Abstract
Objectives
To conduct a systematic review and meta-analysis on data related to macular pigment optical density (MPOD) and visual function in adults with healthy eyes.
Methods
MEDLINE®, Cochrane, and Commonwealth of Agriculture Bureau abstracts databases were searched for English-language publications between 1946 and August 2018. Included studies examined correlation of MPOD and visual function in adults with healthy eyes at all timepoints and all designs, except for case–control, case reports, and reviews. Visual function outcomes of interest included photostress recovery, contrast sensitivity, visual acuity, glare sensitivity/disability, and dark adaptation. Random effects model meta-analyses combined study-level correlation (r).
Results
Twenty-two publications were included. In meta-analysis MPOD was found to be significantly correlated with contrast sensitivity at 30′ (two studies, summary r: 0.37; 95% CI 0.15, 0.56), and at 1° eccentricity with a spatial frequency of 7, 11, and 21 cpd (three studies, summary r: 0.31; 95% CI 0.06, 0.52), with photostress recovery at a 1° eccentricity with a moderate background, 10 cpd, and 16% contrast (two studies, summary r: −0.17; 95% CI −0.31, −0.02), and at 30′ (four studies, summary r: −0.57; 95% CI −0.78, −0.24), and with glare disability at 30′ eccentricity with a log scale at 460 nm (three studies, summary r = 0.47; 95% CI 0.32; 0.59). There were insufficient data for meta-analysis for other visual functions.
Conclusions
Our review identifies a link between MPOD and visual function with significant correlations with photostress recovery, glare disability, and contrast sensitivity.
Similar content being viewed by others
Introduction
Extensive epidemiological observation indicates that fruits and vegetables rich in carotenoids provide a variety of health benefits, to include ocular health [1,2,3]. The relationshibpe between the ditary carotenoids, lutein and zeaxanthin, and visular function is particular interest because of the more than 600 carotenoids found in nature they are selectively taken up ocular tissue to include the macula where they are referred to as macular pigment [4, 5]. As macular pigment, lutein and zeaxanthin are thought to be protective as blue light filters, antioxidants, and anti-inflammatory agents [6]. These may also have structural roles to modulate the function of synaptic membranes and enhance gap junction communication [7,8,9]. There is a variety of evidence indicating that these bioactives may have a beneficial role in visual function through macular pigment [6]. Visual function has been shown to improve as a function of macular pigment optical density (MPOD) [10, 11]. For example, the increased visual processing that was associated with macular carotenoid supplementation [11, 12]. Higher MPOD was found to be inversely related to visual discomfort from bright light conditions, e.g., glare [13,14,15], with individuals able to tolerate more light averting their gaze due to discomfort. In addition, glare disability (whereby glare results in “washed out” vision due to light scattering in the eye) is significantly, inversely related to MPOD [13, 14, 16].
Given this, lutein and zeaxanthin have been proposed to be considered for a recommended intake for optimal ocular health and prevention of eye disease [17]. Recommended dietary intakes are made based on the current scientific knowledge on nutritional needs of healthy populations. Lutein and zeaxanthin are not essential nutrients; however, evidence is accumulating to suggest that they are important to optimize ocular health and function. Of note, the precise mechanisms by which they may influence ocular health are open to further investigations. As a first step toward developing lutein/zeaxanthin dietary guidelines, this study will examine the evidence linking MPOD with visual function in adult populations with healthy eyes. This would be an important initial step toward establishing a recommended intake. Therefore, this study aims to evaluate the association between MPOD and visual function outcomes among populations relevant to the general adult population.
Methods
This systematic review examines the correlation between MPOD and visual function. We followed both a prespecified standard protocol published in PROSPERO (www.crd.york.ac.uk/PROSPERO) and the standards for conduct and reporting for systematic reviews as detailed in the preferred reporting items for systematic reviews and meta-analyses guidance [18].
Search strategy
We conducted a literature search in MEDLINE®, Cochrane, and Commonwealth of Agriculture Bureau abstracts databases for studies reporting outcomes of MPOD and visual function in healthy human eyes published between 1946 and August 2018. The search strategy combined terms of macular pigment and visual function (Supplementary Table 1). We did not limit our search by type of study design, or visual function. We supplemented the search with articles identified during the evidence map.
Study selection
Citations that resulted from the literature search were independently screened in duplicate in Abstrackr [19] using the prespecified PICO study eligibility criteria, and any discrepancies were resolved during team conferences. The full-text articles corresponding to the accepted abstracts were retrieved and screened for eligibility.
Study inclusion criteria
We included studies of all study designs (including randomized trials, cohort studies, and cross-sectional studies), except case–control, case reports, and reviews, and of any study duration that reported an association between MPOD and visual functions in adults (≥18 years old) with healthy eyes (i.e., without eye diseases). If a study included a mixed population of eye disease and healthy eyes, data on the association between MPOD and visual functions among the healthy population were included when available. We included studies conducted in any country and setting that used any MPOD method of measurement (i.e., heterochromic flicker photometry) and which reported any level of macular pigment eccentricity. Visual function outcomes of interest included photostress recovery, contrast sensitivity (CS), visual acuity, glare sensitivity/disability, dark adaptation, as well as other measures described in individual studies.
Study exclusion criteria
We excluded studies with less than ten subjects and non-English-language publications. We also excluded studies that did not report a correlation for MPOD and visual function. Studies on subjects with diagnosed eye disease (including but not limited to macular degeneration, glaucoma, cataract, and retinopathy) at baseline, diabetes, and cognitive impairment were excluded, unless they provide subgroup data for a healthy population.
Data extraction and quality assessment
Data from each study were extracted independently in a standardized, pre-piloted form by one of the four investigators and confirmed by at least one other. The form was designed to capture all relevant elements including study characteristics; method of assessing MPOD; participant characteristics; eye tested (left, right, or dominant); degrees of retinal eccentricity at which MPOD was measured; visual function; type of correlation; association between MPOD and outcome; and potential confounding variables adjusted for. We captured all reported study-level correlation (r) or any other association metric reported between MPOD and visual function outcomes for all reported timepoints and degrees of eccentricity.
Two reviewers independently assessed the risk of biases for each included study. The risk of bias assessment followed the Agency for Healthcare Research and Quality Methods Guide for Effectiveness and Comparative Effectiveness Review and assessed the following domains: risk of selection, performance, attrition, detection, and selective outcome reporting biases. Each methodological quality item was assessed by outcome and is reported as Yes, No, or Unclear.
Data synthesis
We consulted with technical experts on which visual function we could combine, and we grouped studies with the same degrees of eccentricity assessing similar outcomes. When intervention trials reported the correlation of MPOD and visual function both at baseline and post intervention, we choose baseline correlation as the primary correlation.
We performed random effects correlation meta-analysis when at least two sufficiently homogeneous studies reported similar outcomes at the same degrees of eccentricity. When sufficient studies were available subgroups analysis (age, gender, weight status, family history of eye disease) was considered. We tested between-study statistical heterogeneity using the Q statistic and quantified its extent with I2. We used the I2 threshold values of 25, 50, and 75% to grade statistical heterogeneity as low, moderate, and high, respectively. When at least two studies reporting the same outcomes and eccentricities were not available, we synthesized the results qualitatively.
Results
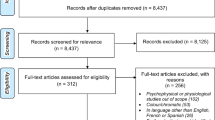
Twenty-two publications met the inclusion criteria (Table 1, Fig. 1, Supplementary Table 2) [11, 12, 14, 16, 20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37]. Fifteen studies were conducted in the USA, three in Ireland, one in England, one in Spain, one in Romania, and one did not clearly specify the country. Most studies reported including both men and women, and total enrolled participants ranged from 26 to 150. Four studies included a mix of Caucasian (75–83.3%), Black (7.7–17.5%), Hispanic (5.5–7.7%), and Asian (0–3.9%), one study (Kvansakul et al. [26]) included only Caucasians, and the rest did not report data on the race of the participants. No studies reported separate analysis by race. All but one of the studies, which used autofluorescence, used heterochromatic flicker photometry (HFP) to measure MPOD.
Abstracts identified (n = 4147); abstracts not meeting criteria (n = 4091); full-text articles retrieved (n = 57); full-text articles excluded after screening (n = 35); full-text articles meeting study eligibility criteria (n = 22); eligible cross-sectional studies (n = 11), randomized controlled trials (n = 9); and prospective observational studies (n = 2).
Contrast sensitivity
Four studies reported on an association between CS and MPOD [25, 27, 32, 36]. Two studies with a total of 209 participants measured MPOD at 30′ eccentricity [25, 36] and three studies with a total of 315 participants measured MPOD at 1° eccentricity [25, 27, 32].
Meta-analysis of two studies with a total of 209 participants reported a significant positive association between CS and MPOD measured at 30′ with moderate heterogeneity (summary r: 0.37; 95% CI 0.15, 0.56; I2 = 61.3%) [25, 36]. Meta-analysis of three studies [25, 27, 32] with a total of 315 participants did not find a correlation between CS and MPOD measured at 1° eccentricity and a spatial frequency of 4 cycles per degree (cpd) (summary r: 0.28; 95% CI −0.05, 0.56; I2 = 86%), but found a significant positive correlation with high heterogeneity between CS and MPOD measured at 1° eccentricity and a spatial frequency of 7 cpd (summary r: 0.31; 95% CI 0.06, 0.52; I2 = 86%), 11 cpd (summary r: 0.32; 95% CI 0.09, 0.52; I2 = 73%), and 21 cpd (summary r: 0.30; 95% CI 0.03, 0.54; I2 = 80%) (Table 2).
Another study found no association between CS at 0.25°and baseline MPOD, when it compared participants in the highest and the lowest tertile [28].
Photostress recovery
Seven studies reported on an association between photostress recovery and MPOD [11, 16, 23, 25, 33,34,35]. Two studies with a total of 176 participants measured MPOD at 1° eccentricity [25, 34]. One of the two studies reported results for various backgrounds, cpd, and % contrast [25]. A significant random effects correlation was only found in meta-analysis for a moderate background, 10 cpd, and 16% contrast (summary change: −0.17; 95% CI −0.31, −0.02; I2 = 0.0%) without heterogeneity (Table 3).
Meta-analysis of four studies with a total of 285 participants that reported on an association between photostress recovery and MPOD measured at 30′ eccentricity found a significant negative correlation with high heterogeneity (summary change: −0.57; 95% CI −0.78, −0.24; I2 = 89.6%) [16, 25, 33, 35]. Another study that reported only linear results found a significant correlation between MPOD at 30′ eccentricity and photostress recovery over the study period [23].
One study that measured MPOD averaged over a spatial distribution (10°, 20°, 30°, 2.75°, 2.75°) found a significant negative association between MPOD and photostress recovery (p < 0.0001) [11]. A second study measured the association of photostress recovery and MPOD at 15’ and 2° eccentricity but found no significant correlation for either measures [25]. A third study that measured MPOD at 0.25°, 0.5°, 1.0°, 1.75°, and 3° eccentricity also did not find a significant association at any of the eccentricities [27].
Glare disability
Five studies examined the correlation between glare disability and MPOD at 30′ eccentricity [16, 23, 25, 33, 35], two studies examined glare disability with MPOD at 2° eccentricity [25, 31], and two studies examined glare disability with MPOD at 1° eccentricity [25, 34]. Of five studies examining glare disability at 30′ eccentricity, two studies reported in log scale of glare disability using two different types of light. The summary meta-analysis of these two studies found no significant correlation with high heterogeneity (summary r = 0.54; 95% CI −0.13; 0.88; I2 = 94.2%). The summary meta-analysis of three studies examining log scale of glare disability at 460 nm showed a significant correlation between MPOD and glare disability with moderate heterogeneity (summary r = 0.47; 95% CI 0.32; 0.59; I2 = 69.0%).
Two studies examined glare disability with MPOD at 2° eccentricity [25, 31]. Of these, one study used three different spatial frequencies (3, 6, 9 cpd) for glare disability. The meta-analysis of these two studies found no significant correlation between different spatial frequency of glare disability and MPOD at 2° eccentricity with high heterogeneity (Table 4). Two studies examined glare disability with MPOD at 1° eccentricity [25, 34]. These studies evaluated different spatial frequency for glare disability. The meta-analysis of these two studies found no significant correlation between different spatial frequency of glare disability and MPOD at 1° eccentricity (data not shown).
One other study that measured glare disability at 0.5° eccentricity did not find a significant correlation between MPOD and glare disability [37].
Best corrected visual acuity (BCVA)
One cross-sectional study examined the relationship between MPOD and BCVA in adults (n = 142) [27]. The spatial profile of MPOD was measured—at 0.25°, 0.5°, 1.0°, 1.75°, and 3° retinal eccentricity. Using Pearson partial correlations to adjust for confounding variables, these researchers found that BCVA was positively associated with MPOD at each eccentricity (p ≤ 0.01 for all). The strength of these associations ranged from r = 0.237 to 0.308. The Pearson r value between BCVA and MPOD at 0.25° eccentricity was 0.268 (p = 0.01). Another study, which tested the effect of MPOD augmentation on visual function in healthy adults (n = 120), reported a qualitative positive association between BCVA and MPOD at baseline (V1) [28]. At V1, adults in the highest tertile for MPOD (0.25° retinal eccentricity) had a slightly higher BCVA (113 ± 3) than those in the lowest MPOD tertile (BCVA = 112 ± 3) (p = 0.045).
Contrast threshold (CT)
One cross-sectional study examined the relationship between MPOD and CT, accounting for fruit and vegetable intake, and serum lutein and zeaxanthin status, in younger (20–35 years old; n = 54) and older adults (45–65 years old; n = 54) [22]. MPOD was measured at the 1.0° central target. CT was determined with six different sizes of annular stimuli at 6.3°, 4.0°, 2.5°, 1.6°, 1.0°, and 0.7° visual angles, with and without glare light conditions. CS is the inverse of CT. Spearman’s rho correlation coefficients were used to adjust for fruit plus vegetable intake, serum lutein, and serum lutein and zeaxanthin/cholesterol and triglycerides. In younger adults, CT and MPOD were not associated at any condition. In older adults, CT and MPOD were inversely associated for all visual angles with or without glare (p < 0.03 for all). Without glare, the strength of the relationship ranged from −0.333 (p < 0.001) at 1.6° visual angle to −0.219 (p = 0.023) at 4.0° visual angle. With glare, the strength of the relationship ranged from −0.287 (p = 0.003) at 2.5° visual angle to −0.216 (p = 0.025) at 4.0° visual angle. When results from younger and older adults were combined into one analysis (n = 108), CT and MPOD were significantly inversely associated for all visual angles in conditions with or without glare (p < 0.03 for all). Without glare, Spearman r values were −0.152 (p = 0.026), −0.160 (p = 0.018), −0.238 (p < 0.001), −0.213 (p = 0.002), −0.245 (p < 0.001), and −0.213 (p = 0.007) at 6.3°, 4.0°, 2.5°, 1.6°, 1.0°, and 0.7° visual angles, respectively. With glare, Spearman r values were −0.182 (p = 0.012), −0.170 (p = 0.008), −0.181 (p = 0.009), −0.177 (p = 0.002), −0.205 (p = 0.009), and −0.176 (p = 0.012) at 6.3°, 4.0°, 2.5°, 1.6°, 1.0°, and 0.7° visual angles, respectively.
Another study [26] measured MPOD at 2.5° eccentricity and reported on the association with contrast acuity threshold. It did not find a significant correlation between MPOD and contrast acuity threshold.
Critical flicker fusion (CFF)
Two studies examined the relationship between MPOD and CFF threshold [11, 12]. In the first study at baseline, among 92 young adults ages (18–32 years), CFF threshold was significantly correlated with MPOD (r = 0.34, p < 0.01), but no correlation between MPOD and CFF was assessed at follow-up [12]. In the second study, at baseline, among 48 healthy young adults, MPOD was significantly correlated with CFF (r = 0.31; p = 0.035), but no correlation between MPOD and CFF was assessed at follow-up [11].
Dark adaptation
Two cross-sectional studies and one longitudinal study examined the relationship between MPOD and dark adaptation measured as elapsed time required detecting mesopic targets of fixed intensity [14, 30]. In the first study, among 27 healthy subjects with a mean age of 30 years, MPOD and elapsed time showed significant correlations for all three cd/m2 luminance conditions, namely 1 cd/m2 (r = −0.39, p = 0.04); 0.1 cd/m2 (r =̶ −0.68, p = 0.0001); and 0.05 cd/m2 (r = −0.76, p < 0.0001) [14].
In the second study, among 33 subjects (aged 15–68) there was no correlation between different measures of dark adaptation, including cone time constant in minutes (r = −0.02, p = 0.91); cone threshold as log cd/m2 (r = −0.01, p = 0.96); rod–cone break in minutes (r = 0.13, p = 0.45); slope of the second rod component (r = 0.32, p = 0.07); rod–rod break in minutes (r = 0.11, p = 0.52); slope of the third rod component (r = 0.31, p = 0.08); and the last threshold 30 min after the onset of bleach T30 (r = −0.24, p = 0.18) [30].
In the longitudinal study [28], among 121 subjects, those in the highest tertile of MPOD were significantly more capable of adjusting to sudden changes in light/dark adaptation.
Hyperacuity
One cross-sectional study examined the relationship between MPOD at 30′ eccentricity and hyperacuity in young adults (n = 40) [21]. Hyperacuity was measured under two light conditions: (1) white (460 nm), which is filtered by MPOD, and (2) yellow (570 nm), which is not filtered by MPOD. Hyperacuity was not significantly related to MPOD in either white (r = −0.226, p = 0.16) or yellow conditions (r = 0.147, p = 0.36).
Parafoveal scotopic sensitivity
One cross-sectional study examined the relationship between MPOD and parafoveal scotopic sensitivity in young (n = 10) and older (n = 27) adults. MPOD was measured at a centrally fixated stimulus 1° in diameter [24]. Parafoveal scotopic sensitivity of the older subjects was positively related to MPOD (r = 0.40, p < 0.02) but not in young adults (r = 0.20).
Photopic increment sensitivity
One cross-sectional study examined the relationship between MPOD (1°) and photopic sensitivity in young (24–36 years, n = 10) and older (60–84 years, n = 27) adults [24]. Photopic sensitivity measured at 440 and 550 nm was not related to MPOD in young adults (r = 0.11 and 0.18, respectively) but was significantly and positively related to MPOD in older adults with correlation coefficients of 0.53 and 0.49 at 440 and 550 nm, respectively, (p < 0.001).
Resolution acuity
One cross-sectional study examined the relationship between MPOD and resolution acuity in young adults (18–30 years, n = 40). MPOD was measured at 30′ eccentricity [21]. Resolution acuity was determined under two light conditions: (1) white (460 nm), which is filtered by MPOD, and (2) yellow (570 nm), which is not filtered by MPOD. Resolution acuity was not significantly related to MPOD in either white (r = 0.003, p = 0.98) or yellow conditions (r = 0.03, p = 0.85).
Spatial resolution
One cross-sectional study examined the relationship between MPOD and spatial resolution (ability to differentiate spatial boundaries) over a range of spatial frequencies (5.5, 7.33, 9.17, 11, and 18.33 cpd) in adults (n = 25) [14]. As a function of increasing MPOD, spatial resolution increased. The strength of association (Pearson’s r value) between MPOD and the ability to detect the spatial resolution ranged from −0.24 (for 5.5 cpd) to −0.77 (for 18.33 cpd). The detection threshold data for 9.17, 11, and 18.33 cpd, which were strongly associated with MPOD (r = −0.49, −0.55, −0.77, respectively, p ≤ 0.013 for all). For the two lowest spatial frequency values tested, 5.5 and 7.33 cpd, there was no statistically significant association with MPOD (r = −0.24 and −0.37, respectively).
Temporal CS
A randomized, placebo-controlled study, which tested the effect of lutein and zeaxanthin supplementation on visual processing speed in young healthy adults (18–32 years, n = 102), performed correlational analysis between MPOD and temporal CS at baseline [12]. MPOD was measured at 30′ retinal eccentricity. Both foveal and parafoveal temporal CS were measured at 0.4, 1.0, and 1.4 log frequency. Using Pearson product moment correlations (one-tailed criteria), researchers found that MPOD was positively associated with temporal CS. For foveal temporal CS, r values were 0.29, 0.27, and 0.26 at 1.4, 1.0, and 0.4 log hertz, respectively, (p < 0.005 for all). For parafoveal temporal CS, r values were 0.21 (p < 0.025) at 1.4 log hertz, and 0.26 (p < 0.005) at both 1.0 and 0.4 log hertz.
Discussion
This systematic review found a total of 22 studies that met the inclusion criteria. Meta-analysis was conducted for CS, glare disability, and photostress recovery. Meta-analysis found a significant positive association between CS and MPOD measured at 30′ and measured at 1° eccentricity with a spatial frequency of 7, 11, or 21 cpd. Meta-analysis of studies that reported on an association between photostress recovery and MPOD measured at 30′ eccentricity found a significant negative correlation. Meta-analysis examining the correlation between glare disability and MPOD at 30′ eccentricity found no significance, while meta-analysis of studies examining log scale of glare disability at 460 nm showed a significant correlation between MPOD and glare disability.
In this evaluation, we report statistically significant relationships between MPOD and measures of visual function (photostress recovery, glare disability, CS). Given that MPOD can be augmented through increased dietary intakes, these findings have meaningful significance for day-to-day activities and functions. There are several mechanisms of action by which lutein and zeaxanthin are involved in visual function. The high metabolic activity and exposure to light of the retina makes this tissue particularly vulnerable to oxidative stress. Lutein and zeaxanthin, being antioxidants, confer protection in this role. In terms of photostress recovery and glare disability, short-wave light filtration would appear to be a reasonable explanation for performance enhancement. However, for CS, light filtration cannot explain a significant enhancement. These effects are more likely due to more efficient communication among visual neurons [1].
To our knowledge this is the first systematic review to examine the association between MPOD and visual function. Studies that met the inclusion criteria were mostly conducted in the same laboratories, so similar methods for the measurements of MPOD and visual function were used across studies. All but one of the studies, which used autofluorescence, used HFP to measure MPOD.
Our conclusions are limited by a few caveats largely due to the lack of reported data in publications on the correlation of interest. The limitations of this review are largely reflective of the lack of data on the correlation of interest reported in publications. Few studies reported on the association of interest. The limited number of studies included in each meta-analysis may have contributed to higher heterogeneity. In addition, although the correlation between MPOD and visual function were analyzed cross-sectionally, studies used different designs to assess this outcome that may have contributed to high heterogeneity. There were studies that we were unable to include because although they clearly stated that researchers measured both MPOD and at least one of our visual functions of interest they did not assess the relationship between the two. Of the included studies, few reported the same MPOD eccentricities measurements for the same visual outcomes, so we were unable to combine them in meta-analysis which resulted in some meta-analysis with only two studies. Due to the limitations in total number of studies there was also insufficient data for subgroup analysis considering age, sex, weight status, and family history of eye disease.
These results are particularly compelling given that lutein and zeaxanthin as sole components of MPOD are believed to provide ocular protection through their roles as blue light filters, antioxidants, and anti-inflammatory agents [6]. These results are first step in providing information toward developing dietary lutein/zeaxanthin dietary in promoting visual health.
Our evaluation suggests that higher MPOD may be beneficial for visual function. This may be due to lutein and zeaxantin’s role as antioxidants. Among the carotenoids, lutein and zeaxanthin are preferentially taken up into the macula. Based on lutein and zeaxanthin intakes related to a decreased risk of age-related macular degeneration [2], average lutein and zeaxanthin intakes are considered to be low [3]. Lutein and zeaxanthin are not essential nutrients. However, evidence is accumulating to suggest that they are important to optimize ocular health. Although the precise mechanisms by which lutein and zeaxanthin may be influencing neural health remain to be investigated, efforts may be warranted to establish recommended intakes for these dietary carotenoids.
Summary
What was known before
-
There was a variety of epidemiological observational evidence indicating that bioactives may have a beneficial role in visual function through increasing macular pigment.
What this study adds
-
Using correlation meta-analysis, our review identified a link between MPOD and visual function with significant correlations with photostress recovery, glare disability, and CS.
Change history
12 October 2020
The original HTML version of this Article was updated shortly after publication to change the copyright line (the Authors opted to change the licence type from standard to Open Access).
References
Lamport DJ, Saunders C, Butler LT, Spencer JP. Fruits, vegetables, 100% juices, and cognitive function. Nutr Rev. 2014;72:774–89.
Hu D, Huang J, Wang Y, Zhang D, Qu Y. Fruits and vegetables consumption and risk of stroke: a meta-analysis of prospective cohort studies. Stroke. 2014;45:1613–9.
Liu RH. Health-promoting components of fruits and vegetables in the diet. Adv Nutr. 2013;4:384S–92.
Bone RA, Landrum JT, Tarsis SL. Preliminary identification of the human macular pigment. Vis Res. 1985;25:1531–5.
Bernstein PS, Khachik F, Carvalho LS, Muir GJ, Zhao DY, Katz NB. Identification and quantitation of carotenoids and their metabolites in the tissues of the human eye. Exp Eye Res. 2001;72:215–23.
Johnson EJ. Role of lutein and zeaxanthin in visual and cognitive function throughout the lifespan. Nutr Rev. 2014;72:605–12.
Gruszecki W, Sujak A, Strzalka K, Radunz A, Schmid G. Organisation of xanthophyll-lipid membranes studied by means of specific pigment antisera, spectrophotometry and monomolecular layer technique lutein versus zeaxanthin. Z Naturforsch C J Biosci. 1999;54:517–25.
Gruszecki WI. Carotenoid orientation. In: Norman I Krinsky, Susan T Mayne, Helmut Sies, editors. Carotenoids in health and disease. New York, NY: Marcel Dekker, Inc. 2004.
Stahl W, Sies H. Effects of carotenoids and retinoids on gap junctional communication. BioFactors Oxf Engl. 2001;15:95–8.
Hammond BR, Wooten BR. CFF thresholds: relation to macular pigment optical density. Ophthalmic Physiol Opt. 2005;25:315–9.
Stringham JM, Stringham NT, O’Brien KJ. Macular carotenoid supplementation improves visual performance, sleep quality, and adverse physical symptoms in those with high screen time exposure. Foods Basel Switz. 2017;6:47.
Bovier ER, Renzi LM, Hammond BR. A double-blind, placebo-controlled study on the effects of lutein and zeaxanthin on neural processing speed and efficiency. PLoS ONE. 2014;9:e108178.
Stringham JM, Fuld K, Wenzel AJ. Spatial properties of photophobia. Investig Ophthalmol Vis Sci. 2004;45:3838–48.
Stringham JM, Garcia PV, Smith PA, Hiers PL, McLin LN, Kuyk TK, et al. Macular pigment and visual performance in low-light conditions. Investig Ophthalmol Vis Sci. 2015;56:2459–68.
Stringham JM, Snodderly DM. Enhancing performance while avoiding damage: a contribution of macular pigment. Investig Ophthalmol Vis Sci. 2013;54:6298–306.
Stringham JM, Hammond BR Jr. The glare hypothesis of macular pigment function. Optom Vis Sci. 2007;84:859–64.
Wallace TC, Blumberg JB, Johnson EJ, Shao A. Dietary bioactives: establishing a scientific framework for recommended intakes. Adv Nutr. 2015;6:1–4.
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097.
Wallace B, Small K, Brodley C, Lau J, Trikalinos T. Deploying an interactive machine learning system in an evidence-based practice center: abstrackr. In Proceedings of the ACM International Health Informatics Symposium. Miami, FL USA, 2012. p. 819–24.
Bovier ER, Hammond BR. A randomized placebo-controlled study on the effects of lutein and zeaxanthin on visual processing speed in young healthy subjects. Arch Biochem Biophys. 2015;572:54–7.
Engles M, Wooten B, Hammond B. Macular pigment: a test of the acuity hypothesis. Investig Ophthalmol Vis Sci. 2007;48:2922–31.
Estevez-Santiago R, Olmedilla-Alonso B, Beltrán-de-Miguel B. Assessment of lutein and zeaxanthin status and dietary markers as predictors of the contrast threshold in 2 age groups of men and women. Nutr Res. 2016;36:719–30.
Hammond BR, Fletcher LM, Roos F, Wittwer J, Schalch W. A double-blind, placebo-controlled study on the effects of lutein and zeaxanthin on photostress recovery, glare disability, and chromatic contrast. Investig Ophthalmol Vis Sci. 2014;55:8583–9.
Hammond BR Jr, Wooten BR, Snodderly DM. Preservation of visual sensitivity of older subjects: association with macular pigment density. Investig Ophthalmol Vis Sci. 1998;39:397–406.
Hammond BR, Fletcher LM, Elliott JG. Glare disability, photostress recovery, and chromatic contrast: relation to macular pigment and serum lutein and zeaxanthin. Investig Ophthalmol Vis Sci. 2013;54:476–81.
Kvansakul J, Rodriguez-Carmona M, Edgar DF, Barker FM, Köpcke W, Schalch W, et al. Supplementation with the carotenoids lutein or zeaxanthin improves human visual performance. Ophthalmic Physiol Opt. 2006;26:362–71.
Loughman J, Akkali MC, Beatty S, Scanlon G, Davison PA, O’Dwyer V, et al. The relationship between macular pigment and visual performance. Vis Res. 2010;50:1249–56.
Nolan JM, Loughman J, Akkali MC, Stack J, Scanlon G, Davison P, et al. The impact of macular pigment augmentation on visual performance in normal subjects: COMPASS. Vis Res. 2011;51:459–69.
Nolan JM, Power R, Stringham J, Dennison J, Stack J, Kelly D, et al. Enrichment of macular pigment enhances contrast sensitivity in subjects free of retinal disease: central retinal enrichment supplementation trials—report 1. Investig Ophthalmol Vis Sci. 2016;57:3429–39.
Patryas L, Parry NRA, Carden D, Aslam T, Murray IJ. The association between dark adaptation and macular pigment optical density in healthy subjects. Graefes Arch Clin Exp Ophthalmol. 2014;252:657–63.
Putnam CM, Bassi CJ. Macular pigment spatial distribution effects on glare disability. J Optom. 2015;8:258–65.
Renzi LM, Hammond BR. The effect of macular pigment on heterochromatic luminance contrast. Exp Eye Res. 2010;91:896–900.
Stringham JM, Hammond BR. Macular pigment and visual performance under glare conditions. Optom Vis Sci. 2008;85:82–8.
Stringham JM, Garcia PV, Smith PA, McLin LN, Foutch BK. Macular pigment and visual performance in glare: benefits for photostress recovery, disability glare, and visual discomfort. Investig Ophthalmol Vis Sci. 2011;52:7406–15.
Stringham JM, O’Brien KJ, Stringham NT. Macular carotenoid supplementation improves disability glare performance and dynamics of photostress recovery. Eye Vis Lond Engl. 2016;3:30.
Stringham JM, O’Brien KJ, Stringham NT. Contrast sensitivity and lateral inhibition are enhanced with macular carotenoid supplementation. Investig Ophthalmol Vis Sci. 2017;58:2291–5.
Tudosescu R, Alexandrescu CM, Istrate SL, Vrapciu AD, Ciuluvică RC, Voinea L. Correlations between internal and external ocular factors and macular pigment optical density. Rom J Ophthalmol. 2018;62:42–7.
Acknowledgements
We wish to thank James M. Stringham, Ph.D., at Duke University and John M. Nolan, Ph.D., at the Waterford Institute of Technology for their scientific guidance.
Funding
This work was supported by the International Life Sciences Institute North America Bioactive Committee. ILSI North America is a public, nonprofit science organization that provides a forum to advance understanding of scientific issues related to the nutritional quality and safety of the food supply. ILSI North America receives support primarily from its industry membership.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
EJJ’s current position is at Ocean Spray Corp but maintains an appointment at Friedman School at Tufts University at which the work was performed.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Johnson, E.J., Avendano, E.E., Mohn, E.S. et al. The association between macular pigment optical density and visual function outcomes: a systematic review and meta-analysis. Eye 35, 1620–1628 (2021). https://doi.org/10.1038/s41433-020-01124-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-020-01124-2