Abstract
Increasing use of diagnostic genomic sequencing is pushing health services to confront the issue of opportunistic genomic screening (OGS). To date, OGS has been offered concomitant with diagnostic testing. In contrast, we piloted a service offering OGS after return of diagnostic testing results. Evaluation was designed to provide insights for future models of service and included patient surveys at three time points, semi-structured interviews with genetic counsellors (GCs) and a focus group with medical scientists. Uptake was relatively low: 83 of 200 patients approached (42%) attended the OGS service, with 81 accepting OGS. Whilst many who declined to attend the service cited practical barriers, others gave reasons that indicated this was a considered decision. Despite specific genetic counselling, one third of patients did not understand the scope of re-analysis. Yet after post-test counselling, all respondents with novel pathogenic additional findings (AF) understood the implications and reported relevant follow-up. Recall was high: five months after last contact, 75% recalled being offered OGS without prompting. GC interviews and patient survey responses provide insights into complexities that influence patient support needs, including diagnostic status and AF result type. There was no consensus among patients or professionals about when to offer OGS. There was a clear preference for multiple, flexible methods of information provision; achieving this whilst balancing patient support needs and resource requirements is a challenge requiring further investigation. Decisions about whether, when and how to offer OGS are complex; our study shows the two-step approach warrants further exploration.
Similar content being viewed by others
Introduction
The advent of genomic testing for diagnostic purposes has brought with it the possibility of opportunistic genomic screening (OGS) for future disease. That is, a deliberate, pre-emptive analysis of the genome for conditions unrelated to the initial indication for testing [1]. OGS has been contentious, particularly in collectively funded healthcare systems, with extensive ethical debate regarding logistics, cost, medicolegal issues, and psychological impact [2]. The American College of Medical Genetics and Genomics [3] position is that patients should be offered OGS for medically actionable disease-causing gene variants, which we term ‘additional findings’ (AF), unrelated to the initial indication for testing. A more cautious approach has been taken in Europe [4] and Canada [5], where professional organisations stress the necessity of dedicated counselling and informed patient choices. In Australia, clinical laboratories do not routinely offer OGS to those undergoing genomic diagnostic tests [6] and policy makers have identified the need to develop and promote a national, principles-based approach [7]. The European Society of Human Genetics have called for OGS pilot and evaluation studies to support decision-making about OGS [4].
Process evaluation of pilots is essential to inform future implementations of OGS beyond a research context. To date, the majority of reported OGS delivery is the ‘concurrent model’, wherein OGS is offered in the same consultation as diagnostic testing [8,9,10] or at the time of result delivery [11]. This method of service delivery offers system efficiency benefits, with fewer contacts required with clinicians and integrated analysis and reporting for medical scientists. However, it potentially reduces informed decision-making about OGS—in the context of undergoing genetic analysis for a diagnosis, patients may be more likely to be eager for any information without fully understanding the potential implications of AF [12]. Poor recall of OGS decision-making in this model has been reported [13]. Genetic counsellors (GC) report that returning results from diagnostic testing and OGS at the same time is challenging, particularly when both were significant [14], suggesting this be done in separate appointments. The French Society of Predictive and Personalised Medicine [15] suggests performing OGS for cancer predisposition genes simultaneously with diagnostic testing and allowing patients to either confirm or refuse OGS results at the time of result delivery. A pilot of a similar model [16] resulted in several cases where clinically important medical information was known to health professionals that the patient chose not to know.
The process by which OGS is offered and provided is likely to influence outcomes such as patient uptake, knowledge, decisional conflict and regret, and participation in medical follow-up for pathogenic AF. To address the challenges inherent in providing informed consent concurrently for two different genomic analyses with very distinct consequences, we proposed an alternative approach where OGS is both offered and performed after the primary clinical test has been completed and results returned [17]. To evaluate this two-step model, we established a proof-of-concept service. Adults who had previously received the results of clinical exome sequencing from their clinician were offered OGS on stored data for 27 adult-onset conditions. The service provided pre-test genetic counselling to all those who accepted the invitation to learn about the OGS service and post-test genetic counselling to those who proceeded with OGS. We have previously reported our evaluation of resource implications of different service delivery models, drawing on data from this study [18]. Here we report the evaluation of our proof-of-concept service, using mixed methods to explore key elements of delivery including patient uptake, decision-making, understanding, recall and patient and professional perspective on the service and preferences for future models.
Methods
The protocol has been described in detail elsewhere [17]. The following summarises key aspects of the clinical procedures and study methodology. We use OGS to refer to the process and AF for the test result.
Patient participants
Two hundred patients were randomly selected using the approach previously described [17] from a database of patients who received genomic sequencing within clinical care through a Melbourne Genomics Health Alliance funded clinical service design project. Eligibility criteria were: adults over 18 years who had undergone germline exome sequencing for diagnosis of their own clinical condition or as a parent of an affected child (trio testing); consent to be recontacted for further research; English language ability. Potential patient participants received information about OGS and were asked to indicate if they wished to be contacted by a counsellor to learn more. It was emphasised that agreeing to be contacted did NOT oblige them to accept analysis for AF.
Clinical procedures
In-person genetic counselling appointments were arranged for those who expressed interest in knowing more about the OGS service. The research consent form and a decision support tool [17] (Supplementary File 1) were sent prior to the appointment. Pre-test counselling was conducted in genetic services at metropolitan public hospitals in Victoria, Australia, by experienced GCs trained in OGS [17]. Patients who elected to proceed with OGS signed a clinical consent form for reanalysis of their stored genomic data. Genomic data was then analysed by an accredited clinical laboratory for 58 genes known to cause 27 adult-onset conditions with funded management pathways available in the Victorian public healthcare system (Supplementary File 2). OGS results of were discussed at multidisciplinary meetings. Results were returned to patients by a GC either by phone if the patient had a negative or known result, or in person if there was a pathogenic AF.
Data collection
Data were collected from patients, GCs and medical laboratory scientists.
Patient participant data
Patient demographics, initial testing outcome and hypothetical interest in reanalysis for AF were obtained from the primary study database [17]. Uptake of the OGS service, reasons for decision (including reasons for declining) and OGS decision were recorded in a database specific to this study.
Surveys, either electronic or hard copy according to preference, were administered to patient participants at three time points as previously described [17]: survey 1 was administered to all patient participants after pre-test counselling and focused on decision making and understanding; survey 2 was administered only to those who accepted OGS after return of results and focused on understanding of result and decision regret; survey 3 was a short phone survey administered to all patient participants at least 5 months after last contact with a GC and focused on recall and decision regret. Questions on preferences for future service provision were included in S1 and S2.
Professional participant data
Semi-structured interviews were conducted with six of the ten GCs who provided counselling for the OGS service. Interviews focused on their experiences providing OGS to patients, views on workforce support needs and on future delivery of OGS. A focus group was conducted with ten medical laboratory scientists involved in the OGS service exploring the processes used for AF identification and reporting, and perspectives on OGS in the future.
Data analysis
Survey data were captured and managed using REDCap [19, 20] electronic data capture tools hosted at Murdoch Children’s Research Institute. Descriptive analysis of quantitative data was performed with Stata [21], including participation and response statistics, and univariate analysis of demographic features with a Pearson-Chi squared test. Results with a p value less than 0.05 were deemed statistically significant. Qualitative data from GC interviews and the focus group were recorded and transcribed verbatim. Transcripts from GC interviews and the medical scientists focus group and open-ended responses from patient surveys were imported into the qualitative data analysis package NVivo [22]. Transcripts were analysed using an inductive deductive hybrid approach to thematic coding [23]. Deductive coding was used to assign the content of transcripts into pre-determined categories e.g. patient understanding of results; inductive coding was then used to identify emergent concepts within each category. Open-ended survey responses were analysed inductively. All qualitative analyses were synthesised and discussed within the study team.
Results
Participation in the OGS service and evaluation activities is described in Fig. 1. Note that denominators given for survey responses in text vary slightly, due to missing answers.
Uptake of offer to attend the OGS service
Patients offered the opportunity to attend the OGS service were well distributed across socio-demographic variables (income and educational levels) (Supplementary File 3). Of the 200 approached, 83 accepted the offer to attend (42% uptake). There were no significant differences in patient characteristics (age, sex, location, country of birth, education, previous diagnosis, time from completing initial testing to being approached) between those who accepted and those who declined the opportunity to attend the OGS service (Supplementary File 3). Eighty per cent of those who attended the OGS service were undiagnosed after diagnostic genomic testing. Of those who did not attend the service, 51% provided reasons (Fig. 2). Practical considerations e.g. travel time were most commonly cited but concerns about the value or impact of OGS or a preference to deal with medical issues as they arise were also nominated. Thirty-one respondents who declined the OGS offer cited only practical considerations that could be overcome with a true open-ended OGS offer (i.e. only ‘logistics’ or ‘not the right time’). Adding this to the 83 who attended the OGS service gives a theoretical maximum uptake of 57%. Four couples tested as part of a trio opted-in to the service of the 12 couples randomised to be approached. The small number of trio participants mean we are unable to explore differences between this group and adults who’d had diagnostic testing themselves.
Decision-making and uptake of OGS (analysis for AF)
During diagnostic testing, patients were asked about their hypothetical interest in undergoing reanalysis for AF for serious, actionable conditions. Responses available for 146 of the 200 people approached showed 95% (n = 138) expressed hypothetical interest. After genetic counselling, 81 of the 83 patients consented to OGS, giving an actual uptake of reanalysis for AF from the same population of 41% (81/200).
The majority of patients (95%) in survey 1 indicated the level of information they received prior to making a decision about OGS was ‘about right’ (n = 79/83). Over 90% (73/78) reported the pre-test discussion with the GC was helpful. Open comments indicated patients valued the opportunity to ask questions. One patient commented that, due to dyslexia, they prefer discussions to reading. Two commented that the discussion was a formality, as they had made their decision already and two said a phone consultation would be sufficient. Whilst the amount of information in the decision support materials was considered to be ‘about right’ (67/79; 85%), some (21/78, 27%) perceived it to be biased in favour of OGS. Few respondents (3/82, 4%) had high decisional conflict.
Pre-test understanding
Patient survey 1 included six questions to assess understanding of OGS. Most respondents correctly understood the implications of a pathogenic AF for themselves and their family. Patients were asked to indicate using a slider how likely a pathogenic AF result was. Less than a quarter nominated a value of 10% or lower, i.e. within the range regarded as ‘correct’. A third of respondents did not understand that OGS was for a select group of conditions with known treatment or intervention: 23 thought analysis would encompass all known genetic conditions and five thought it related to the initial diagnostic testing.
Return of results—patient and professional perspectives
Of the 81 patients who accepted OGS, six had novel pathogenic AF (Supplementary File 4), all of whom completed survey 2. All survey 2 respondents correctly recalled their AF results. Most respondents with a negative AF result and all with a novel pathogenic AF result understood the implications of their results (Table 1). Five of those with novel pathogenic AF results clearly detailed an appropriate follow-up plan for screening or treatment of themselves and relevant family members, while the remaining respondent indicated they were aware of the pathogenic AF result and that no action was required.
The majority of patient participants who completed Survey 2 indicated they received sufficient information and had no remaining concerns about their result (44/49 90%; 42/48 88%). There were a small number of comments from people with no pathogenic AF found (n = 5) suggesting a phone call did not completely meet their needs. In addition, comments from two people suggested they were still conflating diagnostic testing and OGS: ‘current health problems were not picked up’; ‘I still have no answers’.
GCs when interviewed also expressed concerns about patient understanding that OGS was distinct from diagnostic testing. Three GCs mentioned patients were sometimes aware of the distinction but also saw it as their ‘last chance’ to get an answer. One GC reported some patients were surprised when their results were negative, expecting answers to an existing condition, and ‘still found a lot of people conflating the two’ tests—diagnostic and OGS. One GC reported some patients seemed falsely reassured initially if their results were negative.
‘There were definitely some people who made a bit of an offhand comment like a woman saying, I don’t need to worry about having the testing that my daughter’s having now. We really did have to clarify, actually this doesn’t rule out things.’—GC01
Decision recall and regret
Almost 75% of survey 3 patient respondents immediately remembered being offered OGS five or more months after their last GC contact, and this number rose to 86% after prompting. All patient respondents remembered their decision regarding reanalysis of their data. Two (one without a pathogenic AF and one with a pathogenic AF) had difficulty remembering the outcomes of their testing; the patient who had a pathogenic AF had previously reported being linked-in to appropriate management. Most survey 3 respondents (38/41, 93%) had low decision regret; three respondents had moderate-high decisional regret, one who had not opted-in to OGS and two who did not have pathogenic AF results.
Future OGS service provision
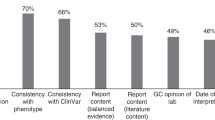
There was no consensus on future provision of an OGS service across participating professional or patient respondents. At the survey 2 timepoint, the majority of patient respondents (86%) believed OGS should be offered in the future but there was no clear preference for when the offer should be made nor how information about OGS should be provided (Fig. 3). Of note, patient preferences changed between the survey 1 and survey 2 timepoints, with more patients preferring a delayed offer of OGS and telehealth provision at survey 2. Drawing on their experience of the pilot service, patients made a number of suggestions for future service delivery. Comments included the need for range of approaches to information delivery (Tables 2 and 3) and support needed, particularly in the context of negative diagnostic and negative OGS results (Table 2).
GC similarly did not express a clear preference for any one timepoint for offering OGS (Table 3). Comments noted offering OGS at the same time as diagnostic testing would be ‘much more cost effective’ and ‘solve the problem’ of ‘people not wanting to come in for multiple rounds of testing’. Offering OGS after diagnostic testing was perceived as less overwhelming for patients, especially those who maybe already dealing with a health crisis. GCs further felt it would allow patients to distinguish between the two tests more clearly. GCs discussed a range of approaches to providing patients with information about OGS, although they primarily raised these as adjuncts to, rather than substitutes for counselling (Table 3).
Medical scientists were undecided about how best to provide OGS, mainly because of resource limitations. They saw the benefits of process efficiency when diagnostic and OGS analyses were conducted together, however, they were adamant that OGS analysis should not be performed at the expense of timely delivery of diagnostic testing (Table 3).
Training needs
Medical scientists and GCs thought that additional training would be necessary ahead of implementation of OGS. Medical scientists discussed the different mindsets required for diagnostic curation versus identification and reporting of AF. Diagnostic curation includes doing ‘your due diligence’ to interpret variants where there are conflicting reports of pathogenicity in the context of a clinical phenotype, whereas identification and reporting of AF should be focused purely on reporting variants of known pathogenicity in people at population risk. Specific training in reporting for OGS or a separate curation workforce were suggested as solutions to this conflict. GCs indicated further training would be valuable in areas such as techniques on challenging a patient’s decision about OGS to elaborate on their values, and the depth of information to share with patients.
Discussion
This study is the first to provide insights into a novel model of OGS service delivery, whereby diagnostic testing and OGS are treated as distinct events and offered asynchronously. Our process evaluation of this proof-of-concept OGS service explores how such a service might be offered in future, including stakeholder perspectives, in the context of existing service infrastructure. The results provide unique insights into several aspects of OGS service delivery that can assist decision-makers to determine which process might be best suited to their objectives and health services.
Firstly, although theoretical interest in OGS was very high (95%), actual uptake by the same patients was considerably lower (42%). This contrasts with studies offering OGS concurrent with diagnostic testing which have reported uptakes ranging from 69 to 94% [9, 16, 24]. Our 42% uptake may reflect the opportunity to more fully consider the implications of OGS. Our novel two-step study design enabled us to explore the reasons why people were NOT interested in learning about the OGS service. The reasons cited by some of those who declined suggested they made a considered decision that aligned with their values. This supports the possibility that the low rate of declining in models where OGS is offered concurrent with diagnostic testing is due to acceptance of OGS without full consideration due to convenience of the concomitant offer. Others who declined our OGS service did cite practical constraints that could theoretically be accommodated with a true two-step model with an open-ended offer, or through provision of telehealth consultations. Even so, speculatively, resolving these barriers would result in a maximum uptake of 57%.
Our results suggest separating the decision about diagnostic testing from the decision about OGS leads to high patient comprehension, illustrated by high recall of participation and understanding of results. Evaluation of concurrent delivery models has shown not all patients participate in follow-up for their pathogenic AF [16, 24]. Patients with novel pathogenic AF identified through our two-step service all reported pursuing appropriate management for their AF. Despite the provision of OGS specific face-to-face genetic counselling and decision support materials, a small group showed persistent misunderstanding of the extent of testing and confusion between diagnostic testing and OGS, although this proportion was lower than reported in studies of concurrent models [13, 25]. Further research is required to understand the reasons for such misunderstandings, as well as strategies to address them.
Patient and professional participants were open to a range of options for future OGS service delivery. In terms of the timing of the offer, this suggests OGS can be offered at whichever time best supports the local health service to meet its priorities. Concurrent offers may result in high uptake; an open-ended offer made after the decision about diagnostic testing would provide patients time to consider OGS. Our study used a resource intensive model of information provision and support which was not intended to be scalable but to offer insights for future service delivery. Most patients wanted to access information in more than one way, stressing the need for a flexible approach, which was also emphasised by participating GCs (Fig. 3, Tables 2, 3). Digital decision aids could make information about OGS accessible to patients at a time that suits them. The Genomics ADvISOR decision aid, for example, has been shown to reduce subsequent counselling time and improve knowledge of analysis for AF [26]. Chatbots are another way to provide flexible access to information about OGS and can support genetic counselling through collecting family history information relevant to the OGS offer [27]. A range of approaches for result delivery were also favoured. Of note, a reduction in preference for face-to-face contact was noted as patients experienced different modes of contact throughout the service. High acceptance of telehealth for return of results has been noted previously [28], as has the association between experience of a model of care and acceptance of it [29]. This study was conducted prior to the significant shift to telehealth caused by the COVID-19 pandemic; it is likely that acceptance of telehealth would now be higher [30]. Many patients in our study indicated they would have been happy to receive negative AF results without GC involvement. However, it is worth noting that 10% of patients without a pathogenic AF in our study wanted further guidance on results or future management. Our study contained a high proportion of people who continue on their ‘diagnostic odyssey’ [31], remaining undiagnosed after diagnostic testing. Complexity has been noted to influence patient preference for mode of service delivery (29). Complexity in our study thus relates not only to type of result (pathogenic AF, no AF) but also to the patient’s diagnostic and medical status. Specific approaches for identifying complex situations and strategies for managing these may be informed by further research and by existing service delivery models or evidence from the reproductive genetic screening setting. Offering a variety of options for information provision and return of results is likely to improve engagement, decision-making and comprehension of OGS and reserve GC availability for patients who require increased levels of assistance, maximising service viability.
Investigating the process of a two-step model for OGS allowed us to explore OGS specifically, separated from diagnostic testing, providing insights into the decision making of those who accepted and those who declined the offer of an OGS service. Though our patient cohort reflects a range of income and education levels, it was restricted to those who spoke English. Decision support materials were provided to patients, but their use and impact were not evaluated. Larger studies than ours are required to investigate outcomes such as health impacts and healthcare costs associated with OGS.
Conclusion
The decision to provide OGS is multi-dimensional. Evidence about the outcomes of OGS is needed to make decisions about whether to implement OGS and outcomes are impacted by how OGS is offered. That is, outcomes are not independent of process. This study is the first to evaluate a novel two-step process for offering OGS, providing insights into decision-making, uptake and delivery specific to OGS.
The predominant model explored to date for OGS is to offer it concurrent with diagnostic testing. This makes sense in research and in user-pays models of healthcare. However, in socialised healthcare systems these distinct decisions do not need to be conflated. Our study demonstrates that separating them in time (enabling OGS-specific genetic counselling) results in relatively low uptake of OGS but a high recall of OGS decision-making and comprehension of results. Nonetheless, the scope of OGS was misunderstood by one third of participants; it remains to be seen if alternative, more scale-able models of information provision can improve knowledge of the OGS offer. The use of alternative models to support decision-making and return of results could also minimise some of the resource implications of separating the OGS decision in time from diagnostic testing. The two-step approach we explored does not provide a solution to all the challenges of delivering OGS; decisions about whether, when and how to offer OGS remain complex and linked to health service resourcing and priorities. Our study does provide evidence that the two-step approach warrants further exploration as an alternative to the status quo whilst highlighting outstanding OGS challenges, in particular how best to meet patient information and support needs while balancing resource requirements.
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Wright DC, Burton DH, Hall A, Moorthie DS, Pokorska-Bocci DA, Sagoo DG, et al. Next steps in the sequence: the implications of whole genome sequencing for health in the UK. PHG Foundation 2011.
Mackley MP, Fletcher B, Parker M, Watkins H, Ormondroyd E. Stakeholder views on secondary findings in whole-genome and whole-exome sequencing: a systematic review of quantitative and qualitative studies. Genet Med. 2017;19:283–93.
ACMG Board of Directors. ACMG policy statement: updated recommendations regarding analysis and reporting of secondary findings in clinical genome-scale sequencing. Genet Med. 2015;17:68–9.
de Wert G, Dondorp W, Clarke A, Dequeker EMC, Cordier C, Deans Z, et al. Opportunistic genomic screening. Recommendations of the European Society of Human Genetics. Eur J Hum Genet. 2021;29:365–77.
Boycott K, Hartley T, Adam S, Bernier F, Chong K, Fernandez BA, et al. The clinical application of genome-wide sequencing for monogenic diseases in Canada: Position Statement of the Canadian College of Medical Geneticists. J Med Genet. 2015;52:431–7.
Vears DF, Borry P, Savulescu J, Koplin JJ. Old challenges or new issues? genetic health professionals’ experiences obtaining informed consent in diagnostic genomic sequencing. AJOB Empir Bioeth. 2021;12:12–23.
Australian Government Department of Health and Aged Care. National Health Genomics Policy Framework 2018–2021—Implementation plan. 2018. https://www.health.gov.au/resources/publications/national-health-genomics-policy-framework-2018-2021-implementation-plan?language=en.
Bowdin SC, Hayeems RZ, Monfared N, Cohn RD, Meyn MS. The SickKids genome clinic: developing and evaluating a pediatric model for individualized genomic medicine. Clin Genet. 2016;89:10–9.
Shahmirzadi L, Chao EC, Palmaer E, Parra MC, Tang S, Gonzalez KDF. Patient decisions for disclosure of secondary findings among the first 200 individuals undergoing clinical diagnostic exome sequencing. Genet Med. 2014;16:395–9.
Genomics England. Additional findings. https://www.genomicsengland.co.uk/initiatives/100000-genomes-project/additional-findings.
Roche MI, Griesemer I, Khan CM, Moore E, Lin FC, O’Daniel JM, et al. Factors influencing NCGENES research participants’ requests for non-medically actionable secondary findings. Genet Med. 2019;21:1092–9.
Bergner AL, Bollinger J, Raraigh KS, Tichnell C, Murray B, Blout CL, et al. Informed consent for exome sequencing research in families with genetic disease: the emerging issue of incidental findings. Am J Med Genet A. 2014;164A:2745–52.
Ballard LM, Horton RH, Dheensa S, Fenwick A, Lucassen AM. Exploring broad consent in the context of the 100,000 Genomes Project: a mixed methods study. Eur J Hum Genet. 2020;28:732–41.
Rost C, Dent KM, Botkin J, Rothwell E. Experiences and lessons learned by genetic counselors in returning secondary genetic findings to patients. J Genet Couns. 2020;29:1234–44.
Pujol P, Vande Perre P, Faivre L, Sanlaville D, Corsini C, Baertschi B, et al. Guidelines for reporting secondary findings of genome sequencing in cancer genes: the SFMPP recommendations. Eur J Hum Genet. 2018;26:1732–42.
Horiuchi Y, Matsubayashi H, Kiyozumi Y, Nishimura S, Higashigawa S, Kado N, et al. Disclosure of secondary findings in exome sequencing of 2480 Japanese cancer patients. Hum Genet. 2021;140:321–31.
Martyn M, Kanga-Parabia A, Lynch E, James PA, Macciocca I, Trainer AH, et al. A novel approach to offering additional genomic findings—a protocol to test a two-step approach in the healthcare system. J Genet Couns. 2019;28:388–97.
Vu M, Degeling K, Martyn M, Lynch E, Chong B, Gaff C, et al. Evaluating the resource implications of different service delivery models for offering additional genomic findings. Genet Med. 2021;23:606–13.
Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O’Neal L, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–81.
StataCorp LLC. Stata statistical software. College Station, TX; 2017.
QSR International. NVivo. 2020. https://www.qsrinternational.com/nvivo-qualitative-data-analysis-software/home.
Fereday J, Muir-Cochrane E. Demonstrating rigor using thematic analysis: a hybrid approach of inductive and deductive coding and theme development. Int J Qual Methods. 2006;5:80–92.
Bishop C, Strong K, Dimmock D. Choices of incidental findings of individuals undergoing genome wide sequencing, a single center’s experience. Clin Genet. 2017;91:137–40.
Sapp JC, Johnston JJ, Driscoll K, Heidlebaugh AR, Miren Sagardia A, Dogbe DN, et al. Evaluation of recipients of positive and negative secondary findings evaluations in a hybrid CLIA-research sequencing pilot. Am J Hum Genet. 2018;103:358–66.
Bombard Y, Clausen M, Mighton C, Carlsson L, Casalino S, Glogowski E, et al. The Genomics ADvISER: development and usability testing of a decision aid for the selection of incidental sequencing results. Eur J Hum Genet. 2018;26:984–95.
Ireland D, Bradford D, Lynch E, Martyn M, Hansen D, Gaff C. Edna: how can a chatbot augment genomic counselling for Additional Findings? 2020. http://hdl.handle.net/102.100.100/368370?index=1.
Ebbert JO, Ramar P, Tulledge-Scheitel SM, Njeru JW, Rosedahl JK, Roellinger D, et al. Patient preferences for telehealth services in a large multispecialty practice. J Telemed Telecare. 2023;29:298–303.
Listorti E, Ferrara L, Adinolfi A, Gerardi MC, Ughi N, Tozzi VD, et al. Joining telehealth in rheumatology: a survey on the role played by personalized experience from patients’ perspective. BMC Health Serv Res. 2023;23:682.
Banbury A, Smith AC, Taylor ML, Der Vartanian C, Ng K, Vitangcol K, et al. Cancer care and management during COVID-19: a comparison of in-person, video and telephone consultations. J Telemed Telecare. 2022;28:733–9.
Macnamara EF, Schoch K, Kelley EG, Fieg E, Brokamp E, Undiagnosed Diseases Network. et al. Cases from the undiagnosed diseases network: the continued value of counseling skills in a new genomic era. J Genet Couns. 2019;28:194–201.
Acknowledgements
We would like to thank Kelly Ormond for co-facilitating the OGS training workshop provided to participating genetic counsellors. The authors would like to thank the healthcare and laboratory professionals who were critical to design and implementation of the service and all of the participants—professional and patient—for their time and valuable insights into their experiences with this service.
Funding
This study was funded by the State Government of Victoria (Department of Health and Human Services) and the ten member organisations of the Melbourne Genomics Health Alliance. Work undertaken at the Murdoch Children’s Research Institute was supported by the Victorian Government’s Operational Infrastructure Support Program. Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
EL and CG were responsible for conception and design of the OGS service and were involved in interpreting results. MM and CG were responsible for conception and design of the evaluation. AKP was involved in design of the evaluation and data collection materials. MM, LL and RW were involved in acquisition of study data and in data analysis and interpretation. AJ was involved in second checking of data analysis and in synthesis of results. All authors were involved in drafting the paper and have read and approved the final paper and agree to be accountable to all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Ethical approval for the pilot and evaluation activities was obtained from Melbourne Health Human Research Ethics Committee (13/MH/326). A focus group with medical laboratory scientific staff was approved by the Royal Children’s Hospital Human Research Ethics Committee (QA/66211/RCHM-2020).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martyn, M., Lee, L., Jan, A. et al. Evaluation of a two-step model of opportunistic genomic screening. Eur J Hum Genet 32, 656–664 (2024). https://doi.org/10.1038/s41431-024-01592-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41431-024-01592-0
This article is cited by
-
What’s new in EJHG in June 2024?
European Journal of Human Genetics (2024)