Abstract
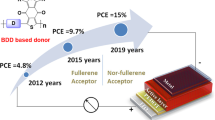
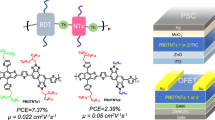
Medium-bandgap polymer donors (such as PBDB-T) and their derivatives are promising donor materials for organic photovoltaics (OPVs). In this study, we aimed to develop a facile synthetic approach for PBDB-T using a C–H/C–I direct arylation reaction in monomer synthesis and C–H/C–Br direct arylation for polymerization. Polycondensation and end-capping methods using direct arylation reactions have been established, enabling the synthesis of polymers without Br termini. The OPV performance of the synthesized polymer was evaluated and compared to that of a benchmark polymer. The synthesized polymer exhibited lower OPV performance than the benchmark polymer despite having similar molecular weights. Possible factors contributing to low OPV performance and future challenges include structural defects, impurities, and large molecular weight distribution.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Grimsdale AC, Chan KL, Martin RE, Jokisz PG, Holmes AB. Synthesis of light-emitting conjugated polymers for applications in electroluminescent devices. Chem Rev. 2009;109:897–1091.
Jiang T, Liu Y, Ren Z, Yan S. The design, synthesis and performance of thermally activated delayed fluorescence macromolecules. Polym Chem. 2020;11:1555–71.
Beaujuge PM, Fréchet JMJ. Molecular design and ordering effects in π-functional materials for transistor and solar cell applications. J Am Chem Soc. 2011;133:20009–29.
Takimiya K, Nakano M. Thiophene-fused naphthalene diimides: New building blocks for electron deficient p-functional materials. Bull Chem Soc Jpn. 2018;91:121–40.
Iino H, Hanna JI. Liquid crystalline organic semiconductors for organic transistor applications. Polym J. 2017;49:23–30.
Liu C, Wang K, Gong X, Heeger AJ. Low bandgap semiconducting polymers for polymeric photovoltaics. Chem Soc Rev. 2016;45:4825–46.
Holliday S, Li Y, Luscombe CK. Recent advances in high performance donor-acceptor polymers for organic photovoltaics. Prog Polym Sci. 2017;70:34–51.
Saito M, Ohkita H, Osaka I. π-Conjugated polymers and molecules enabling small photon energy loss simultaneously with high efficiency in organic photovoltaics. J Mater Chem A. 2020;8:20213–37.
Sun L, Xu X, Song S, Zhang Y, Miao C, Liu X, et al. Medium‐Bandgap conjugated polymer donors for organic photovoltaics. Macromol Rapid Commun. 2019;40:1900074.
Qian D, Ye L, Zhang M, Liang Y, Li L, Huang Y, et al. Design, application, and morphology study of a new photovoltaic polymer with strong aggregation in solution state. Macromolecules. 2012;45:9611–7.
Zhang S, Qin Y, Zhu J, Hou J. Over 14% efficiency in polymer solar cells enabled by a chlorinated polymer donor. Adv Mater. 2018;30:1800868.
Armin A, Li W, Sandberg OJ, Xiao Z, Ding L, Nelson J, et al. A history and perspective of non‐fullerene electron acceptors for organic solar cells. Adv Energy Mater. 2021;11:2003570.
Zheng Z, Yao H, Ye L, Xu Y, Zhang S, Hou J. PBDB-T and its derivatives: A family of polymer donors enables over 17% efficiency in organic photovoltaics. Mater Today. 2020;35:115–30.
Yuan J, Huang T, Cheng P, Zou Y, Zhang H, Yang JL. et al. Enabling low voltage losses and high photocurrent in fullerene-free organic photovoltaics. Nat Commun. 2019;10:570.
Kuwabara J. Direct arylation polycondensation for synthesis of optoelectronic materials. Polym J. 2018;50:1099–106.
Mainville M, Leclerc M. Direct (Hetero)arylation: a tool for low-cost and eco-friendly organic photovoltaics. ACS Appl Polym Mater. 2021;3:2–13.
Kuwabara J, Kanbara T. Step‐economical synthesis of conjugated polymer materials composed of three components: donor, acceptor, and π units. Macromol Rapid Commun. 2021;42:2000493.
Sakamoto J, Rehahn M, Wegner G, Schlüter AD. Suzuki polycondensation: Polyarylenes à la carte. Macromol Rapid Commun. 2009;30:653–87.
Carsten B, He F, Son HJ, Xu T, Yu L. Stille polycondensation for synthesis of functional materials. Chem Rev. 2011;111:1493–528.
Satoh T, Miura M. Catalytic direct arylation of heteroaromatic compounds. Chem Lett. 2007;36:200–5.
Ackermann L, Vicente R, Kapdi AR. Transition-metal-catalyzed direct arylation of (hetero)arenes by C-H bond cleavage. Angew Chem, Int Ed. 2009;48:9792–827.
Liégault B, Lapointe D, Caron L, Vlassova A, Fagnou K. Establishment of broadly applicable reaction conditions for the palladium-catalyzed direct arylation of heteroatom-containing aromatic compounds. J Org Chem. 2009;74:1826–34.
Rossi R, Bellina F, Lessi M, Manzini C. Cross-coupling of heteroarenes by C-H functionalization: Recent progress towards direct arylation and heteroarylation reactions involving heteroarenes containing one heteroatom. Adv Synth Catal. 2014;356:17–117.
Segawa Y, Maekawa T, Itami K. Synthesis of extended π-systems through C-H activation. Angew Chem Int Ed. 2015;54:66–81.
Po R, Bernardi A, Calabrese A, Carbonera C, Corso G, Pellegrino A. From lab to fab: how must the polymer solar cell materials design change?—an industrial perspective. Energy Environ Sci. 2014;7:925–43.
Po R, Bianchi G, Carbonera C, Pellegrino A. ‘All that glisters is not gold’: An analysis of the synthetic complexity of efficient polymer donors for polymer solar cells. Macromolecules. 2015;48:453–61.
Wang Q, Takita R, Kikuzaki Y, Ozawa F. Palladium-catalyzed dehydrohalogenative polycondensation of 2-bromo-3-hexylthiophene: An efficient approach to head-to-tail poly(3-hexylthiophene). J Am Chem Soc. 2010;132:11420–1.
Lu W, Kuwabara J, Kanbara T. Polycondensation of dibromofluorene analogues with tetrafluorobenzene via direct arylation. Macromolecules. 2011;44:1252–5.
Matsidik R, Komber H, Luzio A, Caironi M, Sommer M. Defect-free naphthalene diimide bithiophene copolymers with controlled molar mass and high performance via direct arylation polycondensation. J Am Chem Soc. 2015;137:6705–11.
Wakioka M, Ozawa F. Highly efficient catalysts for direct arylation polymerization (DArP). Asian J Org Chem. 2018;7:1206–16.
Kuwabara J, Kanbara T. Facile synthesisof π-conjugated polymers via direct arylation polycondensation. Bull Chem Soc Jpn. 2019;92:152–61.
Leclerc M, Brassard S, Beaupré S. Direct (hetero)arylation polymerization: toward defect-free conjugated polymers. Polym J. 2020;52:13–20.
Michelin RA, Zanotto L, Braga D, Sabatino P, Angelici RJ. Transition-metal-promoted cyclization reactions of isocyanide ligands. Synthesis of cyclic aminooxycarbene complexes of platinum(II) and x-ray structure of trans-[(PPh3)2Pt[CN(C6H4Me-p)CH2CH2O]Br]BF4. Inorg Chem. 1988;27:85–92.
Braunecker WA, Oosterhout SD, Owczarczyk ZR, Larsen RE, Larson BW, Ginley DS, et al. Ethynylene-linked donor–acceptor alternating copolymers. Macromolecules. 2013;46:3367–75.
Xu S, Zhou Z, Fan H, Ren L, Liu F, Zhu X, et al. An electron-rich 2-alkylthieno[3,4-b]thiophene building block with excellent electronic and morphological tunability for high-performance small-molecule solar cells. J Mater Chem A. 2016;4:17354–62.
MacDowell DWH, Wisowaty JC. Thiophene analogs of anthraquinone. J Org Chem. 1972;37:1712–7.
Faradhiyani A, Zhang Q, Maruyama K, Kuwabara J, Yasuda T, Kanbara T. Synthesis of bithiazole-based semiconducting polymers via Cu-catalysed aerobic oxidative coupling. Mater Chem Front. 2018;2:1306–9.
Kuwabara J, Tsuchida W, Guo S, Hu Z, Yasuda T, Kanbara T. Synthesis of conjugated polymers via direct C–H/C–Cl coupling reactions using a Pd/Cu binary catalytic system. Polym Chem. 2019;10:2298–304.
Kobayashi K, Sugie A, Takahashi M, Masui K, Mori A. Palladium-catalyzed coupling reactions of bromothiophenes at the C−H bond adjacent to the sulfur atom with a new activator system, AgNO3/KF. Org Lett. 2005;7:5083–5.
Mori A, Sugie A. Palladium-catalyzed CH arylation and dehydrogenative homocoupling of heteroaromatic compounds and application to the design of advanced organic materials. Bull Chem Soc Jpn. 2008;81:548–61.
Kuwabara J, Nohara Y, Choi SJ, Fujinami Y, Lu W, Yoshimura K, et al. Direct arylation polycondensation for the synthesis of bithiophene-based alternating copolymers. Polym Chem. 2013;4:947–53.
Kuwabara J, Fujie Y, Maruyama K, Yasuda T, Kanbara T. Suppression of homocoupling side reactions in direct arylation polycondensation for producing high performance OPV materials. Macromolecules. 2016;49:9388–95.
Fujinami Y, Kuwabara J, Lu W, Hayashi H, Kanbara T. Synthesis of thiophene- and bithiophene-based alternating copolymers via Pd-catalyzed direct C-H arylation. ACS Macro Lett. 2012;1:67–70.
Iizuka E, Wakioka M, Ozawa F. Mixed-ligand approach to palladium-catalyzed direct arylation polymerization: Synthesis of donor-acceptor polymers with dithienosilole (DTS) and thienopyrroledione (TPD) units. Macromolecules. 2015;48:2989–93.
Wakioka M, Morita H, Ichihara N, Saito M, Osaka I, Ozawa F. Mixed-ligand approach to palladium-catalyzed direct arylation polymerization: synthesis of donor–acceptor polymers containing unsubstituted bithiophene units. Macromolecules. 2020;53:158–64.
Kim Y, Cook S, Kirkpatrick J, Nelson J, Durrant JR, Bradley DDC, et al. Effect of the end group of regioregular poly(3-hexylthiophene) polymers on the performance of polymer/fullerene solar cells. J Phys Chem C. 2007;111:8137–41.
Park JK, Jo J, Seo JH, Moon JS, Park YD, Lee K, et al. End-capping effect of a narrow bandgap conjugated polymer on bulk heterojunction solar cells. Adv Mater. 2011;23:2430–5.
Kuwabara J, Yasuda T, Takase N, Kanbara T. Effects of the terminal structure, purity, and molecular weight of an amorphous conjugated polymer on its photovoltaic characteristics. ACS Appl Mater Interfaces. 2016;8:1752–8.
Asai H, Xue D, Kamiya S, Kuwabara J, Kanbara T, Marumoto K. Direct evidence of the internal deterioration mechanism due to molecular chain ends in polymer solar cells by operando spin detection. ACS Appl Polym Mater. 2022;4:607–17.
Tong M, Cho S, Rogers JT, Schmidt K, Hsu BBY, Moses D, et al. Higher molecular weight leads to improved photoresponsivity, charge transport and interfacial ordering in a narrow bandgap semiconducting polymer. Adv Funct Mater. 2010;20:3959–65.
Osaka I, Saito M, Mori H, Koganezawa T, Takimiya K. Drastic change of molecular orientation in a thiazolothiazole copolymer by molecular-weight control and blending with PC61BM leads to high efficiencies in solar cells. Adv Mater. 2012;24:425–30.
Ashraf RS, Schroeder BC, Bronstein HA, Huang Z, Thomas S, Kline RJ, et al. The influence of polymer purification on photovoltaic device performance of a series of indacenodithiophene donor polymers. Adv Mater. 2013;25:2029–34.
Acknowledgements
The authors thank the Chemical Analysis Center of University of Tsukuba for the measurements of NMR and MALDI-TOF-MS spectra. The authors also thank Prof. Y. Nishihara and Prof. H. Mori of Okayama University for the measurement of high-temperature GPC. This work was partly supported by the SEI GROUP CSR Foundation and JST A-STEP Grant Number JPMJTM20BT.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kuwabara, J., Hiyaji, K., Guo, S. et al. Direct arylation polycondensation for the synthesis of medium-bandgap polymer donors (PBDB-T) for organic photovoltaics. Polym J 55, 395–404 (2023). https://doi.org/10.1038/s41428-022-00712-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-022-00712-1