Abstract
Aims
To present current expert practice patterns and to formulate a consensus for the management of HSV and VZV AU by uveitis specialists worldwide.
Methods
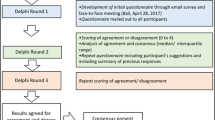
A two-round online modified Delphi survey with masking of the study team was conducted. Responses were collected from 76 international uveitis experts from 21 countries. Current practices in the diagnosis and treatment of HSV and VZV AU were identified. A working group (The Infectious Uveitis Treatment Algorithm Network [TITAN]) developed data into consensus guidelines. Consensus is defined as a particular response towards a specific question meeting ≥75% of agreement or IQR ≤ 1 when a Likert scale is used.
Results
Unilaterality, increased intraocular pressure (IOP), decreased corneal sensation and diffuse or sectoral iris atrophy are quite specific for HSV or VZV AU from consensus opinion. Sectoral iris atrophy is characteristic of HSV AU. Treatment initiation is highly variable, but most experts preferred valacyclovir owing to simpler dosing. Topical corticosteroids and beta-blockers should be used if necessary. Resolution of inflammation and normalisation of IOP are clinical endpoints.
Conclusions
Consensus was reached on several aspects of diagnosis, choice of initial treatment, and treatment endpoints for HSV and VZV AU. Treatment duration and management of recurrences varied between experts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.
References
Rathinam SR, Namperumalsamy P. Global variation and pattern changes in epidemiology of uveitis. Indian J Ophthalmol. 2007;55:173–83.
Siverio Júnior CD, Imai Y, Cunningham ETJ. Diagnosis and management of herpetic anterior uveitis. Int Ophthalmol Clin. 2002;42:43–8.
Zandi S, Bodaghi B, Garweg JG. Review for disease of the year: treatment of viral anterior uveitis: a perspective. Ocul Immunol Inflamm. 2018;26:1135–42.
Tsirouki T, Dastiridou A, Symeonidis C, Tounakaki O, Brazitikou I, Kalogeropoulos C, et al. A focus on the epidemiology of uveitis. Ocul Immunol Inflamm. 2018;26:2–16.
Chee S-P, Jap A. Viral anterior uveitis. In: Biswas J, Majumder PD, editors. Uveitis: an update. New Delhi: Springer; 2016.
Abdulaal MR, Abiad BH, Hamam RN. Uveitis in the aging eye: incidence, patterns, and differential diagnosis. J Ophthalmol. 2015;2015:509456.
Sakai J-I, Usui Y, Suzuki J, Kezuka T, Goto H. Clinical features of anterior uveitis caused by three different herpes viruses. Int Ophthalmol. 2019;39:2785–95.
Takase H, Kubono R, Terada Y, Imai A, Fukuda S, Tomita M, et al. Comparison of the ocular characteristics of anterior uveitis caused by herpes simplex virus, varicella-zoster virus, and cytomegalovirus. Jpn J Ophthalmol. 2014;58:473–82.
Chronopoulos A, Roquelaure D, Souteyrand G, Seebach JD, Schutz JS, Thumann G. Aqueous humor polymerase chain reaction in uveitis—utility and safety. BMC Ophthalmol. 2016;16:1–7.
Wensing B, Mochizuki M, De Boer JH. Clinical characteristics of herpes simplex virus associated anterior uveitis. Ocul Immunol Inflamm. 2018;26:333–37.
Neumann R, Barequet D, Rosenblatt A, Amer R, Ben-Arie-Weintrob Y, Hareuveni-Blum T, et al. Herpetic anterior uveitis—analysis of presumed and PCR proven cases. Ocul Immunol Inflamm. 2019;27:211–18.
Qian Z, Fan H, Tao Y, Li W, Gu W. Herpetic anterior uveitis in a Chinese referral center: clinical manifestations and laboratory test results. Ocul Immunol Inflamm. 2020;28:758–63.
Testi I, Aggarwal K, Jaiswal N, Dahiya N, Thng ZX, Agarwal A, et al. Antiviral therapy for Varicella Zoster Virus (VZV) and Herpes Simplex Virus (HSV)-induced anterior uveitis: a systematic review and meta-analysis. Front Med. 2021;8:686427.
Kalezic T, Mazen M, Kuklinski E, Asbell P. Herpetic eye disease study: lessons learned. Curr Opin Ophthalmol. 2018;29:340–46.
Anon. A controlled trial of oral acyclovir for iridocyclitis caused by herpes simplex virus. The Herpetic Eye Disease Study Group. Arch Ophthalmol. 1996;114:1065–72.
Centre for Evidence-Based Medicine. Oxford Centre for Evidence-Based Medicine—levels of evidence. 2009. http://www.cebm.net/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/.
Niederberger M, Köberich S. Coming to consensus: the Delphi technique. Eur J Cardiovasc Nurs. 2021;20:692–95.
Nasa P, Jain R, Juneja D. Delphi methodology in healthcare research: how to decide its appropriateness. World J Methodol. 2021;11:116–29.
Radosavljevic A, Agarwal M, Chee SP, Zierhut M. Epidemiology of viral induced anterior uveitis. Ocul Immunol Inflamm. 2022;30:297–309.
Keorochana N, Treesit I, Funarunart P. Characteristics and clinical outcomes of hypertensive anterior uveitis. Ocul Immunol Inflamm. 2020;28:538–48.
Accorinti M, Petitti L, Gaeta A, Giannini D, De Geronimo D. Viral acute anterior uveitis: clinical signs useful for differential diagnosis. Ocul Immunol Inflamm. 2020;29:1355–62.
Groen-Hakan F, Babu K, Tugal-Tutkun I, Pathanapithoon K, de Boer JH, Smith JR, et al. Challenges of diagnosing viral anterior uveitis. Ocul Immunol Inflamm. 2017;25:710–20.
Choi W, Kang HG, Choi EY, Kim SS, Kim CY, Koh HJ, et al. Clinical utility of aqueous humor polymerase chain reaction and serologic testing for suspected infectious uveitis: a single-center retrospective study in South Korea. BMC Ophthalmol. 2020;20:242.
De Groot-Mijnes JDF, Chan ASY, Chee SP, Verjans GMGM. Immunopathology of virus-induced anterior uveitis. Ocul Immunol Inflamm. 2018;26:338–46.
Pleyer U, Chee S-P. Current aspects on the management of viral uveitis in immunocompetent individuals. Clin Ophthalmol. 2015;9:1017–28.
Yoo W-S, Kim G-N, Chung I, Cho M-C, Han YS, Kang SS, et al. Clinical characteristics and prognostic factors in hypertensive anterior uveitis diagnosed with polymerase chain reaction. Sci Rep. 2021;11:8814.
Kongyai N, Sirirungsi W, Pathanapitoon K, Tananuvat N, Kunavisarut P, Leechanachai P, et al. Viral causes of unexplained anterior uveitis in Thailand. Eye. 2012;26:529–34.
van Velzen M, Missotten T, van Loenen FB, Meesters RJW, Luider TM, Baarsma GS, et al. Acyclovir-resistant herpes simplex virus type 1 in intra-ocular fluid samples of herpetic uveitis patients. J Clin Virol. 2013;57:215–21.
Neoh C, Harding SP, Saunders D, Wallis S, Tullo AB, Nylander A, et al. Comparison of topical and oral acyclovir in early herpes zoster ophthalmicus. Eye. 1994;8:688–91.
Beutner KR. Valacyclovir: a review of its antiviral activity, pharmacokinetic properties, and clinical efficacy. Antivir Res. 1995;28:281–90.
Miserocchi E, Modorati G, Galli L, Rama P. Efficacy of valacyclovir vs acyclovir for the prevention of recurrent herpes simplex virus eye disease: a pilot study. Am J Ophthalmol. 2007;144:547–51.
Schuster AK, Harder BC, Schlichtenbrede FC, Jarczok MN, Tesarz J. Valacyclovir versus acyclovir for the treatment of herpes zoster ophthalmicus in immunocompetent patients. Cochrane Database Syst Rev. 2016;11:CD11503.
Gershon AA, Breuer J, Cohen JI, Cohrs RJ, Gershon MD, Gilden D, et al. Varicella zoster virus infection. Nat Rev Dis Prim. 2015;1:15016.
Sung VCT, Barton K. Management of inflammatory glaucomas. Curr Opin Ophthalmol. 2004;15:136–40.
Chang JH, McCluskey P, Missotten T, Ferrante P, Jalaludin B, Lightman S. Use of ocular hypotensive prostaglandin analogues in patients with uveitis: does their use increase anterior uveitis and cystoid macular oedema? Br J Ophthalmol. 2008;92:916–21.
Markomichelakis NN, Kostakou A, Halkiadakis I, Chalkidou S, Papakonstantinou D, Georgopoulos G. Efficacy and safety of latanoprost in eyes with uveitic glaucoma. Graefes Arch Clin Exp Ophthalmol. 2009;247:775–80.
Chan NS-W, Ti S-E, Chee S-P. Decision-making and management of uveitic cataract. Indian J Ophthalmol. 2017;65:1329–39.
Funding
HNS’s work is supported by the NIH Intramural Research Program (IRP). The funding organisation had no role in the design or conduct of this research. RA has received NMRC Clinician Scientist Award from National Medical Research Council, Singapore, but the funding organisation has no role in the design or conduct of this research.
Author information
Authors and Affiliations
Consortia
Contributions
All authors contributed to the intellectual development of this paper. RA and VG conceived and planned the study. ZXT and IP wrote the first draft of the paper. ZXT, IP, IT, and KC performed the literature review. ZXT, IP, IT, KC, MW, SPC, ADD, JHK, BB, JET, TBA, MDdS, JRS, PM, RLDN, DAJ, JHdB, HNS, DAG, MK, JLD, JTR, NPJ, QDN, CP, RA, and VG contributed to interpreting the results and provided critical feedback to the paper. The final version of the paper has been seen and approved by all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41433_2023_2630_MOESM1_ESM.pdf
Currently available evidence for Herpes Simplex Virus and Varicella-Zoster Virus anterior uveitis management- Studies graded by level of evidence
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thng, Z.X., Putera, I., Testi, I. et al. The Infectious Uveitis Treatment Algorithm Network (TITAN) Report 1—global current practice patterns for the management of Herpes Simplex Virus and Varicella Zoster Virus anterior uveitis. Eye 38, 61–67 (2024). https://doi.org/10.1038/s41433-023-02630-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-023-02630-9