Abstract
Orbital decompression surgery and medical therapy for thyroid eye disease (TED) have evolved over the past 150 years and afforded the opportunity to restore pre-disease appearance and visual function. This manuscript explores the past 150 years of surgical innovation for the treatment of TED. The “Age of Surgical Heroism” spans the time from 1888 to 1979 during which the pioneers of orbital decompression developed lateral orbitotomy, transcranial decompression, paranasal sinus decompression, and transantral decompression despite an incomplete understanding of the pathophysiology of both TED and a limited ability to non-invasively assess their patients. The “Age of Surgical Refinement” dawned with the development of computed tomography and represents the years 1979–2000. During this time, the “swinging eyelid” approach for two- and three-wall decompressions was introduced, a combined orbital-extradural four wall decompression procedure was developed, fat decompression was explored, and endoscopic decompression techniques were advanced. At the beginning of the 21st century, our understanding of the orbital pathophysiology of TED evolved significantly. Clinicians recognized the age-related phenotype of TED based largely on the relative contribution of extraocular muscle enlargement vs. orbital fat expansion. The “Modern Age” of Customized Orbital Decompression features both “medical decompression” during the active phase of TED and, in the stable phase, customized surgical plans incorporating individual patients’ anatomy, orbital pathology, and surgical goals that collectively maximize therapeutic benefit while minimizing therapeutic morbidity.
Similar content being viewed by others
Introduction
Prior to the development of orbital decompression surgery, it was widely accepted that in those severely affected by thyroid eye disease (TED) “protrusion of the eyes progressed until ophthalmitis resulted, and death invariably occurred from orbital infection and resultant meningitis. Many enucleations were performed, but the usual termination was an infected orbit, intracranial extension of the infection, and death” [1]. Fortunately, the evolution of TED decompression surgery and medical therapy has afforded the opportunity to restore both appearance and visual function. This manuscript explores the past 150 years of surgical innovation for the treatment of TED.
Age of surgical heroism: 1888–1979
The “Age of surgical heroism” spans the ~ 100 years during which the pioneers of orbital decompression introduced a series of surgical procedures despite an incomplete understanding of the pathophysiology of both TED proptosis and dysthyroid optic neuropathy (DON) and a limited ability to non-invasively assess the patients. Beginning in the late 1880s, the surgical approaches to the orbit were limited to those most familiar to the surgeon. However, with the advent of general anesthesia and antisepsis in the mid-19th century, the morbidity and mortality of larger procedures decreased and surgical innovations followed [2, 3].
Lateral orbitotomy
The lateral orbitotomy was the first approach to surgical decompression. Kronlein, a Swiss neurosurgeon, described the first lateral orbitotomy procedure for the removal of orbital tumors while sparing the globe in 1889 [4, 5]. In 1890, Dollinger of Budapest adopted Kroenlein’s approach to perform decompression of the orbit, allowing orbital contents to prolapse into the temporalis fossa [6]. The effect was minimal, yielding 2 mm of proptosis reduction, but he had introduced the concept of orbital decompression surgery.
Orbital fat decompression
In 1920, Moore at Moorfields Eye Hospital referenced the first attempt at orbital fat decompression. He experimented with this procedure based on observations he had made during the post-mortem examination of a TED patient with severe proptosis and increased orbital fat. Moore wrote, “It is difficult to identify what is an excess of fat in a cavity which is normally full of it. In this case, however, the orbit was certainly full to overflowing with it, and nothing else abnormal was present.” [7] He subsequently performed orbital fat removal on a patient with marked proptosis that limited closure of the eyelids.
Transcranial decompression
In 1931, Naffzinger at the University of California San Francisco described a transcranial approach with complete removal of the orbital roof, the roof of the optic canal and opening the annulus of Zinn. Employing this technique, Naffzinger was able to arrest “swelling of the (optic) nerve head and hemorrhages”, and improve vision [1]. He later extended his surgical procedure to include the orbital roof, medial to ethmoid and sphenoid cells, and lateral to the greater wing of the sphenoid [8]. The extended Naffzinger transcranial orbital decompression, achieved a 6–9 mm reduction in proptosis. He described postoperative pulsatile proptosis, “of which the patient was unaware” and was of limited duration [1].
Paranasal sinus decompression
In 1929, Hirch, an otolaryngologist, operated on a 24-year-old man through the “canine fossa” and removed the orbital floor through the maxillary sinus as well as orbital fat, citing Moore’s successful fat decompression [7, 9]. Using this approach, he documented 3 mm proptosis reduction [9]. Sewall, also an otolaryngologist, reported a new transcutaneous frontoethmoidal decompression technique in the 1920s and 30 s. He performed surgery on the “ethmosphenoid-frontal group of sinuses” as a “technique for removal of part of the optic foramen wall for relief of pressure on the optic nerve” caused by severe sinusitis [10]. He later employed this technique to perform orbital decompression for TED. Compared with the transcranial approach favored by Naffzinger, Sewall contended that his approach was safer, easier, and gave the surgeon increased space for decompression [11].
Transantral decompression
In 1957, Walsh and Ogura at Washington University in St. Louis, Missouri published their, “Transantral Orbital Decompression for Malignant Exophthalmos” [12]. By their method, the “floor and medial wall of the orbit are removed after the ethmoid air cells have been exenterated.” Walsh and Ogura described removal of ridge of bone against the orbital plate, currently termed “the orbital strut.” They documented a side-by-side comparison of the efficacy of the external and transantral decompression procedures by performing both methods on the same patient. They reported near equal proptosis reduction in both orbits several months after surgery. Using this technique, Walsh and Ogura were able to achieve an average reduction of 4–7 mm of proptosis [12]. They noted the advantages of the transantral method included no external incision and the ability to perform the procedure under local anesthesia. In their initial report, no cases experienced postoperative diplopia, but they did note residual cheek numbness in some. The authors also noted an inverse relationship between the general sense of preoperative orbital tension and the ultimate response to decompression surgery. A provocative footnote to their 1957 paper was the case description of pituitary external beam irradiation used to treat hypothyroidism in one of their orbital decompression patients [12]. The improvement in orbital inflammation was attributed to the effect of the successful surgery, however, the results may be equally be ascribed to the effect of poorly collimated radiation fields that may have inadvertently delivered the first therapeutic orbital radiation for TED.
The age of surgical refinement 1979–2000
The Age of Surgical Refinement dawned with the development of CT scan technology. Previously, plain film X-rays and ultimately tomography were used to visualize the orbit. Plain films were taken from multiple views to reveal all the walls of the orbit, the optic canal, and the superior orbital fissure. Tomography enhanced the plain film studies by defocusing the image plane to allow better recognition of pathological changes in bone structure or soft-tissue density [13]. However, the details of the orbital soft tissues were severely lacking. Houndsfield first wrote of “computerized transverse axial scanning (tomography)” in 1972. The development of CT scanning won Houdsfield and Cormack the Nobel Prize and was ~ 100 times more sensitive than conventional X-ray systems [14]. In 1979, Trokel and Hilal demonstrated the use of commuted tomography in the orbit by examining 603 patients using 4 mm slices instead of the conventional 8–13 mm slice. They examined orbital CT scans from patients with enlarged EOMs, and established the diagnostic radiographic features of TED [15].
Two and three-wall decompression
In 1981, McCord introduced the “swinging eyelid” approach, performed by exposing the orbital floor and medial wall through a lateral canthotomy and cantholysis, followed by swinging or everting the lower lid to extend the incision into the inferior fornix [16]. McCord described using this technique for a two-wall (antral-ethmoidal) or three-wall (antral-ethmoidal-lateral) decompression. Decompressions were customized to the severity of the case. A two-wall decompression resulted in a 4–7 mm decrease in proptosis, whereas a three-wall procedure could produce 6–8 mm of proptosis reduction. McCord felt that the surgical effect of decompression was inversely related to the degree of soft-tissue fibrosis within the orbit. Of note, all patients were treated with maximal medial therapy before surgery.
Four wall decompression
Kennerdell and Maroon developed a combined orbital-extradural decompression procedure in 1982 for patients requiring > 10 mm proptosis reduction. This procedure incorporated removal of a portion of all four orbital walls as well as incisions of the periorbita to allow orbital fat to prolapse into the newly created space. Kennerdell reported seven cases with 10–17 mm of proptosis reduction using this technique [17].
Fat decompression
Fat decompression is infrequently mentioned in the literature [7, 9] until 1988 when Olivari published his results removing infraorbital fat through a transpalpebral incision [18]. Olivari identified the contribution of fat expansion to proptosis in select patients. In their series of patients, they removed an average of 6 cc orbital fat and documented an average proptosis reduction of 5.9 mm [18]. Trokel and Kazim followed with two reports refining the technique for the safe removal of intra- and extraconal fat as well as establishing the preoperative indications for the procedure. It was determined that the procedure was most effective in cases of orbital fat expansion as compared to those with predominant extraocular muscle (EOM) enlargement and that the surgical effect is typically 1 mm of proptosis reduction for each 1 cc of orbital fat volume removed [19]. More unexpected was the improvement in diplopia in 11% of cases and reversal of DON in selected cases [20, 21].
Endoscopic decompression
Endoscopic transnasal decompression was described in 13 orbits by Kennedy in 1990 [22]. The advantages of endoscopic surgery and an extra orbital approach were evident for those patients with tight orbits or a globe at risk including those with prior corneal transplant or glaucoma filtering procedure in which increased introrbital pressure pressure and manipulation should be minimized.
Deep lateral wall decompression
In 1998, Goldberg published his technique of removing bone along the deep lateral wall. Although these were the surfaces historically removed in both Naffziger and the Kennerdell and Maroon procedures [4, 6], Goldberg posited that there is considerable room for orbital expansion in the lateral orbit alone if the thicker, deep areas of lateral wall are removed [23]. By removing the deep lateral wall, one can maximize proptosis reduction through a small transcutaneous lateral approach.
Transcaruncle orbitotomy
The transcaruncle orbitotomy was introduced in 2000 by Shorr and Baylis [24]. In order to achieve continuous exposure of the medial and inferior orbit during orbital wall fracture repair and orbital expansion, the traditional transconjunctival approach was extended through the caruncle [24]. This method has the advantages of being simple, providing immediate access to the medial wall, and requiring no additional equipment as is the case with endoscopic surgery. The limitations however are apparent when operating within a tight orbit, making apical visualization difficult. As well, because there is no sinonasal dissection, patients may be predisposed to postoperative sinus infections.
Endoscopic medial wall, floor, and lateral wall decompression
As the popularity of endoscopic floor and medial wall decompressions increased, the concept of a “balanced decompression”, which includes the lateral floor and may decrease postoperative diplopia while increasing the decompression effect, was introduced [25]. In 2008, Dubin published a series of patients who underwent image-guided endoscopic balanced orbital decompression [26]. The indications for surgery were optic neuropathy or a desired proptosis reduction > 6 mm. The authors found that all patients had either resolution or improvement in their optic neuropathy and the mean proptosis reduction was 6.2 mm. The incorporation of intraoperative image guidance assists with surgical anatomy and may be a useful adjunct to this procedure [26].
Modern principles of TED management
In the present era, TED is managed based upon the phase of the disease. The acute phase is most commonly treated medically, employing corticosteroids (CS), orbital radiotherapy (ORT), and other more recently developed immunomodulatory agents such as rituximab to suppress the clinical signs and symptoms of inflammation and potentially limit the duration of the disease. Surgical decompression in the acute phase of TED is reserved for cases of medically non-responsive optic neuropathy. In the stable phase, rehabilitative surgery is sequenced to include orbital fat and/or bone decompression, strabismus surgery, and lastly eyelid surgery as required.
Pathophysiology of orbitopathy
At the beginning of the 21st century, a refined understanding of the orbital pathophysiology of TED and DON developed. Clinicians recognized the age-related phenotype of TED relative to the contribution of EOM enlargement vs. orbital fat expansion. From a mechanical perspective, the clinical signs and symptoms of TED are owing to expansion of the soft-tissue volume within the relatively static bony orbital volume. The soft-tissue expansion displaces the globe anteriorly, impedes venous outflow, and induces compressive optic neuropathy. The mechanism of DON is hypothesized to be multifactorial and includes the consequences of optic nerve compression, stretch, and inflammation. Although generally accepted that pathologically enlarged rectus muscles compressing the optic nerve produces DON, in 2000, Kazim et al. documented reversal of DON in five patients after only orbital fat decompression with no bony removal, demonstrating the unique contribution of orbital fat expansion to DON. Each of these patients had enlarged EOMs but also had evidence of an expanded fat compartment [20]. Orepesa et al. [27] showed that in compressive DON, whereas the medial rectus is most likely to be enlarged across the entire orbit, the superior rectus–levator complex is significantly greater in those with TED compressive optic neuropathy than in those with non-compressive TED at the posterior 1/3 of the orbit. As well, an inferior visual field defect has been shown to be the most typical TED-DON-associated visual field change [28]. The diagnosis of DON relies on multiple factors including vision, pupillary findings, color vision, and visual field testing. A mathematical formula to diagnose DON was developed in 2018 and uses relative afferent pupillary defect, mean deviation on Humphrey visual field, percentage of color plates, and summed limitation of ductions to predict the presence of DON [29].
Medical decompression
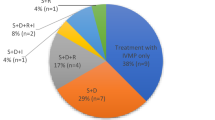
During the active phase, surgical treatment aimed at reducing orbital soft-tissue volume (fat decompression) or expanding orbital capacity (bony orbital decompression) may be effective, but has limitations, including increased surgical risk and less predictable postoperative outcomes. Moreover, decompression surgery does not routinely shorten the course of the orbitopathy. It is not uncommon to see patients in whom successful orbital decompression surgery reverses compressive optic neuropathy only to be followed by unrelenting progressive orbitopathy filling the newly expanded orbital bony volume and in turn producing recurrent optic nerve compression. Alternatively, during the active, progressive phase of TED, “medical decompression” is advocated. This term refers to the reduction of orbital soft-tissue volume by immunomodulation, most commonly utilizing CS and ORT. Historically, oral CS have been used extensively to treat TED, although the results of several prospective clinical trials suggest that CS given intravenously (IVCS) may be more effective with less morbidity, improved quality of life, and fewer subsequent surgeries [30, 31]. Severe hepatotoxicity is a potential complication of IVCS therapy, but appears to be dose dependent and is thought to occur only in patients receiving a cumulative dose > 8 g of methylprednisolone [32]. Similarly, evidence suggests that ORT is appropriate in patients with early, progressive, moderate to severe, active TED. Patients with mild or inactive disease are not expected to benefit from the treatment when compared with the natural history of the disease, and the risks may outweigh the benefits in these patients. Accordingly, a positive response to CS typically suggests therapeutic potential for ORT. ORT is more effective when administered in conjunction with CS, although it can serve as an independent modality in patients for whom CS are contraindicated. Similarly, it can also be concluded that corticosteroid therapy is more effective, in the short and long term, when administered in conjunction with ORT.
In the authors’ experience with this treatment paradigm, 95% of patients suffering TED-DON experienced a reversal of optic neuropathy without recurrence [33]. After treatment with combined ORT and CS, TED-DON reversed and the orbitopathy has stabilized, orbital decompression surgery can be performed electively at a time that the functional and cosmetic results of surgery are most predictable, or surgery avoided altogether. In the authors reported experience, 78% of patients suffering TED-DON avoided the need for either urgent or elective surgical decompression [33]. Orbital decompression remains a valuable treatment option for TED-DON unresponsive to CS. There is ample evidence demonstrating that the combination of ORT and CS is an effective alternative in patients with steroid-responsive TED-DON. Furthermore, the administration of ORT and CS early in the course of disease in those patients with peripheral diplopia may prevent the development of TED-DON [34].
Modern age of customized orbital decompression 2000 present
Advances in orbital imaging, expanded understanding of the pathophysiology of TED, and refinement of surgical techniques set the stage for the era of customized orbital decompression. What remained was the development of a systematic approach to constructing an individualized surgical plan for each patient. As previously described, the notion of individualized surgical plans was alluded to in Naffziger’s and McCord’s manuscripts, but owing to the limitations of the time remained undeveloped. As currently conceptualized, the authors view the following principles as key to customized orbital decompression:
- 1.
Treatment of active phase TED is primarily medical, whereas stable phase TED is treated with elective surgery.
- 2.
All cases of proptosis are different. The relative contribution of volume expansion of the orbital fat and the extraocular muscles dictates the most appropriate operation.
- 3.
Hertel measurements are not the best guide to surgical planning, and normative Hertel averages are of no value.
- 4.
Pre-disease photos help to define the amount of proptosis reduction needed.
In preoperative assessment, surgeons should first determine whether the operative indication is proptosis or DON, and then determine the desired proptosis reduction. Using preoperative imaging, surgeons should evaluate the ratio of EOM to fat expansion and note the size and health of the sinuses. Finally, both surgeons and patients should understand the risk of postoperative strabismus and the type anticipated: esotropia from the medial wall, hypotropia from the floor, and torsional diplopia from extensive floor and medial wall. When discussing risks with patients, surgical morbidities associated with orbital decompression include sinusitis, over correction of proptosis, altered vocal quality, infraorbital anesthesia, or pain, globe ptosis, cerebrospinal fluid leak, optic nerve injury, and increased lid retraction or lagophthalmos.
Given these considerations, one may perform a custom decompression by indication (see Fig. 1). We would further add the following surgical tips. When decompressing the orbital floor, we leave the floor lateral to the infraorbital nerve intact. Removal of this bone produces little additional volume expansion and increases the risk of infraorbital anesthesia. We leave the anterior floor medially to avoid hypoglobus and torsional diplopia. When deciding whether to remove the “strut”, consider the surgical goal. Remove the strut to maximize the decompression for treatment of proptosis or DON. In a study of endoscopic two- and three-wall decompressions with and without strut removal, the mean reduction in proptosis was 3.60 mm for those with a three-wall decompression with strut preservation vs. 7.65 mm for those with a three-wall decompression with strut removal [35]. Similarly, in most cases we remove the periosteum to maximize the surgical effect. However, in a small minority in which concern regarding the development of postoperative diplopia exists, we will leave a strip of periosteum over the medial rectus muscle (“sling”) to reduce the risk of postoperative diplopia. Doing so does however reduce the magnitude of proptosis reduction as does preserving the orbital strut. Failures of orbital decompression may occur owing to preserved orbital bones, progressive orbitopathy/recurrent DON, reformation of preoperative periosteal dimensions, or due to an unsuspected alternate etiology of optic neuropathy.
Custom orbital decompression by indication. If the indication is proptosis and a 6 mm or more decompression is warranted, for a patient with large EOMs, we recommend a three-wall decompression of the lateral wall, medial wall, floor, and fat decompression. In a patient with small EOMs, we recommend a fat decompression plus the lateral wall. For patients with proptosis but requiring < 6 mm of decompression, if the EOMs are large, we recommend a lateral wall plus medial and floor but sparing the strut. If the EOMs are small, we recommend a fat decompression with the lateral wall. If the decompression desired is 3–4 mm and the EOMs are small to medium fat decompression alone is indicated. When the indication for decompression is DON, if the patient is in the active phase, we recommend a medial wall and floor decompression including the strut followed by radiotherapy. For patients in the stable phase, we recommend a medial wall and floor decompression including the strut. Surgeons may add lateral wall and fat decompression if there is excess proptosis
Conclusion
The surgical approach to orbital decompression has evolved dramatically over the past 150 years. The authors hope that future work will focus on the predictability of the decompressive effect, mitigating late recurrence of proptosis, limiting postoperative diplopia, and standardizing more predictable simultaneous decompression, EOM, and eyelid surgeries.
References
Naffziger HC. Progressive exophthalmos following thyroidectomy; its pathology and treatment. Ann Surg. 1931;94:582–6.
Semmelweis IP. Die aetiologie, der bergriff und die prophylaxis des kindbettfiebers. Am J Obstet Gynecol. 1995;172:236–27.
Lister J. On the antiseptic principle in the practice of surgery. Br Med J. 1867;2:246–8.
Kronlein R. Zur pathologie and operative Behandlung der dermoidcysten der orbita. Klin Chir. 1889;4:149–63.
Alper MG. Pioneers in the history of orbital decompression for Graves’ ophthalmopathy. R.U. Kroenlein (1847-1910), O. Hirsch (1877-1965) and H.C. Naffziger (1884-1961). Doc Ophthalmol. 1995;89:163–71.
Dollinger, J. Die Druckenflastung der Augenhoehe durch Entfernung der aeusseren Orbitalwand bei hochgradigem Exophthalmus (Morbus Basedowii) und konsekutiver Hornhauterkrankung. Deutsche Medizin Wochenschr, 1911.
Foster Moore R. A note on the exophthalmos and limitation of the eye movements of Graves’s disease. Lancet. 1920;196:701.
Naffziger HC. Progressive exophthalmos associated with disorders of the thyroid gland. Ann Surg. 1938;108:529–44.
Hirsch O. Surgical decompression of malignant exophthalmos. Arch Otolaryngol. 1950;51:325–34.
Sewall E. External operation on the ethmosphenoidfrontal group of sinuses under local anesthesia: technic for removal of part of optic foramen wall for relief of pressure on optic nerve. Arch Otolaryngol. 1926;4:377–411.
Sewall E. Operative control of progressive exophthalmos. Arch Otolaryngol. 1936;24:621–4.
Walsh TE, Ogura JH. Transantral orbital decompression for malignant exophthalmos. Laryngoscope. 1957;67:544–68.
Trokel SL. Radiological techniques in the diagnosis of ocular and orbital tumors. Int Ophthalmol Clin. 1972;12:145–62.
Hounsfield GN. Computerized transverse axial scanning (tomography): part I. Description of system. 1973. Br J Radiol. 1995;68:H166–72.
Trokel SL, Hilal SK. Recognition and differential diagnosis of enlarged extraocular muscles in computed tomography. Am J Ophthalmol. 1979;87:503–12.
McCord CD Jr., Moses JL. Exposure of the inferior orbit with fornix incision and lateral canthotomy. Ophthalmic Surg. 1979;10:53–63.
Kennerdell JS, Maroon JC. An orbital decompression for severe dysthyroid exophthalmos. Opthalmology. 1982;89:467–72.
Olivari N. Transpalpebral decompression of endocrine ophthalmopathy (Graves’ disease) by removal of intraorbital fat: experience with 147 operations over 5 years. Plast Reconstr Surg. 1991;87:627–41.
Trokel S, Kazim M, Moore S. Orbital fat removal. Decompression for Graves orbitopathy. Ophthalmology. 1993;100:674–82.
Kazim M, et al. Reversal of dysthyroid optic neuropathy following orbital fat decompression. Br J Ophthalmol. 2000;84:600–5.
Prat MC, et al. Orbital fat decompression for thyroid eye disease: retrospective case review and criteria for optimal case selection. Ophthalmic Plast Reconstr Surg. 2015;31:215–8.
Kennedy DW, et al. Endoscopic transnasal orbital decompression. Arch Otolaryngol Neck Surg. 1990;116:275–82.
Goldberg RA, Kim AJ, Kerivan KM. The lacrimal keyhole, orbital door jamb, and basin of the inferior orbital fissure. Three areas of deep bone in the lateral orbit. Arch Ophthalmol. 1998;116:1618–24.
Shorr N, et al. Transcaruncular approach to the medial orbit and orbital apex. Ophthalmology. 2000;107:1459–63.
Kacker A, et al. “Balanced” orbital decompression for severe Graves’ orbitopathy: technique with treatment algorithm. Otolaryngol Head Neck Surg. 2003;128:228–35.
Dubin MR, et al. Image-guided endoscopic orbital decompression for Graves’ orbitopathy. Ann Otol Rhinol Laryngol. 2008;117:177–85.
Oropesa, S, et al. Predominant contribution of superior rectus-levator complex enlargement to optic neuropathy and inferior visual field defects in thyroid eye disease. Ophthalmic Plast Reconstr Surg. 2018. https://doi.org/10.1097/IOP.0000000000001224
Choi CJ, et al. Patterns of visual field changes in thyroid eye disease. Orbit. 2017;36:201–7.
Callahan AB, et al. The columbia thyroid eye disease-compressive optic neuropathy diagnostic formula. Ophthalmic Plast Reconstr Surg. 2018;34:S68–s71.
Kahaly GJ, et al. Randomized, single blind trial of intravenous versus oral steroid monotherapy in Graves’ orbitopathy. J Clin Endocrinol Metab. 2005;90:5234–40.
Bartalena L, et al. Efficacy and safety of three different cumulative doses of intravenous methylprednisolone for moderate to severe and active Graves’ orbitopathy. J Clin Endocrinol Metab. 2012;97:4454–63.
Marino M, et al. Acute and severe liver damage associated with intravenous glucocorticoid pulse therapy in patients with Graves’ ophthalmopathy. Thyroid. 2004;14:403–6.
Gold KG, et al. Orbital radiotherapy combined with corticosteroid treatment for thyroid eye disease-compressive optic neuropathy. Ophthalmic Plast Reconstr Surg. 2018;34:172–7.
Shams PN, et al. Reduced risk of compressive optic neuropathy using orbital radiotherapy in patients with active thyroid eye disease. Am J Ophthalmol. 2014;157:1299–305.
Finn AP, et al. A retrospective review of orbital decompression for thyroid orbitopathy with endoscopic preservation of the inferomedial orbital bone strut. Ophthalmic Plast Reconstr Surg. 2017;33:334–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no disclosures related to the content of this manuscript.
Rights and permissions
About this article
Cite this article
Tooley, A.A., Godfrey, K.J. & Kazim, M. Evolution of thyroid eye disease decompression—dysthyroid optic neuropathy. Eye 33, 206–211 (2019). https://doi.org/10.1038/s41433-018-0259-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-018-0259-0
This article is cited by
-
Comparison of surgical effect in active and inactive Dysthyroid Optic Neuropathy after Endoscopic Transnasal Medial Orbital Decompression
Graefe's Archive for Clinical and Experimental Ophthalmology (2024)
-
Consideration of specific key points improves outcome of decompression treatment in patients with endocrine orbitopathy: pre-/post-OP comparison and biomechanical simulation
European Journal of Medical Research (2022)
-
Management of patients with dysthyroid optic neuropathy treated with intravenous corticosteroids and/or orbital decompression surgery
Graefe's Archive for Clinical and Experimental Ophthalmology (2022)
-
Modified endoscopic transnasal orbital apex decompression in dysthyroid optic neuropathy
Eye and Vision (2021)
-
Dysthyroid optic neuropathy: evaluation and management
Journal of Endocrinological Investigation (2021)