Abstract
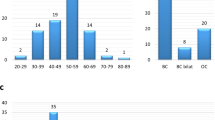
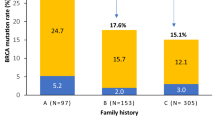
Mutations in BRCA1 and BRCA2 significantly elevate the risk of developing breast and ovarian cancer. Limited data exists regarding the prevalence of BRCA mutations, and optimal, cost-effective testing strategies in developing countries like India. This study aimed to evaluate the utility of a Next Generation Sequencing (NGS) panel for BRCA1/2 mutation testing among women diagnosed with, or at risk of developing hereditary breast and ovarian cancers. We also aimed to identify population specific BRCA1/2 mutation hotspots, to enable the development of more affordable testing strategies. We identified 921 patients with breast and ovarian cancer who underwent mutation testing. The target enrichment was followed by targeted NGS in 772 patients and an allele-specific PCR (ASPCR) based genotyping for BRCA1:c.68_69delAG (or 185delAG), was carried out in 149 patients. We identified 188 (20.4%) patients with BRCA1/2 variants: 118 (62.8%) with pathogenic/likely pathogenic and 70 (37.2%) with VUS. The 185delAG was identified as a recurrent mutation in the Southern Indian population, accounting for 24.6% of the pathogenic variants. In addition, a family history of breast, ovary, pancreas, or prostate (BOPP) cancer was found to be associated with an increased risk of identifying a deleterious BRCA1/2 variant [OR = 2.11 (95% CI 1.45–3.07) p ≤ 0.001]. These results suggest that Targeted NGS is a sensitive and specific strategy for BRCA testing. For Southern Indian patients, a two-tiered approach can be considered: Initial screening with ASPCR for BRCA1 185delAG followed by NGS for those testing negative. Expanding the gene panel and identifying other population-specific mutation hot spots is a promising area with potential for improvements in testing and treatment strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets that have been generated and/or analyzed in the course of the current study are accessible through the corresponding author and can be obtained upon request.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
Cancer Today [Internet]. 2020. Available from: http://gco.iarc.fr/today/home.
Shi T, Wang P, Xie C, Yin S, Shi D, Wei C, et al. BRCA1 and BRCA2 mutations in ovarian cancer patients from China: ethnic-related mutations in BRCA1 associated with an increased risk of ovarian cancer. Int J Cancer. 2017;140:2051–9.
Tutt ANJ, Garber JE, Kaufman B, Viale G, Fumagalli D, Rastogi P, et al. Adjuvant olaparib for patients with BRCA1- or BRCA2-mutated breast cancer. N Engl J Med. 2021;384:2394–405.
Moore K, Colombo N, Scambia G, Kim BG, Oaknin A, Friedlander M, et al. Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl J Med. 2018;379:2495–505.
Robson M, Im SA, Senkus E, Xu B, Domchek SM, Masuda N, et al. Olaparib for metastatic breast cancer in patients with a germline BRCA mutation. N Engl J Med. 2017;377:523–33.
Pujade-Lauraine E, Ledermann JA, Selle F, Gebski V, Penson RT, Oza AM, et al. Olaparib tablets as maintenance therapy in patients with platinum-sensitive, relapsed ovarian cancer and a BRCA1/2 mutation (SOLO2/ENGOT-Ov21): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2017;18:1274–84.
Malvia S, Bagadi SA, Dubey US, Saxena S. Epidemiology of breast cancer in Indian women. Asia Pac J Clin Oncol. 2017;13:289–95.
Vaidyanathan K, Lakhotia S, Ravishankar HM, Tabassum U, Mukherjee G, Somasundaram K. BRCA1 and BRCA2 germline mutation analysis among Indian women from south India: identification of four novel mutations and high-frequency occurrence of 185delAG mutation. J Biosci. 2009;34:415–22.
Friedenson B. The BRCA1/2 pathway prevents hematologic cancers in addition to breast and ovarian cancers. BMC Cancer. 2007;7:152.
Yoshida K, Miki Y. Role of BRCA1 and BRCA2 as regulators of DNA repair, transcription, and cell cycle in response to DNA damage. Cancer Sci. 2004;95:866–71.
Singh J, Thota N, Singh S, Padhi S, Mohan P, Deshwal S, et al. Screening of over 1000 Indian patients with breast and/or ovarian cancer with a multi-gene panel: prevalence of BRCA1/2 and non-BRCA mutations. Breast Cancer Res Treat. 2018;170:189–96.
Chheda P, Pande S, Dama T, Vinarkar S, Chanekar M, Limaye S, et al. Spectrum of germline BRCA mutations in hereditary breast and ovarian cancer syndrome in Indian population: a central reference laboratory experience. Cancer Res Stat Treat. 2020;3:32.
Kadri MSN, Patel KM, Bhargava PA, Shah FD, Badgujar NV, Tarapara BV, et al. Mutational landscape for indian hereditary breast and ovarian cancer cohort suggests need for identifying population specific genes and biomarkers for screening. Front Oncol. 2021;10:568786.
Gupta S, Rajappa S, Advani S, Agarwal A, Aggarwal S, Goswami C, et al. Prevalence of BRCA1 and BRCA2 mutations among patients with ovarian, primary peritoneal, and fallopian tube cancer in india: a multicenter cross-sectional study. JCO Global Oncol. 2021; Available from: https://doi.org/10.1200/GO.21.00051.
Lincoln SE, Kobayashi Y, Anderson MJ, Yang S, Desmond AJ, Mills MA, et al. A systematic comparison of traditional and multigene panel testing for hereditary breast and ovarian cancer genes in more than 1000 patients. J Mol Diagn. 2015;17:533–44.
Buys SS, Sandbach JF, Gammon A, Patel G, Kidd J, Brown KL, et al. A study of over 35,000 women with breast cancer tested with a 25-gene panel of hereditary cancer genes. Cancer. 2017;123:1721–30.
Harismendy O, Ng PC, Strausberg RL, Wang X, Stockwell TB, Beeson KY, et al. Evaluation of next generation sequencing platforms for population targeted sequencing studies. Genome Biol. 2009;10:R32.
Mu W, Lu HM, Chen J, Li S, Elliott AM. Sanger confirmation is required to achieve optimal sensitivity and specificity in next-generation sequencing panel testing. J Mol Diagn. 2016;18:923–32.
Daly MB, Pilarski R, Yurgelun MB, Berry MP, Buys SS, Dickson P, et al. NCCN guidelines insights: genetic/familial high-risk assessment: breast, ovarian, and pancreatic, version 1.2020. J Natl Compr Canc Netw. 2020;18:380–91.
Chapla A, Mruthyunjaya MD, Asha HS, Varghese D, Varshney M, Vasan SK, et al. Maturity onset diabetes of the young in India – a distinctive mutation pattern identified through targeted next-generation sequencing. Clin Endocrinol. 2015;82:533–42.
Chang X. wANNOVAR: annotating genetic variants for personal genomes via the web. J Med Genet. 2012;49:433–6.
McNeill A. No April fools in clinical genomics. Eur J Hum Genet. 2022;30:389–90.
VarSome [Internet]. 2020. VarSome Citations. Available from: https://varsome.com/about/general/varsome-citations/.
Lek M, Karczewski KJ, Minikel EV, Samocha KE, Banks E, Fennell T, et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature 2016;536:285–91.
Jasin M. Homologous repair of DNA damage and tumorigenesis: the BRCA connection. Oncogene. 2002;21:8981–93.
Yoshida R. Hereditary breast and ovarian cancer (HBOC): review of its molecular characteristics, screening, treatment, and prognosis. Breast Cancer. 2021;28:1167–80.
Hu C, Hart SN, Gnanaolivu R, Huang H, Lee KY, Na J, et al. A population-based study of genes previously implicated in breast cancer. N Engl J Med. 2021;384:440–51.
Díez O, Osorio A, Durán M, Martinez-Ferrandis JI, de la Hoya M, Salazar R, et al. Analysis of BRCA1 and BRCA2 genes in Spanish breast/ovarian cancer patients: a high proportion of mutations unique to Spain and evidence of founder effects. Hum Mutat. 2003;22:301–12. https://pubmed.ncbi.nlm.nih.gov/12955716/.
Alvarez C, Tapia T, Perez-Moreno E, Gajardo-Meneses P, Ruiz C, Rios M, et al. BRCA1 and BRCA2 founder mutations account for 78% of germline carriers among hereditary breast cancer families in Chile. Oncotarget. 2017;8:74233–43. https://pubmed.ncbi.nlm.nih.gov/29088781/.
Turbitt E, Lin JL. Genetic testing decisions in non-western cultures: an opportunity for intergenerational decision making. Eur J Hum Genet. 2022;30:391.
Saxena S, Chakraborty A, Kaushal M, Kotwal S, Bhatanager D, Mohil RS, et al. Contribution of germline BRCA1 and BRCA2 sequence alterations to breast cancer in Northern India. BMC Med Genet. 2006;7:75.
Mannan AU, Singh J, Lakshmikeshava R, Thota N, Singh S, Sowmya TS, et al. Detection of high frequency of mutations in a breast and/or ovarian cancer cohort: implications of embracing a multi-gene panel in molecular diagnosis in India. J Hum Genet. 2016;61:515–22.
Kim H, Choi DH. Distribution of BRCA1 and BRCA2 mutations in Asian patients with breast cancer. J Breast Cancer. 2013;16:357–65.
Bougie O, Weberpals JI. Clinical considerations of BRCA1- and BRCA2-mutation carriers: a review. Int J Surg Oncol. 2011;2011:e374012.
Rajkumar T, Meenakumari B, Mani S, Sridevi V, Sundersingh S. Targeted resequencing of 30 genes improves the detection of deleterious mutations in South Indian Women with Breast and/or Ovarian Cancers. Asian Pac J Cancer Prev. 2015;16:5211–7.
Chakraborty A, Banerjee D, Basak J, Mukhopadhyay A. Absence of 185delAG and 6174delT mutations among breast cancer patients of eastern India. Asian Pac J Cancer Prev. 2015;16:7929–33.
Kreiss Y, Barak F, Baruch RG, Levy-Lahad E, Pras E, Friedman E. The founder mutations in the BRCA1, BRCA2, and ATM genes in Moroccan Jewish women with breast cancer. Genet Test. 2000;4:403–7.
The frequency of founder mutations in the BRCA1, BRCA2, and APC genes in Australian Ashkenazi Jews - Bahar - 2001 - Cancer - Wiley Online Library. 2023. Available from: https://acsjournals.onlinelibrary.wiley.com/doi/full/10.1002/1097-0142%2820010715%2992%3A2%3C440%3A%3AAID-CNCR1340%3E3.0.CO%3B2-O.
Berman DB, Wagner-Costalas J, Schultz DC, Lynch HT, Daly M, Godwin AK. Two distinct origins of a common BRCA1 mutation in breast-ovarian cancer families: a genetic study of 15 185delAG-mutation kindreds. Am J Hum Genet. 1996;58:1166.
Laitman Y, Feng BJ, Zamir IM, Weitzel JN, Duncan P, Port D, et al. Haplotype analysis of the 185delAG BRCA1 mutation in ethnically diverse populations. Eur J Hum Genet. 2013;21:212–6.
Stone Inscription of Oldest Synagogue In Tamil Nadu Found | Chennai News - Times of India. 2023. Available from: https://timesofindia.indiatimes.com/city/chennai/stone-inscription-of-oldest-synagogue-in-tamil-nadu-found/articleshow/92837874.cms.
Waldman YY, Biddanda A, Dubrovsky M, Campbell CL, Oddoux C, Friedman E, et al. The genetic history of Cochin Jews from India. Hum Genet. 2016;135:1127–43.
Kadalmani K, Deepa S, Bagavathi S, Anishetty S, Thangaraj K, Gajalakshmi P. Independent origin of 185delAG BRCA1 mutation in an Indian family. Neoplasma 2007;54:51–6.
Acknowledgements
We would like to thank all our patients and their families for their consent and participation in the study. We acknowledge Mrs. Manika Varshini and Ms. Judith Evangeline for their involvement in the initial stages of the study.
Funding
There was no specific funding for this manuscript.
Author information
Authors and Affiliations
Contributions
Study conception and design: Dr. Aaron Chapla and Dr. Ashish Singh. NGS and ASPCR design and standardization: Dr. Aaron Chapla, Mrs. Deny Varghese, Mr. Parthiban R. Material preparation, data collection, analysis: Dr. Ajoy Oommen John, Dr. Ashish Singh, Dr. Anjana Joel, Ms. Pratibha Yadav, Dr. Divya Bala Thumaty, Dr. Fibi Ninan K, Dr. Josh Thomas Georgy, Dr. Anish Jacob Cherian, Dr. Shawn Thomas, Dr. Anitha Thomas, Dr. Vinotha Thomas, Dr. Abraham Peedicayil, Ms. Lavanya Ravichandran, Mr. Jabasteen Johnson, Dr. Bijesh Yadav, Dr. Patricia S, Dr. Selvamani B, Dr. Deepak Abraham, Dr. MJ Paul, Dr. Raju Titus Chacko, Dr. Nihal Thomas, Dr. Ashish Singh, Dr. Aaron Chapla. First draft of the manuscript was written by Dr. Ajoy Oommen John, Dr. Aaron Chapla, Dr. Ashish Singh and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study was approved by the institutional review board IRB no 13205 (retro) dated 22.07.2020.
Consent for participation
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
John, A.O., Singh, A., Yadav, P. et al. The BRCA mutation spectrum among breast and ovarian cancers in India: highlighting the need to screen BRCA1 185delAG among South Indians. Eur J Hum Genet (2024). https://doi.org/10.1038/s41431-024-01596-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41431-024-01596-w