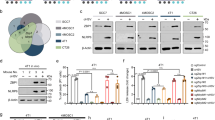
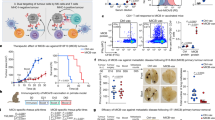
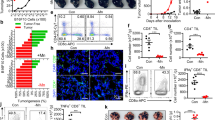
Abstract
Boosting tumor immunosurveillance with vaccines has been proven to be a feasible and cost-effective strategy to fight cancer. Although major breakthroughs have been achieved in preventative tumor vaccines targeting oncogenic viruses, limited advances have been made in curative vaccines for virus-irrelevant malignancies. Accumulating evidence suggests that preconditioning tumor cells with certain cytotoxic drugs can generate whole-cell tumor vaccines with strong prophylactic activities. However, the immunogenicity of these vaccines is not sufficient to restrain the outgrowth of existing tumors. In this study, we identified arsenic trioxide (ATO) as a wide-spectrum cytotoxic and highly immunogenic drug through multiparameter screening. ATO preconditioning could generate whole-cell tumor vaccines with potent antineoplastic effects in both prophylactic and therapeutic settings. The tumor-preventive or tumor-suppressive benefits of these vaccines relied on CD8+ T cells and type I and II interferon signaling and could be linked to the release of immunostimulatory danger molecules. Unexpectedly, following ATO-induced oxidative stress, multiple cell death pathways were activated, including autophagy, apoptosis, necroptosis, and ferroptosis. CRISPR‒Cas9-mediated knockout of cell death executors revealed that the absence of Rip3, Mlkl, or Acsl4 largely abolished the efficacy of ATO-based prophylactic and therapeutic cancer vaccines. This therapeutic failure could be rescued by coadministration of danger molecule analogs. In addition, PD-1 blockade synergistically improved the therapeutic efficacy of ATO-based cancer vaccines by augmenting local IFN-γ production.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All requests for raw and analyzed data and materials will be promptly reviewed by the Institute of Systems Medicine, Chinese Academy of Medical Sciences, to verify whether the request is subject to any intellectual property or confidentiality obligations. Any data and materials that can be shared will be released via a material transfer agreement. All raw RNA-sequencing data can be found at the NCBI Sequence Read Archive (accession number: PRJNA818773).
References
Song M, Vogelstein B, Giovannucci EL, Willett WC, Tomasetti C. Cancer prevention: molecular and epidemiologic consensus. Science. 2018;361:1317–8. https://doi.org/10.1126/science.aau3830
Finn OJ. The dawn of vaccines for cancer prevention. Nat Rev Immunol. 2018;18:183–94. https://doi.org/10.1038/nri.2017.140
Garland SM, Kjaer SK, Munoz N, Block SL, Brown DR, DiNubile MJ, et al. Impact and effectiveness of the quadrivalent human papillomavirus vaccine: a systematic review of 10 years of real-world experience. Clin Infect Dis. 2016;63:519–27. https://doi.org/10.1093/cid/ciw354
McMahon BJ, Bulkow LR, Singleton RJ, Williams J, Snowball M, Homan C, et al. Elimination of hepatocellular carcinoma and acute hepatitis B in children 25 years after a hepatitis B newborn and catch-up immunization program. Hepatology. 2011;54:801–7. https://doi.org/10.1002/hep.24442
Sun X, Zeng L, Huang Y. Transcutaneous delivery of DNA/mRNA for cancer therapeutic vaccination. J Gene Med. 2019;21:e3089. https://doi.org/10.1002/jgm.3089
Peng M, Mo Y, Wang Y, Wu P, Zhang Y, Xiong F, et al. Neoantigen vaccine: an emerging tumor immunotherapy. Mol Cancer. 2019;18:128. https://doi.org/10.1186/s12943-019-1055-6
Jou J, Harrington KJ, Zocca MB, Ehrnrooth E, Cohen EEW. The changing landscape of therapeutic cancer vaccines-novel platforms and neoantigen identification. Clin Cancer Res. 2021;27:689–703. https://doi.org/10.1158/1078-0432.CCR-20-0245
Cheever MA, Higano CS. PROVENGE (Sipuleucel-T) in prostate cancer: the first FDA-approved therapeutic cancer vaccine. Clin Cancer Res. 2011;17:3520–6. https://doi.org/10.1158/1078-0432.CCR-10-3126
Saxena M, van der Burg SH, Melief CJM, Bhardwaj N. Therapeutic cancer vaccines. Nat Rev Cancer. 2021;21:360–78. https://doi.org/10.1038/s41568-021-00346-0
van der Burg SH, Arens R, Ossendorp F, van Hall T, Melief CJ. Vaccines for established cancer: overcoming the challenges posed by immune evasion. Nat Rev Cancer. 2016;16:219–33. https://doi.org/10.1038/nrc.2016.16
Zitvogel L, Apetoh L, Ghiringhelli F, Kroemer G. Immunological aspects of cancer chemotherapy. Nat Rev Immunol. 2008;8:59–73. https://doi.org/10.1038/nri2216
Ma Y, Kepp O, Ghiringhelli F, Apetoh L, Aymeric L, Locher C, et al. Chemotherapy and radiotherapy: cryptic anticancer vaccines. Semin Immunol. 2010;22:113–24. https://doi.org/10.1016/j.smim.2010.03.001
Ma Y, Pitt JM, Li Q, Yang H. The renaissance of anti-neoplastic immunity from tumor cell demise. Immunol Rev. 2017;280:194–206. https://doi.org/10.1111/imr.12586
Casares N, Pequignot MO, Tesniere A, Ghiringhelli F, Roux S, Chaput N, et al. Caspase-dependent immunogenicity of doxorubicin-induced tumor cell death. J Exp Med. 2005;202:1691–701. https://doi.org/10.1084/jem.20050915
Min L, Teijeira A, Sanchez-Paulete AR, Ochoa MC, Alvarez M, Otano I, et al. Cellular cytotoxicity is a form of immunogenic cell death. J Immunother Cancer. 2020;8. https://doi.org/10.1136/jitc-2019-000325.
Vacchelli E, Ma Y, Baracco EE, Sistigu A, Enot DP, Pietrocola F, et al. Chemotherapy-induced antitumor immunity requires formyl peptide receptor 1. Science. 2015;350:972–8. https://doi.org/10.1126/science.aad0779
Chiang CL, Benencia F, Coukos G. Whole tumor antigen vaccines. Semin Immunol. 2010;22:132–43. https://doi.org/10.1016/j.smim.2010.02.004
Chiang CL, Coukos G, Kandalaft LE. Whole tumor antigen vaccines: where are we? Vaccines (Basel). 2015;3:344–72. https://doi.org/10.3390/vaccines3020344
Galaine J, Turco C, Vauchy C, Royer B, Mercier-Letondal P, Queiroz L, et al. CD4 T cells target colorectal cancer antigens upregulated by oxaliplatin. Int J Cancer. 2019;145:3112–25. https://doi.org/10.1002/ijc.32620
Lhuillier C, Rudqvist NP, Yamazaki T, Zhang T, Charpentier M, Galluzzi L, et al. Radiotherapy-exposed CD8+ and CD4+ neoantigens enhance tumor control. J Clin Invest. 2021;131. https://doi.org/10.1172/JCI138740.
Rehman H, Silk AW, Kane MP, Kaufman HL. Into the clinic: Talimogene laherparepvec (T-VEC), a first-in-class intratumoral oncolytic viral therapy. J Immunother Cancer. 2016;4:53. https://doi.org/10.1186/s40425-016-0158-5
Sasso MS, Mitrousis N, Wang Y, Briquez PS, Hauert S, Ishihara J, et al. Lymphangiogenesis-inducing vaccines elicit potent and long-lasting T cell immunity against melanomas. Sci Adv. 2021;7. https://doi.org/10.1126/sciadv.abe436
Larocca CA, LeBoeuf NR, Silk AW, Kaufman HL. An update on the role of talimogene laherparepvec (T-VEC) in the treatment of melanoma: best practices and future directions. Am J Clin Dermatol. 2020;21:821–32. https://doi.org/10.1007/s40257-020-00554-8
Stojdl DF, Lichty B, Knowles S, Marius R, Atkins H, Sonenberg N, et al. Exploiting tumor-specific defects in the interferon pathway with a previously unknown oncolytic virus. Nat Med. 2000;6:821–5. https://doi.org/10.1038/77558
Laheru D, Lutz E, Burke J, Biedrzycki B, Solt S, Onners B, et al. Allogeneic granulocyte macrophage colony-stimulating factor-secreting tumor immunotherapy alone or in sequence with cyclophosphamide for metastatic pancreatic cancer: a pilot study of safety, feasibility, and immune activation. Clin Cancer Res. 2008;14:1455–63. https://doi.org/10.1158/1078-0432.CCR-07-0371
Salgia R, Lynch T, Skarin A, Lucca J, Lynch C, Jung K, et al. Vaccination with irradiated autologous tumor cells engineered to secrete granulocyte-macrophage colony-stimulating factor augments antitumor immunity in some patients with metastatic non-small-cell lung carcinoma. J Clin Oncol. 2003;21:624–30. https://doi.org/10.1200/JCO.2003.03.091
Chen Z, Chen SJ. Poisoning the Devil. Cell. 2017;168:556–60. https://doi.org/10.1016/j.cell.2017.01.029
Lo-Coco F, Avvisati G, Vignetti M, Thiede C, Orlando SM, Iacobelli S, et al. Retinoic acid and arsenic trioxide for acute promyelocytic leukemia. N. Engl J Med. 2013;369:111–21. https://doi.org/10.1056/NEJMoa1300874
Michaud M, Martins I, Sukkurwala AQ, Adjemian S, Ma Y, Pellegatti P, et al. Autophagy-dependent anticancer immune responses induced by chemotherapeutic agents in mice. Science. 2011;334:1573–7. https://doi.org/10.1126/science.1208347
Yang H, Ma Y, Chen G, Zhou H, Yamazaki T, Klein C, et al. Contribution of RIP3 and MLKL to immunogenic cell death signaling in cancer chemotherapy. Oncoimmunology. 2016;5:e1149673. https://doi.org/10.1080/2162402X.2016.1149673
Wang Q, Wang Y, Ding J, Wang C, Zhou X, Gao W, et al. A bioorthogonal system reveals antitumour immune function of pyroptosis. Nature. 2020;579:421–6. https://doi.org/10.1038/s41586-020-2079-1
Efimova I, Catanzaro E, Van der Meeren L, Turubanova VD, Hammad H, Mishchenko TA, et al. Vaccination with early ferroptotic cancer cells induces efficient antitumor immunity. J Immunother Cancer. 2020;8. https://doi.org/10.1136/jitc-2020-001369
Liao P, Wang W, Wang W, Kryczek I, Li X, Bian Y, et al. CD8(+) T cells and fatty acids orchestrate tumor ferroptosis and immunity via ACSL4. Cancer Cell. 2022. https://doi.org/10.1016/j.ccell.2022.02.003
Michaud M, Sukkurwala AQ, Di Sano F, Zitvogel L, Kepp O, Kroemer G. Synthetic induction of immunogenic cell death by genetic stimulation of endoplasmic reticulum stress. Oncoimmunology. 2014;3:e28276. https://doi.org/10.4161/onci.28276
Aaes TL, Kaczmarek A, Delvaeye T, De Craene B, De Koker S, Heyndrickx L. Vaccination with necroptotic cancer cells induces efficient anti-tumor immunity. Cell Rep. 2016;15:274–87. https://doi.org/10.1016/j.celrep.2016.03.037
Obeid M, Tesniere A, Ghiringhelli F, Fimia GM, Apetoh L, Perfettini JL. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med. 2007;13:54–61. https://doi.org/10.1038/nm1523
Wang Y, Gao W, Shi X, Ding J, Liu W, He H, et al. Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of a gasdermin. Nature. 2017;547:99–103. https://doi.org/10.1038/nature22393
Sarhan J, Liu BC, Muendlein HI, Li P, Nilson R, Tang AY, et al. Caspase-8 induces cleavage of gasdermin D to elicit pyroptosis during Yersinia infection. Proc Natl Acad Sci USA. 2018;115:E10888–97. https://doi.org/10.1073/pnas.1809548115
Stutz A, Horvath GL, Monks BG, Latz E. ASC speck formation as a readout for inflammasome activation. Methods Mol Biol. 2013;1040:91–101. https://doi.org/10.1007/978-1-62703-523-1_8
Lin CC, Hsu C, Hsu CH, Hsu WL, Cheng AL, Yang CH. Arsenic trioxide in patients with hepatocellular carcinoma: a phase II trial. Invest N. Drugs. 2007;25:77–84. https://doi.org/10.1007/s10637-006-9004-9
Owonikoko TK, Zhang G, Kim HS, Stinson RM, Bechara R, Zhang C, et al. Patient-derived xenografts faithfully replicated clinical outcome in a phase II co-clinical trial of arsenic trioxide in relapsed small cell lung cancer. J Transl Med. 2016;14:111. https://doi.org/10.1186/s12967-016-0861-5
Workenhe ST, Mossman KL. Oncolytic virotherapy and immunogenic cancer cell death: sharpening the sword for improved cancer treatment strategies. Mol Ther. 2014;22:251–6. https://doi.org/10.1038/mt.2013.220
Deng L, Liang H, Xu M, Yang X, Burnette B, Arina A, et al. STING-dependent cytosolic DNA sensing promotes radiation-induced Type I interferon-dependent antitumor immunity in immunogenic tumors. Immunity. 2014;41:843–52. https://doi.org/10.1016/j.immuni.2014.10.019
Ott PA, Hu Z, Keskin DB, Shukla SA, Sun J, Bozym DJ, et al. An immunogenic personal neoantigen vaccine for patients with melanoma. Nature. 2017;547:217–21. https://doi.org/10.1038/nature22991
Hu Z, Leet DE, Allesoe RL, Oliveira G, Li S, Luoma AM, et al. Personal neoantigen vaccines induce persistent memory T cell responses and epitope spreading in patients with melanoma. Nat Med. 2021;27:515–25. https://doi.org/10.1038/s41591-020-01206-4
Wagner S, Mullins CS, Linnebacher M. Colorectal cancer vaccines: Tumor-associated antigens vs neoantigens. World J Gastroenterol. 2018;24:5418–32. https://doi.org/10.3748/wjg.v24.i48.5418
Zhang Z, Zhang H, Li D, Zhou X, Qin Q, Zhang Q. Caspase-3-mediated GSDME induced Pyroptosis in breast cancer cells through the ROS/JNK signalling pathway. J Cell Mol Med. 2021;25:8159–68. https://doi.org/10.1111/jcmm.16574
Jin Y, Li H, Xie G, Chen S, Wu S, Fang X. Sevoflurane combined with ATP activates caspase-1 and triggers caspase-1-dependent pyroptosis in murine J774 macrophages. Inflammation. 2013;36:330–6. https://doi.org/10.1007/s10753-012-9550-6
Rogers C, Rogers C, Erkes DA, Nardone A, Aplin AE. Gasdermin pores permeabilize mitochondria to augment caspase-3 activation during apoptosis and inflammasome activation. Nat Commun. 2019;10:1689. https://doi.org/10.1038/s41467-019-09397-2
Liao Z, Li S, Liu R, Feng X, Shi Y, Wang K, et al. Autophagic degradation of Gasdermin D protects against nucleus pulposus cell pyroptosis and retards intervertebral disc degeneration in vivo. Oxid Med Cell Longev. 2021;2021:5584447. https://doi.org/10.1155/2021/5584447
Funding
YM is supported by the National Science and Technology Innovation 2030 Major Project of China (2022ZD0205700), Natural Science Foundation of China (NSFC, 81972701), CAMS Innovation Fund for Medical Sciences (CIFMS; 2021-I2M-1-074, 2022-I2M-2-004), National Special Support Program for High-level Talents, China Ministry of Science and Technology (National Key Research and Development Program, Grant 2017YFA0506200), and Innovative and Entrepreneurial Team Program (Jiangsu Province).
Author information
Authors and Affiliations
Contributions
Study conception and design: YM. Data collection: JC, YM, SZ, ZJ, XZ. Analysis and interpretation of results: JC, YM, PL, HY. Draft manuscript preparation: YM, JC. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, J., Jin, Z., Zhang, S. et al. Arsenic trioxide elicits prophylactic and therapeutic immune responses against solid tumors by inducing necroptosis and ferroptosis. Cell Mol Immunol 20, 51–64 (2023). https://doi.org/10.1038/s41423-022-00956-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-022-00956-0
Keywords
This article is cited by
-
Tumorous IRE1α facilitates CD8+T cells-dependent anti-tumor immunity and improves immunotherapy efficacy in melanoma
Cell Communication and Signaling (2024)
-
CRISPR–Cas9 applications in T cells and adoptive T cell therapies
Cellular & Molecular Biology Letters (2024)
-
Arsenic trioxide augments immunogenic cell death and induces cGAS-STING-IFN pathway activation in hepatocellular carcinoma
Cell Death & Disease (2024)
-
Immunogenic cell death in cancer: concept and therapeutic implications
Journal of Translational Medicine (2023)
-
Immunological consequences of arsenic trioxide-induced necrosis
Cellular & Molecular Immunology (2023)