Abstract
Objective
To examine the time to onset of disease in the fellow eye of patients with unilateral DMO in routine clinical practice and to identify risk factors for development of bilateral DMO.
Design
Retrospective cohort study.
Participants
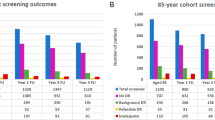
One hundred forty treatment-naive patients 18 years or older with unilateral DMO presenting to Cole Eye Institute between January 2012 and July 2021.
Methods
Records of patients with unilateral DMO were reviewed for development of DMO in the fellow eye. Demographic, diabetic, ocular, and systemic characteristics were collected at initial DMO diagnosis date. Bivariate and multivariate analyses were performed and significant factors were modelled using Kaplan-Meier curves.
Results
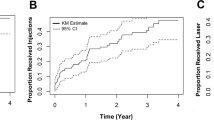
Fifty patients with conversion to bilateral DMO and 90 patients without conversion were identified. Average time to bilateral DMO was 15.0 ± 15.7 months. 64% of patients converted within 1 year and 90% converted within 3 years. HbA1c (p = 0.003), diabetic retinopathy duration (p = 0.029), and diabetic foot disease (DFD) (p = 0.002) were identified as significant risk factors for conversion. Patients with better visual acuity at time of initial diagnosis and history of panretinal photocoagulation (PRP) (p = 0.044) or focal laser (p = 0.035) in the primary eye were also more likely to convert.
Conclusions
Participants were most likely to develop fellow eye DMO within the first year after initial DMO diagnosis. In routine clinical practice, poor glycaemic control and DFD were risk factors associated with bilateral eye involvement. Clinicians may consider screening the fellow eye of high-risk individuals at each appointment within the first year of diagnosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Cheung N, Mitchell P, Wong TY. Diabetic retinopathy. Lancet. 2010;376:124–36. https://doi.org/10.1016/s0140-6736(09)62124-3.
Browning DJ, Stewart MW, Lee C. Diabetic macular oedema: evidence-based management. Indian J Ophthalmol. 2018;66:1736–50. https://doi.org/10.4103/ijo.IJO_1240_18.
Yau JWY, Rogers SL, Kawasaki R, Lamoureux EL, Kowalski JW, Bek T, et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35:556–64. https://doi.org/10.2337/dc11-1909.
Dhoot DS. Incidence of new diabetic macular oedema in fellow eyes of patients in the VISTA and VIVID studies. investigative. 2020;61:278.
Fraser C, Amico D. Diabetic retinopathy: prevention and treatment. UpToDate. Wolters Klumer; Waltham, MA: UpToDate Inc. 2018.
Miljanovic B, Glynn RJ, Nathan DM, Manson JE, Schaumberg DA. A prospective study of serum lipids and risk of diabetic macular oedema in type 1. Diabetes Diabetes. 2004;53:2883–92. https://doi.org/10.2337/diabetes.53.11.2883.
Man REK, Fenwick EK, Sabanayagam C, Li L-J, Tey CS, Soon HJT, et al. Differential impact of unilateral and bilateral classifications of diabetic retinopathy and diabetic macular oedema on vision-related quality of life. Investig Ophthalmol Vis Sci. 2016;57:4655–60. https://doi.org/10.1167/iovs.16-20165.
Di Y, Li Z, Ye J, Li L, Li B, Yu R. The fellow eye effect of unilateral intravitreal conbercept injections in eyes with diabetic macular oedema. Acta Diabetol. 2020;57:1001–7. https://doi.org/10.1007/s00592-020-01511-x.
Chalam KV, Bressler SB, Edwards AR, Berger BB, Bressler NM, Glassman AR, et al. Retinal thickness in people with diabetes and minimal or no diabetic retinopathy: Heidelberg Spectralis optical coherence tomography. Invest Ophthalmol Vis Sci. 2012;53:8154–61. https://doi.org/10.1167/iovs.12-10290.
Cheung N, Cheung CMG, Talks SJ, Wong TY. Management of diabetic macular ooedema: new insights and global implications of DRCR protocol V. Eye. 2020;34:999–1002. https://doi.org/10.1038/s41433-019-0738-y.
Hereen T. Eye. 2021. https://cran.r-project.org/web/packages/eye/vignettes/eye.html.
Lois N, Cook JA, Wang A, Aldington S, Mistry H, Maredza M, et al. Evaluation of a new model of care for people with complications of diabetic retinopathy: the EMERALD study. Ophthalmology. 2021;128:561–73. https://doi.org/10.1016/j.ophtha.2020.10.030.
Agarwal D, Udeh B, Campbell J, Bena J, Rachitskaya A. Follow-up appointment delay in diabetic macular oedema patients. Ophthalmic Surg Lasers Imaging Retina. 2021;52:200–6. https://doi.org/10.3928/23258160-20210330-04.
Heier JS, Korobelnik J-F, Brown DM, Schmidt-Erfurth U, Do DV, Midena E, et al. Intravitreal aflibercept for diabetic macular oedema: 148-week results from the VISTA and VIVID studies. Ophthalmology. 2016;123:2376–85. https://doi.org/10.1016/j.ophtha.2016.07.032.
Bressler NM, Odia I, Maguire M, Glassman AR, Jampol LM, MacCumber MW, et al. Association between change in visual acuity and change in central subfield thickness during treatment of diabetic macular oedema in participants randomized to aflibercept, bevacizumab, or ranibizumab: A post hoc analysis of the protocol T randomized clinical trial: A post hoc analysis of the protocol T randomized clinical trial. JAMA Ophthalmol. 2019;137:977–85. https://doi.org/10.1001/jamaophthalmol.2019.1963.
Chou T-H, Wu P-C, Kuo JZ-C, Lai C-H, Kuo C-N. Relationship of diabetic macular ooedema with glycosylated haemoglobin. Eye. 2009;23:1360–3. https://doi.org/10.1038/eye.2008.279.
Muste JC, Valentim CCS, Iyer AI, Mammo DA, Alsaloum P, Xu CM, et al. Progression to vision-threatening retinopathy complications following panretinal laser for proliferative diabetic retinopathy. Ophthalmic Surg Lasers Imaging Retina. 2022;53:186–93. https://doi.org/10.3928/23258160-20220316-03.
Zhang X, Zeng H, Bao S, Wang N, Gillies MC. Diabetic macular oedema: new concepts in patho-physiology and treatment. Cell Biosci. 2014;4:27 https://doi.org/10.1186/2045-3701-4-27.
Kelkar A, Webers C, Shetty R, Kelkar J, Labhsetwar N, Pandit A, et al. Factors affecting compliance to intravitreal anti-vascular endothelial growth factor therapy in Indian patients with retinal vein occlusion, age-related macular degeneration, and diabetic macular oedema. Indian J Ophthalmol. 2020;68:2143–7. https://doi.org/10.4103/ijo.IJO_1866_19.
Figueira J, Henriques J, Carneiro Â, Marques-Neves C, Flores R, Castro-Sousa JP, et al. Guidelines for the management of center-involving diabetic macular oedema: treatment options and patient monitorization. Clin Ophthalmol. 2021;15:3221–30. https://doi.org/10.2147/OPTH.S318026.
Author information
Authors and Affiliations
Contributions
PA was responsible for protocol design, data collection and analysis, and manuscript writing. MWR was responsible for data collection and analysis. VB, BLK, AKW, BJL, CCSV, CMX, and JCM were responsible for data collection. MK was responsible for manuscript writing and editing. RPS was responsible for designing the project and for providing feedback on the study.
Corresponding author
Ethics declarations
Competing interests
RPS reports personal fees from Genentech/Roche, Alcon/Novartis, Zeiss, Bausch+Lomb, Regeneron Pharmaceuticals, Inc, Gyroscope and Asceplix and grants from Apellis and Graybug. Other authors have no financial disclosures to report.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alsaloum, P., Russell, M.W., Blaga, V. et al. Time to fellow eye involvement in patients with unilateral diabetic macular oedema. Eye 37, 2761–2767 (2023). https://doi.org/10.1038/s41433-023-02410-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-023-02410-5