Abstract
Long noncoding RNAs (LncRNAs) have been reported to play critical roles in gastric cancer, but true biomarkers remain unknown. In this study, we found a new lncRNA LINC00355 that was involved in malignant progression of gastric cancer (GC) and further revealed its role and mechanism. Differentially expressed lncRNAs were identified through bioinformatics, and qRT-PCR was used to validate the expression of LINC00355 in gastric cancer tissues and cells. The biological role of LINC00355 in GC was detected by gene overexpression and knockdown experiments. Subcellular fractionation, qRT-PCR, and FISH were performed to detect the subcellular localization. Co-IP and western blotting were used to study the ubiquitination-mediated regulation of P53 and the expression of the E3 ligases RAD18 and UBE3C. The results showed that LINC00355 was significantly increased in gastric cancer cell lines and patient tissues and closely correlated with late stages, distant metastasis, and poor prognosis of patients. High expression of LINC00355 promoted the proliferation and invasion of gastric cancer cells in vivo and in vitro. Mechanistic studies found that LINC00355 that mainly located in the nucleus, acting as a transcriptional activator, promoted transcription of RAD18 and UBE3C, which both bind to P53 and mediate the ubiquitination and degradation of P53. Furthermore, LINC00355 overexpression enhanced the ubiquitination process, and LINC00355 knockdown alleviated it. These results indicated that LINC00355 induces gastric cancer cell proliferation and invasion by promoting transcription of RAD18 and UBE3C, which mediates ubiquitination of P53 and thereby plays a critical role in survival and tumorigenicity of gastric cancer cells. LINC00355 may represent a new mechanism for GC progression and provide a potential marker for GC diagnosis and treatment.
Similar content being viewed by others
Introduction
Gastric cancer (GC) is the fifth most frequently diagnosed cancer and the third leading cause of cancer-related death worldwide1. Although there are many available methods for screening gastric cancer, many patients are still diagnosed at advanced stages, and some patients are even diagnosed with metastasis in lymph nodes or other organs2,3. Surgery resection and chemotherapy are the main methods for treating gastric cancer, but patients with advanced gastric cancer do not have the option of surgery, and the prognosis is very poor4,5. Therefore, elucidating the molecular biological mechanism of gastric cancer is indispensable for finding a sensitive diagnostic method for early gastric cancer and improving prognosis.
Long noncoding RNA (lncRNA) is a class of molecules that have transcripts longer than 200 nt, lack an open-reading frame, and have limited protein-coding function6. In the human genome, as common epigenetic regulatory molecules, lncRNAs play an important role in epigenetics and are involved in transcriptional regulation, RNA shearing and modification, mRNA stabilization, translational regulation, protein stabilization and transport, chromosome formation, and structural stability7,8. They participate in regulating embryo development, tissue differentiation, organ formation, and the occurrence and development of some diseases9,10. The dysregulation of lncRNAs can promote tumorigenesis and enhance the development of cancer by regulating proliferation, invasion, and metastasis11,12.
Many lncRNAs have been found to act as oncogenes and tumor suppressors, and many lncRNAs act by interacting with familiar oncogenes or tumor suppressors13,14,15. P53, encoded by the TP53 gene, is an important tumor suppressor that has the nickname of “gene guardian,” and its transcriptional activity is crucial for cell-cycle progression, apoptosis, and DNA repair16,17,18. Although TP53 is largely accepted as a tumor-suppressor gene, oncogenic effects of mutant P53 proteins, such as deregulated metabolic pathways, increased tumor invasion, and enhanced chemotherapy resistance, have also been reported, indicating a gain-of-function role for mutant P53, including in gastric cancer18,19. Post-translational modification of P53, mainly including ubiquitination, phosphorylation, and acetylation, is one of the important mechanisms of regulating P53 activity20. The ubiquitin–proteasome pathway plays a crucial role in regulating P53 protein levels21. It is well known that MDM2, functioning as an E3 ubiquitin ligase, promotes the degradation of P53, and there are also a number of other E3 ubiquitin ligases that bind to P53 to facilitate proteasome-mediated ubiquitination of P5322,23.
In this study, we screened lncRNA profiles using microarray analysis and identified a novel lncRNA, LINC00355, in gastric cancer tissue from patients. It was highly expressed in gastric cancer tissues compared with adjacent normal gastric mucosal epithelial tissues, and was associated with poor prognosis in patients. High expression of LINC00355 promoted gastric cancer cell proliferation and tumorigenicity in immunodeficient mice. High-throughput RNA sequencing upon LINC00355 knockdown revealed that the expression of RAD18 and UBE3C is positively correlated with LINC00355 expression. We predicted substrates of the E3 ubiquitin ligases RAD18 and UBE3C using UbiBrowser (http://ubibrowser.ncpsb.org) and found that P53 was their only “highly probable” substrate. Our data showed that LINC00355 enhanced the interaction between P53 and the E3 ubiquitin ligases RAD18 and UBE3C, thereby facilitating ubiquitination and degradation of P53. Therefore, LINC00355 acts as an oncogene and mediates gastric cancer cell proliferation and invasion by promoting ubiquitination of P53.
Results
LINC00355 is highly expressed in gastric cancer
We performed an integrative analysis of TCGA stomach cancer and normal tissue RNA sequencing data and gastric cancer microarray profiles comprising GSE58828 and GSE27342 from GEO datasets. Bioinformatics analysis showed that there were 353, 37, and 69 upregulated lncRNAs in the TCGA, GSE27342, and GSE58828 datasets, respectively (Fig. 1a). FEZF1-AS1 and LINC00355 were only two overlapping lncRNAs (Fig. 1b, left panel). A previous study reported that FEZF1-AS1 promoted gastric cancer cell proliferation and that high expression of FEZF1-AS1 predicted poor prognosis in gastric cancer patients24. We also found that 167 lncRNAs in GSE27342, 18 lncRNAs in GSE58828, and 310 lncRNAs in TCGA were downregulated, and that LINC00982 was the only overlapping lncRNA (Fig. 1b, right panel). It was reported that LINC00982 was expressed at low levels in gastric cancer tissues, and that decreased LINC00982 expression was negatively correlated with patient prognosis25. Therefore, we presumed that LINC00355, a novel lncRNA, might have an important role in the development and prognosis of gastric cancer.
LINC00355 is upregulated in gastric cancer and predicts poor prognosis in patients
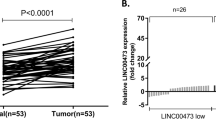
To investigate the relevance between LINC00355 and gastric cancer development, we examined LINC00355 expression in gastric cancer tissue by qRT-PCR from 72 clinical gastric cancer patients and the corresponding paracarcinoma tissue. The results showed that the expression of LINC00355 in gastric cancer tissues was significantly higher than that in adjacent normal tissues (Fig. 2a), and its level was positively associated with depth of invasion and TNM stage (Fig. 2b, c). The survival analysis based on patients’ clinical data showed that high expression of LINC00355 significantly shortened the survival time of patients (Fig. 2d), which suggested that high expression of LINC00355 predicted poor prognosis of gastric cancer.
a LINC00355 expression was analyzed by qRT-PCR in gastric cancer and adjacent non-tumor tissues (n = 72). The results are presented as the fold change in tumor tissues relative to normal tissues. b, c The expression of LINC00355 was positively associated with depth of invasion and TNM stage. d LINC00355 low-expression patients had a longer survival than LINC00355 high-expression patients. Error bars indicate the means ± S.E.M. *P < 0.05, **P < 0.01.
The correlational analysis between LINC00355 expression and clinicopathological factors of gastric cancer patients determined that higher LINC00355 expression levels were significantly correlated with advanced pathologic stage (P = 0.000), invasion depth (P = 0.000), and distant metastasis (P = 0.029) in gastric cancer patients, but it was not correlated with other factors, including histologic differentiation, lymphatic metastasis, sex, and age (Table 1).
LINC00355 promotes malignant phenotypes of gastric cancer cell lines
To determine the role of LINC00355 in the progression of gastric cancer, we examined the expression of LINC00355 in gastric cancer cell lines and the effects of its gain and loss of function. Our data showed that LINC00355 was obviously more highly expressed in MGC803, BGC823, HGC27, SGC7901, and AGS cells than in the normal gastric epithelium cell line GES-1 (Fig. 3a). We silenced the expression of LINC00355 with siRNA in MGC803 and BGC823 cells and overexpressed LINC00355 with the pc-DNA3.1-LINC00355 plasmid in AGS cells. The CCK8 assay showed that knockdown of LINC00355 expression significantly inhibited the proliferation of MGC803 and BGC823 cell lines compared with control cells, while overexpression of LINC00355 clearly increased the proliferation of AGS cell lines (Fig. 3b). In addition, through the EdU experiments, we got consistent results (Supplementary Fig. 1) Similarly, the colony-formation assay revealed that LINC00355 knockdown decreased clone formation of MGC803 and BGC823 cells, while LINC00355 overexpression increased the number of clones of AGS cells (Fig. 3c). Furthermore, the results of the Transwell assay and wound-scratch assay revealed that knockdown of LINC00355 dramatically inhibited cell migration and wound healing compared with the control cells in MGC803 and BGC823 cells. In contrast, overexpression of LINC00355 increased AGS cell migration and wound healing compared with that of the corresponding control groups (Fig. 3d, e). We used qRT-PCR to confirm the expression efficiencies of LINC00355 transfected with siRNA in MGC803 cells and LINC00355 plasmid in AGS cells (Fig. 3f). These results suggest that increased expression of LINC00355 in gastric cancer cells is involved in gastric cancer cell proliferation, migration, and invasion, thereby promoting oncogenesis and progression of gastric cancer.
a Analysis of LINC00355 expression levels in gastric cancer cell lines compared with GES-1 cells by qRT-PCR. b Cell proliferation was detected by CCK8 assay colony-formation assays after BGC823, and MGC803 cells were transfected with siRNAs against LINC00355, and AGS cells were transfected with LINC00355 plasmid. c Colony-formation assays in BGC823 and MGC803 cells transfected with knockdown of LINC00355, and overexpression plasmid of LINC00355 in AGS got consistent results with CCK8 assay. d Cell invasion was measured by Transwell assay, and cell lines were treated the same as in b and c. e Cell migration was monitored by wound-scratch assay, and cell lines were treated the same as in b and c. f Transfection efficiencies of LINC00355 were tested by qRT-PCR in BGC823 MGC803 cells transfected with siRNA and LINC00355 cDNA plasmid in AGS cells. The experiments were repeated three times. Error bars indicate the means ± S.E.M. *P < 0.05, **P < 0.01.
LINC00355 contributes to gastric cancer xenograft tumorigenesis
To further explore whether LINC00355 could promote gastric cancer tumorigenesis in vivo, we established xenograft tumor models in NYG mice with MGC803 cells stably transfected with LINC00355 shRNA and AGS cells stably transfected with pc-DNA3.1-LINC00355, as well as their corresponding controls. Consistent with the in vitro results, 3 weeks after the injection, the tumor sizes and weights of the shRNA LINC00355 group were far less than those of the control group. In the LINC00355-overexpression group, the volume and weight of tumors were much larger than those of the control group (Fig. 4a–c). In addition, we detected Ki-67 expression in tumor tissues, and the results showed that Ki-67 expression in tumors derived from control mice was stronger than that in tumors derived from the LINC00355-knockdown group, while Ki-67 expression in tumors derived from the LINC00355-overexpression group was stronger than that in the control tissue (Fig. 4d). Furthermore, TUNEL experiments showed that tissues from the LINC00355-knockdown group had more apoptotic cells than the control, while tissues from the overexpressed LINC00355 group had fewer apoptotic cells than the control group (Fig. 4e). Thus, these results provide further evidence that LINC00355 plays a tumor-promoting role in gastric cancer in vivo and in vitro.
a Tumor size is shown in groups of LINC00355 knockdown in MGC803, LINC00355 overexpression in AGS cells, and their corresponding controls. b Tumor volumes of mice were measured every 3 days after injection with human gastric cell lines with LINC00355 knockdown and overexpression. c Tumor weights were compared between groups. d Representative images of tumor sections underwent IHC staining using antibodies against Ki-67. e The tumor sections were observed with TUNEL staining to determine the apoptotic cells. The experiments were repeated three times. Error bars indicate the means ± S.E.M. *P < 0.05, **P < 0.01.
LINC00355 regulates the expression of the E3 ubiquitin ligases RAD18 and UBE3C
To further study the mechanism by which LINC00355 promotes the progression of gastric cancer, we investigated its subcellular localization. The results of subcellular fractionation showed that LINC00355 was mainly localized to the nucleus. In line with the data, FISH also showed that LINC00355 was localized preferentially to the nucleus (Fig. 5a, b). To determine the downstream target of LINC00355, we performed RNA transcript high-throughput sequencing. GO and pathway enrichment analysis showed that the top 1 and 2 differentially expressed genes were enriched in protein monoubiquitination and polyubiquitination (Fig. 5c). We analyzed the data profile, tested a panel of downstream molecules, and found that the E3 ubiquitin ligases RAD18 and UBE3C are positively correlated with the level of LINC00355 expression at both the mRNA and protein levels (Fig. 5d–e). This result suggested that LINC00355 upregulates RAD18 and UBE3C that may enhance ubiquitination of their substrates. We predicted potential substrates of RAD18 and UBE3C through the UbiBrowser website (http://ubibrowser.ncpsb.org) and found that P53 was the only “highly probable” substrate of both RAD18 and UBE3C (Fig. 5f).
a Subcellular fractionation assay revealed the nucleus localization of LINC00355. b FISH assay further confirmed that LINC00355 was mainly localized in the nucleus. c Gene ontology (GO) biological process was analyzed to find downstream targets of LINC00355 through RNA sequencing. d The relative mRNA expression of RAD18 and UBE3C after knockdown and overexpression of LINC00355. e The protein levels of RAD18 and UBE3C after knockdown and overexpression of LINC00355. f The UbiBrowser website predicted the potential substrates of E3 ligases RAD18 and UBE3C, and P53 was the common and unique “highly probable” substrate. g The P53 protein levels increased upon LINC00355 knockdown and decreased upon its overexpression. Cells being treated with MG132, a proteasome inhibitor, the P53 protein levels did not obviously change whatever LINC00355 knockdown or overexpression, compared to controls. The experiments were repeated three times. Error bars indicate the means ± S.E.M. *P < 0.05, **P < 0.01.
To test the effect of LINC00355 on P53 expression, we performed P53 western blot analysis in cell lines with LINC00355 knockdown and overexpression, and the results suggested that LINC00355 downregulated the expression of P53 at the protein level but not at the mRNA level (Fig. 5d, g). We also found that cell lines treated with the proteasome inhibitor MG132 attenuated the decrease in P53 protein levels caused by LINC00355 overexpression and the increase in P53 protein levels caused by LINC00355 knockdown (Fig. 5g). Taken together, we deduced that LINC00355 downregulated P53 by mediating proteasome-mediated ubiquitination, thereby promoting gastric cancer progression.
LINC00355 promotes RAD18 and UBE3C-mediated ubiquitination and degradation of P53
To demonstrate that RAD18 and UBE3C are indeed involved in the ubiquitination of P53, we first proved that both RAD18 and UBE3C interacted with P53, and that these two E3 ligases interacted with each other using a Co-IP assay (Fig. 6a, b). Second, ubiquitination experiments showed that overexpression of RAD18 or UBE3C resulted in increased ubiquitination of P53 in HEK293T cell lines (Fig. 6c). In addition, overexpression of both RAD18 and UBE3C dramatically enhanced the ubiquitination of P53 to a greater extent than either of them alone (Fig. 6d). The above results suggested that RAD18 and UBE3C are E3 ubiquitin ligases targeting P53 for degradation. Third, we overexpressed LINC00355 in HEK293T cell lines and found increased ubiquitination degradation of P53. In contrast, knockdown of LINC00355 led to reduced ubiquitination of P53 (Fig. 6e). Furthermore, we found that both RAD18 and UBE3C synergized the effect of LINC00355 on the ubiquitination and degradation of P53 (Fig. 6f). Above all, we conclude that LINC00355 promotes RAD18 and UBE3C-mediated ubiquitination and degradation of P53, and that the combination of RAD18 and UBE3C further accelerates the process.
a Both RAD18 and UBE3C could bind to P53. Lysates from HEK293T cell lines that had been transfected with the indicated expression constructs were subjected to IP with anti-Flag antibody followed by immunoblotting analysis with Flag and Myc antibodies. b These two E3 ligases RAD18 and UBE3C could interact with each other using UBE3C IP and immunoblotting analysis with RAD18 and UBE3C antibodies. c RAD18 and UBE3C promoted the ubiquitination and degradation of P53. Lysates were IP with anti-Flag antibody followed by immunoblotting analysis with HA, Flag, and Myc antibodies. d The combination of RAD18 and UBE3C could further heighten the ubiquitination degradation of P53 than only overexpressing one of them. e Overexpression of LINC00355 enhanced the ubiquitination and degradation of P53, and knockdown of LINC00355 decreased this process of P53. f LINC00355 promoted the ubiquitination degradation of P53 mediated by RAD18 and UBE3C. g The schematic of the molecular mechanisms of LINC00355 in gastric cancer cells. The experiments were repeated three times.
Discussion
Increasing studies suggest that abnormal expression of lncRNAs is closely related to the occurrence of various human diseases, especially tumorigenesis and cancer progression9,26,27. Many studies have found that lncRNAs are involved in some tumor pathological processes and play an important role in gastric tumorigenesis. For example, lncRNA HAGLROS suppresses mTOR pathway-mediated autophagy to promote tumorigenesis and progression, and lncRNA HOXC-AS3 promotes gastric cancer cell proliferation and migration through transcriptional activation of some related gene28,29.
In this study, we identified LINC00355, as a novel and highly expressed lncRNA, in gastric cancer patients by bioinformatics analysis, and tested the role and mechanism of LINC00355 in gastric cancer by assessing clinical patient tissue and cell lines. Sufficient evidence from gain- and loss-of-function experiments in vitro and in vivo showed that LINC00355 promoted the malignant progression of gastric cancer as an oncogene. Moreover, the high expression of LINC00355 is closely correlated with gastric cancer patients’ TNM stages, distant metastasis, and poor prognosis. Furthermore, we found that LINC00355 contributed to the phenotype of gastric cancer by inducing increased transcription of the E3 ligases RAD18 and UBE3C, both of which mediated ubiquitination and degradation of P53.
Many lncRNAs have been found to act as transcriptional and post-transcriptional regulatory genes that regulate processes, including transcription, mRNA splicing, mRNA degradation, and translation30,31. In our study, RNA-seq analyses revealed that RAD18 and UBE3C were potential downstream targets of LINC00355. Then, we demonstrated that LINC00355 regulated RAD18 and UBE3C at both the mRNA and protein levels, which suggested that LINC00355 co-activated their transcription. Previous studies have shown that RAD18 is a key regulator of the DNA transport synthesis (TLS) pathway that has been reported to be abnormally expressed in many cancers, such as glioma. RAD18 acts as a promoter in glioma progression and reduces the sensitivity of glioma cells to radiation by downregulating P5332,33. RAD18 is involved in the expression of various proteases and is an important component of the ubiquitin–proteasome system34. It was reported that RAD18 promotes ubiquitination degradation of FANCD2 and PCNA proteins, thereby inhibiting the apoptosis of cancer cells35,36. Another E3 ubiquitin ligase UBE3C belongs to the ubiquitin ligase HECT family of proteins; it is highly expressed in melanoma, renal cell carcinoma, endometrial cancer, and colorectal cancer, and participates in the occurrence and development of tumors by enhancing epithelial–mesenchymal transformation or activating the Wnt/beta-catenin pathway37,38,39.
In this study, we found that RAD18 and UBE3C, as cooperative E3 ligases, facilitate proteasome-mediated degradation of P53 via ubiquitination, and that LINC00355 further promoted the ubiquitylation process and decreased the protein expression of wild-type P53, thereby promoting tumorigenesis. It is well known that P53, as a tumor-suppressor protein, accumulates in cells mainly in response to DNA damage and oncogene activation. It acts as a nuclear transcription factor that transactivates genes involved in apoptosis, cell-cycle regulation, and numerous other processes40. MDM2 binds and ubiquitylates P53 in the nucleus, resulting in P53 nuclear export and degradation41. Emerging evidence has revealed additional activities of P53 in the cytoplasm, where it triggers apoptosis and inhibits autophagy. As an important tumor-suppressor gene, its loss or mutation often leads to tumorigenesis42. The nuclear versus cytoplasmic effects of P53 are determined by multiple post-translational modifications that affect its interactions with other proteins, its shuttling between the cytoplasm and the nucleus, and its biological activities. In our study, we point out that P53 is ubiquitylated not only by MDM2 but also by two other E3 ligases, RAD18 and UBE3C. These two ligases are located in the nucleoplasm, the subcellular location of P53, where they pass ubiquitin molecules to P53 and contribute to its degradation. LINC00355 promotes RAD18 and UBE3C to decrease P53 and blocks its function as a “genomic guardian” to induce malignant phenotypes of gastric cancer.
Conclusions
This study demonstrates that significantly highly expressed LINC00355 in gastric cancer induces the cell proliferation and invasion by promoting transcription of RAD18 and UBE3C in which both of them facilitate the ubiquitination and degradation of P53. Therefore, LINC00355 plays a critical role in survival and tumorigenicity of gastric cancer cells, and has a great prospect to become a potential biomarker of gastric cancer.
Our data are consistent with the newly recent reports about LINC00355 acting as an oncogene in other cancers, including bladder cancer, head and neck squamous cell carcinoma, and lung adenocarcinoma43,44,45.
Methods
Tissue collection and ethics statement of study subjects
A total of 72 gastric cancer tissues in this study were selected from resection of gastric cancer and adjacent noncancerous tissues of patients who underwent surgery at Nanjing First Hospital of Jiangsu Province in China between 2013 and 2015. Samples were collected during surgery, and liquid nitrogen was used immediately for rapid freezing storage. Patients were diagnosed by pathological biopsy, and no local or systemic treatment was performed before surgery. The protocol used in this study was approved by the Research Ethics Committee of Nanjing Medical University. All patients provided written informed consent for publication. All tissue samples we obtained were preserved in RNA Transport (OMEGA Engineering Inc., Norwalk, CT, USA) and immediately frozen at −80 °C until required.
Cell lines
Human gastric cancer cell lines MGC803, BGC823, SGC7901, HGC27, AGS, normal gastric epithelium cell line GES-1, and human embryonic kidney (HEK) 293T cell lines were obtained from the Chinese Academy of Sciences Committee on Type Culture Collection Cell Bank (Shanghai, China). They were grown in high-glucose DMEM or RPMI-1640 medium (Gibco, Life Technologies, CA, USA) supplemented with 10% fetal bovine serum (FBS) and penicillin (100 IU/mL) and streptomycin (100 mg/mL) (Gibco, Life Technologies, California, USA) and maintained in a humidified incubator at 37 °C with 5% CO2. The cell lines were authenticated by STR profiling, and mycoplasma test was negative.
Microarray analysis
Microarray datasets of gastric cancer gene expression were downloaded from The Cancer Genome Atlas (TCGA) and National Center for Biotechnology Information (NCBI) Gene Expression Omnibus (GEO) dataset to test gene differential expression. Cluster 3.0 software was used (http://hemi.biocuckoo.org) to generate differentially expressed gene heat maps. Differential expression of lncRNAs was determined from the intersection between GSE58828, GSE27342, and TCGA datasets.
RNA extraction and qRT-PCR analysis
The total RNA was extracted using TRIzol reagent (Invitrogen, Grand Island, NY, USA) and measured by a NanoDrop ND-1000 Spectrophotometer (Agilent, CA, USA) according to the manufacturer’s instructions. cDNA was synthesized using the PrimeScript RT reagent Kit (Takara Bio USA, Inc., Mountain View, CA, USA). To detect the expression level of LINC00355 and other related genes in tissues and cells, qRT-PCR was used with the SYBR green method following the manufacturer’s instructions (LightCycler 480, Roche, Basel, Switzerland). GAPDH was used as a control. Primers were designed in NCBI. Three times biological replicates, error bars indicate means ± S.E.M.
Cell viability and clone-formation assay
Cell Counting Kit 8 (CCK8, Corning Co., NY, USA) was used to detect the proliferative ability of cells. A total of 3 × 10³ cells per well were seeded in 96-well plates, and the wavelength was measured at 450 nm after 2 h of treatment. The cellular ability curve was drawn based on the absorbance value.
5-ethynyl-2-deoxyuridine (EdU) assay was performed according to the manufacturer’s instructions (BeyoClick™ EdU Cell Proliferation Kit with Alexa Fluor 488, C0071S, Beyotime, Shanghai, China); BGC823, MGC803, and AGS cells were incubated with EdU for 2 h.
For the colony-formation assay, 1000 cells per well were seeded in six-well plates and cultured in medium containing 10% FBS for 2 weeks. Colonies were fixed with paraformaldehyde for 30 min and then stained with 0.1% crystal violet for 30 min. The number of clones was counted using naked-eye observation and was analyzed statistically. Three times biological replicates, error bars indicate means ± S.E.M.
Transwell assay and wound-healing assay
For the Transwell invasion assay, cell lines transfected with siRNAs for 24 h were seeded in the upper chamber with 4 × 104 cells and cultured with 300 μl of medium containing 0.1% FBS. The lower chamber contained medium supplemented with 10% FBS. Twenty-four hours later, cells in the upper chamber were gently wiped with cotton swabs, and the remaining cells were washed with PBS three times, fixed with methanol, and stained with 0.1% crystal violet. The number of invading cells was counted under a microscope and analyzed statistically.
The wound-healing assay was used to detect the migration capabilities of gastric cancer cell lines. Cells were inoculated into six-well plates for 12–24 h, transfected with siRNAs, control siRNAs, pc-DNA3.1-LINC00355, and pc-DNA3.1 vector and cultured for another 24 h. The cell layer was scraped using a 200-μl sterile plastic tip, and the cells were washed away with medium. The width of the scratch was photographed under the microscope, and changes in the scratch were observed again 48 h later. Biological replicates were counted three times.
Subcellular fractionation and fluorescence in situ hybridization (FISH)
Subcellular fractionation of cytoplasmic and nuclear fractions was performed by using the PARIS Kit (Life Technologies) according to the manufacturer’s instructions to determine the cellular localization of LINC00355. All procedures were performed according to the manufacturer’s protocol. The total RNA was extracted from the nucleus and cytoplasm, and qRT-PCR analysis was conducted to determine the relative levels of LINC00355, GAPDH, and U6. The Ribo Fluorescent In Situ Hybridization Kit and Ribo lncRNA FISH Probe Mix (Ribo, Guangzhou, China) were used to perform the FISH assay according to the manufacturer’s protocol. Biological replicates were counted three times.
RNA sequencing
Total RNA from control, LINC00355 knockdown (48 h after siRNA transfection) in MGC803 cells was isolated with RNeasy Mini Kit (Qiagen, Hilden, Germany). The experiment was performed with three independent biological replicates for each condition. The nine libraries were constructed with an Illumina TruSeq Stranded Total RNA Preparation Kit (Illumina, RS-122-2301), which contained Ribo-Zero Gold, which facilitates the depletion of rRNA. RNA-seq data are included in Supplementary Table 1.
Western blot analysis and immunoprecipitation (IP)
Western blot analysis was conducted according to the method described previously46. The cells were lysed using RIPA protein extraction reagent supplemented with a protease inhibitor cocktail and phenylmethylsulfonyl fluoride (PMSF) (Roche, Basel, Switzerland). Protein concentration was measured using the Beyotime Protein Assay Kit (Beyotime). Approximately 20–40 μg of protein extracts were separated by 6–15% SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to PVDF membranes (Millipore, Burlington, MA, USA); the membranes were incubated with specific primary antibodies (1:500–1000) and secondary antibodies (1:2000–5000). ECL chromogenic substrate was used to visualize the bands. GAPDH was used as an endogenous control.
For the IP assay, cells were washed twice with precooled PBS, lysed with IP lysis buffer for 30 min, scraped, and centrifuged at 13,000 g/min for 10 min. The supernatant was transferred to a new EP tube, and a small amount of lysate was used as an input. For the Flag IP experiment, the remaining lysate was incubated with anti-Flag M2 Magnetic Beads (CST, Danvers, MA, USA) for 2 h at 4 °C with gentle rotation. Before incubation, the beads were washed three times with wash buffer. For the general IP experiment, the remaining lysate was incubated with the special primary antibody overnight at 4 °C with gentle rotation, and then protein A/G was added and incubated with rotation for 1 h at 4 °C. The supernatant was discarded after washing and resuspended in 50 μl of 2× SDS, and the supernatant was moved to a new tube and boiled in a 98 °C metal bath for 10 min. Immunocomplexes were analyzed by SDS/PAGE and immunoblotted with primary antibodies and the corresponding secondary antibodies. Biological replicates were counted three times.
Establishment of xenografts and in vivo studies
Immune-deficient 4-week-old male NYG mice were obtained from Nanjing Medical University Experimental Animal Center, raised under specific nonpathogenic conditions, and performed on the basis of the protocol approved by the Animal Experimental Ethics Committee of Nanjing Medical University. During the whole experiment, we always complied with the “Guide for the Care and Use of Laboratory Animals” prepared by the National Academy of Sciences (USA) and published by the National Institutes of Health (USA). LINC00355 shRNA and Ctrl shRNA were stably transfected into MGC803 cells. A total of 4 × 106 cells were injected subcutaneously on each side per mouse. The tumor size and volume were recorded three times a week, the tumor sizes were measured by calipers, and the tumor volumes were calculated from the length (the longest diameter across the tumor) and width (the corresponding perpendicular diameter) using the following formula: π/6 × length × width2 28. After 3 weeks of injection, the animals were sacrificed, and tumors were removed. Parts of the tissue were fixed with paraformaldehyde for immunohistochemical (IHC) and terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) analysis, and the rest were preserved at −80 °C for qRT-PCR or western blot analysis.
IHC analysis and TUNEL assay
Ki-67 expression was detected with IHC analysis in xenograft mouse tumor tissue. Both the intensity and extent of immunoreactivity were evaluated according to standard protocols as described previously47.
Antibodies and reagents
The following antibodies used in this study were purchased from the indicated sources: P53 (CST 2524S), RAD18 (CST 9040), UBE3C (Abcam ab226173), GAPDH (Abmart P30008M), HA-Tag (CST 3724S), FLAG (CST 14793S), UB (CST 3933S), and Myc (CST 2272S); Antibody diluent was purchased from Beyotime, China. The transfection reagent Lipofectamine™ 2000 was from Invitrogen.
Statistical analysis
All statistical analyses were performed using SPSS 22.0 software (IBM SPSS, Chicago, IL, USA). The survival curves are drawn with Kaplan–Meier survival plots and tested using log-rank tests. Differences between groups were assessed by two-sided Student’s t test. The Chi-square test was used to analyze the pathologic features of LINC00355 expression in gastric cancer. P value < 0.05 standing for significant, <0.01 standing for highly significant.
Change history
30 January 2024
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1038/s41420-024-01828-4
References
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Machlowska, J., Maciejewski, R. & Sitarz, R. The pattern of signatures in gastric cancer prognosis. Int. J. Mol. Sci. 19, https://doi.org/10.3390/ijms19061658 (2018).
Lansdorp-Vogelaar, I. & Kuipers, E. J. Screening for gastric cancer in western countries. Gut 65, 543–544 (2016).
Russo, A. E. & Strong, V. E. Gastric cancer etiology and management in Asia and the West. Annu. Rev. Med. 70, 353–367 (2019).
Jiang, Y. et al. Association of adjuvant chemotherapy with survival in patients with stage II or III gastric cancer. JAMA Surg. 152, e171087 (2017).
Kopp, F. & Mendell, J. T. Functional classification and experimental dissection of long noncoding RNAs. Cell 172, 393–407 (2018).
Sun, Q., Hao, Q. & Prasanth, K. V. Nuclear long noncoding RNAs: key regulators of gene expression. Trends Genet. 34, 142–157 (2018).
Quinn, J. J. & Chang, H. Y. Unique features of long non-coding RNA biogenesis and function. Nat. Rev. Genet. 17, 47–62 (2016).
Dey, B. K., Mueller, A. C. & Dutta, A. Long non-coding RNAs as emerging regulators of differentiation, development, and disease. Transcription 5, e944014 (2014).
Fernandes, J. C. R., Acuna, S. M., Aoki, J. I., Floeter-Winter, L. M. & Muxel, S. M. Long non-coding RNAs in the regulation of gene expression: physiology and disease. Noncoding RNA 5, https://doi.org/10.3390/ncrna5010017 (2019).
Prensner, J. R. & Chinnaiyan, A. M. The emergence of lncRNAs in cancer biology. Cancer Discov. 1, 391–407 (2011).
Gutschner, T. & Diederichs, S. The hallmarks of cancer: a long non-coding RNA point of view. RNA Biol. 9, 703–719 (2012).
Arun, G., Diermeier, S. D. & Spector, D. L. Therapeutic targeting of long non-coding RNAs in cancer. Trends Mol. Med. 24, 257–277 (2018).
Niknafs, Y. S. et al. The lncRNA landscape of breast cancer reveals a role for DSCAM-AS1 in breast cancer progression. Nat. Commun. 7, 12791 (2016).
Malik, R. et al. The lncRNA PCAT29 inhibits oncogenic phenotypes in prostate cancer. Mol. Cancer Res. 12, 1081–1087 (2014).
Kandoth, C. et al. Mutational landscape and significance across 12 major cancer types. Nature 502, 333–339 (2013).
Chen, J. The cell-cycle arrest and apoptotic functions of p53 in tumor initiation and progression. Cold Spring Harb. Perspect. Med. 6, a026104 (2016).
Mantovani, F., Collavin, L. & Del Sal, G. Mutant p53 as a guardian of the cancer cell. Cell Death Differ. 26, 199–212 (2019).
Shin, Y. J. et al. Prognostic implications and interaction of L1 methylation and p53 expression statuses in advanced gastric cancer. Clin. Epigenetics 11, 77 (2019).
Dai, C. & Gu, W. p53 post-translational modification: deregulated in tumorigenesis. Trends Mol. Med. 16, 528–536 (2010).
Pant, V. & Lozano, G. Limiting the power of p53 through the ubiquitin proteasome pathway. Genes Dev. 28, 1739–1751 (2014).
Marine, J. C. & Lozano, G. Mdm2-mediated ubiquitylation: p53 and beyond. Cell Death Differ. 17, 93–102 (2010).
Sane, S. & Rezvani, K. Essential roles of E3 ubiquitin ligases in p53 regulation. Int. J. Mol. Sci. 18, https://doi.org/10.3390/ijms18020442 (2017).
Liu, Y. W. et al. LincRNAFEZF1-AS1 represses p21 expression to promote gastric cancer proliferation through LSD1-mediated H3K4me2 demethylation. Mol. Cancer 16, 39 (2017).
Fei, Z. H. et al. Upregulated expression of long non-coding RNA LINC00982 regulates cell proliferation and its clinical relevance in patients with gastric cancer. Tumour Biol. 37, 1983–1993 (2016).
Zhang, K. et al. Genome-wide lncRNA microarray profiling identifies novel circulating lncRNAs for detection of gastric cancer. Theranostics 7, 213–227 (2017).
Fattahi, S. et al. LncRNAs as potential diagnostic and prognostic biomarkers in gastric cancer: a novel approach to personalized medicine. J. Cell Physiol. 235, 3189–3206 (2020).
Chen, J. F. et al. STAT3-induced lncRNA HAGLROS overexpression contributes to the malignant progression of gastric cancer cells via mTOR signal-mediated inhibition of autophagy. Mol. Cancer 17, 6 (2018).
Zhang, E. et al. A novel long noncoding RNA HOXC-AS3 mediates tumorigenesis of gastric cancer by binding to YBX1. Genome Biol. 19, 154 (2018).
O’Leary, V. B. et al. PARTICLE, a triplex-forming long ncRNA, regulates locus-specific methylation in response to low-dose irradiation. Cell Rep. 11, 474–485 (2015).
Wu, H. et al. Unusual processing generates SPA LncRNAs that sequester multiple RNA binding proteins. Mol. Cell 64, 534–548 (2016).
Durando, M., Tateishi, S. & Vaziri, C. A non-catalytic role of DNA polymerase eta in recruiting Rad18 and promoting PCNA monoubiquitination at stalled replication forks. Nucleic Acids Res. 41, 3079–3093 (2013).
Wu, B. et al. High expression of RAD18 in glioma induces radiotherapy resistance via down-regulating P53 expression. Biomed. Pharmacother. 112, 108555 (2019).
Hibbert, R. G., Huang, A., Boelens, R. & Sixma, T. K. E3 ligase Rad18 promotes monoubiquitination rather than ubiquitin chain formation by E2 enzyme Rad6. Proc. Natl Acad. Sci. USA 108, 5590–5595 (2011).
Han, J. et al. SIVA1 directs the E3 ubiquitin ligase RAD18 for PCNA monoubiquitination. J. Cell Biol. 205, 811–827 (2014).
Branzei, D., Seki, M. & Enomoto, T. Rad18/Rad5/Mms2-mediated polyubiquitination of PCNA is implicated in replication completion during replication stress. Genes Cells 9, 1031–1042 (2004).
Wang, M., Cheng, D., Peng, J. & Pickart, C. M. Molecular determinants of polyubiquitin linkage selection by an HECT ubiquitin ligase. EMBO J. 25, 1710–1719 (2006).
Tang, L. et al. Ubiquitin ligase UBE3C promotes melanoma progression by increasing epithelial-mesenchymal transition in melanoma cells. Oncotarget 7, 15738–15746 (2016).
Wen, J. L. et al. UBE3C promotes growth and metastasis of renal cell carcinoma via activating Wnt/beta-catenin pathway. PLoS ONE 10, e0115622 (2015).
O’Brate, A. & Giannakakou, P. The importance of p53 location: nuclear or cytoplasmic zip code? Drug Resist Updat 6, 313–322 (2003).
Inoue, T., Geyer, R. K., Howard, D., Yu, Z. K. & Maki, C. G. MDM2 can promote the ubiquitination, nuclear export, and degradation of p53 in the absence of direct binding. J. Biol. Chem. 276, 45255–45260 (2001).
Green, D. R. & Kroemer, G. Cytoplasmic functions of the tumour suppressor p53. Nature 458, 1127–1130 (2009).
Yan, L., Wang, P., Fang, W. & Liang, C. Cancer-associated fibroblasts-derived exosomes-mediated transfer of LINC00355 regulates bladder cancer cell proliferation and invasion. Cell Biochem. Funct. https://doi.org/10.1002/cbf.3462 (2019).
Lu, S., Sun, Z., Tang, L. & Chen, L. LINC00355 promotes tumor progression in HNSCC by hindering MicroRNA-195-mediated suppression of HOXA10 expression. Mol. Ther. Nucleic Acids 19, 61–71 (2020).
Liang, Y. et al. A novel long non-coding RNA LINC00355 promotes proliferation of lung adenocarcinoma cells by down-regulating miR-195 and up-regulating the expression of CCNE1. Cell Signal 66, 109462 (2020).
Taniue, K. et al. Long noncoding RNA UPAT promotes colon tumorigenesis by inhibiting degradation of UHRF1. Proc. Natl Acad. Sci. USA 113, 1273–1278 (2016).
Yang, F., Tang, X. Y., Liu, H. & Jiang, Z. W. Inhibition of mitogen-activated protein kinase signaling pathway sensitizes breast cancer cells to endoplasmic reticulum stress-induced apoptosis. Oncol. Rep. 35, 2113–2120 (2016).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (no. 31401094, no. 31700716, no. 81772978, and no. 81972626), the Natural Science Foundation of Jiangsu Province, China (no. BK20181365), and Jiangsu Provincial Special Program of Medical Science (no. BE2017611).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
All the authors are aware of and agree to the content of the paper and their being listed as a co-author of the paper.
Ethics approval and consent to participate
The use of clinical samples was approved by the Ethics Committee of Nanjing Medical University and informed consent was obtained from all patients.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Ivano Amelio
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.1038/s41420-024-01828-4"
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, W., Jin, Y., Wu, P. et al. RETRACTED ARTICLE: LINC00355 induces gastric cancer proliferation and invasion through promoting ubiquitination of P53. Cell Death Discov. 6, 99 (2020). https://doi.org/10.1038/s41420-020-00332-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-020-00332-9