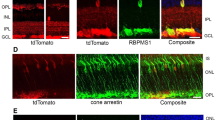
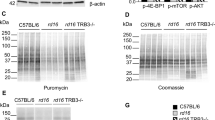
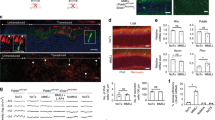
Abstract
Therapies for genetic disorders caused by mutated mitochondrial DNA are an unmet need, in large part due barriers in delivering DNA to the organelle and the absence of relevant animal models. We injected into mouse eyes a mitochondrially targeted Adeno-Associated-Virus (MTS-AAV) to deliver the mutant human NADH ubiquinone oxidoreductase subunit I (hND1/m.3460 G > A) responsible for Leber’s hereditary optic neuropathy, the most common primary mitochondrial genetic disease. We show that the expression of the mutant hND1 delivered to retinal ganglion cells (RGC) layer colocalizes with the mitochondrial marker PORIN and the assembly of the expressed hND1 protein into host respiration complex I. The hND1-injected eyes exhibit hallmarks of the human disease with progressive loss of RGC function and number, as well as optic nerve degeneration. We also show that gene therapy in the hND1 eyes by means of an injection of a second MTS-AAV vector carrying wild-type human ND1 restores mitochondrial respiratory complex I activity, the rate of ATP synthesis and protects RGCs and their axons from dysfunction and degeneration. These results prove that MTS-AAV is a highly efficient gene delivery approach with the ability to create mito-animal models and has the therapeutic potential to treat mitochondrial genetic diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Kauppila JHK, Baines HL, Bratic A, Simard ML, Freyer C, Mourier A, et al. A Phenotype-Driven Approach to Generate Mouse Models with Pathogenic mtDNA Mutations Causing Mitochondrial Disease. Cell Rep. 2016;16:2980–2990.
Jurkute N, Yu-Wai-Man P. Leber hereditary optic neuropathy: bridging the translational gap. Curr Opin Ophthalmol. 2017;28:403–409.
Yu-Wai-Man P, Griffiths PG, Hudson G, Chinnery PF. Inherited mitochondrial optic neuropathies. J Med Genet. 2009;46:145–58.
Rasool N, Lessell S, Cestari DM. Leber Hereditary Optic Neuropathy: Bringing the Lab to the Clinic. Semin Ophthalmol. 2016;31:107–16.
Harding AE, Sweeney MG, Govan GG, Riordan-Eva P. Pedigree analysis in Leber hereditary optic neuropathy families with a pathogenic mtDNA mutation. Am J Hum Genet. 1995;57:77–86.
Yu-Wai-Man P, Votruba M, Moore AT, Chinnery PF. Treatment strategies for inherited optic neuropathies: past, present and future. Eye. 2014;28:521–37.
Newman NJ, Biousse V. Hereditary optic neuropathies. Eye. 2004;18:1144–60.
Jankauskaite E, Bartnik E, Kodron A. Investigating Leber’s hereditary optic neuropathy: Cell models and future perspectives. Mitochondrion. 2017;32:19–26.
Klopstock T, Metz G, Yu-Wai-Man P, Buchner B, Gallenmuller C, Bailie M, et al. Persistence of the treatment effect of idebenone in Leber’s hereditary optic neuropathy. Brain. 2013;136:e230.
Guy J, Feuer WJ, Davis JL, Porciatti V, Gonzalez PJ, Koilkonda RD, et al. Gene Therapy for Leber Hereditary Optic Neuropathy: Low- and Medium-Dose Visual Results. Ophthalmology. 2017;124:1621–1634.
Yu-Wai-Man P, Newman NJ, Carelli V, Moster ML, Biousse V, Sadun AA, et al. Bilateral visual improvement with unilateral gene therapy injection for Leber hereditary optic neuropathy. Sci Transl Med. 2020;12:eaaz7423.
Weiss JN, Levy S, Benes SC. Stem Cell Ophthalmology Treatment Study (SCOTS): bone marrow-derived stem cells in the treatment of Leber’s hereditary optic neuropathy. Neural Regen Res. 2016;11:1685–1694.
Rahman S. Emerging aspects of treatment in mitochondrial disorders. J Inherit Metab Dis. 2015;38:641–53.
Guy J, Qi X, Pallotti F, Schon EA, Manfredi G, Carelli V, et al. Rescue of a mitochondrial deficiency causing Leber Hereditary Optic Neuropathy. Ann Neurol. 2002;52:534–42.
Qi X, Sun L, Lewin AS, Hauswirth WW, Guy J. The mutant human ND4 subunit of complex I induces optic neuropathy in the mouse. Investig Ophthalmol Vis Sci. 2007;48:1–10.
Koilkonda R, Yu H, Talla V, Porciatti V, Feuer WJ, Hauswirth WW, et al. LHON gene therapy vector prevents visual loss and optic neuropathy induced by G11778A mutant mitochondrial DNA: biodistribution and toxicology profile. Investig Ophthalmol Vis Sci. 2014;55:7739–53.
Cwerman-Thibault H, Augustin S, Lechauve C, Ayache J, Ellouze S, Sahel JA, et al. Nuclear expression of mitochondrial ND4 leads to the protein assembling in complex I and prevents optic atrophy and visual loss. Mol Ther Methods Clin Dev. 2015;2:15003.
Baracca A, Solaini G, Sgarbi G, Lenaz G, Baruzzi A, Schapira AH, et al. Severe impairment of complex I-driven adenosine triphosphate synthesis in leber hereditary optic neuropathy cybrids. Arch Neurol. 2005;62:730–6.
Cwerman-Thibault H, Augustin S, Ellouze S, Sahel JA, Corral-Debrinski M. Gene therapy for mitochondrial diseases: Leber Hereditary Optic Neuropathy as the first candidate for a clinical trial. C R Biol. 2014;337:193–206.
Feuer WJ, Schiffman JC, Davis JL, Porciatti V, Gonzalez P, Koilkonda RD, et al. Gene Therapy for Leber Hereditary Optic Neuropathy: Initial Results. Ophthalmology. 2016;123:558–70.
Koilkonda RD, Yu H, Chou TH, Feuer WJ, Ruggeri M, Porciatti V, et al. Safety and effects of the vector for the Leber hereditary optic neuropathy gene therapy clinical trial. JAMA Ophthalmol. 2014;132:409–20.
Guy J, Feuer WJ, Porciatti V, Schiffman J, Abukhalil F, Vandenbroucke R, et al. Retinal ganglion cell dysfunction in asymptomatic G11778A: Leber hereditary optic neuropathy. Investig Ophthalmol Vis Sci. 2014;55:841–8.
Koilkonda RD, Guy J. Leber’s Hereditary Optic Neuropathy-Gene Therapy: From Benchtop to Bedside. J Ophthalmol. 2011;2011:179412.
Bacman SR, Williams SL, Pinto M, Peralta S, Moraes CT. Specific elimination of mutant mitochondrial genomes in patient-derived cells by mitoTALENs. Nat Med. 2013;19:1111–3.
Mok BY, de Moraes MH, Zeng J, Bosch DE, Kotrys AV, Raguram A, et al. A bacterial cytidine deaminase toxin enables CRISPR-free mitochondrial base editing. Nature. 2020;583:631–637.
Lee H, Lee S, Baek G, Kim A, Kang BC, Seo H, et al. Mitochondrial DNA editing in mice with DddA-TALE fusion deaminases. Nat Commun. 2021;12:1190.
Yu H, Koilkonda RD, Chou TH, Porciatti V, Ozdemir SS, Chiodo V, et al. Gene delivery to mitochondria by targeting modified adenoassociated virus suppresses Leber’s hereditary optic neuropathy in a mouse model. Proc Natl Acad Sci USA. 2012;109:E1238–47.
Yu H, Ozdemir SS, Koilkonda RD, Chou TH, Porciatti V, Chiodo V, et al. Mutant NADH dehydrogenase subunit 4 gene delivery to mitochondria by targeting sequence-modified adeno-associated virus induces visual loss and optic atrophy in mice. Mol Vis. 2012;18:1668–83.
Yu H, Koilkonda RD, Chou TH, Porciatti V, Mehta A, Hentall ID, et al. Consequences of zygote injection and germline transfer of mutant human mitochondrial DNA in mice. Proc Natl Acad Sci USA. 2015;112:E5689–98.
Yu H, Porciatti V, Lewin A, Hauswirth W, Guy J. Longterm Reversal of Severe Visual Loss by Mitochondrial Gene Transfer in a Mouse Model of Leber Hereditary Optic Neuropathy. Sci Rep. 2018;8:5587.
Yu H, Sant DW, Wang G, Guy J. Mitochondrial Transfer of the Mutant Human ND6T14484C Gene Causes Visual Loss and Optic Neuropathy. Transl Vis Sci Technol. 2020;9:1.
Porciatti V. The mouse pattern electroretinogram. Doc Ophthalmol. 2007;115:145–53.
Yang X, Chou TH, Ruggeri M, Porciatti V. A new mouse model of inducible, chronic retinal ganglion cell dysfunction not associated with cell death. Investig Ophthalmol Vis Sci. 2013;54:1898–904.
Ruggeri M, Wehbe H, Jiao S, Gregori G, Jockovich ME, Hackam A, et al. In vivo three-dimensional high-resolution imaging of rodent retina with spectral-domain optical coherence tomography. Investig Ophthalmol Vis Sci. 2007;48:1808–14.
Weber-Lotfi F, Ibrahim N, Boesch P, Cosset A, Konstantinov Y, Lightowlers RN, et al. Developing a genetic approach to investigate the mechanism of mitochondrial competence for DNA import. Biochim Biophys Acta. 2009;1787:320–7.
Koulintchenko M, Temperley RJ, Mason PA, Dietrich A, Lightowlers RN. Natural competence of mammalian mitochondria allows the molecular investigation of mitochondrial gene expression. Hum Mol Genet. 2006;15:143–54.
Hussain SA, Yalvac ME, Khoo B, Eckardt S, McLaughlin KJ. Adapting CRISPR/Cas9 System for Targeting Mitochondrial Genome. Front Genet. 2021;12:627050.
Wang Y, Hu LF, Cui PF, Qi LY, Xing L, Jiang HL. Pathologically Responsive Mitochondrial Gene Therapy in an Allotopic Expression-Independent Manner Cures Leber’s Hereditary Optic Neuropathy. Adv Mater. 2021;33:e2103307.
Harvey AR, Kamphuis W, Eggers R, Symons NA, Blits B, Niclou S, et al. Intravitreal injection of adeno-associated viral vectors results in the transduction of different types of retinal neurons in neonatal and adult rats: a comparison with lentiviral vectors. Mol Cell Neurosci. 2002;21:141–57.
Martin KR, Quigley HA, Zack DJ, Levkovitch-Verbin H, Kielczewski J, Valenta D, et al. Gene therapy with brain-derived neurotrophic factor as a protection: retinal ganglion cells in a rat glaucoma model. Investig Ophthalmol Vis Sci. 2003;44:4357–65.
Nickells RW, Schmitt HM, Maes ME, Schlamp CL. AAV2-Mediated Transduction of the Mouse Retina After Optic Nerve Injury. Investig Ophthalmol Vis Sci. 2017;58:6091–6104.
Shevtsova Z, Malik JM, Michel U, Bahr M, Kugler S. Promoters and serotypes: targeting of adeno-associated virus vectors for gene transfer in the rat central nervous system in vitro and in vivo. Exp Physiol. 2005;90:53–9.
Ojala D, Montoya J, Attardi G. tRNA punctuation model of RNA processing in human mitochondria. Nature. 1981;290:470–4.
Falkenberg M, Larsson NG, Gustafsson CM. DNA replication and transcription in mammalian mitochondria. Annu Rev Biochem. 2007;76:679–99.
Facucho-Oliveira JM, St John JC. The relationship between pluripotency and mitochondrial DNA proliferation during early embryo development and embryonic stem cell differentiation. Stem Cell Rev Rep. 2009;5:140–58.
Montoya J, Gaines GL, Attardi G. The pattern of transcription of the human mitochondrial rRNA genes reveals two overlapping transcription units. Cell. 1983;34:151–9.
Yu-Wai-Man P, Votruba M, Burte F, La Morgia C, Barboni P, Carelli V. A neurodegenerative perspective on mitochondrial optic neuropathies. Acta Neuropathol. 2016;132:789–806.
Finsterer J, Zarrouk-Mahjoub S. Leber’s hereditary optic neuropathy is multiorgan not mono-organ. Clin Ophthalmol. 2016;10:2187–2190.
Pilz YL, Bass SJ, Sherman J. A Review of Mitochondrial Optic Neuropathies: From Inherited to Acquired Forms. J Optometry. 2017;10:205–214.
Sadun AA, La Morgia C, Carelli V. Mitochondrial optic neuropathies: our travels from bench to bedside and back again. Clin Exp Ophthalmol. 2013;41:702–12.
Wang MY, Sadun AA. Drug-related mitochondrial optic neuropathies. J Neuro-ophthalmol. 2013;33:172–8.
Kroemer G, Galluzzi L, Brenner C. Mitochondrial membrane permeabilization in cell death. Physiol Rev. 2007;87:99–163.
Sadun AA. Mitochondrial optic neuropathies. J Neurol Neurosurg Psychiat. 2002;72:423–5.
Sadun AA, Win PH, Ross-Cisneros FN, Walker SO, Carelli V. Leber’s hereditary optic neuropathy differentially affects smaller axons in the optic nerve. Trans Am Ophthalmol Soc. 2000;98:223–32.
Wallace DC, Lott MT. Leber Hereditary Optic Neuropathy: Exemplar of an mtDNA Disease. Handb Exp Pharmacol. 2017;240:339–376.
Theodorou-Kanakari A, Karampitianis S, Karageorgou V, Kampourelli E, Kapasakis E, Theodossiadis P, et al. Current and Emerging Treatment Modalities for Leber’s Hereditary Optic Neuropathy: A Review of the Literature. Adv Ther. 2018;35:1510–1518.
Asanad S, Meer E, Tian JJ, Fantini M, Nassisi M, Sadun AA. Leber’s hereditary optic neuropathy: Severe vascular pathology in a severe primary mutation. Intractable Rare Dis Res. 2019;8:52–55.
Yu-Wai-Man P, Chinnery PF Leber Hereditary Optic Neuropathy. In Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Mirzaa G et al., editors. Seattle (WA): GeneReviews((R)); 2021.
Yu-Wai-Man P, Turnbull DM, Chinnery PF. Leber hereditary optic neuropathy. J Med Genet. 2002;39:162–9.
Yu-Wai-Man P, Griffiths PG, Chinnery PF. Mitochondrial optic neuropathies—disease mechanisms and therapeutic strategies. Prog Retin Eye Res. 2011;30:81–114.
Carelli V, Ross-Cisneros FN, Sadun AA. Mitochondrial dysfunction as a cause of optic neuropathies. Prog Retin Eye Res. 2004;23:53–89.
Carelli V, Ghelli A, Ratta M, Bacchilega E, Sangiorgi S, Mancini R, et al. Leber’s hereditary optic neuropathy: biochemical effect of 11778/ND4 and 3460/ND1 mutations and correlation with the mitochondrial genotype. Neurology. 1997;48:1623–32.
Lodi R, Taylor DJ, Tabrizi SJ, Kumar S, Sweeney M, Wood NW, et al. In vivo skeletal muscle mitochondrial function in Leber’s hereditary optic neuropathy assessed by 31P magnetic resonance spectroscopy. Ann Neurol. 1997;42:573–9.
Lodi R, Carelli V, Cortelli P, Iotti S, Valentino ML, Barboni P, et al. Phosphorus MR spectroscopy shows a tissue specific in vivo distribution of biochemical expression of the G3460A mutation in Leber’s hereditary optic neuropathy. J Neurol Neurosurg Psychiatry. 2002;72:805–7.
Yu-Wai-Man P. Therapeutic Approaches to Inherited Optic Neuropathies. Semin Neurol. 2015;35:578–86.
Yuan J, Zhang Y, Liu H, Wang D, Du Y, Tian Z, et al. Seven-Year Follow-up of Gene Therapy for Leber’s Hereditary Optic Neuropathy. Ophthalmology. 2020;127:1125–1127.
Acknowledgements
The authors acknowledge the defining contributions of John Guy, MD (deceased), who invented mito-targeting AAV, designed and performed the research. His tireless efforts made this study possible. We also thank Dr. Marco Ruggeri and the Ophthalmic Biophysics Center of the University of Miami for providing OCT analysis techniques; Dr. Alfred S. Lewin and Mr. Vince A. Chiodo at the University of Florida for the MTSAAV package. The authors declare no competing interests. This study is supported by the National Eye Institute R01 EY 027414, R01 EY017141, R24 EY028785, P30 EY014801 Bascom Palmer Eye Institute Core Grant, and institutional support from the Retina Research Foundation (GR015009). R.K. Lee was partially supported by the Walter G. Ross Foundation, the Camiener Foundation Glaucoma Research Fund, and the Guiterrez Family Research Fund.
Author information
Authors and Affiliations
Contributions
JG and HY designed research; YL, JDE, DEA, SV, JG, and HY performed research; WWH contributed to new reagents/analytic tools; YL, NS, VP, RKL, and HY analyzed data; and VP, RKL, and HY wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, Y., Eastwood, J.D., Alba, D.E. et al. Gene therapy restores mitochondrial function and protects retinal ganglion cells in optic neuropathy induced by a mito-targeted mutant ND1 gene. Gene Ther 29, 368–378 (2022). https://doi.org/10.1038/s41434-022-00333-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41434-022-00333-6
This article is cited by
-
Mitochondria and the eye—manifestations of mitochondrial diseases and their management
Eye (2023)
-
Developments in the Treatment of Leber Hereditary Optic Neuropathy
Current Neurology and Neuroscience Reports (2022)