Abstract
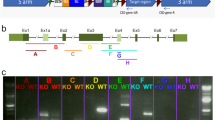
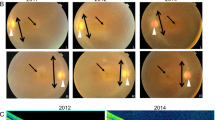
Bardet–Biedl syndrome (BBS) is a rare ciliopathy for which there are no current effective treatments. BBS is a genetically heterogeneous disease, though the M390R mutation in BBS1 is involved in ~25% of all genetic diagnoses of BBS. The principle features of BBS include retinal degeneration, obesity, male infertility, polydactyly, intellectual disability, and renal abnormalities. Patients with mutations in BBS genes often present with night blindness within the first decade of life, which progresses to complete blindness. This is due to progressive loss of photoreceptor cells. Male infertility is caused by a lack of spermatozoa flagella, rendering them immobile. In this study, we have crossed the wild-type human BBS1 gene, driven by the CAG promoter, onto the Bbs1M390R/M390R mouse model to determine if ectopic expression of BBS1 rescues male infertility and retinal degeneration. qRT-PCR indicates that the BBS1 transgene is expressed in multiple tissues throughout the mouse, with the highest expression seen in the testes, and much lower expression in the eye and hypothalamus. Immunohistochemistry of the transgene in the eye showed little if any expression in the photoreceptor outer nuclear layer. When male Bbs1M30R/M390R;BBS1TG+ mice are housed with WT females, they are able to sire offspring, indicating that the male infertility phenotype of BBS is rescued by the transgene. Using electroretinography (ERGs) to measure retinal function and optical coherence tomography to measure retinal thickness, we show that the transgene does not confer protection against retinal degeneration in Bbs1M300R/M390R;BBS1TG+ mice. The results of this study indicate that the male infertility aspect of BBS is an attractive target for gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sheffield V, Zhang Q, Heon E, Drack A, Stone E, Carmi R. The Bardet-Biedl syndrome. 3rd ed. Erickson R, Wynshaw-Boris A, editors. Oxford University Press; 2016.
Forsythe E, Beales P. Bardet-Biedl syndrome. Eur J Hum Genet. 2013;21:8–13.
Farag T, Teebi A. High incidence of Bardet Biedl syndrome among the Bedouin. Clin Genet. 1989;36:463–5.
Schaefer E, Delvalee C, Mary L, Stoetzel C, Geoffroy V, Marks-Delesalle C, et al. Identification and characterization of known biallelic mutations in the IFT27 (BBS19) gene in a novel family with Bardet-Biedl syndrome. Front Genet. 2019;10:1–6.
Davis R, Swiderski R, Rahmouni K, Nishimura D, Mullins R, Agassandian K, et al. A knockin mouse model of the Bardet-Biedl syndrome 1 M390R mutation has cilia defects, ventriculomegaly, retinopathy, and obesity. PNAS. 2007;4:19422–7.
Azari A, Aleman T, Cideciyan A, Schwartz S, Windsor E, Sumaroka A, et al. Retinal disease expression in Bardet-Biedl syndrome-1 (BBS1) is a spectrum from maculopathy to retina-wide degeneration. Investig Ophthalmol Vis Sci. 2006;47:5004–10.
Cox K, Kerr N, Kedrov M, Nishimura D, Jennings B, Stone E, et al. Phenotypic expression of Bardet-Biedl syndrome in patients homozygous for the common M390R mutation in the BBS1 gene. Vis Res. 2012;75:77–87.
Jin H, Nachury M. The BBSome. Curr Biol. 2009;19:482–73.
Seo S, Guo D, Bugge K, Morgan D, Rahmouni K, Sheffield V. Requirement of Bardet-Biedl syndrome proteins for leptin receptor signaling. Hum Mol Genet. 2009;18:1323–31.
Young R. The renewal of photoreceptor cell outer segments. J Cell Biol. 1967;33:61–72.
Nguyen J, Hicks D. Renewal of photoreceptor outer segments and their phagocytosis by the retinal pigment epithelium. Int Rev Cytol. 2000;196:245–313.
Tian G, Lodowski K, Lee R, Imanishi Y. Retrograde intraciliary trafficking of opsin during the maintenance of cone‐shaped photoreceptor outer segments of Xenopus laevis. J Comp Neurol. 2014;522:3577–89.
Datta P, Allamargot C, Hudson J, Anderson E, Bhattarai S, Drack A, et al. Accumulation of non-outer segment proteins in the outer segment underlies photoreceptor degeneration in Bardet-Biedl syndrome. PNAS. 2015;112:E4400–9.
Hsu Y, Garrison J, Kim G, Schmitz A, Searby C, Zhang Q, et al. BBSome function is required for both the morphogenesis and maintenance of the photoreceptor outer segment. PLOS Genet. 2017;13:e1007057.
Agarwal A, Mulgund A, Hamada A, Chyatte M. A unique view on male infertility around the globe. Reprod Biol Endocrinol. 2015;13:32–40.
Zhang Q, Nishimura D, Vogel T, Shao J, Swiderski R, Yin T, et al. BBS7 is required for BBSome formation and its absence in mice results in Bardet-Biedl syndrome phenotypes and selective abnormalities in membrane protein trafficking. J Cell Sci. 2013;126:2372–80.
Smalley E, First AAV. gene therapy poised for landmark approval. Nat Biotechnol. 2017;35:998–9.
Cai X, Conley S, Naash M. RPE65: role in the visual cycle, human retinal disease, and gene therapy. Ophthalmic Genet. 2009;30:57–62.
Simons D, Boye S, Hauswirth W, Wu S. Gene therapy prevents photoreceptor death and preserves retinal function in a Bardet-Biedl syndrome mouse model. PNAS. 2011;108:6276–81.
Seo S, Mullins R, Dumitrescu A, Bhattarai S, Gratie D, Wang K, et al. Subretinal gene therapy of mice with Bardet-Biedl syndrome type 1. Investig Ophthalmology Vis Sci. 2013;11:6118–32.
Zincarelli C, Soltys S, Rengo G, Rabinowitz J. Analysis of AAV serotypes 1-9 mediated gene expression and tropism in mice after systemic injection. Mol Ther. 2008;16:1073–80.
Mattapallil J, Wawrousek E, Chan C, Zhao H, Roychoudhury J, Ferguson T, et al. The Rd8 mutation of the Crb1 gene is present in vendor lines of C57BL/6N mice and embryonic stem cells, and confounds ocular induced mutant phenotypes. Immunol Microbiol. 2012;53:2921–7.
Schielzeth H, Dingemanse N, Nakagawa S, Westneat D, Allegue H, Teplitsky C, et al. Robustness of linear mixed‐effects models to violations of distributional assumptions. Methods Ecol Evol. 2020;11:1141–52.
Knief U, Forstmeier W. Violating the normality assumption may be the lesser of two evils. bioRxiv. 2018.
Du Plessis S, Cabler S, McAlister D, Sabanegh E, Agarwal A. The effect of obesity on sperm disorders and male infertility. Nat Rev Urol. 2010;7:153–61.
Jodar M, Soler-Ventura A, Oliva R. Semen proteomics and male infertility. J Proteom. 2017;162:125–34.
Pereira R, Barros RS, Sousa A. Major regulatory mechanisms involved in sperm motility. Asian J Androl. 2017;19:5–14.
Goyal R, Kotru M, Gogia A, Sharma S. Qualitative defects with normal sperm counts in a patient attending infertility clinic. Indian J Pathol Microbiol. 2018;61:233–5.
Levine H, Jorgensen N, Martino-Andrade A, Mendiola J, Weksler-Derri D, Mindlis I, et al. Temporal trends in sperm count: a systematic review and meta-regression analysis. Hum Reprod Updat. 2017;23:646–59.
Skakkebaek N. Sperm counts, testicular cancers, and the environment. BMJ. 2017;359:j4517.
Ikawa M, Tergaonkar V, Ogura A, Ogonuki N, Inoue K, Verma I. Restoration of spermatogenesis by lentiviral gene transfer: offspring from infertile mice. PNAS. 2002;99:7524–9.
Jain T, Grainger D, Ball G, Biggons W, Rebar R, Robins J, et al. 30 years of data: impact of the United States in vitro fertilization data registry on advancing fertility care. Fertil Steril. 2019;111:477–88.
Li X, Sun T, Wang X, Tang J, Liu Y. Restore natural fertility of Kitw/Kitwv mouse with nonobstructive azoospermia through gene editing on SSCs mediated by CRISPR-Cas9. Stem Cell Res Ther. 2019;10:271–9.
Dyer O. Scientists call for moratorium on editing heritable genes. BMJ. 2019;364:1256.
Xiong W, Wu D, Xue U, Wang S, Chung M, Ji X, et al. AAV cis-regulatory sequences are correlated with ocular toxicity. PNAS. 2019;116:5785–94.
Funding
This work was funded by the National Institutes of Health, Grant numbers R01 EY011298 (VCS) and R01 EY017168 (VCS), as well as the Roy J. Carver Charitable Trust (VCS). This work also benefitted from the University of Iowa Visual Science Core Facilities funded by NIH P30EY025580 (VCS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Cring, M.R., Meyer, K.J., Searby, C.C. et al. Ectopic expression of BBS1 rescues male infertility, but not retinal degeneration, in a BBS1 mouse model. Gene Ther 29, 227–235 (2022). https://doi.org/10.1038/s41434-021-00241-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41434-021-00241-1