Abstract
Background
Bevacizumab is a beneficial therapy in several advanced cancer types. Predictive biomarkers to better understand which patients are destined to benefit or experience toxicity are needed. Associations between bevacizumab induced hypertension and survival have been reported but with conflicting conclusions.
Methods
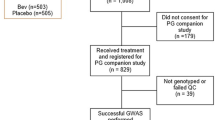
We performed post-hoc analyses to evaluate the association in 3124 patients from two phase III adjuvant breast cancer trials, E5103 and BEATRICE. Differences in invasive disease-free survival (IDFS) and overall survival (OS) between patients with hypertension and those without were compared. Hypertension was defined as systolic blood pressure (SBP) ≥ 160 mmHg (n = 346) and SBP ≥ 180 mmHg (hypertensive crisis) (n = 69). Genomic analyses were performed to evaluate germline genetic predictors for the hypertensive crisis.
Results
Hypertensive crisis was significantly associated with superior IDFS (p = 0.015) and OS (p = 0.042), but only IDFS (p = 0.029; HR = 0.28) remained significant after correction for prognostic factors. SBP ≥ 160 mmHg was not associated with either IDFS or OS. A common single-nucleotide polymorphism, rs6486785, was significantly associated with hypertensive crisis (p = 8.4 × 10−9; OR = 5.2).
Conclusion
Bevacizumab-induced hypertensive crisis is associated with superior outcomes and rs6486785 predicted an increased risk of this key toxicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Unidentified genome wide genotyping data and raw sequence data can be accessed at dbGaP (accession number phs003201.v1.p1).
References
Garcia J, Hurwitz HI, Sandler AB, Miles D, Coleman RL, Deurloo R, et al. Bevacizumab (Avastin(R)) in cancer treatment: A review of 15 years of clinical experience and future outlook. Cancer Treat Rev. 2020;86:102017.
Miles DW, Chan A, Dirix LY, Cortes J, Pivot X, Tomczak P, et al. Phase III study of bevacizumab plus docetaxel compared with placebo plus docetaxel for the first-line treatment of human epidermal growth factor receptor 2-negative metastatic breast cancer. J Clin Oncol. 2010;28:3239–47.
Miller K, Wang M, Gralow J, Dickler M, Cobleigh M, Perez EA, et al. Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. N Engl J Med. 2007;357:2666–76.
Robert NJ, Dieras V, Glaspy J, Brufsky AM, Bondarenko I, Lipatov ON, et al. RIBBON-1: randomized, double-blind, placebo-controlled, phase III trial of chemotherapy with or without bevacizumab for first-line treatment of human epidermal growth factor receptor 2-negative, locally recurrent or metastatic breast cancer. J Clin Oncol. 2011;29:1252–60.
Miller KD, O’Neill A, Gradishar W, Hobday TJ, Goldstein LJ, Mayer IA, et al. Double-Blind Phase III Trial of Adjuvant Chemotherapy With and Without Bevacizumab in Patients With Lymph Node-Positive and High-Risk Lymph Node-Negative Breast Cancer (E5103). J Clin Oncol. 2018;36:2621–9.
Bell R, Brown J, Parmar M, Toi M, Suter T, Steger GG, et al. Final efficacy and updated safety results of the randomized phase III BEATRICE trial evaluating adjuvant bevacizumab-containing therapy in triple-negative early breast cancer. Ann Oncol. 2017;28:754–60.
Cameron D, Brown J, Dent R, Jackisch C, Mackey J, Pivot X, et al. Adjuvant bevacizumab-containing therapy in triple-negative breast cancer (BEATRICE): primary results of a randomised, phase 3 trial. Lancet Oncol. 2013;14:933–42.
Slamon D, Swain S, Buyse M, Martin M, Geyer C, Im Y-H, et al. Abstract S1-03: Primary results from BETH, a phase 3 controlled study of adjuvant chemotherapy and trastuzumab ± bevacizumab in patients with HER2-positive, node-positive or high risk node-negative breast cancer. Cancer Res. 2013;73:S1–03. -S1-
Ranpura V, Pulipati B, Chu D, Zhu X, Wu S. Increased risk of high-grade hypertension with bevacizumab in cancer patients: a meta-analysis. Am J Hypertens. 2010;23:460–8.
Li M, Kroetz DL. Bevacizumab-induced hypertension: Clinical presentation and molecular understanding. Pharm Ther. 2018;182:152–60.
Oza AM, Selle F, Davidenko I, Korach J, Mendiola C, Pautier P, et al. Efficacy and Safety of Bevacizumab-Containing Therapy in Newly Diagnosed Ovarian Cancer: ROSiA Single-Arm Phase 3B Study. Int J Gynecol Cancer. 2017;27:50–8.
Coleman RL, Brady MF, Herzog TJ, Sabbatini P, Armstrong DK, Walker JL, et al. Bevacizumab and paclitaxel-carboplatin chemotherapy and secondary cytoreduction in recurrent, platinum-sensitive ovarian cancer (NRG Oncology/Gynecologic Oncology Group study GOG-0213): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2017;18:779–91.
Schneider BP, Wang M, Radovich M, Sledge GW, Badve S, Thor A, et al. Association of vascular endothelial growth factor and vascular endothelial growth factor receptor-2 genetic polymorphisms with outcome in a trial of paclitaxel compared with paclitaxel plus bevacizumab in advanced breast cancer: ECOG 2100. J Clin Oncol. 2008;26:4672–8.
Gampenrieder SP, Romeder F, Muss C, Pircher M, Ressler S, Rinnerthaler G, et al. Hypertension as a predictive marker for bevacizumab in metastatic breast cancer: results from a retrospective matched-pair analysis. Anticancer Res. 2014;34:227–33.
Hurwitz HI, Douglas PS, Middleton JP, Sledge GW, Johnson DH, Reardon DA, et al. Analysis of early hypertension and clinical outcome with bevacizumab: results from seven phase III studies. Oncologist. 2013;18:273–80.
Liau CT, Chou WC, Wei KC, Chang CN, Toh CH, Jung SM. Female sex, good performance status, and bevacizumab-induced hypertension associated with survival benefit in Asian patients with recurrent glioblastoma treated with bevacizumab. Asia Pac J Clin Oncol. 2018;14:e8–e14.
Osterlund P, Soveri LM, Isoniemi H, Poussa T, Alanko T, Bono P. Hypertension and overall survival in metastatic colorectal cancer patients treated with bevacizumab-containing chemotherapy. Br J Cancer. 2011;104:599–604.
Pant S, Martin LK, Geyer S, Wei L, Van Loon K, Sommovilla N, et al. Treatment-related Hypertension as a Pharmacodynamic Biomarker for the Efficacy of Bevacizumab in Advanced Pancreas Cancer: A Pooled Analysis of 4 Prospective Trials of Gemcitabine-based Therapy With Bevacizumab. Am J Clin Oncol. 2016;39:614–8.
Lombardi P, Rossini D, Crespi V, Germani MM, Bergamo F, Pietrantonio F, et al. Bevacizumab-induced hypertension as a predictor of clinical outcome in metastatic colorectal cancer: An individual patient data-based pooled analysis of two randomized studies and a systematic review of the literature. Cancer Treat Rev. 2022;103:102326.
Tada T, Kumada T, Hiraoka A, Hirooka M, Kariyama K, Tani J, et al. Adverse events as potential predictive factors of therapeutic activity in patients with unresectable hepatocellular carcinoma treated with atezolizumab plus bevacizumab. Cancer Med. 2023;12:7772–83.
Duco MR, Murdock JL, Reeves DJ. Vascular endothelial growth factor inhibitor induced hypertension: Retrospective analysis of the impact of blood pressure elevations on outcomes. J Oncol Pharm Pr. 2022;28:265–73.
Khan KI, Ramesh P, Kanagalingam S, Zargham Ul Haq F, Victory Srinivasan N, Khan AI, et al. Bevacizumab-Induced Hypertension as a Potential Physiological Clinical Biomarker for Improved Outcomes in Patients With Recurrent Glioblastoma Multiforme: A Systematic Review. Cureus. 2022;14:e29269.
Robinson ES, Khankin EV, Karumanchi SA, Humphreys BD. Hypertension induced by vascular endothelial growth factor signaling pathway inhibition: mechanisms and potential use as a biomarker. Semin Nephrol. 2010;30:591–601.
Schneider BP, Li L, Shen F, Miller KD, Radovich M, O’Neill A, et al. Genetic variant predicts bevacizumab-induced hypertension in ECOG-5103 and ECOG-2100. Br J Cancer. 2014;111:1241–8.
Whelton PK, Carey RM, Aronow WS, Casey DE Jr, Collins KJ, Dennison Himmelfarb C, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2018;71:e13–e115.
Schneider BP, Li L, Radovich M, Shen F, Miller KD, Flockhart DA, et al. Genome-Wide Association Studies for Taxane-Induced Peripheral Neuropathy in ECOG-5103 and ECOG-1199. Clin Cancer Res. 2015;21:5082–91.
Schneider BP, Lai D, Shen F, Jiang G, Radovich M, Li L, et al. Charcot-Marie-Tooth gene, SBF2, associated with taxane-induced peripheral neuropathy in African Americans. Oncotarget. 2016;7:82244–53.
Lacruz ME, Kluttig A, Kuss O, Tiller D, Medenwald D, Nuding S, et al. Short-term blood pressure variability - variation between arm side, body position and successive measurements: a population-based cohort study. BMC Cardiovasc Disord. 2017;17:31.
Lu Y, Linderman GC, Mahajan S, Liu Y, Huang C, Khera R, et al. Quantifying Blood Pressure Visit-to-Visit Variability in the Real-World Setting: A Retrospective Cohort Study. Circ Cardiovasc Qual Outcomes. 2023;16:e009258.
Muntner P, Shimbo D, Carey RM, Charleston JB, Gaillard T, Misra S, et al. Measurement of Blood Pressure in Humans: A Scientific Statement From the American Heart Association. Hypertension. 2019;73:e35–e66.
Jee SH, He J, Whelton PK, Suh I, Klag MJ. The effect of chronic coffee drinking on blood pressure: a meta-analysis of controlled clinical trials. Hypertension. 1999;33:647–52.
Fryar CD, National Center for Health Statistics, Nutrition Examination S. Hypertension prevalence and control among adults: United States, 2015-2016. Hyattsville, MD: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics; 2017.
Lakatta EG, Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part I: aging arteries: a “set up” for vascular disease. Circulation. 2003;107:139–46.
Higashi Y, Kihara Y, Noma K. Endothelial dysfunction and hypertension in aging. Hypertens Res. 2012;35:1039–47.
Stauffer BL, Westby CM, DeSouza CA. Endothelin-1, aging and hypertension. Curr Opin Cardiol. 2008;23:350–5.
Yeung KR. Intergenerational and genetic influence on blood pressure in pregnancy and beyond. Australia: Western Sydney University; 2015.
Venkatesha S, Toporsian M, Lam C, Hanai J, Mammoto T, Kim YM, et al. Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat Med. 2006;12:642–9.
Surendran P, Feofanova EV, Lahrouchi N, Ntalla I, Karthikeyan S, Cook J, et al. Discovery of rare variants associated with blood pressure regulation through meta-analysis of 1.3 million individuals. Nat Genet. 2020;52:1314–32.
Zhou R, Tomkovicz VR, Butler PL, Ochoa LA, Peterson ZJ, Snyder PM. Ubiquitin-specific peptidase 8 (USP8) regulates endosomal trafficking of the epithelial Na+ channel. J Biol Chem. 2013;288:5389–97.
Ali F, Khan A, Muhammad SA, Abbas SQ, Hassan SSU, Bungau S. Genome-wide Meta-analysis Reveals New Gene Signatures and Potential Drug Targets of Hypertension. ACS Omega. 2022;7:22754–72.
Plummer C, Michael A, Shaikh G, Stewart M, Buckley L, Miles T, et al. Expert recommendations on the management of hypertension in patients with ovarian and cervical cancer receiving bevacizumab in the UK. Br J Cancer. 2019;121:109–16.
Funding
This study was conducted by the ECOG-ACRIN Cancer Research Group (Peter J. O’Dwyer, MD and Mitchell D. Schnall, MD, PhD, Group Co-Chairs) and supported by the National Cancer Institute of the National Institutes of Health under the following award numbers: U10CA180820 and U10CA180794. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Additional support was provided by the Breast Cancer Research Foundation. Susan G. Komen for the Cure (SAC110056) (BPS) and Vera Bradley Foundation Center (BPS). Bevacizumab and matching placebo were provided free of charge by Roche/Genentech.
Author information
Authors and Affiliations
Contributions
Conceptualization, FS and BPS; Methodology, FS, JJS, SP, EC, LG, GX, GC, NK and BPS; Formal Analysis, JJS and SP; Investigation, FS, JJS, SP and BPS; Resources, AO, DC, TMS, KDM, GWS and BPS; Data Curation, AO; Writing—Original Draft, all authors; Funding Acquisition, BPS.
Corresponding author
Ethics declarations
Competing interests
BPS has received research support from Roche/Genentech for drug supply. The remaining authors declare no competing financial interests.
Ethics approval and consent to participate
This study involves secondary research of coded biological samples and clinical data for which patient consent was not required. It was determined exempt from Institutional Review Board (IRB) review as described in 45CFR 46.104 from the Indiana University IRB (application# 1311859668).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shen, F., Jiang, G., Philips, S. et al. Germline predictors for bevacizumab induced hypertensive crisis in ECOG-ACRIN 5103 and BEATRICE. Br J Cancer 130, 1348–1355 (2024). https://doi.org/10.1038/s41416-024-02602-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-024-02602-0