Abstract
Background
The early-onset rectal cancer with rapidly increasing incidence is considered to have distinct clinicopathological and molecular profiles with high-risk features. This leads to challenges in developing specific treatment strategies for early-onset rectal cancer patients and questions of whether early-onset locally advanced rectal cancer (LARC) needs aggressive neoadjuvant treatment.
Methods
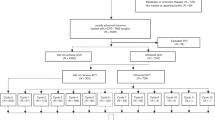
In this post hoc analysis of FOWARC trial, we investigated the role of preoperative radiation in early-onset LARC by comparing the clinicopathological profiles and short-term and long-term outcomes between the early-onset and late-onset LARCs.
Results
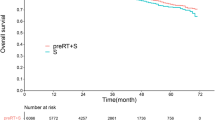
We revealed an inter-tumor heterogeneity of clinical profiles and treatment outcomes between the early-onset and late-onset LARCs. The high-risk features were more prevalent in early-onset LARC. The neoadjuvant radiation brought less benefits of tumor response and more risk of complications in early-onset group (pCR: OR = 3.75, 95% CI = 1.37–10.27; complications: HR = 11.35, 95% CI = 1.46–88.31) compared with late-onset group (pCR: OR = 5.33, 95% CI = 1.83–15.58; complications: HR = 5.80, 95% CI = 2.32–14.49). Furthermore, the addition of radiation to neoadjuvant chemotherapy didn’t improve long-term OS (HR = 1.37, 95% CI = 0.49–3.87) and DFS (HR = 1.05, 95% CI = 0.58–1.90) for early-onset patients.
Conclusion
Preoperative radiation plus chemotherapy may not be superior to the chemotherapy alone in the early-onset LARC. Our findings provide insight into the treatment of early-onset LARC by interrogating the aggressive treatment and alternative regimens.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Sinicrope FA. Increasing incidence of early-onset colorectal cancer. N Engl J Med. 2022;386:1547–58.
Xie Y, Shi L, He X, Luo Y. Gastrointestinal cancers in China, the USA, and Europe. Gastroenterol Rep. 2021;9:91–104.
Sung H, Siegel RL, Rosenberg PS, Jemal A. Emerging cancer trends among young adults in the USA: analysis of a population-based cancer registry. Lancet Public Health. 2019;4:e137–47.
Stoffel EM, Murphy CC. Epidemiology and mechanisms of the increasing incidence of colon and rectal cancers in young adults. Gastroenterology. 2020;158:341–53.
Siegel RL, Wagle NS, Cercek A, Smith RA, Jemal A. Colorectal cancer statistics, 2023. CA Cancer J Clin. 2023;73:233–54.
Bailey CE, Hu CY, You YN, Bednarski BK, Rodriguez-Bigas MA, Skibber JM, et al. Increasing disparities in the age-related incidences of colon and rectal cancers in the United States, 1975-2010. JAMA Surg. 2015;150:17–22.
Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17–48.
Shen D, Wang P, Xie Y, Zhuang Z, Zhu M, Wang X, et al. Clinical spectrum of rectal cancer identifies hallmarks of early-onset patients and next-generation treatment strategies. Cancer Med. 2023;12:3433–41.
Ugai T, Sasamoto N, Lee HY, Ando M, Song M, Tamimi RM, et al. Is early-onset cancer an emerging global epidemic? Current evidence and future implications. Nat Rev Clin Oncol. 2022;19:656–73.
Collaborative REACCT, Zaborowski AM, Abdile A, Adamina M, Aigner F, d’Allens L, et al. Characteristics of early-onset vs late-onset colorectal cancer: a review. JAMA Surg. 2021;156:865–74.
Kneuertz PJ, Chang GJ, Hu C-Y, Rodriguez-Bigas MA, Eng C, Vilar E, et al. Overtreatment of young adults with colon cancer: more intense treatments with unmatched survival gains. JAMA Surg. 2015;150:402–9.
Manjelievskaia J, Brown D, McGlynn KA, Anderson W, Shriver CD, Zhu K. Chemotherapy use and survival among young and middle-aged patients with colon cancer. JAMA Surg. 2017;152:452–9.
Kolarich A, George TJ, Hughes SJ, Delitto D, Allegra CJ, Hall WA, et al. Rectal cancer patients younger than 50 years lack a survival benefit from NCCN guideline-directed treatment for stage II and III disease. Cancer. 2018;124:3510–9.
Cavestro GM, Mannucci A, Balaguer F, Hampel H, Kupfer SS, Repici A, et al. Delphi initiative for early-onset colorectal cancer (DIRECt) international management guidelines. Clin Gastroenterol Hepatol. 2023;21:581–603.e33.
Benson AB, Venook AP, Al-Hawary MM, Azad N, Chen YC, Ciombor KK, et al. NCCN clinical practice guidelines in oncology: rectal cancer. Version 1.2023. 2023.
Yanhong D, Pan C, Ping L, Lei W, Weiqing C, Long C, et al. Modified FOLFOX6 with or without radiation versus fluorouracil and leucovorin with radiation in neoadjuvant treatment of locally advanced rectal cancer: initial results of the Chinese FOWARC multicenter, open-label, randomized three-arm phase III trial. J Clin Oncol. 2016;34:3300–7.
Yanhong D, Pan C, Ping L, Lei W, Weiqing C, Long C, et al. Neoadjuvant modified FOLFOX6 with or without radiation versus fluorouracil plus radiation for locally advanced rectal cancer: final results of the Chinese FOWARC trial. J Clin Oncol. 2019;37:3223–33.
Schrag D, Shi Q, Weiser MR, Gollub MJ, Saltz LB, Musher BL, et al. Preoperative treatment of locally advanced rectal cancer. N Engl J Med. 2023;389:322–34.
Trakarnsanga A, Gonen M, Shia J, Nash GM, Temple LK, Guillem JG, et al. Comparison of tumor regression grade systems for locally advanced rectal cancer after multimodality treatment. J Natl Cancer Inst. 2014;106:dju248.
Bliggenstorfer JT, Bingmer K, Ofshteyn A, Stein SL, Charles R, Steinhagen E. Neoadjuvant radiation above NCCN guidelines for rectal cancer is associated with age under 50 and early clinical stage. Surg Endosc. 2022;36:2925–35.
Keller DS, Berho M, Perez RO, Wexner SD, Chand M. The multidisciplinary management of rectal cancer. Nat Rev Gastroenterol Hepatol. 2020;17:414–29.
Khan SA, Morris M, Idrees K, Gimbel MI, Rosenberg S, Zeng Z, et al. Colorectal cancer in the very young: a comparative study of tumor markers, pathology and survival in early onset and adult onset patients. J Pediatr Surg. 2016;51:1812–7.
Himbert C, Figueiredo JC, Shibata D, Ose J, Lin T, Huang LC, et al. Clinical characteristics and outcomes of colorectal cancer in the ColoCare study: differences by age of onset. Cancers. 2021;13:3817.
Yoshiro I, Kenji K, Yoshiharu S. Treatment of elderly patients with colorectal cancer. BioMed Res Int. 2018;2018:2176056.
Meng L, Thapa R, Delgado MG, Gomez MF, Ji R, Knepper TC, et al. Association of age with treatment-related adverse events and survival in patients with metastatic colorectal cancer. JAMA Netw Open. 2023;6:e2320035.
Shen D, Wang X, Wang H, Xu G, Xie Y, Zhuang Z, et al. Current surveillance after treatment is not sufficient for patients with rectal cancer with negative baseline CEA. J Natl Compr Cancer Netw. 2022;20:653–62.e3.
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. Lancet. 2019;394:1467–80.
Vliegen RF, Beets GL, Lammering G, Dresen RC, Rutten HJ, Kessels AG, et al. Mesorectal fascia invasion after neoadjuvant chemotherapy and radiation therapy for locally advanced rectal cancer: accuracy of MR imaging for prediction. Radiology. 2008;246:454–62.
Glynne-Jones R, Wyrwicz L, Tiret E, Brown G, Rödel C, Cervantes A, et al. Rectal cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv22–40.
Yojiro H, Kei M, Yutaka S, Yoshinori I, Yoichi A, Tetsuya H, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol. 2020;25:1–42.
Eng C, Jácome AA, Agarwal R, Hayat MH, Byndloss MX, Holowatyj AN, et al. A comprehensive framework for early-onset colorectal cancer research. Lancet Oncol. 2022;23:e116–28.
Burnett-Hartman AN, Lee JK, Demb J, Gupta S. An update on the epidemiology, molecular characterization, diagnosis, and screening strategies for early-onset colorectal cancer. Gastroenterology. 2021;160:1041–9.
Hall WA, Smith JJ. Achieving a cure without total mesorectal excision in rectal adenocarcinoma. J Clin Oncol. 2023;41:173–80.
Garcia-Aguilar J, Patil S, Gollub MJ, Kim JK, Yuval JB, Thompson HM, et al. Organ preservation in patients with rectal adenocarcinoma treated with total neoadjuvant therapy. J Clin Oncol. 2022;40:2546–56.
Eric R, Philippe R, Jean-Jacques T, Alain V, Bernard L, Michel R, et al. Organ preservation for rectal cancer (GRECCAR 2): a prospective, randomised, open-label, multicentre, phase 3 trial. Lancet. 2017;390:469–79.
Smith JJ, Dasari A, Shi Q, Garcia-Aguilar J, Sanoff HK, George TJ, et al. Alliance A022104/NRG-GI010: a randomized phase II trial testing the efficacy of triplet versus doublet chemotherapy to achieve clinical complete response in patients with locally advanced rectal cancer—the Janus Rectal Cancer trial. 2023;41:TPS3640-TPS3640.
Funding
This work was supported by the Project 5010 of Clinical Medical Research of Sun Yat-sen University-5010 Cultivation Foundation (No. 2018026, YL), the Sixth Affiliated Hospital of Sun Yat-sen University Clinical Research-“1010” Program (MH; YL), the National Natural Science Foundation of China (No. 81972245, YL; No. 82173067, YL; No. 81902877, HY; No. 82272965, HY), the Natural Science Foundation of Guangdong Province (No. 2022A1515012656, HY; No. 2021A1515010134, MH; No. 2020A1515010036, XL), the Science and Technology Program of Guangzhou (202201011004, HY), the Scientific Research Project of the Sixth Affiliated Hospital of Sun Yat-Sen University (2022JBGS07), the Talent Project of the Sixth Affiliated Hospital of Sun Yat-sen University (No. P20150227202010251, YL), the Excellent Talent Training Project of the Sixth Affiliated Hospital of Sun Yat-sen University (No. R2021217202512965, YL), the Fundamental Research Funds for the Central Universities, Sun Yat-sen University (No. 23ykbj007, HY), the Program of Introducing Talents of Discipline to Universities (YL), the Program of Guangdong Provincial Clinical Research Center for Digestive Diseases (2020B1111170004), and National Key Clinical Discipline (2012).
Author information
Authors and Affiliations
Contributions
HY conceived and supervised the project. JC, KL, TL, and JW analyzed and interpreted the data. JC, KL, JW and HL wrote the manuscript. JC, KL, and TL completed the figures and tables. JC, ZY, ZW, and JH collected and collated the data. JL, XL, and XW surveyed reports or published literature. MH, YL, and HY revised the manuscript with approval from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The Institutional Review Board of the Sixth Affiliated Hospital of Sun Yat-sen University reviewed and approved the study protocol.
Consent for publication
Written informed consent was obtained from all subjects or their representatives for the study participation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cai, J., Lin, K., Luo, T. et al. Neoadjuvant chemotherapy is noninferior to chemoradiotherapy for early-onset locally advanced rectal cancer in the FOWARC trial. Br J Cancer (2024). https://doi.org/10.1038/s41416-024-02652-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41416-024-02652-4