Abstract
Background
Tumor cells continue to evolve the metastatic potential in response to signals provided by the external microenvironment during metastasis. Platelets closely interact with tumor cells during hematogenous metastasis and facilitate tumor development. However, the molecular mechanisms underlying this process are not fully understood.
Methods
RNA-sequencing was performed to screen differentially expressed genes mediated by platelets. The effects of platelet and CD39 on tumor metastasis were determined by experimental metastasis models with WT, NCG and CD39−/− mice.
Results
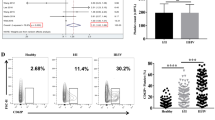
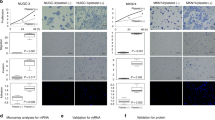
RNA-sequencing results showed that platelets significantly up-regulated CD39 expression in tumor cells. CD39 is a novel immune checkpoint molecule and a key driver of immunosuppression. Our data provided evidence that the expression of CD39 was enhanced by platelets in a platelet-tumor cell contact dependent manner. Although the role of CD39 expressed by immune cells is well established, the effect of CD39 expressed by tumor cells on tumor cell behavior, anti-tumor immunity and tumor metastasis is unclear. We found that CD39 promoted tumor cell invasion, but had no effect on proliferation and migration. Notably, we showed that the ability of platelets to prime tumor cells for metastasis depends on CD39 in the experimental tumor metastasis model. CD39 silencing resulted in fewer experimental metastasis formation, and this anti-metastasis effect was significantly reduced in platelet-depleted mice. Furthermore, overexpression of CD39 in tumor cells promoted metastasis. In order to eliminate the effect of CD39 expressed in cells other than tumor cells, we detected tumor metastasis in CD39−/− mice and obtained similar results. Moreover, overexpression of CD39 in tumor cells inhibited antitumor immunity. Finally, the data from human samples also supported our findings.
Conclusions
Our study shows that direct contact with platelets induces CD39 expression in tumor cells, leading to immune suppression and promotion of metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are available from the corresponding author on reasonable request. The RNA sequencing data have been deposited in the NCBI Sequence Read Archive (SRA) database under the accession number PRJNA1056323 and PRJNA1066901.
References
Yaacoub K, Pedeux R, Tarte K, Guillaudeux T. Role of the tumor microenvironment in regulating apoptosis and cancer progression. Cancer Lett. 2016;378:150–9.
Palumbo, Talmage JS, Massari KE, Jeunesse JV, La CM, Flick MJ, et al. Platelets and fibrin(ogen) increase metastatic potential by impeding natural killer cell-mediated elimination of tumor cells. Blood. 2005;105:178–85.
Placke T, Örgel M, Schaller M, Jung G, Rammensee HG, Kopp HG, et al. Platelet-derived MHC class I confers a pseudonormal phenotype to cancer cells that subverts the antitumor reactivity of natural killer immune cells. Cancer Res. 2012;72:440–8.
Nieswandt B, Hafner M, Echtenacher B, Männel DN. Lysis of tumor cells by natural killer cells in mice is impeded by platelets. Cancer Res. 1999;59:1295–300.
Garner H, de Visser KE. Immune crosstalk in cancer progression and metastatic spread: a complex conversation. Nat Rev Immunol. 2020;20:483–97.
Schmied L, Höglund P, Meinke S. Platelet-Mediated Protection of Cancer Cells From Immune Surveillance - Possible Implications for Cancer Immunotherapy. Front Immunol. 2021;12:640578.
Liu Y, Zhang Y, Ding Y, Zhuang R. Platelet-mediated tumor metastasis mechanism and the role of cell adhesion molecules. Crit Rev Oncol Hematol. 2021;167:103502.
Gay LJ, Brunhilde FH. Contribution of platelets to tumour metastasis. Nat Rev Cancer. 2011;11:123.
Plesner L. Ecto-ATPases: identities and functions. Int Rev Cytol. 1995;158:141–214.
Guo S, Han F, Zhu W. CD39 - A bright target for cancer immunotherapy. Biomed Pharmacother. 2022;151:113066.
Sun P, Zheng X, Li X. The Effects of CD73 on Gastrointestinal Cancer Progression and Treatment. J Oncol. 2022;2022:4330329.
Mandapathil M, Hilldorfer B, Szczepanski MJ, Czystowska M, Szajnik M, Ren J, et al. Generation and accumulation of immunosuppressive adenosine by human CD4+CD25highFOXP3+ regulatory T cells. J Biol Chem. 2010;285:7176–86.
Antonioli L, Pacher P, Vizi ES, Hasko G. CD39 and CD73 in immunity and inflammation. Trends Mol Med. 2013;19:355–67.
Canale FP, Ramello MC, Nunez N, Araujo Furlan CL, Bossio SN, Gorosito Serran M, et al. CD39 Expression Defines Cell Exhaustion in Tumor-Infiltrating CD8(+) T Cells. Cancer Res. 2018;78:115–28.
Borsellino G, Kleinewietfeld M, Di Mitri D, Sternjak A, Diamantini A, Giometto R, et al. Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression. Blood. 2007;110:1225–32.
Gu J, Ni X, Pan X, Lu H, Lu Y, Zhao J, et al. Human CD39hi regulatory T cells present stronger stability and function under inflammatory conditions. Cell Mol Immunol. 2017;14:521–8.
Sun X, Wu Y, Gao W, Enjyoji K, Csizmadia E, Muller CE, et al. CD39/ENTPD1 expression by CD4+Foxp3+ regulatory T cells promotes hepatic metastatic tumor growth in mice. Gastroenterology. 2010;139:1030–40.
Feng L, Sun X, Csizmadia E, Han L, Bian S, Murakami T, et al. Vascular CD39/ENTPD1 directly promotes tumor cell growth by scavenging extracellular adenosine triphosphate. Neoplasia. 2011;13:206–16.
Sun X, Han L, Seth P, Bian S, Li L, Csizmadia E, et al. Disordered purinergic signaling and abnormal cellular metabolism are associated with development of liver cancer in Cd39/ENTPD1 null mice. Hepatology. 2013;57:205–16.
Zhang H, Vijayan D, Li XY, Robson SC, Geetha N, Teng MWL, et al. The role of NK cells and CD39 in the immunological control of tumor metastases. Oncoimmunology. 2019;8:e1593809.
Künzli BM, Rath S, Käser S, Csizmadia E, Enjyoji K, Cowan P, et al. Impact of CD39 and purinergic signalling on the growth and metastasis of colorectal cancer. Purinergic Signal. 2011;7:231–41.
Bastid J, Regairaz A, Bonnefoy N, Déjou C, Giustiniani J, Laheurte C, et al. Inhibition of CD39 enzymatic function at the surface of tumor cells alleviates their immunosuppressive activity. Cancer Immunol Res. 2015;3:254–65.
Li XY, Moesta AK, Xiao C, Nakamura K, Casey M, Zhang H, et al. Targeting CD39 in Cancer Reveals an Extracellular ATP- and Inflammasome-Driven Tumor Immunity. Cancer Discov. 2019;9:1754–73.
Piranlioglu R, Lee E, Ouzounova M, Bollag RJ, Vinyard AH, Arbab AS, et al. Primary tumor-induced immunity eradicates disseminated tumor cells in syngeneic mouse model. Nat Commun. 2019;10:1430.
Labelle M, Begum S, Hynes RO. Direct signaling between platelets and cancer cells induces an epithelial-mesenchymal-like transition and promotes metastasis. Cancer cell. 2011;20:576–90.
Zhang X, Yang Y, Jing L, Zhai W, Zhang H, Ma Q, et al. Pyruvate Kinase M2 Contributes to TLR-Mediated Inflammation and Autoimmunity by Promoting Pyk2 Activation. Front Immunol. 2021;12:680068.
Huang B, Zhao J, Li H, He KL, Chen Y, Chen SH, et al. Toll-like receptors on tumor cells facilitate evasion of immune surveillance. Cancer Res. 2005;65:5009–14.
Flanagan MB, Dabbs DJ, Brufsky AM, Beriwal S, Bhargava R. Histopathologic variables predict Oncotype DX recurrence score. Mod Pathol. 2008;21:1255–61.
Huong PT, Nguyen LT, Nguyen XB, Lee SK, Bach DH. The Role of Platelets in the Tumor-Microenvironment and the Drug Resistance of Cancer Cells. Cancers (Basel). 2019;11:240.
Jackson SW, Hoshi T, Wu Y, Sun X, Enjyoji K, Cszimadia E, et al. Disordered purinergic signaling inhibits pathological angiogenesis in cd39/Entpd1-null mice. Am J Pathol. 2007;171:1395–404.
Li J, Wang L, Chen X, Li L, Zhang Y. CD39/CD73 up-regulation on myeloid-derived suppressor cells via TGF-β-mTOR-HIF-1 signaling in patients with non-small cell lung cancer. Oncoimmunology. 2017;6:e1320011.
Mbeunkui F, Johann DJ Jr. Cancer and the tumor microenvironment: a review of an essential relationship. Cancer Chemother Pharmacol. 2009;63:571–82.
Joyce JA, Pollard JW. Microenvironmental regulation of metastasis. Nat Rev Cancer. 2009;9:239–52.
Yan M, Jurasz P. The role of platelets in the tumor microenvironment: From solid tumors to leukemia. Biochim Biophys Acta. 2016;1863:392–400.
Ballerini P, Dovizio M, Bruno A, Tacconelli S, Patrignani P. P2Y12 Receptors in Tumorigenesis and Metastasis. Front Pharmacol. 2018;9:66.
Shi L, Feng M, Du S, Wei X, Song H, Yixin X, et al. Adenosine Generated by Regulatory T Cells Induces CD8(+) T Cell Exhaustion in Gastric Cancer through A2aR Pathway. Biomed Res Int. 2019;2019:4093214.
Deaglio S, Dwyer KM, Gao W, Friedman D, Usheva A, Erat A, et al. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med. 2007;204:1257–65.
Acknowledgements
We thank the members of Institute of Immunology and Molecular Medicine for discussions and technical help; Wei Wang and Yuzhong Wang for their assistance in tumor tissues collection.
Funding
The present study was funded by the National Natural Science Foundation of China (82003027, 81874169, 82171810), Doctoral Startup Fund of Jining Medical University (2017JYQD24). Grants from Tai Shan Young Scholar Foundation of Shandong Province (tsqn202211234). Project of Shandong Province Higher Educational Youth Innovation Science and Technology Program (2021KJ074).
Author information
Authors and Affiliations
Contributions
Zhaochen Ning led the main research work. Keyan Liu, Hui Zhang and Xiaotong Wang participated in discussions on study design, analysis, and interpretation. Guanjun Dong contributed to revise the manuscript. Huabao Xiong contributed to the overall design and guidance of the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
All experiments involving animals were approved by the Medical Animal Care & Welfare Committee of Jining Medical University. Human tissue samples were acquired following the relevant guidelines and regulations approved by the Ethics Committee of the Affiliated Hospital of Jining Medical University. Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ning, Z., Liu, K., Zhang, H. et al. Platelets induce CD39 expression in tumor cells to facilitate tumor metastasis. Br J Cancer (2024). https://doi.org/10.1038/s41416-024-02640-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41416-024-02640-8