Abstract
The chemokine, CCL5, is a key mediator for the recruitment of immune cells into tumors and tissues. Akt/NF-κB signaling is significantly activated by CCL5. However, the role of NF-κB inactivation in apoptosis induced by negative regulation of CCL5 remains unclear. Here, we analyzed the effect of cordycepin on NF-κB activity in SKOV-3 cells and found that cordycepin-mediated inhibition of NF-κB signaling induced apoptosis in SKOV-3 cells via the serial activation of caspases. In addition, immune-blotting analysis showed that CCL5 is highly expressed in SKOV-3 cells. In addition to activating caspases, we show that, cordycepin prevents TNF-α-induced increase in CCL5, Akt, NF-κB, and c-FLIPL activation and that CCL5 siRNA could inhibit Akt/NF-κB signaling. Moreover, cordycepin negatively regulated the TNF-α-mediated IκB/NF-κB pathway and c-FLIPL activation to promote JNK phosphorylation, resulting in caspase-3 activation and apoptosis. Also, we show that c-FLIPL is rapidly lost in NF-κB activation-deficient. siRNA mediated c-FLIP inhibition increased JNK. SP600125, a selective JNK inhibitor, downregulated p-JNK expression in cordycepin-treated SKOV-3 cells, leading to suppression of cordycepin-induced apoptosis. Thus, these results indicate that cordycepin inhibits CCL5-mediated Akt/NF-κB signaling, which upregulates caspase-3 activation in SKOV-3 cells, supporting the potential of cordycepin as a therapeutic agent for ovarian cancer.
Similar content being viewed by others
Introduction
Cordycepin, 3′-Deoxyadenosine, is a known polyadenylation inhibitor with various pharmacological activities, such as anti-proliferative, anti-cancer, and anti-inflammatory effects1,2,3,4,5,6,7,8. Cordycepin is an active small molecule implicated in regulating various physiological functions by immune-activation and also presents various properties, including anti-viral, anti-infection, anti-inflammatory, anti-aging, anti-cancer properties, and enhances sexual performance9,10,11,12,13. Already, cordycepin has been shown to induce cancer cell death in a large spectrum of tumor cell lines, including breast14, colon15, and oral squamous cell carcinoma8. However, the effects of cordycepin in ovarian carcinoma cells are not clear until now. In some tissues, inflammatory conditions increase the risk of certain cancer. Cytokines and chemokines are involved in an intensive dialog enhancing angiogenesis, tumor metastasis, and the subversion of adaptive immunity, as well as changing responses to hormone therapy or to chemotherapy16. CCL5 belongs to the CC-chemokine family and plays a critical role in the migration and metastasis of human malignant tumor cells17. The activity of CCL5 is mediated by binding to CCR1, CCR3, and mainly CCR518, 19. In the cancer microenvironment, cancer cell stimulates de novo secretion of CCL5 from cancer stem like cells (CSLCs), and CCL5 acts as a paracrine or autocrine signaling to promote tumor cell migration, invasion, and metastasis20, 21.
Akt /protein kinase B (PKB) is a crucial node in diverse signaling pathways essential in both normal cellular physiology, as well as various disease states. Akt signaling controls cell proliferation and anti-apoptosis, cell growth, glucose metabolism, cell migration, and metastasis. Akt is an integrative regulator of tumor survival and apoptosis, and it is also activated downstream of PI3K and is down-regulated by the cancer suppressor PTEN22. Akt functions through its ability to activate many key pro-oncogenic target genes that induce cell growth or antagonize apoptotic pathways.
Nuclear factor-κB (NF-κB) comprises a family of transcription factors that regulate the transcription of cytokines, antimicrobial effectors, and genes that control cellular differentiation, growth, and proliferation in cancer stem cells23. Inducible NF-κB activation relies upon phosphorylation-triggered proteasomal degradation of the inhibitor of NF-κB proteins (IκBs) that retain inactive NF-κB dimers in the cytosol in unstimulated cells24. Recent work suggests a role for NF-κB in the propagation of ovarian cancer cells, but the significance and mechanism of NF-κB in ovarian cancer remains poorly understood. The NF-κB pathway is overactivated in aggressive ovarian cancer25.
In this study, we used inflammatory mediator, TNF-α, which has been shown to participate in both the initiation and progression of cancer, and demonstrated that CCL5 is highly expressed in an ovarian cancer cell line under these conditions. We then investigated the functional mechanisms underlying the stimulation of the NF-κB signaling pathway by CCL5 in ovarian cancer cells. Herein, we show that cordycepin prevents constitutively Akt-mediated NF-κB transcription factor activation by downregulating CCL5 and that the consequent activation of JNK signaling pathway causes cancer cell death.
Results
Cordycepin inhibits the cell viability of ovarian cancer cells
To investigate the effects of cordycepin on the proliferation of cancer cells. SKOV-3, MDAH-2774, and OVCAR-3 human ovarian cancer cells were treated directly with cordycepin at 0, 20, 40, 60, and 100 μg/mL for 24 or 48 h. As shown in Fig. 1a, cordycepin dose-dependently inhibited the cell viability of SKOV-3, MDAH-2774, OVCAR-3 during 24 h and 48 h incubation. At 40 μg/mL, 48 h treatment of cordycepin decreased approximately half of the SKOV-3, MDAH-2774, OVCAR-3 cell population (Fig. 1a). These results were consistent with cell viability of SKOV-3, MDAH-2774, OVCAR-3 cells treated with cordycepin. To observe the cell death of cordycepin-treated cancer cells, the morphologies of ovarian cancer cells were compared to those of untreated control cells by using light microscopy (Nikon TS-100, Nikon, Tokyo, Japan). The morphology of SKOV-3, MDAH-2774, and OVCAR-3 cells changed drastically after 60 μg/mL cordycepin treatment for 48 h. Multiple cells began to detach from the surface of the culture plate and appeared buoyant. Moreover, the cells appeared to be shrunken, resulting in reduced cell volume. These morphological changes preceded apoptosis. On the other hand, 40 μg/mL cordycepin induced less drastic changes at 48 h (Fig. 1b).
a Inhibition of the growth of SKOV-3, MDAH-2774, OVCAR-3 ovarian cancer cells by cordycepin. Ovarian cancer cells were exposed to 0, 20, 40, 60, and 100 μg/mL cordycepin for 24 and 48 h. b Microscopic images of SKOV-3, MDAH-2774, OVCAR-3 cells treated with cordycepin for 48 h. Magnification ×400. c Apoptosis analysis of ovarian cancer cells exposed to cordycepin. The statistics were shown the percentages of the cells represented by mainly early and late apoptosis which was apparent when the percentage of live cells markedly decreased. Data are presented as means ± standard deviations from triplicate experiments. Statistical significance was considered as **p < 0.01 and ***p < 0.001 vs. vehicle treatment
Cordycepin induces apoptotic changes in ovarian cancer cells
The apoptotic effect of cordycepin on SKOV-3, MDAH-2774, OVCAR-3 cells were analyzed with Annexin V. For the evaluation of apoptosis, we used a Muse Annexin V and Dead Cell kit to measure the changes in cell apoptosis after 48 h. Ovarian cancer cell lines were treated for 48 h with 0, 20, 40, 60, and 100 μg/mL cordycepin. Total fractions of apoptosis (early and late apoptosis) were increased by cordycepin treatment in dose-dependent manner. The viable fractions of SKOV-3, MDAH-2774 and OVCAR-3 cells were reduced from 95, 94 and 97% in control group to 88%, 46% and 89% in cordycepin (100 μg/mL)-treated group, respectively (Fig. 1c).
Effect of cordycepin on gene expression profiles
SKOV-3 cells were chosen for further study based on the results of cell viability test and FACs analysis. And SKOV-3 cells which have multiple characteristics of high grade serous, clear cell and endometrioid types were considered appropriate for a comprehensive view of anti-cancer effect of cordycepin across different ovarian cancer cell types. To assess the putative genes involved in cordycepin induced anti-tumor activity, we performed a gene expression microarray using SKOV-3 cancer cells treated with cordycepin. Among the 63,242 unique genes (using Agilent’s Human GE 8 × 60 K Microarray) tested, 30,858 genes were expressed in cells treated with 100 μg/mL of cordycepin. Among these 30,858 genes, 2561 and 1942 genes were upregulated and downregulated, respectively, by treatment with 100 μg/mL cordycepin compared to the untreated control at 48 h. Genes upregulated or downregulated by a 2-fold were further analyzed. Biologically relevant features were constructed by using the Database for Annotation, Visualization, and Integrated Discovery (DAVID) tools (http://david.abcc.ncifcrf.gov/). Gene lists corresponding to 2-fold up- and downregulation in the cordycepin-induced ovarian cancer cells were uploaded to DAVID for Gene Ontology analysis (Fig. 2a). To compare the results obtained upon cordycepin treatment with putative genes that belonged in apoptosis, we clarified candidate genes by the GeneCards database (http://www.genecards.org/) (Fig. 2b). The intersection obtained by hierarchical clustering is showed along with the gene lists in Fig. 2c. The signaling network of genes in response to apoptosis induced by cordycepin is presented in Fig. 2d.
a Results of a gene ontology analysis by using microarray approaches in response to 100 μg/mL of cordycepin. Gene lists corresponding to two-fold upregulation and downregulation in cordycepin-treated SKOV-3 cells for 48 h were developed by using DAVID for Gene Ontology analysis (http://david.abcc.ncifcrf.gov/). b Immune response- and apoptosis-related genes and their hierarchical clustering in response to cordycepin. A dendrogram of hierarchical clustering revealed genes that were altered more than two-fold owing to apoptosis in response to cordycepin. c Gene lists (>2-fold, <2-fold, and apoptosis-related genes) are shown and are intersected individually by using Venn diagrams. d Signaling network of the immune response- and apoptosis-related genes in response to cordycepin. Nodes colored by using a Qiagen IPA were the genes in the apoptosis regulatory network in the cordycepin-treated SKOV-3 cells (red: upregulated genes, green: downregulated genes)
Cordycepin downregulates AKT/NF-κB signaling pathway and upregulates cleaved caspase-3
Next, to investigate whether cordycepin suppresses the NF-κB signaling pathway, the expression of pro-inflammatory chemokine CCL5, IκB, NF-κB, c-FLIPL, Bax, cleaved PARP-1, and cleaved caspase-3 was determined in SKOV-3 cells exposed to cordycepin (Fig. 3a). Western blot analysis was used to detect the expression of NF-κB, c-FLIPL, Bax, cleaved PARP-1, and cleaved caspase-3 in SKOV-3 cells cultured in the presence of 60 and 100 μg/mL of cordycepin. As shown in Fig. 3a, increasing amounts of cordycepin resulted in a dose-dependent reduction in CCL5 expression, whereas NF-κB expression changed slightly at 60 μg/mL cordycepin, and decreased at 100 μg/mL cordycepin. Meanwhile, cordycepin treatment resulted in increased expression of cleaved caspase-3. As cordycepin induced apoptosis in ovarian cancer cells, it was appropriate to assess which downstream effectors mediated this process. NF-κB is a transcription factor that plays a important role in cytokine- and LPS-induced gene activation during inflammatory events26. This suggests that cordycepin blocked the inflammation-related signaling pathway such as CCL5, Akt/NF-κB, c-FLIPL, and upregulated caspase-3 activation. The expression levels of inflammation-related proteins in SKOV-3 cells treated by TNF-α and cordycepin were determined by using western blotting assay. TNF-α is a cytokine that induces inflammation in ovarian cancer cells. Thus, we investigated the important role of cordycepin in TNF-α-treated SKOV-3 cells. Inflammation-related proteins such as Akt, IκBα, nuclear NF-κB, and c-FLIPL were activated by TNF-α in SKOV-3 cells, while effectively downregulated by cordycepin in TNF-α treated SKOV-3 cells (Fig. 3b). The cordycepin (100 μg/mL) significantly decreased the expression of phosphorylated AKT and nuclear NF-κB increased by TNF-α treatment. These results indicate that cordycepin downregulated the TNF-α-mediated Akt/NF-κB/ c-FLIPL signaling pathway in ovarian cancer cells.
a Western blots showing the expression of NF-κB-p65, IκB, CCL5, c-FLIPL, caspase-3, and cleaved caspase-3 in response to 0, 60, and 100 μg/mL of cordycepin in SKOV-3 cells. b Effect of TNF-α on NF-kB was observed in SKOV-3 cells exposed to cordycepin. Western blot analysis was used to detect the expression of p-IκB, IκB, cytosolic NF-κB-p65, nuclear NF-κB-p65, p-Akt, and Akt in SKOV-3 cells cultured in the presence of 100 μg/mL of cordycepin. After 24 h, cells were challenged with 10 ng/mL of TNF-α for 24 h alone or in the presence of cordycepin. Data are expressed as means ± SD, * p < 0.05, ** p < 0.01, *** p < 0.001
Cordycepin inhibits Akt/NF-κB signaling pathway through CCL5 and reduces migration
We detected CCL5 expression levels of SKOV-3 cells treated by TNF-α and cordycepin using western blotting assay (Fig. 4a). A significant (p < 0.05) increase of CCL5 release was observed in TNF-α-treated SKOV-3 cells. Meanwhile, the release of CCL5 was effectively downregulated by cordycepin treatment of TNF-α-treated SKOV-3 cells (Fig. 4a). Akt activation was also downregulated by cordycepin and a significant (p < 0.05) decrease of Akt activation was observed in SKOV-3 cells treated with CCL5 siRNA (Fig. 4b); whereas CCL5 overexpression increased Akt activation in cordycepin-treated SKOV-3 cells (Fig. 4c). These results indicated that cordycepin inhibits CCL5-mediated Akt signaling pathway in ovarian cancer cells. Next, we investigated the effect of Akt on NF-κB through cordycepin-regulated CCL5 in ovarian cancer cells. CCL5 effectively enhanced Akt phosphorylation in SKOV-3 cells. When Akt was silenced, cordycepin treatment of both SKOV-3 cells and SKOV-3 cells overexpressing CCL5 decreased nuclear translocation of NF-κB, whereas Akt overexpression increased nuclear translocation of NF-κB in cordycepin-treated SKOV-3 cells (Fig. 4d). To evaluate the potential biological relevance of the regulatory effect of cordycepin, we assessed the effect of CCL5 on the migration of tumor cells. Migration of SKOV-3 cells was measured by using a wound-healing assay. CCL5 overexpression increased cancer cell migration. The treatment of cordycepin (100 μg/mL) significantly suppressed the cell migration in both CCL5-overexpressed and control vector-transfected cancer cells in a dose-dependent manner at 48 h (Fig. 4e). These results indicate that cordycepin attenuates CCL5-mediated Akt/NF-κB phosphorylation to downregulate SKOV-3 cell migration.
a Western blot analysis was used to detect the expression of CCL5 in SKOV-3 cells cultured in the presence of 100 μg/mL cordycepin. After 24 h, cells were challenged with 10 ng/mL of TNF-α for 24 h alone or in the presence of cordycepin. b SKOV-3 cells were treated for 24 h with cordycepin. After 24 h, cells were challenged with siRNA directed against CCL5 (si-CCL5) for 48 h alone or in the presence of cordycepin. c SKOV-3 cells were treated with cordycepin for 6 h, and the expression of CCL5, p-Akt and Akt was measured by western blotting assay. d SKOV-3 cells were treated with cordycepin for 6 h, then the medium was collected 24 h later and the secretion of cytosol and nuclear NF-κB was measured. SKOV-3 cells were incubated with siRNA directed against Akt (si-Akt) or negative control siRNA for 48 h, transfected with Akt and CCL-5-overexpression constructs for 48 h. Representative western blots were shown from 3 independent experiments. e Microscopic images demonstrating the results of the in vitro migration of SKOV-3 and CCL5-overexpressed and knock-downed SKOV-3 cells by using the simple scratch technique of migration assay. Data are expressed as means ± SD, *p < 0.05, **p < 0.01, ***p < 0.001
Cordycepin-mediated Akt/NF-κB/c-FLIPL signaling inhibition activates JNK to induce caspase-3 activation
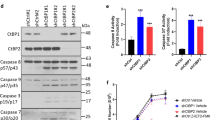
To further investigate whether Akt/NF-κB is functionally linked to JNK signaling, we examined the effect of Akt/NF-κB on JNK. Cordycepin enhanced the expression of p-JNK. Next, we performed a loss-of-function experiment using NF-κB knockdown by cordycepin and the NF-κB inhibitor, PDTC. Both cordycepin and PDTC attenuated c-FLIPL and enhanced p-JNK, and the selective JNK inhibitor, SP600125 (20 μM) reduced the phosphorylation of JNK and the expression of Bax. Immunoblots confirmed the reduction in p-JNK protein in SKOV-3 cells, whereas cordycepin enhanced the p-JNK protein-mediated increase in Bax and cleaved caspase-3 (Fig. 5a). Also, siRNA mediated c-FLIP inhibition increased JNK (Fig. 5b). These results indicate that JNK signaling promotes SKOV-3 cell apoptosis by enhancing caspase-3 activation. Taken together, these results indicate that cordycepin-mediated NF-κB inhibition upregulated p-JNK, leading to the upregulation of cleaved caspase-3. These results indicate that cordycepin suppresses the activation of Akt/NF-κB signaling pathway and c-FLIPL activation, which results in p-JNK upregulation and the subsequent induction of caspase-3 activation.
a SKOV-3 cells were treated with PDTC and SP600125 for 1 h, then cultured with cordycepin. The expression of NF-κB-p65, c-FLIPL, JNK, p-JNK and cleaved caspase-3 was analyzed by western blotting assay. b Cells were treated with si-c-FLIPL for 1 day. The expression of c-FLIPL, JNK and p-JNK. Data are expressed as means ± SD, **p < 0.01, ***p < 0.001
Discussion
Cordycepin has a variety of biological functions, including anti-tumor, antiviral, anti-oxidant, and anti-inflammatory activities1, 13, 27, 28. In the past few years, several reports indicated that cordycepin inhibits the expression of some inflammatory genes by suppressing NF-κB activation29. Meanwhile, other studies reported that cordycepin has anti-cancer and anti-metastatic effects, inhibiting the expression of some critical molecules involved in tumor growth and metastasis by blocking NF-κB activation30, 31.
In the present study, we showed that the CCL5-mediated Akt/NF-κB signaling pathway was involved in human ovarian cancer cells after cordycepin treatment. First, we found that cordycepin-mediated CCL5 inhibition downregulated p-Akt. Second, cordycepin-mediated Akt inactivation by inhibiting CCL5 reduced nuclear NF-κB preceded SKOV-3 ovarian cancer cell apoptosis. Third, cordycepin upregulated p-JNK through the reduction of nuclear NF-κB. Finally, treatment with the JNK inhibitor, SP600125, significantly decreased Bax and cleaved caspase-3. It has been reported that CCL5 acts through PI3K/Akt, which in turn activates IKKα/β and NF-κB, resulting in the activation of αvβ3 integrin and contributing to the migration of human lung cancer cells32. Therefore, to investigate whether cordycepin inhibits CCL5-mediated Akt/NF-κB signaling pathway, we detected CCL5 expression level in SKOV-3 cells treated by TNF-α and cordycepin using western blotting assay (Fig. 4a). CCL5 was increased in TNF-α treated SKOV-3 cells, while it was downregulated by cordycepin in TNF-α treated SKOV-3 cells (Fig. 4a). Akt activation was also downregulated by cordycepin and CCL5 siRNA in SKOV-3 cells (Fig. 4b); whereas CCL5 overexpression increased Akt activation in cordycepin-treated SKOV-3 cells (Fig. 4c). These results indicate that cordycepin could inhibit CCL5-mediated Akt signaling pathway in ovarian cancer cells.
A functional link exists between the Akt and NF-κB pathways and the Akt signaling pathway actively regulates NF-κB33, 34. Therefore, we investigated the effect of Akt on NF-κB through cordycepin-regulated CCL5 in ovarian cancer cells. Our results demonstrated that, when Akt was silenced, cordycepin decreased nuclear translocation of NF-κB, in SKOV-3 cells and SKOV-3 cells overexpressing CCL5, while Akt overexpression increased nuclear translocation of NF-κB in cordycepin-treated SKOV-3 cells (Fig. 4d), suggesting that cordycepin inhibits Akt/NF-κB signaling pathway through CCL5. FLIP is an important mediator of NF-kappaB-controlled antiapoptotic signals35.
We undertook a series of experiments to investigate whether c-FLIP is implicated in the antiapoptotic NF-κB response. Generally, activation of JNK is involved in the induction of apoptosis36, whereas c-FLIP exerts other cellular functions including increased cell proliferation and tumorigenesis37, 38.
Therefore, we evaluated the fundamental role of c-FLIPL in the regulation of JNK signaling in SKOV-3 cells treated with cordycepin. c-FLIPL was dramatically decreased, whereas JNK activation was increased in both PDTC- and cordycepin-treated SKOV-3 cells, whereas NF-κB activation was decreased and caspase-3 activation was increased (Fig. 5a). To further investigate the role of JNK in cordycepin-mediated apoptosis, we used the JNK inhibitor, SP600125, (20 μM) (Fig. 5a). SP600125 decreased the levels of Bax and cleaved caspase-3 in cordycepin-treated SKOV-3 cells. Also, siRNA mediated c-FLIP inhibition increased JNK (Fig. 5b). These results indicate that c-FLIPL may therefore play a key role in the NF-κB-mediated control of death signals and that cordycepin augments Bax and caspase-3 activation through the activation of JNK by inhibiting NF-κB induced c-FLIPL signaling.
We showed that the protein level of p-JNK was dramatically increased by negative regulation of CCL5-mediated Akt/NF-κB expression in cordycepin-treated SKOV-3 cells. We demonstrated that JNK is a critical mediator of cordycepin-induced SKOV-3 cell apoptosis. These findings provide novel insights into the molecular mechanisms of SKOV-3 cell apoptosis. Therefore, controlling CCL5 expression may provide new ways and strategies to enhance SKOV-3 cell apoptosis. Taken together, our results demonstrate that cordycepin mediates Bax’s apoptotic regulation of NF-κB by downregulating CCL5. Also, this systematic investigation study shows the precise molecular mechanism of NF-κB signaling pathway induced by cordycepin and reveals role and potential therapeutic use of cordycepin to inhibit migration in the treatment of ovarian cancer.
Materials and methods
Reagents and chemicals
Fetal bovine serum (FBS), 1% (w/v) penicillin-streptomycin and phosphate-buffered saline (PBS) were obtained from Thermo (Paisley, Scotland, UK). Dulbecco’s Modified Eagle’s Medium (DMEM) was purchased from Sigma-Aldrich (St Louis, MO, USA). Cordycepin (3′-Deoxyadenosine, from Cordyceps militaris), Pyrrolidine dithiocarbamate (PDTC), and SP600125 were purchased from Sigma-Aldrich. TNF-α was purchased from R & D system (R&D Systems, Minneapolis, MN, USA). A Muse Annexin & Dead Cell kit was from Millipore (Millipore, Billerica, MA, USA). Whole cell lysis buffer was purchased from Intron (Seoul, Korea), and transfection reagent Hilymax and CCK-8 were from Dojindo (Dojindo, Japan). Antibodies against CCL5, NF-κB, p-IκB, IκB, p65, p-Akt, Akt, caspase-3, and β-actin were purchased from Cell Signaling (Beverly, MA, USA). Antibodies against PARP-1 and Bax were from Santa Cruz Biotechnology (Dallas, TX, USA).
Cell lines and cell viability assay
The human ovarian-carcinoma-cell line (SKOV-3, MDAH-2774, OVCAR-3) were obtained from the American Type Culture Collection (Rockville, MD, USA). Cells were grown in DMEM medium, supplemented with 10% (v/v) FBS and 1% (w/v) penicillin-streptomycin at 37 °C with 5% (v/v) CO2. Cells (5 × 103 of cells per well) were placed in a 96-well plate. After a 24-h incubation, the cells were treated with cordycepin for 48 h. Cell viability assays were performed as previously described39. In brief, at the end of the treatment, 10 μL of cell-counting kit-8 solution were added to the cell solution and incubated for 1 h at 37 °C. Cell viability was determined by using a microplate reader (Sunrise, Tecan, Switzerland) to measure the absorbance at 450 nm. The assays were performed in triplicate. The appropriate dose was determined by evaluating the cytotoxicity of cordycepin for 48 h.
Cell apoptosis assay
To detect the effect of cordycepin on apoptosis, we analyzed the Muse Annexin V & Dead Cell reagent (Millipore) following the user’s guide and the manufacturer’s instructions. Briefly, cells were treated with cordycepin for 48 h, harvested with trypsin-EDTA and washed twice in PBS. The cell suspension was centrifuged at 2000 rpm for 2 min and 1 × 106 of cells were transferred in suspension with fresh medium containing serum to a new tube Staining protocol included warming the Muse Annexin V and Dead Cell Reagent to room temperature, addition of 100 μL of cells in suspension to each tube, addition of 100 μL of the Muse Annexin V and Dead Cell Reagent to each tube. Measurements were conducted by using an Muse Cell Analyzer (Millipore, Billerica, MA, USA). The statistics were shown the percentages of the cells represented by alive, apoptosis and dead population.
Microarray analysis
For the microarray analysis of the cordycepin-treated SKOV-3 cancer cells, a human twin 44 K cDNA chip was used for the transcription profiling analysis. Total RNA was extracted from vehicle- or 100 μg/mL cordycepin-treated SKOV-3 cancer cells, and cDNA probes were prepared by using reverse transcription of 50 mg of RNA in the presence of aminoallyl dUTP, followed by coupling with Cy3 dye (vehicle) or Cy5 dye (cordycepin-treated). Genes were considered differentially expressed when, after a significance analysis of the microarray (SAM), the global M value, log2 (R/G), exceeded |1.0| (twofold) with a P-value < 0.05. A Student’s t-test was applied to assess the statistical significance of the differential expression of genes after cordycepin treatment. In order to analyze the biological significance of the changes, we categorized the array data into specific gene groups.
Ontology-related network analysis
To study the biological functions of ontology-related regulated genes and proteins through their interaction network, we conducted a bioinformatic network analysis by using an ingenuity pathway analysis (IPA, http://www.ingenuity.com). The IPA identifies a gene interaction network based on a regularly-updated “Ingenuity Pathways Knowledge-base.” The updatable database was retrieved from the biological literature. Network generation was optimized from the inputted expression profile when possible and aimed at the production of highly connected networks.
Fractionation and protein extraction
SKOV-3 cells were incubated with cordycepin for 2 days. The cells were collected with 2 mL of homogenization buffer A (25 mM Tris (pH 7.5), 2 mM EDTA, 0.5 mM EGTA, 1 mM DTT, protease inhibitor cocktail, 1 mM PMSF, and 0.02% Triton X-100) per culture dish, homogenized 15 times using a 15-mL Dounce homogenizer with pestle A, and centrifuged at 100,000 × g for 30 min. The supernatant cytosolic fraction was transferred into a new tube and 500 μL of homogenization buffer B (homogenization buffer A containing 1% Triton X-100) was added to the pellet. The pellet was resuspended by sonication, incubated for 30 min at 4 °C by rocking, and centrifuged at 100,000 × g for 30 min. The supernatant nuclear fraction was transferred into a fresh tube. The samples were prepared for protein analysis by western blotting.
Western blot analysis
The expression of cordycepin-induced apoptosis-related signaling proteins was examined by using western blotting, as described previously40. In brief, 25 μg of the denatured protein was separated by using 12% polyacrylamide gel electrophoresis and transferred onto a nitrocellulose membrane. The nitrocellulose membrane was then stained with Ponceau S to position the proteins. The blotted membrane was blocked for 1 h with 5% (w/v) skimmed milk in TTBS (Tween-20 and Tris-buffered saline), followed by incubation with diluted primary antibodies, CCL5 (1:200), NF-κB(1:1000), p-IκB (1:500), IκB (1:500), p65 (1:1000), p-Akt (1:500), Akt (1:1000), caspase-3 (1:200), Bax (1:1000), PARP (1:1000), and β-actin (1:2000), at room temperature for 2 h or at 4 °C overnight. The membrane was washed three times for 5 min each time with 0.1% (v/v) Tween-20 in TBS before incubation with horseradish-peroxidase (HRP)-conjugated goat anti-mouse IgG or HRP-conjugated rabbit anti-goat IgG with a 1:2000 dilution in TBS containing 5% (w/v) skimmed milk at room temperature for 1 h. The membranes were rinsed three times with TTBS for 5 min each, and an enhanced chemiluminescence system (Thermo Scientific, San Jose, CA, USA) was used to visualize the bands on a ChemiDoc MP system (Bio-Rad, Hercules, CA, USA). Densitometric measurements of bands were performed by using Image J software (National Institutes of Health, Bethesda, MD, USA). The expression levels of proteins were quantitatively analyzed through comparison with actin used as an internal control.
Biochemical analysis
CCL5 and AKT were overexpressed by using lentivirus (LV)-carrying RFP-conjugated full-length CCL5 or Akt (Lenti H1.4-ccl5/RFP, Lenti H1.4-Akt/RFP, Bioneer Corp., Daejeon, Korea). Small interfering RNAs (siRNAs) were purchased from ST Pharm (Seoul, Korea). The nucleotide sequence for Akt siRNAs was 5′-CGU UCU GCU GCG ACA AUG A-3′ and CCL5 siRNA (NM_001278736.1) was 5′-AAG GAA GUC AGC AUG CCU CUA-3′. SKOV-3 cells were seeded (2 × 105 cells/6-well plates). After incubation, the cells were supplied with growth medium containing 10% FBS and were harvested 48 h later for further assays. siRNAs were transfected in SKOV-3 cells using lipofectamine RNAiMAX reagent (Invitrogen, Carlsbad, CA, USA) following the manufacturer’s instructions. Cells were then treated with 60 μg/mL of cordycepin for 48 h.
Migration assay
The migration assay was conducted by using control RFP-vector-transfected and CCL5-RFP transfected cells. Cells were seeded into a 24-well plate. The cell monolayer was scraped with a pipette tip to create a wound. The cells were treated with 100 μg/mL of cordycepin for 48 h. The plates were imaged using the TissueFAXS system (TissueGnostics, Vienna, Austria). Cell migration was analyzed (quantification of the “healed” area and migrated cells) was performed with the HistoQuest software (TissueGnostics). Samples were analyzed with a Zeiss AxioImagerZ1 microscope system with a charge-coupled device camera and the TissueFAXSTM automated acquisition system (TissueGnostics). The percentages of antibody-positive and stemness marker-positive tumors were determined and depicted as scattergrams. Images were digitalized and protein expression was quantified. Statistical flow analysis was performed with the HistoQuestTM software (TissueGnostics).
Statistical analyses
GraphPad Prism software (GraphPad, San Diego, CA, USA) was used for the statistical analyses. Student’s t-test was used to assess the statistical difference between the control and the MRGX-treated groups. P values < 0.05 were considered statistically significant.
References
Chang, W. et al. Cordycepin inhibits vascular smooth muscle cell proliferation. Eur. J. Pharmacol. 597, 64–69 (2008).
Chen, L. S., Stellrecht, C. M. & Gandhi, V. RNA-directed agent, cordycepin, induces cell death in multiple myeloma cells. Br. J. Haematol. 140, 682–391 (2008).
Kim, H. G. et al. Cordycepin inhibits lipopolysaccharide-induced inflammation by the suppression of NF-kappaB through Akt and p38 inhibition in RAW 264.7 macrophage cells. Eur. J. Pharmacol. 545, 192–199 (2006).
Nakamura, K. et al. Antitumor effect of cordycepin (3′-deoxyadenosine) on mouse melanoma and lung carcinoma cells involves adenosine A3 receptor stimulation. Anticancer Res. 26, 43–47 (2006).
Shi, P., Huang, Z., Tan, X. & Chen, G. Proteomic detection of changes in protein expression induced by cordycepin in human hepatocellular carcinoma BEL-7402 cells. Methods Find. Exp. Clin. Pharmacol. 30, 347–353 (2008).
Thomadaki, H., Scorilas, A., Tsiapalis, C. M. & Havredaki, M. The role of cordycepin in cancer treatment via induction or inhibition of apoptosis: implication of polyadenylation in a cell type specific manner. Cancer Chemother. Pharmacol. 61, 251–265 (2008).
Wehbe-Janek, H., Shi, Q. & Kearney, C. M. Cordycepin/Hydroxyurea synergy allows low dosage efficacy of cordycepin in MOLT-4 leukemia cells. Anticancer Res. 27, 3143–3146 (2007).
Wu, W. C., Hsiao, J. R., Lian, Y. Y., Lin, C. Y. & Huang, B. M. The apoptotic effect of cordycepin on human OEC-M1 oral cancer cell line. Cancer Chemother. Pharmacol. 60, 103–111 (2007).
Zhou, X., Meyer, C. U., Schmidtke, P. & Zepp, F. Effect of cordycepin on interleukin-10 production of human peripheral blood mononuclear cells. Eur. J. Pharmacol. 453, 309–317 (2002).
de Julian-Ortiz, J. V., Galvez, J., Munoz-Collado, C., Garcia-Domenech, R. & Gimeno-Cardona, C. Virtual combinatorial syntheses and computational screening of new potential anti-herpes compounds. J. Med. Chem. 42, 3308–3314 (1999).
Sugar, A. M. & McCaffrey, R. P. Antifungal activity of 3’-deoxyadenosine (cordycepin). Antimicrob. Agents Chemother. 42, 1424–1427 (1998).
Pao, H. Y., Pan, B. S., Leu, S. F. & Huang, B. M. Cordycepin stimulated steroidogenesis in MA-10 mouse Leydig tumor cells through the protein kinase C Pathway. J. Agric. Food Chem. 60, 4905–4913 (2012).
Yao, W. L. et al. Cordycepin suppresses integrin/FAK signaling and epithelial-mesenchymal transition in hepatocellular carcinoma. Anticancer Agents Med. Chem. 14, 29–34 (2014).
Choi, S. et al. Cordycepin-induced apoptosis and autophagy in breast cancer cells are independent of the estrogen receptor. Toxicol. Appl. Pharmacol. 257, 165–173 (2011).
Lee, S. Y., Debnath, T., Kim, S. K. & Lim, B. O. Anti-cancer effect and apoptosis induction of cordycepin through DR3 pathway in the human colonic cancer cell HT-29. Food Chem. Toxicol. 60, 439–447 (2013).
Balkwill, F. & Mantovani, A. Inflammation and cancer: back to Virchow? Lancet 357, 539–545 (2001).
Kakinuma, T. & Hwang, S. T. Chemokines, chemokine receptors, and cancer metastasis. J. Leukoc. Biol. 79, 639–651 (2006).
Struyf, S. et al. Diverging binding capacities of natural LD78beta isoforms of macrophage inflammatory protein-1alpha to the CC chemokine receptors 1, 3 and 5 affect their anti-HIV-1 activity and chemotactic potencies for neutrophils and eosinophils. Eur. J. Immunol. 31, 2170–2178 (2001).
Proudfoot, A. E. et al. The BBXB motif of RANTES is the principal site for heparin binding and controls receptor selectivity. J. Biol. Chem. 276, 10620–10626 (2001).
Long, H. et al. Autocrine CCL5 signaling promotes invasion and migration of CD133 + ovarian cancer stem-like cells via NF-kappaB-mediated MMP-9 upregulation. Stem Cells 30, 2309–2319 (2012).
Jotzu, C. et al. Adipose tissue derived stem cells differentiate into carcinoma-associated fibroblast-like cells under the influence of tumor derived factors. Cell Oncol. 34, 55–67 (2011).
Altomare, D. A. & Testa, J. R. Perturbations of the AKT signaling pathway in human cancer. Oncogene 24, 7455–7464 (2005).
Vazquez-Santillan, K., Melendez-Zajgla, J., Jimenez-Hernandez, L., Martinez-Ruiz, G. & Maldonado, V. NF-kappaB signaling in cancer stem cells: a promising therapeutic target? Cell Oncol. 38, 327–339 (2015).
Hoffmann, A. & Baltimore, D. Circuitry of nuclear factor kappaB signaling. Immunol. Rev. 210, 171–186 (2006).
Huang, S., Robinson, J. B., Deguzman, A., Bucana, C. D. & Fidler, I. J. Blockade of nuclear factor-kappaB signaling inhibits angiogenesis and tumorigenicity of human ovarian cancer cells by suppressing expression of vascular endothelial growth factor and interleukin 8. Cancer Res. 60, 5334–5339 (2000).
Viatour, P., Merville, M. P., Bours, V. & Chariot, A. Phosphorylation of NF-kappaB and IkappaB proteins: implications in cancer and inflammation. Trends Biochem. Sci. 30, 43–52 (2005).
Nakamura, K., Shinozuka, K. & Yoshikawa, N. Anticancer and antimetastatic effects of cordycepin, an active component of Cordyceps sinensis. J. Pharmacol. Sci. 127, 53–56 (2015).
Tian, X. et al. Apoptosis and inhibition of proliferation of cancer cells induced by cordycepin. Oncol. Lett. 10, 595–599 (2015).
Kim, K. M. et al. Methanol extract of Cordyceps pruinosa inhibits in vitro and in vivo inflammatory mediators by suppressing NF-kappaB activation. Toxicol. Appl. Pharmacol. 190, 1–8 (2003).
Chang, R. Bioactive polysaccharides from traditional Chinese medicine herbs as anticancer adjuvants. J. Altern. Complement. Med. 8, 559–565 (2002).
Ren, Z. et al. Cordycepin suppresses TNF-alpha-induced NF-kappaB activation by reducing p65 transcriptional activity, inhibiting IkappaBalpha phosphorylation, and blocking IKKgamma ubiquitination. Int. Immunopharmacol. 14, 698–703 (2012).
Huang, C. Y. et al. CCL5 increases lung cancer migration via PI3K, Akt and NF-kappaB pathways. Biochem. Pharmacol. 77, 794–803 (2009).
Hussain, A. R. et al. Cross-talk between NFkB and the PI3-kinase/AKT pathway can be targeted in primary effusion lymphoma (PEL) cell lines for efficient apoptosis. PLoS ONE 7, e39945 (2012).
Bai, D., Ueno, L. & Vogt, P. K. Akt-mediated regulation of NFkappaB and the essentialness of NFkappaB for the oncogenicity of PI3K and Akt. Int. J. Cancer 125, 2863–2870 (2009).
Micheau, O., Lens, S., Gaide, O., Alevizopoulos, K. & Tschopp, J. NF-kappaB signals induce the expression of c-FLIP. Mol. Cell. Biol. 21, 5299–5305 (2001).
Dhanasekaran, D. N. & Reddy, E. P. JNK signaling in apoptosis. Oncogene 27, 6245–6251 (2008).
Safa, A. R. & Pollok, K. E. Targeting the anti-apoptotic protein c-FLIP for cancer therapy. Cancers 3, 1639–1671 (2011).
Bagnoli, M., Canevari, S. & Mezzanzanica, D. Cellular FLICE-inhibitory protein (c-FLIP) signalling: a key regulator of receptor-mediated apoptosis in physiologic context and in cancer. Int. J. Biochem. Cell Biol. 42, 210–213 (2010).
Stockert, J. C., Blazquez-Castro, A., Canete, M., Horobin, R. W. & Villanueva, A. MTT assay for cell viability: intracellular localization of the formazan product is in lipid droplets. Acta Histochem. 114, 785–796 (2012).
Hwang, J. W. et al. Lactobacillus casei extract induces apoptosis in gastric cancer by inhibiting NF-kappaB and mTOR-mediated signaling. Integr. Cancer Ther. 12, 165–173 (2013).
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea funded by the Ministry of Education (2014R1A1A2058114, 2015R1D1A1A01058744, and 2017R1D1A1B03034936). We thank our colleague Prof. Seong-Hoon Lee (Yongin University) for helpful discussion. We thank Editage for English language editing.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Edited by A. Rufini
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cui, Z.Y., Park, S.J., Jo, E. et al. Cordycepin induces apoptosis of human ovarian cancer cells by inhibiting CCL5-mediated Akt/NF-κB signaling pathway. Cell Death Discovery 4, 62 (2018). https://doi.org/10.1038/s41420-018-0063-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-018-0063-4
This article is cited by
-
Based on network pharmacology and molecular docking to explore the potential mechanism of shikonin in periodontitis
BMC Oral Health (2024)
-
Redefining bioactive small molecules from microbial metabolites as revolutionary anticancer agents
Cancer Gene Therapy (2024)
-
Cordycepin from Cordyceps militaris ameliorates diabetic nephropathy via the miR-193b-5p/MCL-1 axis
Chinese Medicine (2023)
-
A novel complementary pathway of cordycepin biosynthesis in Cordyceps militaris
International Microbiology (2023)
-
Cordyceps militaris induces apoptosis in ovarian cancer cells through TNF-α/TNFR1-mediated inhibition of NF-κB phosphorylation
BMC Complementary Medicine and Therapies (2020)