Abstract
Purpose
Expanded preconception carrier screening (ECS) identifies at-risk couples (ARCs) for multiple diseases. ECS reports currently include only pathogenic/likely pathogenic variants (P/LPVs). Variants of unknown significance (VUS) are not reported, unlike genomic or chromosomal array test results in other post/prenatal settings. Couples who are P/LP and VUS carriers (P/LP*VUS) may be at risk, particularly in genes with high P/LP carrier rates. We examined the possible contribution of P/LP*cVUS (coding, nonsynonymous VUS) matings to ECS yield in an Ashkenazi Jewish cohort, a population with well-established preconception screening.
Methods
We analyzed 672 Ashkenazi Jewish genome sequences (225,456 virtual matings) for variants in three different gene sets and calculated the rates of P/LP*P/LP and P/LP*cVUS matings.
Results
Across 180 genes, we identified 4671 variants: 144 (3.1%) P/LP and 1963 (42%) VUS. Across gene sets, the proportion of P/LP*P/LP and P/LP*cVUS ARCs was 2.7–3.8% and 6.8–7.5%, respectively.
Conclusion
Disregarding VUS in ECS may miss ARCs. Even if only 10% of couples currently classified as P/LP*cVUS are ultimately reclassified as P/LP*P/LP, ECS yield would increase by ≈20%. While current understanding of VUS precludes VUS reporting in ECS, these findings underscore the importance of VUS reclassification. This will crucially depend on enlarging population frequency databases, especially of affected individuals.
Similar content being viewed by others
INTRODUCTION
Preconception carrier screening (PCS) aims to identify couples at risk for having offspring with genetic diseases and facilitate informed reproductive choices. PCS was originally limited to a few disorders in specific populations, e.g., Tay–Sachs disease in Ashkenazi Jews (AJ).1 As recently as 2015, the American College of Obstetricians and Gynecologists (ACOG) and the American College of Medical Genetics and Genomics (ACMG) recommendations focused on ethnicity-based testing, with only nine diseases indicated for AJ. Pan-ethnic testing was recommended only for hemoglobinopathies, cystic fibrosis, and spinal muscular atrophy.1 However, next-generation sequencing has led to rapid adoption of expanded carrier screening (ECS), with full sequencing of hundreds of genes.2 In 2017, the ACOG indicated ECS as an acceptable strategy.2 Simulated and real-world ECS results demonstrate greater yield compared with the 2015 ACOG/ACMG recommendations: a 94-gene panel would double the detection rate of hypothetical affected fetuses in an ethnically diverse population.3 Increased yields are achieved by sequencing entire genes (versus targeted variant testing), adding genes with high carrier rates and milder phenotypes (e.g., HFE), and including an ever-growing number of rare disease genes.4,5,6 The major increment in ECS yield is explained by full sequencing of a modest number of genes with high carrier rates.6
Fuller understanding of ECS outcomes should address variants of unknown significance (VUS). As an additional source of what may prove to be pathogenic variants, VUS could significantly affect future ECS yields, particularly in genes with high carrier rates of pathogenic/likely pathogenic variants (P/LPVs). Currently, ECS reports do not include VUS1 and clinical laboratories do not perform prenatal testing for ECS-detected VUS.
We assessed the effect of VUS on detection rates of at-risk couples (ARCs). We compared the yield of complete gene sequencing versus targeted variant testing in the same gene set, and examined the effect of expanding the number of genes sequenced. We based our analysis on 672 genome sequences of AJ, a population typical of PCS.
MATERIALS AND METHODS
See Supplementary Information for details.
Sequencing data
We used phased genome sequences (without copy-number variant [CNV] analysis) of 672 previously published, unrelated AJ samples.7
Genes and variants analyzed
We defined recessive AJ, pan-ethnic, and combined gene sets (Table S1). Genes included were present in at least two of three commercial ECS panels: Baylor,8 Mount Sinai,9 and Jewish Genetic Disease Consortium (JGDC)10 for the AJ gene set, and Baylor,8 Mount Sinai,9 and Counsyl (currently Myriad)11 for the pan-ethnic gene set. The combined gene set is comprised of both AJ and pan-ethnic sets. Variants were classified using InterVar,12 an automated ACMG classifier, and ClinVar.13 Manual reclassification was also performed as follows: (1) variants homozygous in ≥5% of our cohort were reclassified as benign, (2) variants homozygous in 1–5% of our cohort were reclassified as benign if the homozygosity rate was >1% in any population in gnomAD,14 and (3) variants with discrepant InterVar12 and ClinVar13 classifications (Table S2) were reclassified manually (Supplementary Methods).
Virtual matings
We tested all possible 225,456 (672*671/2) theoretical matings, irrespective of gender. When more than one variant in the same gene was present in a sample, the most pathogenic variant was used.
RESULTS
Genes analyzed and variant classification
We analyzed 180 genes underlying autosomal recessive disorders (see “Materials and methods,” Table S1). This combined gene set included 168 genes representing pan-ethnic ECS panels (pan-ethnic gene set) and 55 genes representing AJ ECS panels (AJ gene set). Forty-three genes were included in both pan-ethnic and AJ ECS panels. Following variant classification (see “Materials and methods”) we computed carrier rates and types of ARCs.
In 672 AJ genome sequences, we identified 4671 variants in the exons ± 10 bp of the 180 selected genes. These variants were classified into three categories: P/LP (N = 144, 3.1%), VUS (N = 1963, 42%), and benign/likely benign (B/LB) (N = 2564, 54.9%).
Founder P/LP variants
Founder variants (FVs) were defined as P/LPVs included in AJ targeted variant panels15,16 and with combined >1% carrier frequency per gene.15 Overall, we identified FVs in 33/55 genes in the AJ gene set. FV carrier rates (Table 1) are similar to previous reports.15,16
Four individuals were FV homozygotes, each in a different gene: PAH (phenylketonuria and hyperphenylalaninemia), GBA (Gaucher disease), F11 (factor XI deficiency), and MEFV (familial Mediterranean fever) (Table 1). These diseases have variable phenotypes, and homozygosity for the specific FVs observed can be asymptomatic (Supplementary Results).
Nonfounder P/LP variants in the AJ gene set
We identified 48 nonfounder P/LPVs in the AJ gene set that were heterozygous in 112/672 (16.7%) samples (Table S3). Most of these variants (40/48) are in genes also included in the pan-ethnic gene set, but 20/672 (3%) samples were heterozygous for a nonfounder P/LPV in an AJ-specific gene. There were no homozygotes for nonfounder P/LPVs, nor compound heterozygotes for a nonfounder P/LPV and a FV.
P/LP variants in the pan-ethnic gene set
We identified 84 P/LPVs in the pan-ethnic gene set that were heterozygous in 116/672 (17.3%) samples (Tables S3, S4). Of these, 40/84 were in genes also included in the AJ gene set (Table S3, underlined), and 44/84 were in genes unique to the pan-ethnic gene set (Table S4). One individual was homozygous for a mild phenotype P/LPV in the BTD gene (MIM609019) and one was homozygous for a mild phenotype P/LPV in the BCS1L gene (MIM 603647) (Supplementary Results; Table S4).
Overall, for 180 genes, the mean number of P/LPVs per sample was 1.1 (range 0–6). There were 213/672 (31.7%) samples with no P/LPVs.
VUS
As noted above, 1963/4671 (42.1%) variants were classified as VUS. Of these, 733/1963 (37.3%) were coding VUS (cVUS), i.e., exonic, non-UTR, nonsynonymous VUS.
At-risk couples: virtual matings
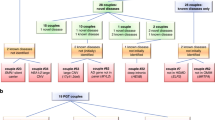
To determine ARCs rates for autosomal recessive conditions, we examined all possible matings between the 672 samples (225,456 theoretical couples). We compared the outcomes of targeted AJ FV testing and full sequencing of the AJ, pan-ethnic, and the combined gene sets. We evaluated the rates of matings where (1) both partners were carriers of a P/LPV in the same gene (P/LP*P/LP) or (2) both partners were carriers of variants in the same gene: one of a P/LPV and the other of a cVUS, (P/LP*cVUS). We did not consider cVUS*cVUS matings to be clinically significant.1
P/LP*P/LP matings
For AJ FVs, 6454/225,456 (2.9%) of possible matings represent P/LP*P/LP ARCs (Table 2). The fully sequenced AJ gene set (which includes both founder and nonfounder variants) resulted in 7839/225,456 (3.5%) ARCs. This difference is partly driven by nonfounder variants in CFTR (MIM 602421) and MEFV (MIM 608107) resulting in variable (including mild) phenotypes. Using the pan-ethnic gene set, 6183/225,456 (2.7%) matings were P/LP*P/LP ARCs. The rate of P/LP*P/LP matings was higher in the AJ gene set (3.5%) than in the pan-ethnic gene set (2.7%), even though the AJ gene set includes threefold fewer genes, due to the contribution of frequent FVs in genes unique to the AJ gene set (2080 ARCs for F11, 190 for LDLR, 45 for GBE1; Table 1).
P/LP*cVUS matings
P/LP*cVUS matings may represent an ARC if the cVUS is ultimately classified as P/LP. Depending on the gene set used, 6.8–7.5% of matings were P/LP*cVUS (Table 2). The proportion of P/LP*cVUS couples was 1.9–2.6 times greater than that of P/LP*P/LP couples, depending on the gene set.
Modeling the effect of cVUS reclassification
We evaluated the effects of cVUS reclassification on P/LP*cVUS frequency. Information on allele frequencies in affected and unaffected individuals is likely to accrue rapidly. Also, allelic data can be assessed by determining homozygosity in unaffected individuals and by using phased data, to determine if a cVUS occurs in trans to a P/LPV in unaffected individuals (in which case it would be considered LB).
Overall, 106/1963 (5.4%) VUS were homozygous in one or more individuals (Table S5), of which 21/106 (19.8%) were homozygous cVUS. Of 1963 VUS, 33 (1.7%) were found in trans to a P/LPV, either a FV or nonfounder variant, including 15/33 (45.4%) which were in trans cVUS (Table S6). Of these 33 variants, 3 were also homozygous in one or more individuals. Overall, 4.9% of cVUS were thus reclassified as LB/B.
Hypothetical fulfillment of ACMG criteria17 PM2 (variant absent in unaffected controls) or PS4 (variant significantly more prevalent in affected individuals versus controls) would respectively change 19/733 (2.6%) and 712/733 (97.1%) of cVUS classifications to P/LP. The much greater potential impact of PS4 fulfillment indicates that obtaining variant frequency data in affected individuals is particularly important for VUS reclassification.
DISCUSSION
We assessed PCS yield using different strategies: AJ FV testing or full sequencing of different gene sets. We focused on the possible contribution of P/LP*cVUS matings to testing yield. Rates of at-risk P/LP*P/LP matings were 2.9% for targeted AJ FV testing, versus 3.5%, 2.7%, and 3.8% for sequencing of AJ, pan-ethnic, and combined gene sets, respectively. This is consistent with previous estimates of 3–4% ARC yield with ECS in AJ.16 Rates of P/LP*cVUS matings were 6.8%, 6.9%, and 7.5% in the AJ, pan-ethnic, and combined gene sets, respectively. The rate of P/LP*cVUS matings is ≈1.9–2.7-fold greater than that of P/LP*P/LP matings. As a screening test, ECS is not designed to detect 100% of ARCs. However, our results indicate that even if only a small proportion of cVUS are ultimately determined to be P/LP, variants currently classified as cVUS could significantly impact ECS yield.
Compared with targeted variant PCS, greater ECS yield has been achieved, as expected, by fully sequencing genes and by increasing the number of genes sequenced. We found that even in a well-characterized population like AJ, where the variant spectrum is well established, full sequencing (of an AJ gene set) increased ARC detection by 21%. This reflects the contribution of rare P/LPVs in genes with high carrier rates for common variants, and is consistent with previous studies demonstrating greater yield by expanding the variant repertoire in AJ.16 In contrast, adding genes (the combined gene set) increased ARC detection by only 8.6%.
In cancer predisposition genes, studies found that 10–25% of VUS will ultimately be reclassified as P/LP.18 There are no comparable data in the preconception field. However, even considering only cVUS, and assuming only 10% of cVUS are reclassified as P/LPVs, the added yield would be ≈19% for the AJ gene set, and ≈20% for the combined gene set. These estimates disregard the possibility that some noncoding VUS could prove to be P/LP.
Variant classification is a major challenge. ACMG classification categories include population data, computational predictions, functional evidence, segregation data, allelic information, and data from disease databases.17 Functional analyses of variant effects are difficult to perform at large scale. More obtainable are cosegregation and allelic data, which inform six ACMG criteria, and frequency in healthy and affected individuals, which inform three ACMG criteria. Our results demonstrate the power of using allelic data in VUS reclassification: we reclassified 4.9% of cVUS as B/LB by analyzing unaffected individuals for homozygosity or for occurrence of a VUS in trans to a P/LPV. Modeling the effect of two frequency criteria showed that hypothetical fulfillment of PS4 (greater prevalence in affecteds versus controls) would reclassify 97.1% of cVUS to P/LP. In contrast, hypothetical fulfillment of PM2 (variant absent in controls) would reclassify only 2.6% of cVUS to P/LP. This underscores the importance of efforts such as ClinVar13 to aggregate variant data in affected individuals.
We note that VUS are reported in other settings, both postnatally (e.g., cancer predisposition in adults, exome sequencing in children) and prenatally (e.g., chromosomal microarrays). We believe ECS results should not, and currently do not, include VUS, which cannot be used for prenatal diagnosis or pregestational testing. Results of this study should not be interpreted as a call to report VUS in ECS, but only as evidence for the importance of further research. Clinically, reanalysis of ECS results prior to each pregnancy may be warranted, similarly to periodic reanalysis of negative exome results in pediatric disease.19
Limitations of this study include omission of X-linked conditions, including fragile X, and large deletions/duplications (e.g., the common SMN1 deletion). AJ samples may be particularly amenable to using homozygosity and occurrence in trans to P/LPVs as methods for VUS reclassification.
Conclusions
ECS detects P/LP*cVUS in ≈7% of couples. Even if only 10% of cVUS are ultimately reclassified as P/LPVs, ARC detection rates would increase by ≈20%. Enlarging population frequency variant databases, particularly for affected individuals, would facilitate VUS reclassification for ECS. Reanalysis of ECS results prior to each pregnancy may also be warranted.
References
Edwards JG, Feldman G, Goldberg J, et al. Expanded carrier screening in reproductive medicine-points to consider. Obstet Gynecol. 2015;125:653–662.
Romero S, Rink B, Biggio JR, et al. Carrier screening in the age of genomic medicine: number 690. Obstet Gynecol. 2017;129:35–40.
Haque IS, Lazarin GA, Kang HP, et al. Modeled fetal risk of genetic diseases identified by expanded carrier screening. JAMA. 2016;316:734–742.
Peyser A, Singer T, Mullin C, et al. Comparing ethnicity-based and expanded carrier screening methods at a single fertility center reveals significant differences in carrier rates and carrier couple rates. Genet Med. 2018;21:1400–1406.
Terhaar C, Teed N, Allen R, et al. Clinical experience with multigene carrier panels in the reproductive setting. Prenat Diagn. 2018;38:572–577.
Guo MH, Gregg AR. Estimating yields of prenatal carrier screening and implications for design of expanded carrier screening panels. Genet Med. 2019;21:1940–1947.
Lencz T, Yu J, Palmer C, et al. High-depth whole genome sequencing of an Ashkenazi Jewish reference panel: enhancing sensitivity, accuracy, and imputation. Hum Genet. 2018;137:343–355.
Baylor Genetics. GeneAware panel listing. https://geneaware.clinical.bcm.edu/GeneAware/Providers/PanelList.aspx. Accessed 4 September 2018.
Sema4. Test catalog. https://sema4.com/products/test-catalog/. Accessed 4 September 2018.
Jewish Genetic Disease Consortium (JGDC). Diseases. https://www.jewishgeneticdiseases.org/jewish-genetic-diseases/. Accessed 4 September 2018.
Counsyl. Foresight™ carrier screen disease list. https://s3.amazonaws.com/static.counsyl.com/website/PDFs/Foresight+Universal+Disease+List.pdf. Accessed 4 September 2018.
Li Q, Wang K. InterVar: clinical interpretation of genetic variants by the 2015 ACMG-AMP guidelines. Am J Hum Genet. 2017;100:267–280.
Landrum MJ, Lee JM, Benson M, et al. ClinVar: improving access to variant interpretations and supporting evidence. Nucleic Acids Res. 2018;46:1062–1067.
Lek M, Karczewski KJ, Minikel EV, et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature. 2016;536:285–291.
The Israeli National Genetic Database (INGD). https://medicine.ekmd.huji.ac.il/En/INGD/Pages/default.aspx. Accessed 22 November 2018.
Baskovich B, Hiraki S, Upadhyay K, et al. Expanded genetic screening panel for the Ashkenazi Jewish population. Genet Med. 2016;18:522–528.
Richards S, Aziz N, Bale S, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–424.
Mersch J, Brown N, Pirzadeh-Miller S, et al. Prevalence of variant reclassification following hereditary cancer genetic testing. JAMA. 2019;320:1266–1274.
Nambot S, Thevenon J, Kuentz P, et al. Clinical whole-exome sequencing for the diagnosis of rare disorders with congenital anomalies and/or intellectual disability: substantial interest of prospective annual reanalysis. Genet Med. 2017;20:645–654.
Bonnefont JP, Djouadi F, Prip-Buus C, et al. Carnitine palmitoyltransferases 1 and 2: biochemical, molecular and medical aspects. Mol Aspects Med. 2004;25:495–520.
Acknowledgements
S.C. thanks the Israel Science Foundation Grant 407/17 and the Abisch Frenkel Foundation. We thank the Ashkenazi Genome Consortium for the AJ genomic data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
S.C. is a paid consultant to MyHeritage. D.M.B. declares stock ownership and compensation from Igentify, where he serves as the Chief Executive Officer. The other authors declare no conflicts of interest.
Ethical approval
Access to the data was provided by The Ashkenazi Genome Consortium (TAGC).7 Written informed consent was obtained from all participants and institutional review board (IRB) approval was obtained from all participating institutions. In the United States: Albert Einstein College of Medicine, New York, NY; Columbia University, New York, NY; Mount Sinai School of Medicine, New York, NY; New York University, New York, NY; North Shore University Hospital–Long Island Jewish Medical Center, NY; Yale University, New Haven, CT; University of Pittsburgh, Pittsburgh, PA; Johns Hopkins University, Baltimore, MD; Cedars-Sinai Medical Center, Los Angeles, CA. In Canada: The University of Toronto, Toronto. In Israel: The Hebrew University of Jerusalem, Jerusalem; Tel Aviv Sourasky Medical Center, Tel Aviv; Rambam Medical Center, Haifa; The National Genetics Ethics Committee of the Israeli Ministry of Health. (See Supplementary materials and methods for references.)
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Fridman, H., Behar, D.M., Carmi, S. et al. Preconception carrier screening yield: effect of variants of unknown significance in partners of carriers with clinically significant variants. Genet Med 22, 646–653 (2020). https://doi.org/10.1038/s41436-019-0676-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41436-019-0676-x
Keywords
This article is cited by
-
Ordering genetic testing by neurologists: points to consider
Journal of Neurology (2023)
-
A capillary electrophoresis-based multiplex PCR assay for expanded carrier screening in the eastern Han Chinese population
npj Genomic Medicine (2022)
-
Assessing clinical utility of preconception expanded carrier screening regarding residual risk for neurodevelopmental disorders
npj Genomic Medicine (2022)
-
Fine-scale population structure and demographic history of British Pakistanis
Nature Communications (2021)