Abstract
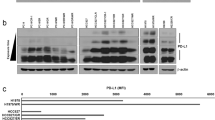
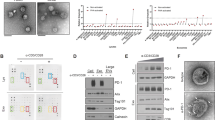
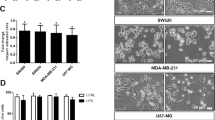
Tumors escape immune attack by upregulating the surface expression of PD-L1, which interacts with PD-1 on T cells to activate immune inhibitory signaling. Anti-PD-1 treatments can effectively block this inhibitory signaling and activate antitumor immune responses. However, anti-PD-1 treatment also has a tumor suppressive effect in patients whose tumor cells do not express PD-L1. The underlying mechanisms are poorly defined. Here, we report that exosomes from bone marrow-derived cells (BMDCs) in tumor-bearing mice, but not in healthy mice, carry PD-L1. PD-L1 on these exosomes is biofunctional and can inhibit CD8+ T cell proliferation and activation in vitro and in vivo. The transfer of exogenous exosomes from BMDCs and the inhibition of the production of endogenous exosomes by BMDCs promote and suppress tumor growth, respectively. PD-L1+ exosomes from BMDCs can be found in tumor tissues. In addition, exosomes from BMDCs promote tumor metastasis in a PD-L1-dependent manner. Therefore, our results indicate that exosomes from BMDCs play important roles in tumor immunosuppression via PD-L1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chen, D. S. & Mellman, I. Elements of cancer immunity and the cancer-immune set point. Nature 541, 321–330 (2017).
Dong, H., Strome, S. E., Salomao, D. R., Tamura, H., Hirano, F. & Flies, D. B. et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat. Med. 8, 793–800 (2002).
Herbst, R. S. et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 515, 563–567 (2014).
Powles, T. et al. MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature 515, 558–562 (2014).
Dong, W. et al. The mechanism of anti-PD-L1 antibody efficacy against PD-L1 negative tumors identifies NK cells expressing PD-L1 as a cytolytic effector. Cancer Discov. 9, 1422–1437 (2019).
Mashouri, L. et al. Exosomes: composition, biogenesis, and mechanisms in cancer metastasis and drug resistance. Mol. Cancer 18, 75 (2019).
Zhou, X. et al. The function and clinical application of extracellular vesicles in innate immune regulation. Cell Mol. Immunol. 17, 323–334 (2020).
Peinado, H. et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 18, 883–891 (2012).
Zhang, F. et al. Specific decrease in B-cell-derived extracellular vesicles enhances post-chemotherapeutic CD8(+) T cell responses. Immunity 50, 738–750 (2019).
Zhang, L. et al. Microenvironment-induced PTEN loss by exosomal microRNA primes brain metastasis outgrowth. Nature 527, 100–104 (2015).
Chen, G. et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 560, 382–386 (2018).
Poggio, M. et al. Suppression of exosomal PD-L1 induces systemic anti-tumor immunity and memory. Cell 177, 414–427 e413 (2019).
Kaplan, R. N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Kuonen, F. et al. Inhibition of the Kit ligand/c-Kit axis attenuates metastasis in a mouse model mimicking local breast cancer relapse after radiotherapy. Clin. Cancer Res. 18, 4365–4374 (2012).
Costa-Silva, B. et al. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat. Cell Biol. 17, 816–826 (2015).
Cai, Z. et al. Activated T cell exosomes promote tumor invasion via Fas signaling pathway. J. Immunol. 188, 5954–5961 (2012).
Xiu, F. M. et al. Surface anchorage of superantigen SEA promotes induction of specific antitumor immune response by tumor-derived exosomes. J. Mol. Med. 85, 511–521 (2007).
Kushida, T. et al. Intra-bone marrow injection of allogeneic bone marrow cells: a powerful new strategy for treatment of intractable autoimmune diseases in MRL/lpr mice. Blood 97, 3292–3299 (2001).
Hong, X. et al. EBAG9 inducing hyporesponsiveness of T cells promotes tumor growth and metastasis in 4T1 murine mammary carcinoma. Cancer Sci. 100, 961–969 (2009).
Trajkovic, K. et al. Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science 319, 1244–1247 (2008).
Ostrowski, M. et al. Rab27a and Rab27b control different steps of the exosome secretion pathway. Nat. Cell Biol. 12, 19–30 (2010).
Coffelt, S. B. et al. IL-17-producing gammadelta T cells and neutrophils conspire to promote breast cancer metastasis. Nature 522, 345–348 (2015).
Kusmartsev, S. & Gabrilovich, D. I. Role of immature myeloid cells in mechanisms of immune evasion in cancer. Cancer Immunol. Immunother. 55, 237–245 (2006).
Talmadge, J. E. Pathways mediating the expansion and immunosuppressive activity of myeloid-derived suppressor cells and their relevance to cancer therapy. Clin. Cancer Res. 13, 5243–5248 (2007).
Noman, M. Z. et al. PD-L1 is a novel direct target of HIF-1alpha, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J. Exp. Med. 211, 781–790 (2014).
Sun, X., Zhang, T., Li, M., Yin, L. & Xue, J. Immunosuppressive B cells expressing PD-1/PD-L1 in solid tumors: a mini review. QJM pii, hcz162 (2019).
Curiel, T. J. et al. Blockade of B7-H1 improves myeloid dendritic cell-mediated antitumor immunity. Nat. Med. 9, 562–567 (2003).
Lin, H. et al. Host expression of PD-L1 determines efficacy of PD-L1 pathway blockade-mediated tumor regression. J. Clin. Invest. 128, 805–815 (2018).
Tang, H. D. et al. PD-L1 on host cells is essential for PD-L1 blockade-mediated tumor regression. J. Clin. Invest. 128, 580–588 (2018).
Acknowledgements
This work was supported by the Basic Public Welfare Projects of Zhejiang Province (LQ18C080001), the Natural Science Foundation of Zhejiang Province (LY19H100003 and LQ20H160018), and the Hangzhou Health Science and Technology Plan (OO20191170).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Sun, Y., Guo, J., Yu, L. et al. PD-L1+ exosomes from bone marrow-derived cells of tumor-bearing mice inhibit antitumor immunity. Cell Mol Immunol 18, 2402–2409 (2021). https://doi.org/10.1038/s41423-020-0487-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-020-0487-7
Keywords
This article is cited by
-
Tumor immune escape: extracellular vesicles roles and therapeutics application
Cell Communication and Signaling (2024)
-
Extracellular vesicle-cell adhesion molecules in tumours: biofunctions and clinical applications
Cell Communication and Signaling (2023)
-
Potential non-invasive biomarkers in tumor immune checkpoint inhibitor therapy: response and prognosis prediction
Biomarker Research (2023)
-
The Bone Marrow Immune Microenvironment in CML: Treatment Responses, Treatment-Free Remission, and Therapeutic Vulnerabilities
Current Hematologic Malignancy Reports (2023)
-
Extracellular vesicle PD-L1 in reshaping tumor immune microenvironment: biological function and potential therapy strategies
Cell Communication and Signaling (2022)