Abstract
Background
Maternal selenium (Se) deficiency is associated with some adverse pregnant outcomes. However, it remains controversial whether maternal Se deficiency during gestation enhances the risks for low-birth-weight (LBW) and small-for-gestational-age (SGA) newborns.
Methods
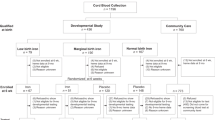
For our cohort study, total 3133 mother-and-infant pairs were selected. Maternal serum Se concentration was detected by graphite furnace atomic absorption spectrometry. According to international references for maternal serum Se concentration, subjects were divided into Se deficiency (<45.0 μg/L), Se insufficiency (45.0-94.9 μg/L) and Se sufficiency (≥95.0 μg/L).
Results
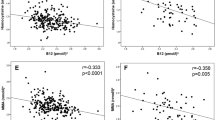
There was a positive relation of maternal serum Se concentration in gestation and neonatal birth weight. Further analysis showed that the risks for LBW and SGA in SD group were significantly higher than that in SI and SS group, the adjusted ORs for LBW and SGA newborns were 1.87 (95%CI: 1.02, 3.45; P = 0.04) and 1.47 (95%CI: 1.07, 2.02; P = 0.02) in SI group, and 3.92 (95%CI: 2.03, 7.57; P < 0.001) and 2.77 (95%CI: 1.92, 4.02; P < 0.001) in SD group compared to SS group. In different gender subgroup, positive relations were observed between maternal Se deficiency and the risk for LBW girls, as well as the risks for both SGA girls and boys.
Conclusion
Maternal Se deficiency in gestation was positively associated with the risk for LBW girls, as well as the risks for both SGA girls and boys.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fairweather-Tait SJ, Bao Y, Broadley MR, Collings R, Ford D, Hesketh JE, et al. Selenium in human health and disease. Antioxid Redox Signal. 2011;14:1337–83.
Rayman MP. The importance of selenium to human health. Lancet. 2000;356:233–41.
Branco V, Godinho-Santos A, Gonçalves J, Lu J, Holmgren A, Carvalho C. Mitochondrial thioredoxin reductase inhibition, selenium status, and Nrf-2 activation are determinant factors modulating the toxicity of mercury compounds. Free Radic Biol Med. 2014;73:95–105.
Schomburg L. Selenium, selenoproteins and the thyroid gland: interactions in health and disease. Nat Rev Endocrinol. 2011;8:160–71.
Ambroziak U, Hybsier S, Shahnazaryan U, Krasnodębska-Kiljańska M, Rijntjes E, Bartoszewicz Z, et al. Severe selenium deficits in pregnant women irrespective of autoimmune thyroid disease in an area with marginal selenium intake. J Trace Elem Med Biol. 2017;44:186–91.
Yang L, Zhao GH, Yu FF, Zhang RQ, Guo X. Selenium and iodine levels in subjects with kashin-Beck disease: a meta-analysis. Biol Trace Elem Res. 2016;170:43–54.
Yang J, Wang T, Wu C, Liu C. Selenium level surveillance for the year 2007 of Keshan disease in Endemic Areas and analysis on surveillance results between 2003 and 2007. Biol Trace Elem Res. 2010;138:53–59.
Wu Q, Rayman MP, Lv H, Schomburg L, Cui B, Gao C, et al. Low population selenium status is associated with increased prevalence of thyroid disease. J Clin Endocr Metab. 2015;100:4037–47.
Türk S, Mändar R, Mahlapuu R, Viitak A, Punab M, Kullisaar T. Male infertility: decreased levels of selenium, zinc and antioxidants. J Trace Elem Med Biol. 2014;28:179–85.
Han YM, Yoon H, Lim S, Sung MK, Shin CM, Park YS, et al. Risk factors for vitamin D, zinc, and selenium deficiencies in Korean patients with inflammatory bowel disease. Gut Liver. 2017;11:363–9.
Sandsveden M, Manjer J. Selenium and breast cancer risk: a prospective nested case-control study on serum selenium levels, smoking habits and overweight. Int J Cancer. 2017;141:1741–50.
Kristal AR, Darke AK, Morris JS, Tangen CM, Goodman PJ, Thompson IM, et al. Baseline selenium status and effects of selenium and vitamin e supplementation on prostate cancer risk. J Natl Cancer Inst. 2014;106:djt456.
Benstoem C, Goetzenich A, Kraemer S, Borosch S, Manzanares W, Hardy G, et al. Selenium and its supplementation in cardiovascular disease-what do we know? Nutrients. 2015;7:3094–118.
Mariath AB, Bergamaschi DP, Rondó PH, Tanaka AC, Hinnig Pde F, Abbade JF, et al. The possible role of selenium status in adverse pregnancy outcomes. Br J Nutr. 2011;105:1418–28.
Vanderlelie J, Perkins AV. Selenium and preeclampsia: a global perspective. Pregnancy Hypertens. 2011;1:213–24.
Kong FJ, Ma LL, Chen SP, Li G, Zhou JQ. Serum selenium level and gestational diabetes mellitus: a systematic review and meta-analysis. Nutr J. 2016;15:94.
Unterscheider J, O’Donoghue K, Daly S, Geary MP, Kennelly MM, McAuliffe FM, et al. Fetal growth restriction and the risk of perinatal mortality-case studies from the multicentre PORTO study. BMC Pregnancy Childbirth. 2014;14:63.
Simeoni U, Armengaud JB, Siddeek B, Tolsa JF. Perinatal origins of adult disease. Neonatology. 2018;113:393–9.
Everson TM, Kappil M, Hao K, Jackson BP, Punshon T, Karagas MR, et al. Maternal exposure to selenium and cadmium, fetal growth, and placental expression of steroidogenic and apoptotic genes. Environ Res. 2017;158:233–44.
Mistry HD, Kurlak LO, Young SD, Briley AL, Pipkin FB, Baker PN, et al. Maternal selenium, copper and zinc concentrations in pregnancy associated with small-for-gestational-age infants. Matern Child Nutr. 2014;10:327–34.
Nazemi L, Shariat M, Chamari M, Chahardoli R, Asgarzadeh L, Seighali F. Comparison of maternal and umbilical cord blood selenium levels in low and normal birth weight neonates. J Family Reprod Health. 2015;9:125–8.
Tao FB, Hao JH, Huang K, Su PY, Cheng DJ, Xing XY, et al. Cohort profile: the China-Anhui birth cohort study. Int J Epidemiol. 2013;42:709–21.
Khalili H, Soudbakhsh A, Hajiabdolbaghi M, Dashti-Khavidaki S, Poorzare A, Saeedi AA, et al. Nutritional status and serum zinc and selenium levels in Iranian HIV infected individuals. BMC Infect Dis. 2008;8:165.
Hashemi SM, Mashhadi M, Moghaddam AA. The relationship between serum selenium and zinc with gastroesophageal cancers in the Southeast of Iran. Indian J Med Paediatr Oncol. 2017;38:169–72.
Mikolajczyk RT, Zhang J, Betran AP, Souza JP, Mori R, Gülmezoglu AM, et al. A global reference for fetal-weight and birthweight percentiles. Lancet. 2011;377:1855–61.
Lee AC, Katz J, Blencowe H, Cousens S, Kozuki N, Vogel JP, et al. National and regional estimates of term and preterm babies born small for gestational age in 138 low-income and middle-income countries in 2010. Lancet Global Health. 2013;1:e26–e36.
Lewandowska M, Sajdak S. The role of early pregnancy maternal selenium levels on the risk for small-for-gestational age newborns. Nutrients. 2019;11:2298.
Khaled Fahim N. Sample size calculation guide—Part 2: How to calculate the sample size for an independent cohort study. Adv J Emerg Med. 2019;3:e12.
Luo BF, Rao HY, Gao YH. Risk factors for familial clustering of hepatitis C virus infection in a Chinese Han population: a cross-sectional study. BMC Public Health. 2018;18:708.
Aggarwal R, Gathwala G, Yadav S. Selenium supplementation for prevention of Late-Onset Sepsis in very low birth weight preterm neonates. J Trop Pediatr. 2016;62:185–93.
Bogden JD, Kemp FW, Chen X, Stagnarogreen A, Stein TP, Scholl TO. Low-normal serum selenium early in human pregnancy predicts lower birth weight. FASEB J. 2007;21:A717.
Alehagen U, Johansson P, Aaseth J, Alexander J, Brismar K. Increase in insulin-like growth factor 1 (IGF-1) and insulin-like growth factor binding protein 1 after supplementation with selenium and coenzyme Q10. A prospective randomized double-blind placebo-controlled trial among elderly Swedish citizens. PLoS ONE. 2017;12:e0178614.
Walenkamp MJ, Losekoot M, Wit JM. Molecular IGF-1 and IGF-1 receptor defects: from genetics to clinical management. Endocr Dev. 2013;24:128–37.
Gohlke BC, Stutte S, Bartmann P, Woelfle J. Does gender-specific BMI development modulate insulin sensitivity in extremely low birth weight infants? J Pediatr Endocrinol Metab. 2009;22:827–35.
Asemi Z, Jamilian M, Mesdaghinia E, Esmaillzadeh A. Effects of selenium supplementation on glucose homeostasis, inflammation, and oxidative stress in gestational diabetes: randomized, double-blind, placebo-controlled trial. Nutrition. 2015;31:1235–42.
Khera A, Vanderlelie JJ, Perkins AV. Selenium supplementation protects trophoblast cells from mitochondrial oxidative stress. Placenta. 2013;34:594–8.
Watson M, van Leer L, Vanderlelie JJ, Perkins AV. Selenium supplementation protects trophoblast cells from oxidative stress. Placenta. 2012;33:1012–9.
Datt C, Chhabra A. Blood selenium level and glutathione peroxidase activity as affected by high level of selenium supplementation in cattle and buffaloes: a comparative study. Indian J Anim Sci. 2004;74:872–7.
Mechri A, Mekhinini A, Othman LB, Fendri C, Gaha L, Kerkeni A. Relationship between glutathione peroxidase activity, glutathione and selenium levels in schizophrenic patients. European Neuropsychopharmacol. 2006;16:S394–5.
Ghneim HK, Alshebly MM. Biochemical markers of oxidative stress in Saudi women with recurrent miscarriage. J Korean Med Sci. 2016;31:98–105.
Motawei SM, Attalla SM, Gouda HE, Harouny MA, Elmansoury AM. The effects of N-acetyl cysteine on oxidative stress among patients with pre-eclampsia. Int J Gynecol Obstet. 2016;135:226–7.
Yoshida A, Watanabe K, Iwasaki A, Kimura C, Matsushita H, Wakatsuki A. Placental oxidative stress and maternal endothelial function in pregnant women with normotensive fetal growth restriction. J Matern Fetal Neonatal Med. 2018;31:1051–7.
Biberoglu E, Biberoglu K, Kirbas A, Daglar K, Genc M, Avci A, et al. Circulating and myometrial markers of oxidative stress in pregnant women with fetal growth restriction. J Obstet Gynaecol Res. 2016;42:29–35.
Rueangdetnarong H, Sekararithi R, Jaiwongkam T, Kumfu S, Chattipakorn N, Tongsong T, et al. Comparisons of the oxidative stress biomarkers levels in gestational diabetes mellitus (GDM) and non-GDM among Thai population: cohort study. Endocr Connect. 2018;7:681–7.
Ferguson KK, Chen YH, VanderWeele TJ, McElrath TF, Meeker JD, Mukherjee B. Mediation of the relationship between maternal phthalate exposure and preterm birth by oxidative stress with repeated measurements across pregnancy. Environ Health Perspect. 2017;125:488–94.
Wu F, Tian FJ, Lin Y, Xu WM. Oxidative stress: placenta function and dysfunction. Am J Reprod Immunol. 2016;76:258–71.
Hu C, Yang Y, Li J, Wang H, Cheng C, Yang L, et al. Maternal diet-induced obesity compromises oxidative stress status and angiogenesis in the porcine placenta by up-regulating Nox2 expression. Oxid Med Cell Longev. 2019;2019:2481592.
Gveric-Ahmetasevic S, Sunjic SB, Skala H, Andrisic L, Stroser M, Zarkovic K, et al. Oxidative stress in small-for-gestational age (SGA) term newborns and their mothers. Free Radic Res. 2009;43:376–84.
Funding
This work was supported by NSFC (81473016 and 81630084), Key Projects of outstanding Youth Talent Support Program in Anhui Provincial University (gxyqZD2016056), and Young Scholars of Wan Jiang in Anhui Province, and National Key Technology R&D Program (2006BAI05A03).
Author information
Authors and Affiliations
Contributions
HW and F-BT designed the research; XZ, Y-JF, and JL performed as measurement, HW, XZ, J-HH, and PZ analyzed data; HW wrote the manuscript; D-XX revised the manuscript. All authors have approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors disclose no conflicts of interest. The funders had no role in the research design, implementation, analysis or interpretation of the data.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, X., Feng, YJ., Li, J. et al. Maternal selenium deficiency during gestation is positively associated with the risks for LBW and SGA newborns in a Chinese population. Eur J Clin Nutr 75, 768–774 (2021). https://doi.org/10.1038/s41430-020-00809-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-020-00809-x