Abstract
Background
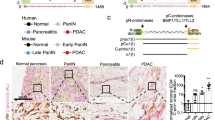
Pancreatic ductal adenocarcinoma (PDAC) is highly malignant with a very poor prognosis due to its silent development and metastatic profile with a 5-year survival rate below 10%. PDAC is characterised by an abundant desmoplastic stroma modulation that influences cancer development by extracellular matrix/cell interactions. Elastin is a key element of the extracellular matrix. Elastin degradation products (EDPs) regulate numerous biological processes such as cell proliferation, migration and invasion. The aim of the present study was to characterise for the first time the effect of two EDPs with consensus sequences “GxxPG” and “GxPGxGxG” (VG-6 and AG-9) on PDAC development. The ribosomal protein SA (RPSA) has been discovered recently, acting as a new receptor of EDPs on the surface of tumour cells, contributing to poor prognosis.
Methods
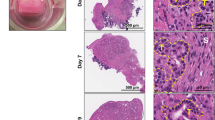
Six week-old female Swiss nude nu/nu (Nu(Ico)-Foxn1nu) mice were subcutaneously injected with human PDAC MIA PaCa-2/eGFP-FLuc+ cells, transduced with a purpose-made lentiviral vector, encoding green fluorescent protein (GFP) and Photinus pyralis (firefly) luciferase (FLuc). Animals were treated three times per week with AG-9 (n = 4), VG-6 (n = 5) or PBS (n = 5). The influence of EDP on PDAC was examined by multimodal imaging (bioluminescence imaging (BLI), fluorescence imaging (FLI) and magnetic resonance imaging (MRI). Tumour volumes were also measured using a caliper. Finally, immunohistology was performed at the end of the in vivo study.
Results
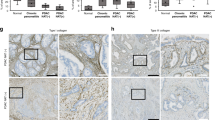
After in vitro validation of MIA PaCa-2 cells by optical imaging, we demonstrated that EDPs exacerbate tumour growth in the PDAC mouse model. While VG-6 stimulated tumour growth to some extent, AG-9 had greater impact on tumour growth. We showed that the expression of the RPSA correlates with a possible effect of EDPs in the PDAC model. Multimodal imaging allowed for longitudinal in vivo follow-up of tumour development. In all groups, we showed mature vessels ending in close vicinity of the tumour, except for the AG-9 group where mature vessels are penetrating the tumour reflecting an increase of vascularisation.
Conclusions
Our results suggest that AG-9 strongly increases PDAC progression through an increase in tumour vascularisation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74:2913–21.
Kamisawa T, Wood LD, Itoi T, Takaori K. Pancreatic cancer. Lancet (Lond, Engl). 2016;388:73–85.
Rawla P, Sunkara T, Gaduputi V. Epidemiology of pancreatic cancer: global trends, etiology and risk factors. World J Oncol. 2019;10:10–27.
Procacci P, Moscheni C, Sartori P, Sommariva M, Gagliano N. Tumor−stroma cross-talk in human pancreatic ductal adenocarcinoma: a focus on the effect of the extracellular matrix on tumor cell phenotype and invasive potential. Cells. 2018;7:1–12.
Feig C, Gopinathan A, Neesse A, Chan DS, Cook N, Tuveson DA. The pancreas cancer microenvironment. Clin Cancer Res. 2012;18:4266–76.
Thomas D, Radhakrishnan P. Tumor-stromal crosstalk in pancreatic cancer and tissue fibrosis. Mol Cancer. 2019;18:14.
Shapiro SD, Endicott SK, Province MA, Pierce JA, Campbell EJ. Marked longevity of human lung parenchymal elastic fibers deduced from prevalence of D-aspartate and nuclear weapons-related radiocarbon. J Clin Invest. 1991;87:1828–34.
Qin Z. Soluble elastin peptides in cardiovascular homeostasis: foe or ally. Peptides. 2015;67:64–73.
Quan T, Wang F, Shao Y, Rittié L, Xia W, Orringer JS, et al. Enhancing structural support of the dermal microenvironment activates fibroblasts, endothelial cells, and keratinocytes in aged human skin in vivo. J Invest Dermatol. 2013;133:658–67.
Robert L, Robert AM, Jacotot B. Elastin-elastase-atherosclerosis revisited. Atherosclerosis. 1998;140:281–95.
Sellami M, Meghraoui-Kheddar A, Terryn C, Fichel C, Bouland N, Diebold MD, et al. Induction and regulation of murine emphysema by elastin peptides. Am J Physiol Lung Cell Mol Physiol. 2016;310:L8–23.
Baló J, Banga I. Elastase and elastase-inhibitor. Nature. 1949;164:491.
Maquart FX, Pasco S, Ramont L, Hornebeck W, Monboisse JC. An introduction to matrikines: Extracellular matrix-derived peptides which regulate cell activity—implication in tumor invasion. Crit Rev Oncol/Hematol. 2004;49:199–202.
Donet M, Brassart-Pasco S, Salesse S, Maquart FX, Brassart B. Elastin peptides regulate HT-1080 fibrosarcoma cell migration and invasion through an Hsp90-dependent mechanism. Br J Cancer. 2014;111:139–48.
Toupance S, Brassart B, Rabenoelina F, Ghoneim C, Vallar L, Polette M, et al. Elastin-derived peptides increase invasive capacities of lung cancer cells by post-transcriptional regulation of MMP-2 and uPA. Clin Exp Metastasis. 2012;29:511–22.
Da Silva J, Lameiras P, Beljebbar A, Berquand A, Villemin M, Ramont L, et al. Structural characterization and in vivo pro-tumor properties of a highly conserved matrikine. Oncotarget. 2018;9:17839–57.
Castronovo V, Claysmith AP, Barker KT, Cioce V, Krutzsch HC, Sobel ME. Biosynthesis of the 67 kDa high affinity laminin receptor. Biochem Biophys Res Commun. 1991;177:177–83.
Digiacomo V, Meruelo D. Looking into laminin receptor: critical discussion regarding the non-integrin 37/67-kDa laminin receptor/RPSA protein. Biol Rev. 2016;91:288–310.
Mecham RP, Hinek A, Griffin GL, Senior RM, Liotta LA. The elastin receptor shows structural and functional similarities to the 67-kDa tumor cell laminin receptor. J Biol Chem. 1989;264:16652–7.
Mercurio AM. Laminin receptors: achieving specificity through cooperation. Trends Cell Biol. 1995;5:419–23.
Lefebvre T, Rybarczyk P, Bretaudeau C, Vanlaeys A, Cousin R, Brassart-Pasco S, et al. TRPM7/RPSA complex regulates pancreatic cancer cell migration. Front Cell Dev Biol. 2020;8:549.
Treadwell JR, Zafar HM, Mitchell MD, Tipton K, Teitelbaum U, Jue J. Imaging tests for the diagnosis and staging of pancreatic adenocarcinoma: a meta-analysis. Pancreas 2016;45:789–95.
Tamm EP, Balachandran A, Bhosale PR, Katz MH, Fleming JB, Lee JH, et al. Imaging of pancreatic adenocarcinoma: update on staging/resectability. Radiol Clin North Am. 2012;50:407–28.
Hee SP, Jeong ML, Hei KC, Sung HH, Joon KH, Byung IC. Preoperative evaluation of pancreatic cancer: comparison of gadolinium-enhanced dynamic MRI with MR cholangiopancreatography versus MDCT. J Magn Reson Imaging. 2009;30:586–95.
Wang Y, Miller FH, Chen ZE, Merrick L, Mortele KJ, Hoff FL, et al. Diffusion-weighted MR imaging of solid and cystic lesions of the pancreas. Radiographics. 2011;31:E47–64.
Mitchell RA, Stanger D, Shuster C, Telford J, Lam E, Enns R. Repeat endoscopic ultrasound-guided fine-needle aspiration in patients with suspected pancreatic cancer: diagnostic yield and associated change in access to appropriate care. Can J Gastroenterol Hepatol. 2016;2016:7678403.
Contag CH, Spilman SD, Contag PR, Oshiro M, Eames B, Dennery P, et al. Visualizing gene expression in living mammals using a bioluminescent reporter. Photochem Photobiol. 1997;66:523–31.
Hilderbrand SA, Weissleder R. Near-infrared fluorescence: application to in vivo molecular imaging. Curr Opin Chem Biol. 2010;14:71–9.
Koh DM, Collins DJ. Diffusion-weighted MRI in the body: applications and challenges in oncology. Am J Roentgenol. 2007;188:1622–35.
Koh DM, Padhani AR. Diffussion-weighted MRI: a new functional clinical technique for tumour imaging. Br J Radiol. 2006;79:633–5.
Leten C, Roobrouck VD, Struys T, Burns TC, Dresselaers T, Vande Velde G, et al. Controlling and monitoring stem cell safety in vivo in an experimental rodent model. Stem Cells. 2014;32:2833–44.
Wu Y, Tan X, Liu P, Yang Y, Huang Y, Liu X, et al. ITGA6 and RPSA synergistically promote pancreatic cancer invasion and metastasis via PI3K and MAPK signaling pathways. Exp Cell Res. 2019;379:30–47.
Long MM, King VJ, Prasad KU, Urry DW. Chemotaxis of fibroblasts toward nonapeptide of elastin. Biochim Biophys Acta. 1988;968:300–11.
Senior RM, Griffin GL, Mecham RP, Wrenn DS, Prasad KU, Urry DW. Val-Gly-Val-Ala-Pro-Gly, a repeating peptide in elastin, is chemotactic for fibroblasts and monocytes. J Cell Biol. 1984;99:870–4.
Mochizuki S, Brassart B, Hinek A. Signaling pathways transduced through the elastin receptor facilitate proliferation of arterial smooth muscle cells. J Biol Chem. 2002;277:44854–63.
Huet E, Brassart B, Cauchard JH, Debelle L, Birembaut P, Wallach J, et al. Cumulative influence of elastin peptides and plasminogen on matrix metalloproteinase activation and type I collagen invasion by HT-1080 fibrosarcoma cells. Clin Exp Metastasis. 2002;19:107–17.
Brassart B, Randoux A, Hornebeck W, Emonard H. Regulation of matrix metalloproteinase-2 (gelatinase A, MMP-2), membrane-type matrix metalloproteinase-1 (MT1-MMP) and tissue inhibitor of metalloproteinases-2 (TIMP-2) expression by elastin-derived peptides in human HT-1080 fibrosarcoma cell line. Clin Exp Metastasis. 1998;16:489–500.
Brassart B, Da Silva J, Donet M, Seurat E, Hague F, Terryn C, et al. Tumour cell blebbing and extracellular vesicle shedding: key role of matrikines and ribosomal protein SA. Br J Cancer. 2019;120:453–65.
Ntayi C, Labrousse AL, Debret R, Birembaut P, Bellon G, Antonicelli F, et al. Elastin-derived peptides upregulate matrix metalloproteinase-2-ediated melanoma cell invasion through elastin-binding protein. J Invest Dermatol. 2004;122:256–65.
Robinet A, Fahem A, Cauchard JH, Huet E, Vincent L, Lorimier S, et al. Elastin-derived peptides enhance angiogenesis by promoting endothelial cell migration and tubulogenesis through upregulation of MT1-MMP. J Cell Sci. 2005;118:343–56.
Dale MA, Xiong W, Carson JS, Suh MK, Karpisek AD, Meisinger TM, et al. Elastin-derived peptides promote abdominal aortic aneurysm formation by modulating M1/M2 macrophage polarization. J Immunol. 2016;196:4536–43.
Hance KA, Tataria M, Ziporin SJ, Lee JK, Thompson RW. Monocyte chemotactic activity in human abdominal aortic aneurysms: role of elastin degradation peptides and the 67-kD cell surface elastin receptor. J Vasc Surg Publ Soc Vasc Surg Int Soc Cardiovasc Surg North Am Chapter. 2002;35:254–61.
Huet E, Brassart B, Wallach J, Debelle L, Haye B, Emonard H, et al. Effect of elastin peptides on the production of matrix metalloproteinase 2 by human skin fibroblasts in culture. J Soc Biol. 2001;195:165–72.
Brassart B, Fuchs P, Huet E, Alix AJP, Wallach J, Tamburro AM, et al. Conformational dependence of collagenase (matrix metalloproteinase-1) up-regulation by elastin peptides in cultured fibroblasts. J Biol Chem. 2001;276:5222–7.
Hinek A, Boyle J, Rabinovitch M. Vascular smooth muscle cell detachment from elastin and migration through elastic laminae is promoted by chondroitin sulfate-induced “shedding” of the 67-kDa cell surface elastin binding protein. Exp Cell Res. 1992;203:344–53.
Coquerel B, Poyer F, Torossian F, Dulong V, Bellon G, Dubus I, et al. Elastin-derived peptides: matrikines critical for glioblastoma cell aggressiveness in a 3-D system. Glia. 2009;57:1716–26.
Fahem A, Robinet A, Cauchard JH, Duca L, Soula-Rothhut M, Rothhut B, et al. Elastokine-mediated up-regulation of MT1-MMP is triggered by nitric oxide in endothelial cells. Int J Biochem Cell Biol. 2008;40:1581–96.
Nackman GB, Karkowski FJ, Halpern VJ, Gaetz HP, Tilson MD. Elastin degradation products induce adventitial angiogenesis in the Anidjar/Dobrin rat aneurysm model. Surgery. 1997;122:39–44.
Debret R, Le Naour RR, Sallenave JM, Deshorgue A, Hornebeck WG, Guenounou M, et al. Elastin fragments induce IL-1β upregulation via NF-κB pathway in melanoma cells. J Invest Dermatol. 2006;126:1860–8.
Szychowski KA, Gmiński J. Impact of elastin-derived VGVAPG peptide on bidirectional interaction between peroxisome proliferator-activated receptor gamma (Pparγ) and beta-galactosidase (β-Gal) expression in mouse cortical astrocytes in vitro. Naunyn Schmiedebergs Arch Pharm. 2019;392:405–13.
Pocza P, Süli-Vargha H, Darvas Z, Falus A. Locally generated VGVAPG and VAPG elastin-derived peptides amplify melanoma invasion via the galectin-3 receptor. Int J Cancer. 2008;122:1972–80.
Timár J, Diczházi C, Ladányi A, Rásó E, Hornebeck W, Robert L, et al. Interaction of tumour cells with elastin and the metastatic phenotype. Ciba Found Symp. 1995;192:321–35.
Yusa T, Blood CH, Zetter BR. Tumor cell interactions with elastin: implications for pulmonary metastasis. Am Rev Respir Dis. 1989;140:1458–62.
Liot S, Balas J, Aubert A, Prigent L, Mercier-Gouy P, Verrier B, et al. Stroma involvement in pancreatic ductal adenocarcinoma: an overview focusing on extracellular matrix proteins. Front Immunol. 2021;12:612271.
Li KY, Yuan JL, Trafton D, Wang JX, Niu N, Yuan CH, et al. Pancreatic ductal adenocarcinoma immune microenvironment and immunotherapy prospects. Chronic Dis Transl Med. 2020;6:6–17.
Vasseur R, Skrypek N, Duchêne B, Renaud F, Martínez-Maqueda D, Vincent A, et al. The mucin MUC4 is a transcriptional and post-transcriptional target of K-ras oncogene in pancreatic cancer. Implication of MAPK/AP-1, NF-κB and RalB signaling pathways. Biochim Biophys Acta. 2015;1849:1375–84.
Timar J, Kashofer K. Molecular epidemiology and diagnostics of KRAS mutations in human cancer. Cancer Metastasis Rev. 2020;39:1029–38.
Buscail L, Bournet B, Cordelier P. Role of oncogenic KRAS in the diagnosis, prognosis and treatment of pancreatic cancer. Nat Rev Gastroenterol Hepatol. 2020;17:153–68.
Sato N, Cheng XB, Kohi S, Koga A, Hirata K. Targeting hyaluronan for the treatment of pancreatic ductal adenocarcinoma. Acta Pharm Sin B 2016;6:101–5.
Delaney LJ, Eisenbrey JR, Brown D, Brody JR, Jimbo M, Oeffinger BE, et al. Gemcitabine-loaded microbubble system for ultrasound imaging and therapy. Acta Biomater. 2021;130:385–94.
Kobes JE, Daryaei I, Howison CM, Bontrager JG, Sirianni RW, Meuillet EJ, et al. Improved treatment of pancreatic cancer with drug delivery nanoparticles loaded with a novel AKT/PDK1 inhibitor. Pancreas. 2016;45:1158–66.
Zhang X, Wojtkowiak JW, Martinez GV, Cornnell HH, Hart CP, Baker AF, et al. MR imaging biomarkers to monitor early response to hypoxia-activated prodrug TH-302 in pancreatic cancer xenografts. PLoS ONE. 2016;11:e0155289.
Kishimoto S, Brender JR, Crooks DR, Matsumoto S, Seki T, Oshima N, et al. Imaging of glucose metabolism by 13C-MRI distinguishes pancreatic cancer subtypes in mice. Elife. 2019;8:e46312.
Kim H, Rigell CJ, Zhai G, Lee SK, Samuel SL, Martin A, et al. Antagonistic effects of anti-EMMPRIN antibody when combined with chemotherapy against hypovascular pancreatic cancers. Mol Imaging Biol. 2014;16:85–94.
Wachsmuth L, Mensen A, Barca C, Wiart M, Tristão-Pereira C, Busato A, et al. Contribution of preclinical MRI to responsible animal research: living up to the 3R principle. Magn Reson Mat Phys Biol Med. 2021;34:469–74.
Kubo A, Kajimura M, Suematsu M. Matrix-assisted laser desorption/ionization (MALDI) imaging mass spectrometry (IMS): a challenge for reliable quantitative analyses. Mass Spectrom (Tokyo, Jpn). 2012;1:A0004–A0004.
Ryan DJ, Spraggins JM, Caprioli RM. Protein identification strategies in MALDI imaging mass spectrometry: a brief review. Curr Opin Chem Biol. 2019;48:64–72.
Bhardwaj A, Srivastava SK, Singh S, Tyagi N, Arora S, Carter JE, et al. MYB promotes desmoplasia in pancreatic cancer through direct transcriptional up-regulation and cooperative action of sonic hedgehog and adrenomedullin. J Biol Chem. 2016;291:16263–70.
Wegner CS, Gaustad JV, Andersen LMK, Simonsen TG, Rofstad EK. Diffusion-weighted and dynamic contrast-enhanced MRI of pancreatic adenocarcinoma xenografts: associations with tumor differentiation and collagen content. J Transl Med. 2016;14:161.
Acknowledgements
We gratefully acknowledge the VIB Bio Imaging Core of KU Leuven for their support and assistance in this work. We thank the PICT-IBiSA Platform of the University of Reims Champagne-Ardenne (France), Dr. Carla Rios Luci and Pr. Stefaan Soenen (NanoHealth and Optical Imaging Group, KU Leuven, Belgium), Dr. Bella Manshian (Translational Cell and Tissue Research, KU Leuven, Belgium) and Aurélie Dupont-Deshorgue (UMR7369) for their skilful technical assistance.
Funding
This work was supported by grants from the European Commission for the PANA project (H2020-NMP-2015-two-stage, grant 686009), the Centre National de la Recherche Scientifique (UMR7369), the University of Reims Champagne-Ardenne, the Region Champagne-Ardenne and the Department Imaging and Pathology of KU Leuven provided a FLOV mandate to partially finance LNs PhD scholarship.
Author information
Authors and Affiliations
Contributions
LN, WG, UH, CG, JW, SB and BB designed, performed and analysed all the experiments. LN wrote the paper with BB, WG, UH and SBP provided essential editorial oversight and critical review. BB and UH jointly supervised this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approvals
This manuscript does not need any ethics approval.
Patient consent
This manuscript does not contain any individual person’s data.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nannan, L., Gsell, W., Belderbos, S. et al. A multimodal imaging study to highlight elastin-derived peptide pro-tumoral effect in a pancreatic xenograft model. Br J Cancer 128, 2000–2012 (2023). https://doi.org/10.1038/s41416-023-02242-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-023-02242-w