Abstract
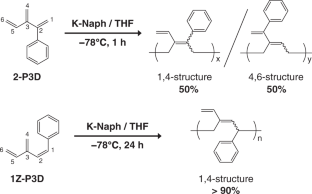
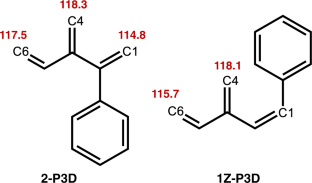
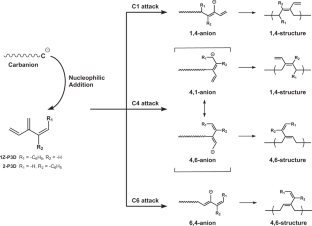
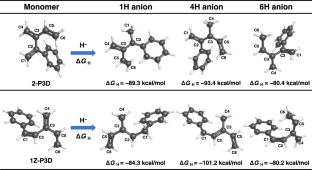
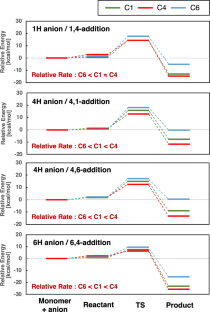
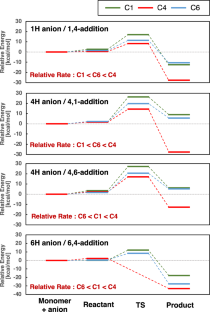
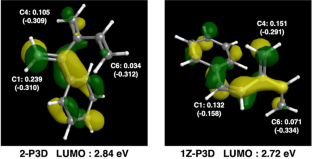
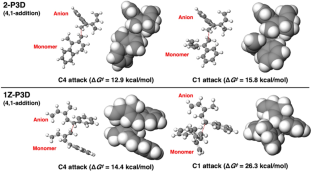
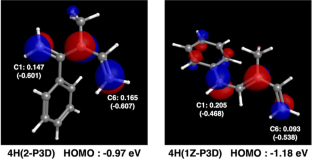
A theoretical study on the anionic polymerization of two phenyl-substituted [3]dendralene derivatives, 2-phenyl[3]dendralene (2-P3D) and (Z)-1-phenyl[3]dendralene (1Z-P3D), was performed using density functional theory (DFT) calculations with ωB97X/6-31 + G(d). The mechanisms of their anionic polymerizations were discussed based on kinetic analysis for chain propagation. In anionic polymerization, 2-P3D and 1Z-P3D were observed to undergo nucleophilic addition exclusively at the C4 position of the [3]dendralene framework, producing 4,1- and 4,6-anions. In the chain propagation of 2-P3D, the 4,1- and 4,6-anions can undergo conjugate addition with comparable activation Gibbs free energies. In contrast, in the chain propagation of 1Z-P3D, the activation Gibbs free energy of the 4,1-addition was much lower than that of the 4,6-addition, indicating that the 4,1-addition was kinetically favorable in the polymerization. These results supported the regioselectivities observed in the anionic polymerizations of 2-P3D and 1Z-P3D. Conformational and molecular orbital analysis showed that the chain end carbanion derived from 1Z-P3D was effectively conjugated with the phenyl substituent; however, that derived from 2-P3D did not. Therefore, the anionic polymerization of phenyl-substituted [3]dendralene derivatives was observed to be strongly influenced by the conjugated structure of the monomer because the position of the phenyl substituent strongly affects the electronic properties of the chain end carbanion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Payne AD, Bojase G, Paddon-Row MN, Sherburn MS. Practical synthesis of the dendralene family reveals alternation in behavior. Angew Chem - Int Ed. 2009;48:4836–9.
Hopf H, Sherburn MS. Dendralenes branch out: cross-conjugated oligoenes allow the rapid generation of molecular. Complex Angew Chem Int Ed. 2012;51:2298–338.
Phelan NF, Orchin M. Cross conjugation. J Chem Educ. 1968;45:633–7.
Limacher PA, Lüthi HP. Cross-conjugation. Wiley Interdiscip Rev Comput Mol Sci. 2011;1:477–86.
Bruschi M, Giuffreda MG, Lüthi HP. Trans versus geminal electron delocalization in tetra- and diethynylethenes: A new method of analysis. Chem - A Eur J. 2002;8:4216–27.
Hosoya H. Cross-conjugation at the heart of understanding the electronic theory of organic chemistry. Curr Org Chem. 2015;19:293–310.
Gu J, Wu W, Stuyver T, Danovich D, Hoffmann R, Tsuji Y, Shaik S. Cross conjugation in polyenes and related hydrocarbons: what can be learned from valence bond theory about single-molecule conductance? J Am Chem Soc. 2019;141(14):6030–47.
Bradford TA, Payne AD, Willis AC, Paddon-Row MN, Sherburn MS. Cross-coupling for cross-conjugation: Practical synthesis and Diels-Alder reactions of [3]dendralenes. Org Lett 2007;9:4861–4.
Toombs-Ruane H, Pearson EL, Paddon-Row MN, Sherburn MS. On the Diels–Alder dimerisation of cross-conjugated trienes. Chem Commun. 2021;48:6639.
Takenaka K, Amamoto S, Kishi H, Takeshita H, Miya M, Shiomi T. Anionic polymerization of 2-phenyl[3]dendralene and 2-(4-methoxyphenyl)[3] dendralene. Macromolecules. 2013;46:7282–9.
Takamura Y, Takenaka K, Toda T, Takeshita H, Miya M, Shiomi T. Anionic polymerization of 2-Hexyl[3]dendralene. Macromol Chem Phys. 2018;219:1700046.
Takagi T, Toda T, Miya M, Takenaka K. Stable and highly regioselective anionic polymerization of (Z)-1-Phenyl[3]dendralene. Macromolecules. 2021;54:4326–32.
Margl P. Mechanisms for anionic butadiene polymerization with alkyl lithium species. Can J Chem. 2009;87:891–903.
Morita H, Van Beylen M. New vistas on the anionic polymerization of styrene in non-polar solvents by means of density functional theory. Polymers. 2016;8:371/1–371/29.
Morita H, Van Beylen M. The mechanism of the propagation in the anionic polymerization of polystyryllithium in non-polar solvents elucidated by density functional theory calculations. A study of the negligible part played by dimeric ion-pairs under usual polymerization condition. Polymers. 2019;11:1022.
Johann T, Leibig D, Grune E, Müller AHE, Frey H. Effect of the substituent position on the anionic copolymerization of styrene derivatives: experimental results and density functional theory calculations. Macromolecules. 2019;52:4545–54.
Kosaka Y, Kawauchi S, Goseki R, Ishizone T. High anionic polymerizability of benzofulvene: new exo-methylene hydrocarbon monomer. Macromolecules. 2015;48:4421–30.
NBO Version 3.1, E. D. Glendening, A. E. Reed, J. E. Carpenter, and F. Weinhold.
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, et al. Gaussian 09, Revision.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Takagi, T., Toda, T., Miya, M. et al. DFT study on the anionic polymerization of phenyl-substituted [3]dendralene derivatives: reactivities of monomer and chain end carbanion. Polym J 54, 643–652 (2022). https://doi.org/10.1038/s41428-022-00615-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-022-00615-1