsMYB112, thereby controlling both vegetative and reproductive development17. In chrysanthemum (Chrysanthemum morifolium), a substantial number of MYB transcription factors have been shown to participate in the determination of flowering time18,19, although little is known about their specific functions.
Chrysanthemum occupies a large share of the global market of cut flowers20,21. Most chrysanthemum varieties are short-day plants and blossom during a relatively short period. The CmMYB2 gene encodes an R2R3 MYB transcription factor; when this gene is constitutively expressed in A. thaliana, flowering is delayed, and all CO, FT, SOC1, LFY, and AP1 genes are downregulated22. However, in this study, when CmMYB2 is overexpressed in chrysanthemum, the flowering time is promoted, while RNAi-enabled knockdown delays flowering. The regulatory mechanism of flowering was further analyzed via transcriptome sequencing. CmMYB2 was found to interact with CmBBX24, which is a zinc-finger transcription factor that has been shown to alter GB synthesis and regulate flowering time23. Thus, the aim of the present study was to characterize in detail the molecular effect of manipulating the expression of CmMYB2. The results showed that CmMYB2 may regulate flowering by inhibiting the activity of the CmBBX24 protein and modulating the GA pathway in chrysanthemum.
Results
Subcellular localization and transactivation activity of CmMYB2
The sequences of the CmMYB2 open reading frame present in the varieties Jinba and Zhongshanzigui were identical stretches of 948 nt, predicted to encode a 315 residue polypeptide. The polypeptide sequence included an R2R3 MYB domain within its N-terminal region and the characteristic GxFMxVVQEMIxxEVRSYM motif within its C-terminal region22. The site of expression of CmMYB2 was deduced by transiently expressing a transgene comprising the CmMYB2 coding sequence fused to GFP and driven by the CaMV 35S promoter in onion epidermal cells. GFP activity was concentrated in the nuclei, while in control transgenic plants harboring the p35S::GFP construct, it was dispersed throughout the nucleus and cytoplasm (Fig. 1a).
a Site of CmMYB2 deposition in transiently transformed onion epidermal cells. Bar: 50 μm. b Transcriptional activation activity of CmMYB2 in yeast. pCL1 and pGBKT7 plasmids were used as the positive and negative controls, respectively. –L/-T: SD media lacking leucine/SD media lacking tryptophan, -AH: SD media lacking both histidine and adenine. c, d Luciferase activity in A. thaliana mesophyll protoplasts expressing 5×GAL4-LUC together with either p35S::GAL4DB-AtARF5, p35S::GAL4DB or p35S::GAL4DB-CmMYB2. c Arabidopsis mesophyll protoplasts imaged after luciferin addition with a charge-coupled device (CCD) camera. d Luciferase activity measured after the introduction of p35S::GAL4DB-CmMYB2 into A. thaliana mesophyll protoplasts
To determine whether CmMYB2 can function as a transcription factor, a transactivation assay was performed in yeast. Yeast cells harboring pCL1 were able to grow freely on synthetic dropout (SD) media lacking histidine and adenine, while cells harboring pGBKT7 were able to grow only on SD media lacking tryptophan. Yeast cells harboring CmMYB2 were unable to activate either the His3 or the Ade2 reporter gene, preventing growth on SD media lacking histidine and adenine (Fig. 1b). The results suggested that CmMYB2 showed no transactivation activity in yeast. A series of equivalent assays was attempted in A. thaliana protoplasts, in which the positive control was p35S::GAL4DB-ARF5 and in which the negative control was p35S::GAL4DB. The reduction in LUC activity exhibited by protoplasts harboring CmMYB2 compared to the activity in protoplasts harboring p35S::GAL4DB suggested that CmMYB2 could act as a transcriptional repressor in A. thaliana protoplasts (Fig. 1c).
Effects of manipulating the expression of CmMYB2 on plant growth and development
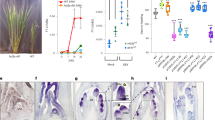
Both CmMYB2 overexpression (OX lines) and CmMYB2 knockdown (RNAi lines) were generated, and their growth and development were monitored at various time points (Fig. 2a). The flowering time of both the OX and RNAi plants differed significantly from that of wild-type (WT) plants. At 80 days after transplanting, OX plants had formed visible flower buds and had entered the flower bud development stage (FBD); whereas this event occurred at 95 days in WT plants and at 99 days in RNAi plants. The differentiation of the first flower buds and beginning of the petal primordium differentiation stage occurred 15 days sooner in OX plants than in WT plants, while the RNAi plants took an additional four days to reach this stage. By 112 days after transplanting, the flowers of OX plants had entered the early opening stage (EO); however, those formed by WT plants did not reach this stage until 127 days after transplantation, and those formed by RNAi plants did not reach it until 130 days after transplantation (Fig. 2b–d). There were no significant differences among the OX, RNAi and WT plants with respect to their overall height, leaf and flower number or diameter of their flowers.
a qRT-PCR analysis of CmMYB2 transcription in WT, OX and RNAi plants. OX-1, 2: two independent CmMYB2 overexpressors, RNAi-1, -2: two independent CmMYB2 RNAi knockdowns. The values are shown as the mean ± SEs (n = 3). b Development of flower buds in transgenic and WT plants. FBD: flower bud development stage, VC: visible color stage, EO: early opening stage, OF: open-flower stage, SF: senescent-flower stage. Bars: 1 cm. c, d Phenotypes of WT, OX and RNAi plants at the reproductive stage. Plants were imaged at (c) 100 days and (d) 120 days after transplanting
Transcription profiling of genes acting downstream of CmMYB2 and associated with flowering time
RNA-seq was implemented to identify differential transcription between WT, OX and RNAi plants (Fig. 3a); Venn diagrams were constructed to show the distribution of these genes visually (Fig. 3b, c). The genes that were either more strongly transcribed in the OX plants than in the WT plants but weakly transcribed in the RNAi plants or vice versa were emphasized. Among the large number of such genes identified, many of their encoded proteins were involved in either the GA synthesis/signaling pathway or the photoperiod pathway; of particular interest were CmGA20ox, CmGA2ox, CmGA3ox, CmGRP, CmGID, and CmDELLA (the GA synthesis and signaling pathway) as well as CmCOL1, CmCOL9, CmPRR7, CmPRR9, and CmCDF1 (the photoperiod pathway) (Table 1). The results of the quantitative real-time PCR (qRT-PCR) assays used to confirm the RNA-seq-based identification are shown in Fig. 4. The analysis confirmed that CmGA20ox (Unigene70017), CmGA2ox (Unigene15204), CmGA3ox (CL3299.Contig1 and Unigene25287), CmGRP (Unigene8840 and CL1665.Contig2), CmGID1 (Unigene22516), CmCOL1 (CL3612.Contig1) and CmPRR7 (CL12170.Contig2) were more strongly transcribed in OX plants than in WT plants but were more weakly transcribed in RNAi plants; moreover, the abundance of CmDELLA (Unigene14903, CL5826.Contig1 and Unigene26087), CmCOL9 (CL11719.Contig1) and CmCDF1 (CL3093.Contig2 and CL3391.Contig1) transcripts was lower in the OX plants than in the WT plants, but it was higher in the RNAi plants.
CmMYB2 alters cellular GA contents through its interaction with CmBBX24
Quantification of the GA1, GA19, and GA20 contents of young leaves of WT, OX and RNAi plants grown under short days showed that these compounds were significantly enhanced in OX plants and significantly reduced in RNAi plants (Table 2). A yeast two-hybrid screen was then performed to identify the proteins able to interact with CmMYB2. CmMYB9A was found to be a potential interacting protein with CmMYB2, and CmBBX24 was also found to interact with CmMYB9A (Table S2). This implied that CmMYB2 may directly interact with CmBBX24. CmBBX24 contained two B-boxes at the N-terminus. CmBBX24boxs was two B-boxes at the N-terminus of CmBBX24 and CmBBX24box2 was the second B-box at the N-terminus of CmBBX24. Yeast cells cotransformed with CmMYB2 and CmBBX24boxs were able to grow on media lacking tryptophan, leucine, histidine and adenine, regardless of whether X-α-gal was supplied (Fig. 5a). Additional experiments showed that CmMYB2 was able to interact with CmBBX24box2 (Fig. 5a). A bimolecular fluorescence complementation (BiFC) assay was used to provide experimental evidence that CmMYB2 could interact with CmBBX24 in vivo. In onion epidermal cells transiently transformed with CmMYB2/cYFP and CmBBX24/nYFP fusion constructs, YFP fluorescence was detected within the nuclei (Fig. 5b), confirming an in vivo interaction between CmMYB2 and CmBBX24.
a A yeast two-hybrid assay showing protein interactions. -TL, SD/-Trp/-Leu; -TLHA, SD/-Trp/-Leu/-Ade/-His; -TLHA + X-α-gal, SD/-Trp/-Leu/-Ade/-His/X-α-gal; vec, empty vector; CmBBX24boxs, two B-boxes in the N-terminus of CmBBX24; CmBBX24box1, the first B-box in the N-terminus of CmBBX24; CmBBX24box2, the second B-box in the N-terminus of CmBBX24; pGBKT7-53 and pGADT7-T served as positive controls, and pGBKT7-Lam and pGADT7-T served as negative controls. b A BiFC assay was used to confirm the interaction between CmMYB2 and CmBBX24, given the results of the yeast two-hybrid assay. YFP: yellow fluorescent protein, Marker: nuclear localization shown by RFP activity, DIC: bright field image, Merge: overlay of YFP, RFP and bright field images
Discussion
CmMYB2 is expressed in the nucleus and acts as a transcriptional repressor
A common feature of MYB transcription factors is their nuclear localization signal peptides24,25,26. The rice gene OsMYB511 is expressed specifically in the nucleus24, as are the Jatropha curcas gene JcR1MYB125 and the Nicotiana benthamiana gene NbPHAN26. Experiments have shown that for NbPHAN, the N-terminal region is responsible for its nuclear localization26. Here, a transient expression experiment based on onion epidermal cells was able to show that CmMYB2 also localizes to the nucleus, as would be expected for a gene product that controls the transcription of other genes. The full wheat TaMYB73 protein, as well as its C-terminal region, exhibits self-activating ability, but its N-terminal region does not27. Previous experiments have shown that the CmMYB19 protein exhibits no transcriptional activation activity when its encoding gene is expressed in yeast28. Here, the outcome of a yeast one-hybrid experiment demonstrated that CmMYB2 similarly failed to exhibit transcriptional activation, while its product was suggested to be able to act as a transcriptional repressor when the gene was expressed in A. thaliana protoplasts.
Involvement of CmMYB2 in the control of flowering time
To transition from the vegetative to the reproductive state, higher plants need to integrate particular environmental signals (such as the photoperiod) with specific endogenous stimuli (such as GA)29,30. Both the photoperiod and the GA pathway, which strongly control the differentiation of flower buds, are highly conserved among plants and have been intensively studied1,31. Here, the RNA-seq approach used to profile the transcriptomes revealed that homologs of a number of known components of the various flowering regulatory pathways were identified as being differentially transcribed between WT plants and those in which the level of expression of CmMYB2 was either increased or decreased; among these homologs were the GA synthesis/signaling genes CmGA20ox, CmGA2ox, CmGA3ox, CmGRP, CmGID, and CmDELLA and the photoperiod pathway genes CmCOL1, CmCOL9, CmPRR7, CmPRR9, and CmCDF1. The strong implication is that CmMYB2 influences flowering time in chrysanthemum by regulating GA and photoperiod pathway genes.
MYB transcription factors have a major impact on plant growth and development, but their contribution to the determination of flowering time is not well understood; much of the relevant data is related to A. thaliana and a small number of crop species. The MYB-related proteins MYB-RELATED PROTEIN 1 (MYR1) and MYB-RELATED PROTEIN 2 (MYR2) have been shown to be repressors of flowering in plants maintained under low-light intensity, since the myr1/myr2 double mutant flowers earlier than WT does32. Moreover, the phenotype resulting from overexpression of MYR1 and MYR2 resembles that of a GA-deficient plant. Here, under short-day conditions, chrysanthemum plants engineered to overexpress CmMYB2 flowered earlier than did WT plants, the latter of which in turn flowered before CmMYB2 knockdown plants did. Altering the level of CmMYB2 expression affected the leaf content of three forms of GA (GA1, GA19, and GA20). CmBBX24 knockdown plants also flower before WT plants, and the transcription of the CmBBX24 gene is affected by the photoperiod; it can also be manipulated by the application of GA23. It was possible to show that CmMYB2 and CmBBX24 interacted both in vitro and in vivo. It is possible that CmMYB2 relieves the CmBBX24 inhibition of the GA pathway based on their interaction and reverse effects on flowering. Unexpectedly, the flowering time of A. thaliana plants constitutively expressing CmMYB2 is delayed when plants are exposed to long-day conditions22. This difference in behavior may be related to the fact that, unlike the short-day plant species Jinba chrysanthemum, flowering of A. thaliana is induced by long days. Heterologous transformation of a gene leading to contrasting flowering times has also been reported in wheat and Arabidopsis33. The details of the mechanism thus need to be elucidated.
In conclusion, overexpression of the chrysanthemum gene CmMYB2, which encodes a MYB protein, accelerated flowering, while its suppression delayed flowering. CmMYB2 probably exerts its effect on flowering time through the GA pathway, since the leaf GA content was responsive to the intensity of CmMYB2 expression. This mode of action is consistent with CmMYB2’s ability to interact with CmBBX24, because the latter protein is also known to regulate flowering through the GA pathway23.
Materials and methods
Plant materials and cultivation conditions
Cuttings of the chrysanthemum variety Jinba at the five- or six-leaf stage were obtained from Nanjing Agricultural University’s Chrysanthemum Germplasm Resource Preservation Center (Nanjing, China) and were grown in a 1:1:1 (v/v/v) nutrient-enriched soil:vermiculite:perlite mixture.
Isolation of CmMYB2
A 1 μg aliquot of RNA extracted from the leaves of a Jinba plant using RNAiso reagent (TaKaRa, Tokyo, Japan) was converted into ss cDNA using M-MLV reverse transcriptase (TaKaRa). The full-length CmMYB2 sequence of Jinba was inferred from the version of the sequence present in the variety Zhongshanzigui22. The full-length CmMYB2 cDNA sequence was subsequently inserted into a pMD19-T easy vector (TaKaRa) for validation by sequencing. The relevant primers used for this procedure are listed in Table S1.
Subcellular localization of CmMYB2
The CmMYB2 open reading frame was amplified using the primer pair CmMYB2-pENTR1A-F/-R, which ensured that the amplicon carried a SalI recognition site at its 5′ end and a NotI recognition site at its 3′ end (the primer details are listed in Table S1). The resulting amplicon was inserted into a pENTR1A vector (Invitrogen, Carlsbad, CA, USA) and subsequently into a pMDC43 vector34 using a reaction based on LR Clonase II (Invitrogen). The CmMYB2 sequence was fused to the N-terminus of GFP, and the fusion construct was placed under the control of the CaMV 35 S promoter, forming a p35S::GFP-CmMYB2 construct. This construct (or, as a control, p35S::GFP) was transformed into onion epidermal cells using a PDS-1000 He-driven particle bombardment device (Bio-Rad, Hercules, CA, USA). The bombarded tissue was cultivated in the dark at 23 °C for 16 h on Murashige and Skoog (MS) media35 before being assayed for GFP activity using confocal laser scanning microscopy (LSM780, Zeiss, Oberkochen, Germany).
Analysis of the transcriptional activity of CmMYB2
The CmMYB2 open reading frame was amplified using the primer pair MYB2-BD-F/-R (see Table S1 for details), which added an NdeI recognition site to the 5’ end and a BamHI recognition site to the 3′ end. After purification, the amplicon was inserted into a pGBKT7 vector (Clontech, Mountain View, CA, USA). A pCL1 vector containing a full-length copy of GAL4 served as a positive control, and an empty pGBKT7 plasmid served as a negative control. The constructs were transformed into bakers’ yeast (Saccharomyces cerevisiae) strain Y2HGold (Clontech) following the “Yeast Transformation System 2” protocol provided by the manufacturer. The pCL1 transformants were incubated on SD/leucine dropout media, while the pGBKT7 and pGBKT7-CmMYB2 transformants were incubated on SD/tryptophan dropout media. After culturing at 30 °C for three days, the transformed cell lines were transferred to SD histidine and adenine dropout media.
Luminescence assays were used to detect the transactivation activity of CmMYB2 in A. thaliana mesophyll protoplasts. The pENTR1A-CmMYB2 construct was previously transformed into p35S::GAL4DB vectors using a reaction based on LR Clonase II. Transient expression of the transgene was achieved as described previously36. A 7.5 μg aliquot of either p35S::GAL4DB-AtARF5 (positive control), p35S::GAL4DB (negative control) or p35S::GAL4DB-CmMYB2 (test construct) was introduced into the protoplasts. A 7.5 μg aliquot of the luciferase (LUC) reporter construct GAL4-LUC was then cotransformed into A. thaliana mesophyll protoplasts. LUC assays were performed as described previously37, except that D-luciferin was replaced by ViviRen Live Cell substrate (www.goldbio.com/). A. thaliana mesophyll protoplasts were cultivated in plates for 16 h in the light at 23 °C. A low-light cooled CCD imaging apparatus (DU934P, Andor, UK) was used to capture LUC images from a 96-well plate. The LUC activity was measured in 10 s intervals, and the luminescence counts were quantified using a 20/20n luminometer (Turner Biosystems Inc., Sunnyvale, CA, USA). Three independent experiments were performed for each assay.
Generation and characterization of chrysanthemum transgenics
The CmMYB2 open reading frame sequence was amplified using primer pair CmMYB2-pBIG-F/R (details given in Table S1), which has a BamHI recognition site at its 5′ end and a SacI recognition site at its 3′ end, and the resulting amplicon was inserted into a pBIG vector (in which the restriction enzyme sites were reconstructed)38. The primer sequences required for the CmMYB2 RNAi fragment were retrieved from wmd3.weigelworld.org. The fragments required to knock down CmMYB2 by RNAi were obtained by following an artificial microRNA cloning protocol (wmd3.weigelworld.org/cgi-bin/webapp39) and were inserted adjacent to the plasmid’s BamHI and SmaI recognition sites (Table S1); the plasmid was subsequently transferred into the pBIG vector. The overexpression and RNAi transgenes were inserted into Agrobacterium tumefaciens EHA105 using the freezing transformation method, after which the transformed bacteria were inserted into Jinba plants via Agrobacterium-mediated transformation40. The growth and development of the two most effective overexpressing (OX-1 and -2) and knockdown (RNAi-1 and -2) plants were assessed at various time points. Flower bud differentiation was divided into five stages: the FBD stage, visible color (VC) stage, EO stage, open-flower (OF) stage and senescent-flower (SF) stage.
RNA-seq analysis
OX-1, RNAi-2, and WT plants were grown under controlled conditions (23°C, 60% relative humidity, and a 16 h photoperiod provided by 150 μmol m−2 s−1 illumination) for four weeks. Their third fully expanded leaf and stem apical meristem were harvested at 8 a.m. (after three hours of light exposure), flash frozen in liquid nitrogen and then stored at −80 °C until needed. RNA was extracted from these explants using RNAiso reagent (TaKaRa). RNA-seq was performed on pooled RNA formed by combining equal quantities; the sequencing was performed by a HiSeq 2000 device (Illumina, San Diego, CA, USA) housed at the Beijing Genomics Institute (Shenzhen, China). Adapter contamination sequences (match length ≥ 10 bp), low-quality reads and noncalled bases were discarded to obtain a set of clean reads for use in subsequent bioinformatic analyses. The transcriptomes were assembled into contigs using the Trinity program41, and the resulting unigenes were annotated using the Kyoto Encyclopedia of Genes and Genomes (KEGG) (www.genome.jp/kegg/kegg1.html), NCBI nonredundant protein (NR) (www.ncbi.nlm.nih.gov/refseq), NCBI nonredundant nucleotide (NT) (www.ncbi.nlm.nih.gov/nuccore), Swiss-Prot (www.uniprot.org), Cluster of Orthologous Groups (COG) (www.ncbi.nlm.nih.gov/COG/) and Gene Ontology (GO) (geneontology.org/page/go-database) databases. Differentially transcribed genes were subjected to both GO functional and KEGG pathway analyses.
Quantitative real-time PCR (qRT-PCR) analysis
Total RNA was extracted using RNAiso reagent (TaKaRa), treated with DNase to remove contaminating genomic DNA and reverse-transcribed using M-MLV reverse transcriptase. The resulting ss cDNA served as a template for a series of qRT-PCRs in which each reaction mixture consisted of 10 μL of SYBR Premix Ex Taq™ II (TaKaRa), 0.4 μL of each primer (10 μM) and 5 μL of 1 ng/μL template. The chrysanthemum EF1α gene (KF305681) served as a reference. The 2−ΔΔCt method was used to calculate the relative abundance of the various transcripts, and each qRT-PCR was carried out in triplicate.
Quantifying the GA content in chrysanthemum leaves
WT, OX. and RNAi plants were grown under an 8-h photoperiod for 30 days. Three of the youngest, fully opened leaves (0.5 g of fresh tissue) were harvested after they had been exposed to 3 h of light, with each line sampled in triplicate. The method used to determine the leaf GA content has been described previously23.
Yeast two-hybrid assays
Each of the sequences of CmBBX24, CmBBX24boxs, CmBBX24box1, and CmBBX24box2 were inserted into pGBKT7 vectors (Clontech), while CmMYB2 was inserted into a pGADT7 vector (Clontech). The primers used are listed in Table S1. The resulting pGBKT7-CmBBX24boxs, pGBKT7-CmBBX24box1, and pGBKT7-CmBBX24box2 constructs were cotransformed together with pGADT7-CmMYB2 into Y2HGold cells (Clontech) according to the “Yeast Transformation System 2” protocol. The transformed cells were subsequently incubated on tryptophan and leucine SD media and on tryptophan, leucine, histidine and adenine SD media in the presence or absence of X-α-gal. pGBKT7-53 and pGADT7-T served as positive controls, and pGBKT7-Lam pGADT7-T served as negative controls.
BiFC assays
The CmBBX24 and CmMYB2 open reading frames were inserted into the pSAT4A-nYFP and pSAT4A-cYFP vectors, respectively42. The primers used are listed in Table S1. The resulting constructs and the p35S::D53-RFP nuclear marker43 were transformed into onion epidermal cells using a PDS-1000He-driven particle bombardment device (Bio-Rad), after which the cells were cultivated on MS media in the dark for 16 h at 23 °C. YFP and RFP activity was observed using confocal laser scanning microscopy (LSM780, Zeiss, Oberkochen, Germany).
References
Fornara, F., De, M. A. & Coupland, G. SnapShot: control of flowering in Arabidopsis. Cell141, 550–550e2 (2010).
Song, Y. H., Ito, S. & Imaizumi, T. Flowering time regulation: photoperiod- and temperature-sensing in leaves. Trends Plant. Sci.18, 575–583 (2013).
Mouhu, K. et al. Identification of flowering genes in strawberry, a perennial SD plant. BMC Plant Biol.9, 122 (2009).
Hyun, Y. et al. Multi-layered regulation of SPL15 and cooperation with SOC1 integrate endogenous flowering pathways at the Arabidopsis shoot meristem. Dev. Cell37, 254–266 (2016).
Jung, J. H., Ju, Y., Seo, P. J., Lee, J. H. & Park, C. M. The SOC1-SPL module integrates photoperiod and gibberellic acid signals to control flowering time in Arabidopsis. Plant J.69, 577–588 (2012).
Oda, A. et al. CsFTL3, a chrysanthemum FLOWERING LOCUS T-like gene, is a key regulator of photoperiodic flowering in chrysanthemums. J. Exp. Bot.63, 1461–1477 (2012).
Wang, Z. Y. & Tobin, E. M. Constitutive expression of the CIRCADIAN CLOCK ASSOCIATED 1 (CCA1) gene disrupts circadian rhythms and suppresses its own expression. Cell93, 1207 (1998).
Kuno, N. et al. The novel MYB protein EARLY-PHYTOCHROME-RESPONSIVE1 is a component of a slave circadian oscillator in Arabidopsis. Plant Cell15, 2476–2488 (2003).
Tominaga, R. et al. Arabidopsis CAPRICE-LIKE MYB 3 (CPL3) controls endoreduplication and flowering development in addition to trichome and root hair formation. Development135, 1335–45 (2008).
Seo, E., Yu, J., Ryu, K. H., Lee, M. M. & Lee, I. WEREWOLF, a regulator of root hair pattern formation, controls flowering time through the regulation of FT mRNA stability. Plant Physiol.156, 1867–77 (2011).
Liu, L. et al. Elevated levels of MYB30 in the phloem accelerate flowering in Arabidopsis through the regulation of FLOWERING LOCUS T. PLoS ONE9, e89799 (2014).
Yan, Y. et al. A MYB-domain protein EFM mediates flowering responses to environmental cues in Arabidopsis. Dev. Cell30, 437–448 (2014).
Abe, M. et al. FE, a phloem-specific Myb-related protein, promotes flowering through transcriptional activation of flowering locus t and flowering locus t interacting protein 1. Plant J.83, 1059–1068 (2015).
Jung, C. et al. Overexpression of AtMYB44 enhances stomatal closure to confer abiotic stress tolerance in transgenic Arabidopsis. Plant Physiol.146, 623–635 (2008).
Liu, S. et al. R2R3 MYB transcription factor PtrMYB192 regulates flowering time in arabidopsis by activating flowering locus c. J. Plant Biol.56, 243–250 (2013).
Zhang, L. et al. The wheat MYB-related transcription factor TaMYB72 promotes flowering in rice. J. Integr. Plant. Biol.58, 701–704 (2016).
Gao, R., Gruber, M. Y., Amyot, L. & Hannoufa, A. SPL13 regulates shoot branching and flowering time in Medicago sativa. Plant. Mol. Biol.96, 1–15 (2018).
Cheng, P. L. et al. A transcriptomic analysis targeting genes involved in the floral transition of winter-flowering chrysanthemum. J. Plant. Growth. Regul.37, 1–13 (2017).
Ren, L. P. et al. Transcriptomic analysis of differentially expressed genes in the floral transition of the summer flowering chrysanthemum. BMC Genomics17, 673 (2016).
Shinoyama, H., Aida, R., Ichikawa, H., Nomura, Y. & Mochizuki, A. Genetic engineering of chrysanthemum (Chrysanthemum morifolium): current progress and perspectives. Plant Biotechnol.29, 323–337 (2012).
da Silva, Teixeira et al. Chrysanthemum biotechnology: Quo vadis?. Crit. ev. Plant. Sci.32, 21–52 (2013).
Shan, H. et al. Heterologous expression of the Chrysanthemum R2R3-MYB transcription factor CmMYB2 enhances drought and salinity tolerance, increases hypersensitivity to ABA and delays flowering in Arabidopsis thaliana. Mol. Biotechnol.51, 160–173 (2012).
Yang, Y. J. et al. A zinc finger protein regulates flowering time and abiotic stress tolerance in Chrysanthemum by modulating gibberellin biosynthesis. Plant Cell26, 2038 (2014).
Huang, P. et al. OsMYB511 encodes a MYB domain transcription activator early regulated by abiotic stress in rice. Genet. Mol. Res.14, 9506–17 (2015).
Li, H. L., Dong, G. & Peng, S. Q. Molecular characterization of the Jatropha curcas JcR1MYB1 gene encoding a putative R1-MYB transcription factor. Genet. Mol. Biol.37, 549 (2014).
Huang, C. et al. NbPHAN, a MYB transcriptional factor, regulates leaf development and affects drought tolerance in Nicotiana benthamiana. Physiol. Plant.149, 297–309 (2013).
He, Y. et al. Ectopic expression of a wheat MYB transcription factor gene, TaMYB73, improves salinity stress tolerance in Arabidopsis thaliana. J. Exp. Bot.63, 1511 (2012).
Wang, Y. et al. CmMYB19 Over-expression improves aphid tolerance in chrysanthemum by promoting lignin synthesis. Int. J. Mol. Sci.18, 619 (2017).
Yant, L., Mathieu, J. & Schmid, M. Just say no: floral repressors help Arabidopsis bide the time. Curr. Opin. Plant. Biol.12, 580–586 (2009).
Wahl, V. et al. Regulation of flowering by trehalose-6-phosphate signaling in Arabidopsis thaliana. Science339, 704 (2013).
Boss, P. K., Bastow, R. M., Mylne, J. S. & Dean, C. Multiple pathways in the decision to flower: enabling, promoting, and resetting. Plant Cell16(Suppl), S18–S31 (2004).
Zhao, C., Hanada, A., Yamaguchi, S., Kamiya, Y. & Beers, E. P. The Arabidopsis Myb genes MYR1 and MYR2 are redundant negative regulators of flowering time under decreased light intensity. Plant J.66, 502–515 (2011).
Zhang, Y. et al. The cyclophilin CYP20-2 modulates the conformation of BRASSINAZOLE-RESISTANT1, which binds the promoter of FLOWERING LOCUS D to regulate flowering in Arabidopsis. Plant Cell25, 2504–21 (2013).
Curtis, M. D. & Grossniklaus, U. A gateway cloning vector set for high-throughput functional analysis of genes in planta. Plant Physiol.133, 462–469 (2003).
Murashige, T. & Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant.15, 473–497 (1962).
Wu, F. H. et al. Tape-Arabidopsis Sandwich - a simpler Arabidopsis protoplast isolation method. Plant Methods5, 16 (2009).
Fujikawa, Y. & Kato, N. Split luciferase complementation assay to study protein-protein interactions in Arabidopsis protoplasts. Plant J.52, 185 (2007).
Becker, D. Binary vectors which allow the exchange of plant selectable markers and reporter genes. Nucleic Acids Res.18, 203 (1990).
Alvarez, J. P. et al. Endogenous and synthetic microRNAs stimulate simultaneous, efficient, and localized regulation of multiple targets in diverse species. Plant Cell18, 1134–51 (2006).
Cui, X. L., Chen, F. D. & Chen, S. M. Establishment of regeneration and transformation system of ground-cover chrysanthemum Yuhuaxunzhang. JNAU China32, 40–46 (2009).
Grabherr, M. G. et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol.29, 644–652 (2011).
Citovsky, V. et al. Subcellular localization of interacting proteins by bimolecular fluorescence complementation in planta. J. Mol. Biol.362, 1120–1131 (2006).
Zhou, F. et al. D14-SCFD3-dependent degradation of D53 regulates strigolactone signalling. Nature504, 406 (2013).
Acknowledgements
This research was supported by the National Natural Science Foundation of China (31572159), the Jiangsu Modern Agricultural Industry Technology System Construction Project (JATS[2018]278), the Fundamental Research Funds for Central Universities (KJQN201815, KYZ201832), the Priority Academic Program Development of Jiangsu Higher Education Institutions and the Jiangsu Planned Projects for Postdoctoral Research Funds.
Author information
Authors and Affiliations
Contributions
F.D.C., J.F.J., and S.M.C. conceived and designed the research; L.Z., Y.X.G., and Z.H.Z. conducted the experiments; Y.N.L., A.P.S., and M.A.J. analyzed the data; and L.Z. wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhu, L., Guan, Y., Liu, Y. et al. Regulation of flowering time in chrysanthemum by the R2R3 MYB transcription factor CmMYB2 is associated with changes in gibberellin metabolism. Hortic Res 7, 96 (2020). https://doi.org/10.1038/s41438-020-0317-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41438-020-0317-1
This article is cited by
-
Decoding the Flower-Anthesis Mechanism in Cestrum nocturnum and Cestrum diurnum
Journal of Plant Growth Regulation (2024)
-
Multi-locus genome-wide association studies reveal the dynamic genetic architecture of flowering time in chrysanthemum
Plant Cell Reports (2024)
-
PagMYB151 facilitates proline accumulation to enhance salt tolerance of poplar
BMC Genomics (2023)
-
An Acer palmatum R2R3-MYB Gene, ApMYB77, Confers Freezing and Drought Tolerance in Arabidopsis thaliana
Journal of Plant Growth Regulation (2023)
-
Overexpression of the CmJAZ1-like gene delays flowering in Chrysanthemum morifolium
Horticulture Research (2021)