Abstract
Design Randomised controlled clinical trial.
Data sources Not applicable.
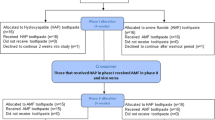
Study aims This split-mouth randomised controlled clinical trial assessed the effects of placing doxycycline hyclate (Atridox) at the implant-abutment interface on the short-term clinical outcomes of dental implants.
Methods The study sample included 20 patients; each patient had two mandibular implants placed, one on either side of the mandible, resulting in a total of 40 implants placed in the study sample. At the time of final prosthesis delivery, doxycycline hyclate (Atridox 10% doxycycline hyclate) was injected at the implant-abutment interface of 20 randomly selected implants on the test side (N = 20) while no intervention was done on the control side (N = 20). The primary outcome measure was changes in pocket probing depth while secondary outcome measures included the incidence of peri-implant mucositis, bleeding on probing, and changes in marginal bone levels on mesial and distal bone aspects of the implant. The outcome measures were assessed at baseline, 3 months, 6 months and 12 months.
Results The results showed favourable effects of prophylactic doxycycline application. Marginal bone levels and pocket probing depths after 6 and 12 months on the test side were less compared to the control side and these differences were statistically significant. The test side also showed fewer implants with bleeding on probing and lower risk of peri-implant mucositis after 3, 6 and 12 months.
Conclusion Prophylactic placement of doxycycline hyclate may reduce peri-implant bone loss and pocket probing depths, and may also reduce the risk of peri-implant mucositis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Giannobile W V, Jung R E, Schwarz F. Evidence-based knowledge on the aesthetics and maintenance of peri-implant soft tissues: Osteology Foundation Consensus Report Part 1 - Effects of soft tissue augmentation procedures on the maintenance of peri-implant soft tissue health. Clin Oral Implants Res 2018; 29 Suppl 15: 7-10.
Cairo F, Barbato L, Tonelli P, Batalocco G, Pagavino G, Nieri M. Xenogeneic collagen matrix versus connective tissue graft for buccal soft tissue augmentation at implant site. A randomized, controlled clinical trial. J Clin Periodontol 2017; 44: 769-776.
Ali K, Kay E J. What are the long-term survival and complication rates of complete-arch fixed implant rehabilitation in edentulous patients? Evid Based Dent 2019; 20: 97-98.
Crossnohere N L, Brundage M, Calvert M J et al. International guidance on the selection of patient-reported outcome measures in clinical trials: a review. Qual Life Res 2021; 30: 21-40.
Geisinger M L, Grosso K C, Kaur M et al. Clinical Decision Making for Primary Peri-Implantitis Prevention: Practical Applications. Clin Adv Periodontics 2021; 11: 43-53.
Ali K, Kay E J. Which type of soft tissue augmentation at dental implant sites is best supported by evidence? Evid Based Dent 2020; 21: 140-141.
Author information
Authors and Affiliations
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Ali, K., McColl, E. Does prophylactic application of doxycycline at the implant-abutment interface improve clinical outcomes of dental implants?. Evid Based Dent 23, 158–159 (2022). https://doi.org/10.1038/s41432-022-0839-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41432-022-0839-5