Abstract
Background
Cancer survivors have increased risk of endocrine complications, but there is a lack of information on the occurrence of specific endocrinopathies at the population-level.
Methods
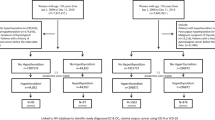
We used data from the California Cancer Registry (2006–2018) linked to statewide hospitalisation, emergency department, and ambulatory surgery databases. We estimated the cumulative incidence of and factors associated with endocrinopathies among adolescents and young adults (AYA, 15–39 years) who survived ≥2 years after diagnosis.
Results
Among 59,343 AYAs, 10-year cumulative incidence was highest for diabetes (4.7%), hypothyroidism (4.6%), other thyroid (2.2%) and parathyroid disorders (1.6%). Hypothyroidism was most common in Hodgkin lymphoma, leukaemia, breast, and cervical cancer survivors, while diabetes was highest among survivors of leukaemias, non-Hodgkin lymphoma, colorectal, cervical, and breast cancer. In multivariable models, factors associated with increased hazard of endocrinopathies were treatment, advanced stage, public insurance, residence in low/middle socioeconomic neighbourhoods, older age, and non-Hispanic Black or Hispanic race/ethnicity. Haematopoietic cell transplant was associated with most endocrinopathies, while chemotherapy was associated with a higher hazard of ovarian dysfunction and hypothyroidism.
Conclusions
We observed a high burden of endocrinopathies among AYA cancer survivors, which varied by treatment and social factors. Evidence-based survivorship guidelines are needed for surveillance of these diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the California Cancer Registry and the California Department of Health Care Access and Innovation. Access to these data sources is granted through an application process by the management or data custodians.
References
Miller KD, Fidler-Benaoudia M, Keegan TH, Hipp HS, Jemal A, Siegel RL. Cancer statistics for adolescents and young adults, 2020. CA Cancer J Clin. 2020;70:443–59.
Moke DJ, Tsai K, Hamilton AS, Hwang A, Liu L, Freyer DR, et al. Emerging cancer survival trends, disparities, and priorities in adolescents and young adults: a California Cancer Registry-Based Study. JNCI Cancer Spectr. 2019;3:pkz031.
Lewis DR, Siembida EJ, Seibel NL, Smith AW, Mariotto AB. Survival outcomes for cancer types with the highest death rates for adolescents and young adults, 1975-2016. Cancer. 2021;127:4277–86.
Diller L, Chow EJ, Gurney JG, Hudson MM, Kadin-Lottick NS, Kawashima TI, et al. Chronic disease in the Childhood Cancer Survivor Study cohort: a review of published findings. J Clin Oncol. 2009;27:2339–55.
Chemaitilly W, Hudson MM. Update on endocrine and metabolic therapy-related late effects observed in survivors of childhood neoplasia. Curr Opin Endocrinol Diabetes Obes. 2014;21:71–6.
Muffly L, Maguire FB, Li Q, Kennedy V, Keegan TH. Late effects in survivors of adolescent and young adult acute lymphoblastic leukemia. JNCI Cancer Spectrum. 2020;4:pkaa025.
Abrahão R, Huynh JC, Benjamin DJ, Li QW, Winestone LE, Muffly L, et al. Chronic medical conditions and late effects after acute myeloid leukaemia in adolescents and young adults: a population-based study. Int J Epidemiol. 2021;50:663–74.
Abrahão R, Li QW, Malogolowkin MH, Alvarez EM, Ribeiro RC, Wun T, et al. Chronic medical conditions and late effects following non-Hodgkin lymphoma in HIV-uninfected and HIV-infected adolescents and young adults: a population-based study. Br J Haematol. 2020;190:371–84.
Keegan THM, Li Q, Steele A, Alvarez EM, Brunson A, Flowers CR, et al. Sociodemographic disparities in the occurrence of medical conditions among adolescent and young adult Hodgkin lymphoma survivors. Cancer Causes Control. 2018;29:551–61.
Friedman DN, Tonorezos ES, Cohen P. Diabetes and metabolic syndrome in survivors of childhood cancer. Horm Res Paediatr. 2019;91:118–27.
Gebauer J, Higham C, Langer T, Denzer C, Brabant G. Long-term endocrine and metabolic consequences of cancer treatment: a systematic review. Endocr Rev. 2019;40:711–67.
Mostoufi-Moab S, Seidel K, Leisenring WM, Armstrong GT, Oeffinger KC, Stovall M, et al. Endocrine abnormalities in aging survivors of childhood cancer: a report from the Childhood Cancer Survivor Study. J Clin Oncol. 2016;34:3240–7.
Pradhan KR, Chen Y, Moustoufi-Moab S, Krull K, Oeffinger KC, Sklar C, et al. Endocrine and metabolic disorders in survivors of childhood cancers and health-related quality of life and physical activity. J Clin Endocrinol Metab. 2019;104:5183–94.
Chemaitilly W, Sklar CA. Childhood cancer treatments and associated endocrine late effects: a concise guide for the pediatric endocrinologist. Horm Res Paediatr. 2019;91:74–82.
Children’s Oncology Group. Long-term follow-up guidelines for survivors of childhood, adolescent, and young adult cancers. Version 5.0. 2021. http://www.survivorshipguidelines.org/. Accessed 04 Apr 2023.
van Dorp W, Mulder RL, Kremer LC, Hudson MM, van den Heuvel-Eibrink MM, van den Berg MH, et al. Recommendations for premature ovarian insufficiency surveillance for female survivors of childhood, adolescent, and young adult cancer: a report from the International Late Effects of Childhood Cancer Guideline Harmonization Group in Collaboration With the PanCareSurFup Consortium. J Clin Oncol. 2016;34:3440–50.
Clement SC, Kremer LCM, Verburg FA, Simmons JH, Goldfarb M, Peeters RP, et al. Balancing the benefits and harms of thyroid cancer surveillance in survivors of childhood, adolescent and young adult cancer: recommendations from the International Late Effects of Childhood Cancer Guideline Harmonization Group in collaboration with the PanCareSurFup Consortium. Cancer Treat Rev. 2018;63:28–39.
Skinner R, Mulder RL, Kremer LC, Hudson MM, Constine LS, Bardi E, et al. Recommendations for gonadotoxicity surveillance in male childhood, adolescent, and young adult cancer survivors: a report from the International Late Effects of Childhood Cancer Guideline Harmonization Group in collaboration with the PanCareSurFup Consortium. Lancet Oncol. 2017;18:e75–90.
van Dalen EC, Mulder RL, Suh E, Ehrhardt MJ, Aune GJ, Bardi E, et al. Coronary artery disease surveillance among childhood, adolescent and young adult cancer survivors: a systematic review and recommendations from the International Late Effects of Childhood Cancer Guideline Harmonization Group. Eur J Cancer. 2021;156:127–37.
Bleyer A, Barr R, Hayes-Lattin B, Thomas D, Ellis C, Anderson B. The distinctive biology of cancer in adolescents and young adults. Nat Rev Cancer. 2008;8:288–98.
Semrad TJ, Li Q, Goldfarb M, Semrad A, Campbell M, Farwell DG, et al. Influence of socioeconomic status on incident medical conditions in two-year survivors of adolescent or young adult differentiated thyroid cancer. J Adolesc Young Adult Oncol. 2021;10:521–33.
Kaiser Permanente Washington Health Research Institute. VOICE Study: valuing opinions and insight from cancer experiences: Kaiser Permanente. 2023. https://www.kpwashingtonresearch.org/voicestudy. Accessed 27 Mar 2023.
California Department of Public Health’s Chronic Disease Surveillance and Research Branch. California Cancer Registry. 2023. https://www.ccrcal.org/learn-about-ccr/. Accessed 28 Sep 2023.
Cree I, Ferlay J, Jakob R. International Classification of Diseases for Oncology, Third Edition, Second Revision. 2019. https://www.who.int/standards/classifications/other-classifications/international-classification-of-diseases-for-oncology. Accessed 23 Mar 2023.
Barr RD, Ries LAG, Trama A, Gatta G, Steliarova-Foucher E, Stiller CA, et al. A system for classifying cancers diagnosed in adolescents and young adults. Cancer. 2020;126:4634–59.
Jensen MV, Rugbjerg K, de Fine Licht S, Johansen C, Schmiegelow K, Andersen KK, et al. Endocrine late effects in survivors of cancer in adolescence and young adulthood: a Danish population-based cohort study. JAMA Network Open. 2018;1:e180349.
de Fine Licht S, Winther JF, Gudmundsdottir T, Holmqvist AS, Bonnesen TG, Asdahl PH, et al. Hospital contacts for endocrine disorders in Adult Life after Childhood Cancer in Scandinavia (ALiCCS): a population-based cohort study. Lancet. 2014;383:1981–9.
National Comprehensive Cancer Network (NCCN). Thyroid carcinoma, Version 1.2023: NCCN.org. 2023. https://www.nccn.org/professionals/physician_gls/pdf/thyroid.pdf. Accessed 14 Mar 2023.
Cianferotti L. Classification of hypoparathyroid disorders. Front Horm Res. 2019;51:127–38.
Yost K, Perkins C, Cohen R, Morris C, Wright W. Socioeconomic status and breast cancer incidence in California for different race/ethnic groups. Cancer Causes Control. 2001;12:703–11.
Lin G, So Y, Johnston G, editors. Analyzing survival data with competing risks using SAS® Software. SAS Global Forum 2012 Conference. Cary, NC: SAS Institute Inc; 2012.
Brennan BM, Shalet SM. Endocrine late effects after bone marrow transplant. Br J Haematol. 2002;118:58–66.
Baker KS, Ness KK, Steinberger J, Carter A, Francisco L, Burns LJ, et al. Diabetes, hypertension, and cardiovascular events in survivors of hematopoietic cell transplantation: a report from the bone marrow transplantation survivor study. Blood. 2007;109:1765–72.
Baker KS, Ness KK, Weisdorf D, Francisco L, Sun CL, Forman S, et al. Late effects in survivors of acute leukemia treated with hematopoietic cell transplantation: a report from the Bone Marrow Transplant Survivor Study. Leukemia. 2010;24:2039–47.
Felicetti F, Manicone R, Corrias A, Manieri C, Biasin E, Bini I, et al. Endocrine late effects after total body irradiation in patients who received hematopoietic cell transplantation during childhood: a retrospective study from a single institution. J Cancer Res Clin Oncol. 2011;137:1343–8.
Tauchmanovà L, Selleri C, Rosa GD, Pagano L, Orio F, Lombardi G, et al. High prevalence of endocrine dysfunction in long-term survivors after allogeneic bone marrow transplantation for hematologic diseases. Cancer. 2002;95:1076–84.
Felicetti F, Gatti F, Faraci D, Rosso D, Zavattaro M, Fortunati N, et al. Impact of allogeneic stem cell transplantation on thyroid function. J Endocrinol Invest. 2023;46:1825–34.
Darzy KH, Shalet SM. Hypopituitarism following radiotherapy revisited. Endocr Dev. 2009;15:1–24.
Jalal N, Haq S, Anwar N, Nazeer S, Saeed U. Radiation induced bystander effect and DNA damage. J Cancer Res Ther. 2014;10:819–33.
Hancock SL, Cox RS, McDougall IR. Thyroid diseases after treatment of Hodgkin’s disease. N Engl J Med. 1991;325:599–605.
Pinnix CC, Cella L, Andraos TY, Ayoub Z, Milgrom SA, Gunther J, et al. Predictors of hypothyroidism in Hodgkin lymphoma survivors after intensity modulated versus 3-dimensional radiation therapy. Int J Radiat Oncol Biol Phys. 2018;101:530–40.
Sklar C, Whitton J, Mertens A, Stovall M, Green D, Marina N, et al. Abnormalities of the thyroid in survivors of hodgkin’s disease: data from the Childhood Cancer Survivor Study. J Clin Endocrinol Metab. 2000;85:3227–32.
Constine LS, Donaldson SS, McDougall IR, Cox RS, Link MP, Kaplan HS. Thyroid dysfunction after radiotherapy in children with Hodgkin’s disease. Cancer. 1984;53:878–83.
Meacham LR, Sklar CA, Li S, Liu Q, Gimpel N, Yasui Y, et al. Diabetes mellitus in long-term survivors of childhood cancer. Increased risk associated with radiation therapy: a report for the childhood cancer survivor study. Arch Intern Med. 2009;169:1381–8.
Dixon SB, Liu Q, Chow EJ, Oeffinger KC, Nathan PC, Howell RM, et al. Specific causes of excess late mortality and association with modifiable risk factors among survivors of childhood cancer: a report from the Childhood Cancer Survivor Study cohort. Lancet. 2023;401:1447–57.
Hudson MM, Mulrooney DA, Bowers DC, Sklar CA, Green DM, Donaldson SS, et al. High-risk populations identified in childhood cancer survivor study investigations: implications for risk-based surveillance. J Clin Oncol. 2009;27:2405–14.
Nandagopal R, Laverdière C, Mulrooney D, Hudson MM, Meacham L. Endocrine late effects of childhood cancer therapy: a report from the Children’s Oncology Group. Horm Res. 2008;69:65–74.
van Iersel L, Li Z, Srivastava DK, Brinkman TM, Bjornard KL, Wilson CL, et al. Hypothalamic-pituitary disorders in childhood cancer survivors: prevalence, risk factors and long-term health outcomes. J Clin Endocrinol Metab. 2019;104:6101–15.
Husebye ES, Pearce SH, Krone NP, Kämpe O. Adrenal insufficiency. Lancet. 2021;397:613–29.
Faria SC, Hwu WJ, Curley SA Metastases to abdominal-pelvic organs. In: Silverman PM, editor. Oncologic imaging: a multidisciplinary approach. Elsevier Health Sciences; 2022. p. 563–75.
Bumpers HL, Hassett JM Jr, Penetrante RB, Hoover EL, Holyoke ED. Endocrine organ metastases in subjects with lobular carcinoma of the breast. Arch Surg. 1993;128:1344–7.
Hull B, Wędrychowicz A, Ossowska M, Furtak A, Skoczeń S, B Starzyk J. Assessment of adrenal function in pediatric cancer survivors. Pediatr Endocrinol Diabetes Metab. 2022;28:250–6.
Doleschal B, Petzer A, Aichberger KJ. Adrenal crisis in metastatic breast cancer. BMJ Case Rep. 2017;2017:bcr2017220284.
Walters AGB, Braatvedt G. Endocrine adverse effects of immune checkpoint inhibitors. Intern Med J. 2021;51:1016–20.
Goldsby RE, Stratton KL, Raber S, Ablin A, Strong LC, Oeffinger K, et al. Long-term sequelae in survivors of childhood leukemia with Down syndrome: a childhood cancer survivor study report. Cancer. 2018;124:617–25.
Sözen M, Vural Ç, Selek A, Kiraz U, Cantürk Z, Çetinarslan B, et al. Infiltration of thyroid papillary cancer tissue with myeloid leukemic cells: a case report. World J Surg Oncol. 2021;19:225.
Nakayama S, Yokote T, Kobayashi K, Hirata Y, Hiraiwa T, Akioka T, et al. Acute myeloid leukemia with infiltration of thyroid gland complicating Hashimoto’s thyroiditis. Endocrine. 2009;36:147–50.
Broersen LH, Pereira AM, Jørgensen JO, Dekkers OM. Adrenal insufficiency in corticosteroids use: systematic review and meta-analysis. J Clin Endocrinol Metab. 2015;100:2171–80.
Raciborska A, Bilska K, Filipp E, Drabko K, Rogowska E, Chaber R, et al. Ovarian function in female survivors after multimodal Ewing sarcoma therapy. Pediatr Blood Cancer. 2015;62:341–5.
Flatt SB, Baillargeon A, McClintock C, Pudwell J, Velez MP. Premature ovarian insufficiency in female adolescent and young adult survivors of non-gynecological cancers: a population-based cohort study. Reprod Health. 2023;20:4.
Coughlin SS. Social determinants of health and cancer survivorship. J Environ Health Sci. 2021;7:11–5.
Seligman HK, Schillinger D. Hunger and socioeconomic disparities in chronic disease. N Engl J Med. 2010;363:6–9.
Castillo DC, Ramsey NL, Yu SS, Ricks M, Courville AB, Sumner AE. Inconsistent access to food and cardiometabolic disease: the effect of food insecurity. Curr Cardiovasc Risk Rep. 2012;6:245–50.
Fitzgerald N, Hromi-Fiedler A, Segura-Pérez S, Pérez-Escamilla R. Food insecurity is related to increased risk of type 2 diabetes among Latinas. Ethn Dis. 2011;21:328–34.
Tatalovich Z, Stinchcomb DG, Mariotto A, Ng D, Stevens JL, Coyle LM, et al. Assessment of interstate residential mobility of SEER patients: SEER and LexisNexis residential address linkage. J Registry Manag. 2022;49:109–13.
Mulder RL, Font-Gonzalez A, Green DM, Loeffen EAH, Hudson MM, Loonen J, et al. Fertility preservation for male patients with childhood, adolescent, and young adult cancer: recommendations from the PanCareLIFE Consortium and the International Late Effects of Childhood Cancer Guideline Harmonization Group. Lancet Oncol. 2021;22:e57–67.
van Iersel L, Mulder RL, Denzer C, Cohen LE, Spoudeas HA, Meacham LR, et al. Hypothalamic-pituitary and other endocrine surveillance among childhood cancer survivors. Endocr Rev. 2022;43:794–823.
International Late Effects of Childhood Cancer Guideline Harmonization Group (IGHG). Long-term follow-up guidelines. 2023. https://www.ighg.org/guidelines/topics/. Accessed 29 Apr 2023.
Funding
Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under Award Number P01CA233432. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The collection of cancer incidence data used in this study was supported by the California Department of Public Health pursuant to California Health and Safety Code Section 103885; Centers for Disease Control and Prevention’s (CDC) National Program of Cancer Registries, under cooperative agreement 5NU58DP006344; the NCI’s Surveillance, Epidemiology and End Results (SEER) Program under contract HHSN261201800032I awarded to the University of California, San Francisco, contract HHSN261201800015I awarded to the University of Southern California, and contract HHSN261201800009I awarded to the Public Health Institute. The ideas and opinions expressed herein are those of the authors and do not necessarily reflect the opinions of the State of California, Department of Public Health, the NCI, and the CDC or their Contractors and Subcontractors.
Author information
Authors and Affiliations
Contributions
Conception and design: Renata Abrahão, Theresa Keegan, Ann Brunson. Administrative support: Theresa Keegan, Ted Wun. Collection and assembly of data: Theresa Keegan, Renata Abrahão. Ann Brunson, Ted Wun, Judy Li, Qian Li. Data analysis and interpretation: Renata Abrahão, Theresa Keegan, Ann Brunson. Manuscript writing: all authors. Final approval of manuscript: all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate statement
This study was approved by the California Committee for the Protection of Human Subjects and Kaiser Permanente Institutional Review Board. We obtained waiver of informed consent as our study does not involve direct data collection from or contact with study participants.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abrahão, R., Brunson, A., Ruddy, K.J. et al. Late endocrine diseases in survivors of adolescent and young adult cancer in California: a population-based study. Br J Cancer 130, 1166–1175 (2024). https://doi.org/10.1038/s41416-024-02594-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-024-02594-x