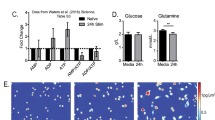
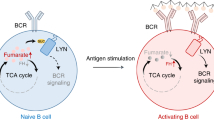
Abstract
Proper control of B cell growth and metabolism is crucial for B-cell-mediated immunity, but the underlying molecular mechanisms remain incompletely understood. In this study, Sin1, a key component of mTOR complex 2 (mTORC2), specifically regulates B cell growth and metabolism. Genetic ablation of Sin1 in B cells reduces the cell size at either the transitional stage or upon antigen stimulation and severely impairs metabolism. Sin1 deficiency also severely impairs B-cell proliferation, antibody responses, and anti-viral immunity. At the molecular level, Sin1 controls the expression and stability of the c-Myc protein and maintains the activity of mTORC1 through the Akt-dependent inactivation of GSK3 and TSC1/2, respectively. Therefore, our study reveals a novel and specific role for Sin1 in coordinating the activation of mTORC2 and mTORC1 to control B cell growth and metabolism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jellusova, J. & Rickert, R. C. The PI3K pathway in B cell metabolism. Crit. Rev. Biochem. Mol. Biol. 51, 359–378 (2016).
Srinivasan, L. et al. PI3 kinase signals BCR-dependent mature B cell survival. Cell 139, 573–586 (2009).
Lam, K. P., Kuhn, R. & Rajewsky, K. In vivo ablation of surface immunoglobulin on mature B cells by inducible gene targeting results in rapid cell death. Cell 90, 1073–1083 (1997).
Kraus, M., Alimzhanov, M. B., Rajewsky, N. & Rajewsky, K. Survival of resting mature B lymphocytes depends on BCR signaling via the Igalpha/beta heterodimer. Cell 117, 787–800 (2004).
Avalos, A. M., Meyer-Wentrup, F. & Ploegh, H. L. B-cell receptor signaling in lymphoid malignancies and autoimmunity. Adv. Immunol. 123, 1–49 (2014).
Khalil, A. M., Cambier, J. C. & Shlomchik, M. J. B cell receptor signal transduction in the GC is short-circuited by high phosphatase activity. Science 336, 1178–1181 (2012).
Patke, A., Mecklenbrauker, I., Erdjument-Bromage, H., Tempst, P. & Tarakhovsky, A. BAFF controls B cell metabolic fitness through a PKC beta- and Akt-dependent mechanism. J. Exp. Med. 203, 2551–2562 (2006).
Konigsberger, S. & Kiefer, F. The BAFFling function of Syk in B-cell homeostasis. EMBO J. 34, 838–840 (2015).
Guo, B., Su, T. T. & Rawlings, D. J. Protein kinase C family functions in B-cell activation. Curr. Opin. Immunol. 16, 367–373 (2004).
Rawlings, D. J., Schwartz, M. A., Jackson, S. W. & Meyer-Bahlburg, A. Integration of B cell responses through Toll-like receptors and antigen receptors. Nat. Rev. Immunol. 12, 282–294 (2012).
Tsui, C. et al. Protein kinase C-beta dictates b cell fate by regulating mitochondrial remodeling, metabolic reprogramming, and heme biosynthesis. Immunity 48, 1144–1159 e1145 (2018).
Su, B. & Jacinto, E. Mammalian TOR signaling to the AGC kinases. Crit. Rev. Biochem. Mol. Biol. 46, 527–547 (2011).
Saxton, R. A. & Sabatini, D. M. mTOR signaling in growth, metabolism, and disease. Cell 168, 960–976 (2017).
Wullschleger, S., Loewith, R. & Hall, M. N. TOR signaling in growth and metabolism. Cell 124, 471–484 (2006).
Cornu, M., Albert, V. & Hall, M. N. mTOR in aging, metabolism, and cancer. Curr. Opin. Genet. Dev. 23, 53–62 (2013).
Wicker, L. S. et al. Suppression of B cell activation by cyclosporin A, FK506 and rapamycin. Eur. J. Immunol. 20, 2277–2283 (1990).
Aagaard-Tillery, K. M. & Jelinek, D. F. Inhibition of human B lymphocyte cell cycle progression and differentiation by rapamycin. Cell. Immunol. 156, 493–507 (1994).
Ersching, J. et al. Germinal Center Selection and Affinity Maturation Require Dynamic Regulation of mTORC1 Kinase. Immunity 46, 1045–1058 e1046 (2017).
Yang, J. et al. Critical roles of mTOR Complex 1 and 2 for T follicular helper cell differentiation and germinal center responses. Elife 5, e17936 (2016).
Zeng, H. et al. mTORC1 and mTORC2 kinase signaling and glucose metabolism drive follicular helper T cell differentiation. Immunity 45, 540–554 (2016).
Zhang, S. et al. B cell-specific deficiencies in mTOR limit humoral immune responses. J. Immunol. 191, 1692–1703 (2013).
Luo, W., Weisel, F. & Shlomchik, M. J. B. Cell receptor and CD40 signaling are rewired for synergistic induction of the c-Myc transcription factor in germinal center B cells. Immunity 48, 313–326 e315 (2018).
Sancak, Y. et al. PRAS40 is an insulin-regulated inhibitor of the mTORC1 protein kinase. Mol. Cell 25, 903–915 (2007).
Jacinto, E. et al. SIN1/MIP1 maintains rictor-mTOR complex integrity and regulates Akt phosphorylation and substrate specificity. Cell 127, 125–137 (2006).
Sarbassov, D. D. et al. Rictor, a novel binding partner of mTOR, defines a rapamycin-insensitive and raptor-independent pathway that regulates the cytoskeleton. Curr. Biol. 14, 1296–1302 (2004).
Jacinto, E. et al. Mammalian TOR complex 2 controls the actin cytoskeleton and is rapamycin insensitive. Nat. Cell Biol. 6, 1122–1128 (2004).
Hara, K. et al. Raptor, a binding partner of target of rapamycin (TOR), mediates TOR action. Cell 110, 177–189 (2002).
Facchinetti, V. et al. The mammalian target of rapamycin complex 2 controls folding and stability of Akt and protein kinase C. EMBO J. 27, 1932–1943 (2008).
Ikenoue, T., Inoki, K., Yang, Q., Zhou, X. & Guan, K. L. Essential function of TORC2 in PKC and Akt turn motif phosphorylation, maturation and signalling. EMBO J. 27, 1919–1931 (2008).
Chen, X. et al. Cryo-EM structure of human mTOR complex 2. Cell Res. 28, 518–528 (2018).
Lazorchak, A. S. et al. Sin1-mTORC2 suppresses rag and il7r gene expression through Akt2 in B cells. Mol. Cell 39, 433–443 (2010).
Lee, K. et al. Requirement for Rictor in homeostasis and function of mature B lymphoid cells. Blood 122, 2369–2379 (2013).
Limon, J. J. et al. mTOR kinase inhibitors promote antibody class switching via mTORC2 inhibition. Proc. Natl Acad. Sci. USA 111, E5076–E5085 (2014).
Holl, T. M., Haynes, B. F. & Kelsoe, G. Stromal cell independent B cell development in vitro: generation and recovery of autoreactive clones. J. Immunol. Methods 354, 53–67 (2010).
Nakano, T., Kodama, H. & Honjo, T. Generation of lymphohematopoietic cells from embryonic stem cells in culture. Science 265, 1098–1101 (1994).
Rickert, R. C., Roes, J. & Rajewsky, K. B lymphocyte-specific, Cre-mediated mutagenesis in mice. Nucleic Acids Res. 25, 1317–1318 (1997).
Engelman, J. A., Luo, J. & Cantley, L. C. The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat. Rev. Genet. 7, 606–619 (2006).
Yang, G., Murashige, D. S., Humphrey, S. J. & James, D. E. A Positive Feedback Loop between Akt and mTORC2 via SIN1 Phosphorylation. Cell Rep. 12, 937–943 (2015).
van Riggelen, J., Yetil, A. & Felsher, D. W. MYC as a regulator of ribosome biogenesis and protein synthesis. Nat. Rev. Cancer 10, 301–309 (2010).
Iritani, B. M. & Eisenman, R. N. c-Myc enhances protein synthesis and cell size during B lymphocyte development. Proc. Natl Acad. Sci. USA 96, 13180–13185 (1999).
Inoki, K., Zhu, T. & Guan, K. L. TSC2 mediates cellular energy response to control cell growth and survival. Cell 115, 577–590 (2003).
Cross, D. A., Alessi, D. R., Cohen, P., Andjelkovich, M. & Hemmings, B. A. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 378, 785–789 (1995).
Sears, R. C. The life cycle of C-myc: from synthesis to degradation. Cell Cycle 3, 1133–1137 (2004).
He, B. et al. Intranasal application of polyethyleneimine suppresses influenza virus infection in mice. Emerg. Microbes Infect. 5, e41 (2016).
Donahue, A. C. & Fruman, D. A. Distinct signaling mechanisms activate the target of rapamycin in response to different B-cell stimuli. Eur. J. Immunol. 37, 2923–2936 (2007).
Chang, X., Lazorchak, A. S., Liu, D. & Su, B. Sin1 regulates Treg-cell development but is not required for T-cell growth and proliferation. Eur. J. Immunol. 42, 1639–1647 (2012).
Lee, K. et al. Mammalian target of rapamycin protein complex 2 regulates differentiation of Th1 and Th2 cell subsets via distinct signaling pathways. Immunity 32, 743–753 (2010).
Frias, M. A. et al. mSin1 is necessary for Akt/PKB phosphorylation, and its isoforms define three distinct mTORC2s. Curr. Biol. 16, 1865–1870 (2006).
Chung, J. B., Silverman, M. & Monroe, J. G. Transitional B cells: step by step towards immune competence. Trends Immunol. 24, 343–349 (2003).
Doughty, C. A. et al. Antigen receptor-mediated changes in glucose metabolism in B lymphocytes: role of phosphatidylinositol 3-kinase signaling in the glycolytic control of growth. Blood 107, 4458–4465 (2006).
Betz, C. et al. Feature Article: mTOR complex 2-Akt signaling at mitochondria-associated endoplasmic reticulum membranes (MAM) regulates mitochondrial physiology. Proc. Natl Acad. Sci. USA 110, 12526–12534 (2013).
Jellusova, J. et al. Gsk3 is a metabolic checkpoint regulator in B cells. Nat. Immunol. 18, 303–312 (2017).
Latronico, M. V., Costinean, S., Lavitrano, M. L., Peschle, C. & Condorelli, G. Regulation of cell size and contractile function by AKT in cardiomyocytes. Ann. N. Y. Acad. Sci. 1015, 250–260 (2004).
Faridi, J., Fawcett, J., Wang, L. & Roth, R. A. Akt promotes increased mammalian cell size by stimulating protein synthesis and inhibiting protein degradation. Am. J. Physiol. Endocrinol. Metab. 285, E964–E972 (2003).
Zhang, F., Lazorchak, A. S., Liu, D., Chen, F. & Su, B. Inhibition of the mTORC2 and chaperone pathways to treat leukemia. Blood 119, 6080–6088 (2012).
Wu, Y. T. et al. mTOR complex 2 targets Akt for proteasomal degradation via phosphorylation at the hydrophobic motif. J. Biol. Chem. 286, 14190–14198 (2011).
Liu, P. et al. Sin1 phosphorylation impairs mTORC2 complex integrity and inhibits downstream Akt signalling to suppress tumorigenesis. Nat. Cell Biol. 15, 1340–1350 (2013).
Liu, P. et al. PtdIns(3,4,5)P3-dependent activation of the mTORC2 kinase complex. Cancer Discov. 5, 1194–1209 (2015).
Dengler, H. S. et al. Distinct functions for the transcription factor Foxo1 at various stages of B cell differentiation. Nat. Immunol. 9, 1388–1398 (2008).
Amin, R. H. & Schlissel, M. S. Foxo1 directly regulates the transcription of recombination-activating genes during B cell development. Nat. Immunol. 9, 613–622 (2008).
Inoue, T. et al. The transcription factor Foxo1 controls germinal center B cell proliferation in response to T cell help. J. Exp. Med. 214, 1181–1198 (2017).
Li, F., Smith, P. & Ravetch, J. V. Inhibitory Fcgamma receptor is required for the maintenance of tolerance through distinct mechanisms. J. Immunol. 192, 3021–3028 (2014).
Acknowledgements
We would like to thank Dr. Kevan M Shokat (UCSF) for generously providing pp242 and Dr. William Sessa (Yale University) for providing the Akt1-Akt2− bone marrow samples. We would like to thank Dr. Biao He (Fudan University) for assisting with the PR8 virus challenge. We also want to thank Dr. David Schatz (Yale University), Dr. Yuan Zhuang (Duke University) for kindly reading the manuscript and providing helpful comments. This study was partially supported by grant PR093728 (DoD to B.S.), the National Natural Science Foundation of China (grant numbers 31470845 and 81430033 to B.S., 31422020 to F.L. and 31600704 to H.H.Z.), grant 13JC1404700 from the Program of Science and Technology Commission of Shanghai Municipality (B.S.), the Ministry of Science and Technology of China (Program 2014CB943600, F.L.), and Chinese Mega Project on Infectious Diseases (No. 2018ZX10302301). F.L. is supported by the “Shu Guang” project of Shanghai Municipal Education Commission and Shanghai Education Development Foundation. A.S.L. is a recipient of Brown-Cox Fellowship from Yale University and is a Leukemia & Lymphoma Society fellow.
Author information
Authors and Affiliations
Contributions
F.L., A.S.L. and B.S. conceived and designed the experiments and wrote the paper. M.L. and A.S.L. performed the experiments, analyzed the data and wrote the paper. X.O. generated the Sin1fl/fl mice. O.A., X.X., D.L., Q.W., L.Y., J.J. and Y.H. performed the experiments. A.S.L., O.A., X.X. and D.L. performed the fetal liver transplantation, in vitro pro-B cell culture and immunoblotting. B cell development analyses were performed by M.L. X.B.L, W.Q.Z and H.H.Z. Flow cytometry, model antigen immunization, the PR8 virus infection assay and B cell metabolic measurements were performed by M.L., X.O., G.Q., Y.F., L.L., S.J., and Q.W. C.H., C.R., and Z.Y. discussed the data.
Corresponding authors
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, M., Lazorchak, A.S., Ouyang, X. et al. Sin1/mTORC2 regulate B cell growth and metabolism by activating mTORC1 and Myc. Cell Mol Immunol 16, 757–769 (2019). https://doi.org/10.1038/s41423-018-0185-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-018-0185-x
This article is cited by
-
Mitochondrial Metabolic Programming and Crosstalk to Nucleus
Current Tissue Microenvironment Reports (2023)
-
miR-582 negatively regulates pre-B cell proliferation and survival through targeting Hif1α and Rictor
Cell Death & Disease (2022)
-
Supplying the trip to antibody production—nutrients, signaling, and the programming of cellular metabolism in the mature B lineage
Cellular & Molecular Immunology (2022)
-
An RNA–RNA crosstalk network involving HMGB1 and RICTOR facilitates hepatocellular carcinoma tumorigenesis by promoting glutamine metabolism and impedes immunotherapy by PD-L1+ exosomes activity
Signal Transduction and Targeted Therapy (2021)