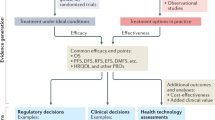
Abstract
Important breakthroughs in medical treatments have improved outcomes for patients suffering from several types of cancer. However, many oncological treatments approved by regulatory agencies are of low value and do not contribute significantly to cancer mortality reduction, but lead to unrealistic patient expectations and push even affluent societies to unsustainable health care costs. Several factors that contribute to approvals of low-value oncology treatments are addressed, including issues with clinical trials, bias in reporting, regulatory agency shortcomings and drug pricing. With the COVID-19 pandemic enforcing the elimination of low-value interventions in all fields of medicine, efforts should urgently be made by all involved in cancer care to select only high-value and sustainable interventions. Transformation of medical education, improvement in clinical trial design, quality, conduct and reporting, strict adherence to scientific norms by regulatory agencies and use of value-based scales can all contribute to raising the bar for oncology drug approvals and influence drug pricing and availability.
Similar content being viewed by others
Background
Important breakthroughs have contributed to an increased life expectancy for patients suffering from several cancers, such as immunotherapy for melanoma [1], second-generation androgen receptor antagonists for prostate cancer [2] and new drugs for myeloma [3]. However, reduction in cancer mortality has been mostly due to prevention, early detection and improved cancer care organisation [4, 5]. In contrast, approval of anticancer drugs by regulatory agencies gives the impression that new drugs are drivers of cancer care improvement. In fact, they often offer limited benefits to patients while the costs of many new cancer treatments are skyrocketing [6, 7]. In the Netherlands, the nationwide prospective cancer registry revealed a median increment in the survival of patients with advanced solid cancers of little more than 1 month over the past 10 years [8]. With value in health care defined as the outcome of an intervention relative to its related costs [9], many new oncological treatments are thus of limited value [10].
The increasing array of new treatments in oncology can lead to unrealistic patient expectations and, if not duly managed, pushes society to unsustainable health care costs. With the current COVID-19 pandemic-driven increased necessity to make value-based choices in all medical fields, we should critically reassess the value of available cancer care and allow only high-value interventions based on properly designed and conducted trials to reach our practice. In this narrative review, we focus on different factors that likely contribute to the use of low-value oncological treatments. Strategies for preventing the advent of low-value oncological care are presented.
Critical interpretation of clinical trials
Relevant outcomes
The evidence upon which any treatment recommendation is based should ideally come from adequately designed, executed and reproduced phase 3 randomised controlled trials (RCTs). Any treatment should result in clear benefits; improve the chances of a cure or to live longer (increase overall survival [OS]) and better lives (improve quality of life [QOL]) [11]. What absolute OS increase is considered relevant is likely to differ between cultures, age groups and countries. As a frame of reference, to be considered for inclusion on the World Health Organization Model List of Essential Medicines, a cancer drug should increase OS by at least 4–6 months as compared to the standard of care [12, 13]. In contrast, in many modern RCTs leading to drug approvals, median OS improvement is mostly <3 months (Table 1) [14,15,16,17,18,19]. An example was the approval of erlotinib in the treatment of advanced pancreatic cancer that increased OS by 2 weeks when combined with gemcitabine, as compared to gemcitabine alone [20].
Improving QOL (assessed in trials as patient-reported outcomes [PROs]) should be the main goal of palliative treatment. Since many trials evaluate surrogate primary endpoints (see below), PRO assessments should be thoroughly and correctly conducted to determine whether the studied intervention delivers meaningful benefit [11]. The Consolidated Standards of Reporting Trials (CONSORT) were extended with specific guidelines to increase the consistency of PRO research and reporting [21]. Several studies have shown that PROs are often not studied and, when addressed, adherence to PRO reporting guidelines is often poor (Table 1) [14, 17, 22,23,24,25,26,27].
Clinical benefit scales based on factors such as OS, QOL, toxicity and symptom control have been developed to translate outcome measures to value [28, 29]. Several studies have assessed whether oncology drug approvals meet such society-defined thresholds of clinical benefit. Most studies show <50% meeting these standards (Table 1) [14, 16, 18, 30,31,32,33,34,35,36,37,38].
Surrogate endpoints
Surrogate endpoints, such as progression-free survival (PFS) or response rate (RR), are increasingly used as primary endpoints in phase 3 oncology RCTs (and in non-randomised studies) (Table 1) [14, 18, 19, 39,40,41,42,43,44,45,46,47]. Their use may contribute to effective treatments reaching patients faster (e.g. recent immunotherapy and targeted therapy approvals for advanced lung cancer [48]). However, a recent retrospective analysis showed a limited median time gain of ~11 months when PFS is studied as opposed to OS [49]. Surrogate endpoints are mostly poorly predictive of outcome parameters such as OS [50] or QOL [51]. The use of surrogate endpoints can therefore result in patient exposure to treatments without proper evidence of an effect on a clinically relevant outcome. Such was the case with bevacizumab in breast cancer, receiving approval after one RCT revealed a positive effect on PFS [52]. After repeat RCTs failed to confirm the magnitude of the initial PFS benefit, showed no improvement in OS but demonstrated increased bevacizumab-related toxicity [53, 54], the approval was revoked. Notably, this treatment option is currently still included in the National Comprehensive Cancer Network (NCCN) breast cancer guideline [55]. Introducing new treatments based on surrogate endpoints can lead to incorrect ‘new standards of care’, to which future potential treatments are being compared in costly clinical trials.
Approvals based on non-phase 3 trials
Even though a phase 3 RCT is considered the gold standard for evidence generation, studies spanning more than a decade have demonstrated that a considerable percentage of oncology drug approvals are based on non-phase 3 RCTs [14, 15, 18, 27, 39, 40, 42, 44, 45, 56, 57] (Table 1). Especially, studies leading to expedited approvals often lack active comparators (see ‘regulatory agencies’ below) [58]. For more than half of FDA oncology drug approvals between 2014 and 2019 that were based on single-arm studies, approved therapies already existed to which these should have been compared [59]. Even though subsequent confirmatory phase 3 RCTs are considered obligatory, they are not always performed [42, 43, 47]. When confirmatory trials do address OS as the primary outcome, these often do not show benefit [47]. A recent example was the approval of olaratumab for treatment of advanced sarcoma in combination with doxorubicin based on a 14-month survival increase in a single open-label phase 1b and phase 2 trial [60]. Resources were wasted on a treatment, of which the results of the ensuing phase 3 RCT failed to show OS improvement [61].
Single RCTs
A new treatment should ideally be approved after demonstrating benefit in a repeated clinical trial. In general, a second positive RCT is considered to confirm the effectiveness of the tested intervention. This is of importance as even with detection of a statistically significant difference between treatment arms in an RCT, there is the possibility of the finding being false positive [62]. Single RCTs are, however, increasingly considered sufficient to lead to drug approvals. A recent study reported that, of 120 FDA approvals (2014–2019), 117 were based on a single RCT [57].
An initially detected effect of an intervention in an RCT is often smaller when the trial is repeated or reanalysed, and large treatment effects in small RCTs are usually not reproducible [63,64,65]. A recent study showed that post-approval updates of RCTs in breast, lung and prostate cancer show a decrease in the initially detected and reported effect upon which the approval was based [66]. The ‘fragility index’, which is the minimum number of changes from non-events to events causing an RCT to lose statistical significance, could be calculated in 17 of 36 oncology RCTs [67]. An outcome change of a mere two events would have 53% of these 17 RCTs lose statistical significance, casting doubt on their robustness. The chance of a properly conducted RCT being false negative is very small (based on the a priori probability of an intervention being effective and the generally accepted threshold of false negativity in RCTs of 20% (power of 80%)). Taking this all together, drug approvals after a single RCT may be based on false positive, exaggerated and/or ‘fragile’ findings.
Non-representative for everyday practice
In clinical trials, the selected population and the context of care delivery are commonly not representative of the general patient population or everyday practice. Fit and relatively young patients are included and intensively monitored during treatment [11]. Even though carrying a higher cancer burden, minorities [68] and older patients [23] are often underrepresented, limiting the broad representability of trial findings. The benefit detected in a clinical trial is usually less and toxicity greater in everyday practice, especially in patients who do not fulfil the original inclusion/exclusion criteria of the respective clinical trial. For example, in ‘real-life data’ in both colorectal cancer [69] and prostate cancer [70], it was observed that treatment outcome was worse than expected in patients not fulfilling the original trial inclusion criteria. A recent study compared trial outcomes of 22 FDA-approved oncology drugs for 29 indications to real-world data obtained in older Medicare beneficiaries [71]. Survival was shorter in Medicare beneficiaries in 28 of 29 indications (median difference −6.3 months, range −28.7 to 2.7 months).
Other important issues in critical evaluation of clinical trials
Many other factors of importance should also be critically assessed when interpreting clinical trials and are beyond the scope of this review. These include the inappropriate use of subgroup analyses [72, 73], the occurrence of inappropriate crossovers [57], the occurrence of informative censoring [74] and the use of inadequate comparator arms [57, 75].
Bias in data publication
Selective or biased reporting in oncological RCTs occurs even in high-impact journals [76, 77]. Spin bias, i.e., misrepresentation of trial findings with the goal of luring readers into believing that the claimed effect is greater than the available data shown, has been reported in almost 60% of RCTs in breast cancer [78] and in 10% of abstracts pertaining to lung cancer trials [79]. Spin bias in abstracts has been shown to influence clinicians’ trial interpretation [80]. It is also directly translated into lay press reporting [81], leading to unfounded expectations of patients, policymakers and physicians [80]. Publication bias, i.e., non-publication or delayed publication of negative studies, needs to be considered whenever we are addressing a potential new treatment (indication), especially since results of non-published studies could be of relevance [82]. Even though the global cancer burden is highest in low- and middle-income countries (LMICs), publication bias by high-impact journals against LMIC oncology RCTs occurs, even as these are more likely to identify high-value therapies [83]. In systematic reviews of cancer trials, publication bias assessments, such as the use of funnel plots, remain underused, which may therefore exaggerate the effects of the reviewed intervention [84]. Harm detected in clinical trials is often underreported [85].
Financial conflicts of interest
Identifying new strategies against disease drives academia and pharma. Aside from academic acknowledgement, funding and promotion of clinical researchers, new treatments, or drug repurposing against both emerging and common diseases, lead to revenue for pharmaceutical companies. However, without industry sponsorship of clinical trials innovation may not reach our patients [86]. Potential financial conflicts of interest (FCI) occur among all stakeholders, investigators and journal editorialists, as well as members of regulatory agencies [6, 87,88,89,90,91]. Studies have shown an association between the likelihood of drug endorsement in consensus guidelines and the FCI of the main author [92, 93].
Regulatory agencies
With the advent of expedited drug approval programmes, regulatory approvals based on limited evidence (e.g. use of surrogate endpoints and non-phase 3 trials) increasingly occur when an intervention serves an ‘unmet clinical need’. This should encompass cancers that have few treatment options with poor disease outcomes [94]. Incorrect use of ‘unmet clinical need’ may, in part, explain the high rate of expedited oncology drug approvals. A recent study showed 95% of 58 FDA approvals from 2012 to 2017 entering an expedited programme [95]. In another study analysing FDA applications for new cancer drugs/indications from 2008 to 2016, 53 of 186 were granted expedited approval [96]. Recently, margetuximab, a monoclonal HER2neu-directed antibody, was granted expedited approval by the FDA for treating advanced breast cancer patients after two prior treatment lines with HER2neu-directed therapy [97]. The drug approval was dated before the publication of the clinical trial [98] and was based on a surrogate endpoint with a PFS benefit of 0.9 months. It is hard to comprehend how, for a disease with many treatment options (next to chemotherapy and hormonal therapy (if applicable) and several FDA-approved HER2neu targeting agents [55]), this would provide in an unmet clinical need warranting expedited approval.
Expedited drug approval pathways require post-approval studies to address evidence gaps. These are often delayed, and if performed, tend to have the same inappropriate characteristics as the pre-approval trials [6, 43, 44, 99,100,101]. A recent overview of 25 years of accelerated approvals of oncology products (all based on surrogate endpoints) showed 51 of 93 initial approvals to confirm benefit in the ensuing years. Thirty-seven (61%) of confirmation studies again used a surrogate endpoint [42]. In another report, for 93 cancer drug indications granted accelerated approval by the FDA, only 20% of confirmatory studies used OS as an endpoint; 41% studied a surrogate with the remaining confirmatory trials not yet reported [43]. Limitations in evidence can therefore persist post approval [102].
Serious side effects may become clear after widespread use after drug approval [85]. For example, real-world post-approval data of ibrutinib, used to treat chronic lymphocytic leukaemia, revealed higher than expected treatment-related cardiac deaths, which may be dose-related [103]. To date, no lower ibrutinib dose trial has been mandated. Another example is the increased risk of myelodysplastic syndrome (MDS) and acute myeloid leukaemia (AML) with the use of poly(ADP-ribose) polymerase (PARP) inhibitors detected in a recent meta-analysis of 28 RCTs and in a study of the WHO’s pharmacovigilance database [104]. This analysis was performed after several cases were detected in the longer follow-up of PARP inhibitor trials. As maintenance PARP inhibitor use in even platinum-sensitive homologous-recombination-proficient ovarian cancer patients has been approved [105], many patients will be exposed to an increased risk of MDS/AML, but with a limited evidence of PFS increase and no proven OS benefit pertaining to their ovarian cancer [106].
Discrepancies between trial inclusion criteria and regulatory agency-approved patient characteristics contribute to the exposure of patients to drugs without supporting data. For example, both enzalutamide [107] and apalutamide [108] were FDA approved for the treatment of men with non-metastatic castration-resistant prostate cancer regardless of prostate-specific antigen (PSA) doubling time, whereas the pivotal trials were limited to men with PSA doubling times of 10 months or less [109, 110]. This broadens prescribing indications to a larger group of lower-risk patients who will be exposed longer to these expensive drugs without proven benefit [111]. A recent study identified 38 approvals (2010–2018) by the FDA, EMA and Pharmaceuticals and Medical Devices Agency; 53% revealed discrepancies between trial inclusion criteria and defined therapeutic indications, and several allow broader prescribing indications [112].
Drug pricing
Conceding the fact that drug pricing, regulatory bodies and financial thresholds for acceptable costs vary between countries, the benefit of many new cancer treatments is in stark contrast to their pricing [7, 113]. No positive association between clinical benefit scales, efficacy or novelty and pricing has been detected [30, 32, 41]. Revenue of approved drugs is such that the need for high drug prices to cover research and development costs may not always be a valid argument [114], especially since the portfolios of pharmaceutical companies overlap, limiting risk and costs of innovation [16, 115].
High drug prices not only lead to cancer treatments being limited to high-income countries but may even delay drugs reaching patients due to unavoidable time-consuming price negotiations [116]. Financial constraint is one of the main determinants of health care availability. With increasing drug prices, even in affluent countries, financial limitations influence cancer drug availability [13, 117].
Raising the bar
Several recommendations can be made that could all contribute to raising the bar in oncology (see Box 1). The first important step is the recognition of the scope of the problems depicted above. The explosive increase in publications on new treatments renders oncologist’s dependent on rapid reading services, abstract reading and expert opinion regarding evidence assessment. A recent study showed that clinicians often overestimate the benefit and underestimate the harm of medical interventions [118]. Unrealistic patient expectations of ‘breakthrough treatments’ [11, 95] occur as a result of direct-to-consumer marketing with a poor translation of scientific data by lay press [81], cancer centres [119] and treating physicians [120, 121]. Medical education needs to be transformed so that we are better prepared for the broader responsibilities and social aspects of modern-day medicine, data interpretation, research and value-weighted treatment selection.
Educational focus on a ‘less is more’ principle for managing patients with advanced cancer should contribute to raising the bar. By adhering to QOL standards, the value of early palliative care with timely refrainment from anticancer therapy should be an integral part of oncology training. Patients suffering from advanced cancer are willing to undergo toxic treatments with a very small chance of benefit near the end of life [122]. However, systemic anticancer treatments often provide limited or no benefit to patients suffering from advanced cancer [123], lead to harm in the last weeks of life, reduce QOL, limit timely hospice care and increase risk of dying in the hospital with subsequently an increased risk of pathological grievance of bereaved [124, 125]. Informing patients of treatment effects in daily practice by quoting point estimates, such as median OS achieved in highly selected patients from RCTs instead of using ‘real-life data’ (if available), could contribute to unrealistic patient expectations [126]. Accurately disclosing prognostic information does not negatively impact patient–physician relationships but may skew low-value life-prolonging care to comfort-oriented care [127]. Therefore, translating complex trial and real-life data to daily clinical practice and adequately and understandably communicating this to patients should receive appropriate attention in oncology training.
Both the medical scientific society and regulatory agencies should aim at improving the quality of clinical trials. The use of surrogate endpoints should be limited to those for which evidence exists of their validity (e.g. metastasis-free survival in localised prostate cancer [128]). Head-to-head comparisons of new drugs and use of the standard of care in control arms should be the norm instead of comparing many new ‘same class’ drugs to inappropriate control arms. Lowering the arbitrary p ≤ 0.05 for rejection of the null hypothesis in RCTs has been proposed as a possible strategy to reduce the risk of false-positive results at the cost of occasionally missing a true and clinically relevant treatment effect [129, 130]. Either the p value for positivity should be lowered or trial replication should, whenever possible, be mandatory before regulatory approval and inclusion into guidelines.
Regulatory agencies should mandate not only confirmatory trials after rapid approvals but should also critically address post-marketing safety concerns. Expedited approval processes should be limited to indications for true unmet clinical needs. When approving drugs, both regulatory agencies and medical societies should adhere to indications as studied, thereby limiting unwarranted prescribing. Retraction of drug approvals should be followed by guideline retractions.
Bias in journal reporting should be eliminated and all data should be available at the time of issuance of both regular approvals and expedited approvals [131]. Data should be presented in a more palatable manner for both physicians and the lay press by employing measures such as absolute risk reductions, number needed to treat and number needed to harm [132]. When applicable, reporting the fragility index, as an indication of trial data robustness, should be considered [67]. Concluding statements in abstracts should be simple and clear and strictly relate to the primary endpoint. Stating FCI’s to readers and audiences should be mandatory, but FCI’s should ideally be avoided.
Our societal responsibility transcends oncological care. Implementation of costly low-value oncological treatments invariably impacts health care accessibility in other medical fields. At least in our country, for the price of one pembrolizumab dose administration [133], four type 1 diabetes mellitus patients can use a flash glucose monitoring system for almost a year to better manage their disease [134], against which they face a lifelong battle. We should, as health care advocates, scrutinise drug pricing and aim for fair distribution of health care spending. Concerted efforts are needed at all levels (physicians, clinical researchers, medical societies, drug companies, regulatory agencies, payers and politicians) for this to be effectively addressed. Integration of value-based scales into regulatory agency policies should raise the bar for drug approvals as well as influence drug pricing and availability. As oncology drugs may often be dosed higher or more frequently than needed, lower doses, alternate dosing schedules and shorter treatment times should be studied [135]. Efforts have been initiated to reduce the use of inappropriate tests, treatments, procedures and costs in oncology, such as the ‘Choosing Wisely’ initiative [136].
The COVID-19 pandemic has impacted health care resources urging the medical community to increasingly adhere to only high-value health care interventions in all medical fields. Strictly selecting high-value cancer care should carry momentum in the oncology landscape far beyond this world disruptive event.
Data availability
Not applicable.
References
Pasquali S, Hadjinicolaou AV, Sileni VC, Rossi CR, Mocellin S. Systemic treatments for metastatic cutaneous melanoma. Cochrane Database Syst. Rev. 2018;2:CD011123.
Gourdin T. Recent progress in treating advanced prostate cancer. Curr Opin Oncol. 2020;32:210–5.
Legarda MA, Cejalvo MJ, Rubia Jdela. Recent advances in the treatment of patients with multiple myeloma. Cancers. 2020;12:3576.
Schilsky RL, Nass S, Le Beau MM, Benz EJ Jr. Progress in cancer research, prevention, and care. N Engl J Med. 2020;383:897–900.
Prasad V. Our best weapons against cancer are not magic bullets. Nature. 2020;577:451.
Lehman R, Gross CP. An international perspective on drugs for cancer: the best of times, the worst of times. JAMA Intern Med. 2019;179:913–4.
Prasad V, De Jesús K, Mailankody S. The high price of anticancer drugs: origins, implications, barriers, solutions. Nat Rev Clin Oncol. 2017;14:381–90.
Fransen H, Aarts M, Brom L, Klinkenberg M, van der Mark M, Pchenitchnikova T, et al. Uitgezaaide kanker in beeld. ISBN 9789072175502. Netherlands: IKNL; 2020. p. 1–61.
Porter ME. What is value in health care? N Engl J Med. 2010;363:2477–81.
Prasad V. Do cancer drugs improve survival or quality of life? BMJ. 2017;359:j4528.
Tannock IF, Amir E, Booth CM, Niraula S, Ocana A, Seruga B, et al. Relevance of randomised controlled trials in oncology. Lancet Oncol. 2016;17:e560–e567.
Shulman LN, Wagner CM, Barr R, Lopes G, Longo G, Robertson J, et al. Proposing essential medicines to treat cancer: methodologies, processes, and outcomes. J Clin Oncol. 2016;34:69–75.
Cortes J, García JMP, Cussac AL, Curigliano G, Saghir NSE, Cardoso F, et al. Enhancing global access to cancer medicines. CA Cancer J Clin. 2020;70:105–24.
Davis C, Naci H, Gurpinar E, Poplavska E, Pinto A, Aggarwal A. Availability of evidence of benefits on overall survival and quality of life of cancer drugs approved by European Medicines Agency: retrospective cohort study of drug approvals 2009-13. BMJ. 2017;359:j4530.
Ladanie A, Schmitt AM, Speich B, Naudet F, Agarwal A, Pereira TV, et al. Clinical trial evidence supporting US Food and Drug Administration approval of novel cancer therapies between 2000 and 2016. JAMA Netw Open. 2020;3:e2024406–15.
Fojo T, Mailankody S, Lo A. Unintended consequences of expensive cancer therapeutics—the pursuit of marginal indications and a me-too mentality that stifles innovation and creativity: the John Conley Lecture. JAMA Otolaryngol Head Neck Surg. 2014;140:1225–36.
Salas-Vega S, Iliopoulos O, Mossialos E. Assessment of overall survival, quality of life, and safety benefits associated with new cancer medicines. JAMA Oncol. 2017;3:382–90.
Meyers DE, Jenei K, Chisamore TM, Gyawali B. Evaluation of the clinical benefit of cancer drugs submitted for reimbursement recommendation decisions in Canada. JAMA Intern Med. 2021;181:499–508.
Paggio JCD, Berry JS, Hopman WM, Eisenhauer EA, Prasad V, Gyawali B, et al. Evolution of the randomized clinical trial in the era of precision oncology. JAMA Oncol. 2021. https://doi.org/10.1001/jamaoncol.2021.0379.
Moore MJ, Goldstein D, Hamm J, Figer A, Hecht JR, Gallinger S, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 2007;25:1960–6.
Calvert M, Blazeby J, Altman DG, Revicki DA, Moher D, Brundage MD, CONSORT PRO Group. Reporting of patient-reported outcomes in randomized trials: the CONSORT PRO extension. JAMA. 2013;309:814–22.
Bylicki O, Gan HK, Joly F, Maillet D, You B, Péron J. Poor patient-reported outcomes reporting according to CONSORT guidelines in randomized clinical trials evaluating systemic cancer therapy. Ann Oncol. 2015;26:231–7.
Sparano F, Aaronson NK, Sprangers MAG, Fayers P, Pusic A, Kieffer JM, et al. Inclusion of older patients with cancer in randomized controlled trials with patient-reported outcomes: a systematic review. BMJ Support Palliat Care. 2019;9:451–63.
Chakraborty R, Cannella L, Cottone F, Efficace F. Quality of patient-reported outcome reporting in randomised controlled trials of haematological malignancies according to international quality standards: a systematic review. Lancet Haematol. 2020;7:e892–e901.
Efficace F, Fayers P, Pusic A, Cemal Y, Yanagawa J, Jacobs M, et al. Quality of patient‐reported outcome reporting across cancer randomized controlled trials according to the CONSORT patient‐reported outcome extension: a pooled analysis of 557 trials. Cancer. 2015;121:3335–42.
Kyte D, Retzer A, Ahmed K, Keeley T, Armes J, Brown JM, et al. Systematic evaluation of Patient-Reported Outcome protocol content and reporting in cancer trials. J Natl Cancer Inst. 2019;111:1170–8.
Safa H, Tamil M, Spiess PE, Manley B, Pow-Sang J, Gilbert SM, et al. Patient-reported outcomes in clinical trials leading to cancer immunotherapy drug approvals from 2011 to 2018: a systematic review. J Natl Cancer Inst. 2020. https://doi.org/10.1093/jnci/djaa174.
Schnipper LE, Davidson NE, Wollins DS, Blayney DW, Dicker AP, Ganz PA, et al. Updating the American Society of Clinical Oncology Value Framework: revisions and reflections in response to comments received. J Clin Oncol. 2016;34:2925–34.
Cherny NI, Dafni U, Bogaerts J, Latino NJ, Pentheroudakis G, Douillard JY, et al. ESMO-Magnitude of Clinical Benefit Scale version 1.1. Ann Oncol. 2017;28:2340–66.
Del Paggio JC, Sullivan R, Schrag D, Hopman WM, Azariah B, Pramesh CS, et al. Delivery of meaningful cancer care: a retrospective cohort study assessing cost and benefit with the ASCO and ESMO frameworks. Lancet Oncol. 2017;18:887–94.
Del Paggio JCD, Azariah B, Sullivan R, Hopman WM, James FV, Roshni S, et al. Do contemporary randomized controlled trials meet ESMO thresholds for meaningful clinical benefit? Ann Oncol. 2016;28:157–62.
Vivot A, Jacot J, Zeitoun J-D, Ravaud P, Crequit P, Porcher R. Clinical benefit, price and approval characteristics of FDA-approved new drugs for treating advanced solid cancer, 2000-15. Ann Oncol. 2017;28:1111–6.
Tibau A, Molto C, Ocana A, Templeton AJ, Carpio LPD, Paggio JCD, et al. Magnitude of clinical benefit of cancer drugs approved by the US Food and Drug Administration. J. Natl Cancer Inst. 2017;110:486–92.
Grössmann N, Paggio JCD, Wolf S, Sullivan R, Booth CM, Rosian K, et al. Five years of EMA-approved systemic cancer therapies for solid tumours—a comparison of two thresholds for meaningful clinical benefit. Eur. J. Cancer. 2017;82:66–71.
Molto C, Hwang TJ, Borrell M, Andres M, Gich I, Barnadas A, et al. Clinical benefit and cost of breakthrough cancer drugs approved by the US Food and Drug Administration. Cancer. 2020;126:4390–9.
Arciero V, Santos SD, Koshy L, Rahmadian A, Saluja R, Everest L, et al. Assessment of Food and Drug Administration– and European Medicines Agency–approved systemic oncology therapies and clinically meaningful improvements in quality of life. JAMA Netw Open. 2021;4:e2033004.
Santos SD, Witzke N, Gyawali B, Arciero VS, Rahmadian AP, Everest L, et al. Reassessing the net benefit of cancer drugs with evolution of evidence using the ASCO Value Framework. J Natl Compr Canc Netw. 2021;26:1–6.
Vokinger KN, Hwang TJ, Grischott T, Reichert S, Tibau A, Rosemann T, et al. Prices and clinical benefit of cancer drugs in the USA and Europe: a cost–benefit analysis. Lancet Oncol. 2020;21:664–70.
Chen EY, Raghunathan V, Prasad V. An overview of cancer drugs approved by the US Food and Drug Administration based on the surrogate end point of response rate. JAMA Intern Med. 2019;28:915–21.
Downing NS, Aminawung JA, Shah ND, Krumholz HM, Ross JS. Clinical trial evidence supporting FDA approval of novel therapeutic agents, 2005-12. JAMA. 2014;311:368–77.
Mailankody S, Prasad V. Five years of cancer drug approvals: innovation, efficacy, and costs. Jama Oncol. 2015;1:539–40.
Beaver JA, Howie LJ, Pelosof L, Kim T, Liu J, Goldberg KB, et al. A 25-year experience of US Food and Drug Administration accelerated approval of malignant hematology and oncology drugs and biologics. JAMA Oncol. 2018;4:849–848.
Gyawali B, Hey SP, Kesselheim AS. Assessment of the clinical benefit of cancer drugs receiving accelerated approval. JAMA Intern Med. 2019;79:906–13.
Johnson JR, Ning Y-M, Farrell A, Justice R, Keegan P, Pazdur R. Accelerated approval of oncology products: the Food and Drug Administration experience. J Natl Cancer Inst. 2011;103:636–44.
Hadidi SA, Ramos CA. Level of evidence for FDA drug approvals in pivotal clinical trials of hematological malignancies. Leuk Lymphoma. 2021;24:1–5.
Naci H, Davis C, Savović J, Higgins JPT, Sterne JAC, Gyawali B, et al. Design characteristics, risk of bias, and reporting of randomised controlled trials supporting approvals of cancer drugs by European Medicines Agency, 2014-16: cross sectional analysis. BMJ. 2019;366:l5221.
Kim C, Prasad V. Cancer drugs approved on the basis of a surrogate end point and subsequent overall survival: an analysis of 5 years of US Food and Drug Administration approvals. JAMA Intern Med. 2015;175:1992–1994.
Singh H, Pazdur R. Contribution of early clinical benefit end points to decreased lung cancer mortality rates. JAMA Oncol. 2021. https://doi.org/10.1001/jamaoncol.2020.8090.
Chen EY, Joshi SK, Tran A, Prasad V. Estimation of study time reduction using surrogate end points rather than overall survival in oncology clinical trials. JAMA Intern Med. 2019;179:642–6.
Haslam A, Hey SP, Gill J, Prasad V. A systematic review of trial-level meta-analyses measuring the strength of association between surrogate end-points and overall survival in oncology. Eur J Cancer. 2019;106:196–211.
Kovic B, Jin X, Kennedy SA, Hylands M, Pedziwiatr M, Kuriyama A, et al. Evaluating progression-free survival as a surrogate outcome for health-related quality of life in oncology: a systematic review and quantitative analysis. JAMA Intern Med. 2018;178:1586–96.
Miller K, Wang M, Gralow J, Dickler M, Cobleigh M, Perez EA, et al. Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. N Engl J Med. 2007;357:2666–76.
Robert NJ, Diéras V, Glaspy J, Brufsky AM, Bondarenko I, Lipatov ON, et al. RIBBON-1: Randomized, double-blind, placebo-controlled, phase III trial of chemotherapy with or without bevacizumab for first-line treatment of human epidermal growth factor receptor 2–negative, locally recurrent or metastatic breast cancer. J Clin Oncol. 2011;29:1252–60.
Pivot X, Schneeweiss A, Verma S, Thomssen C, Passos-Coelho JL, Benedetti G, et al. Efficacy and safety of bevacizumab in combination with docetaxel for the first-line treatment of elderly patients with locally recurrent or metastatic breast cancer: results from AVADO. Eur J Cancer. 2011;47:2387–95.
Gradishar WJ, Moran MS, Aft R, Agnese D, Allison KH, et al. NCCN Breast Cancer Guidelines V2. 2021. https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf. Accessed date 6 June 2021.
Hatswell AJ, Baio G, Berlin JA, Irs A, Freemantle N. Regulatory approval of pharmaceuticals without a randomized controlled study: analysis of EMA and FDA approvals 1999–2014. BMJ Open. 2016;6:e011666.
Hilal T, Gonzalez-Velez M, Prasad V. Limitations in clinical trials leading to anticancer drug approvals by the US Food and Drug Administration. JAMA Intern Med. 2020;180:1108–15.
Naci H, Salcher-Konrad M, Kesselheim AS, Wieseler B, Rochaix L, Redberg RF, et al. Generating comparative evidence on new drugs and devices before approval. Lancet. 2020;395:986–97.
Haslam A, Gill J, Prasad V. The response rate of alternative treatments for drugs approved on the basis of response rate. Int J Cancer. 2021;148:713–22.
Tap WD, Jones RL, Van Tine BA, Chmielowski B, Elias AD, Adkins D, et al. Olaratumab and doxorubicin versus doxorubicin alone for treatment of soft-tissue sarcoma: an open-label phase 1b and randomised phase 2 trial. Lancet. 2016;388:488–97.
Tap WD, Wagner AJ, Schöffski P, Martin-Broto J, Krarup-Hansen A, Ganjoo KN, et al. Effect of doxorubicin plus olaratumab vs doxorubicin plus placebo on survival in patients with advanced soft tissue sarcomas: The ANNOUNCE randomized clinical trial. JAMA. 2020;323:1266–76.
Ioannidis JPA. Why most published research findings are false. PLoS Med. 2005;2:e124.
Ebrahim S, Sohani ZN, Montoya L, Agarwal A, Thorlund K, Mills EJ, et al. Reanalysis of randomized clinical trial data. JAMA. 2014;312:1024–32.
Ioannidis JPA. Contradicted and initially stronger effects in highly cited clinical research. JAMA. 2005;294:218–28.
Nagendran M, Pereira TV, Kiew G, Altman DG, Maruthappu M, Ioannidis JPA, et al. Very large treatment effects in randomised trials as an empirical marker to indicate whether subsequent trials are necessary: meta-epidemiological assessment. BMJ. 2016;355:5432–8.
Elimova E, Moignard S, Li X, Yu M, Xu W, Seruga B, et al. Updating reports of phase 3 clinical trials for cancer. JAMA Oncol. 2021. e206968.
Del Paggio JC, Tannock IF. The fragility of phase 3 trials supporting FDA-approved anticancer medicines: a retrospective analysis. Lancet Oncol. 2019;20:1065–9.
Loree JM, Anand S, Dasari A, Unger JM, Gothwal A, Ellis LM, et al. Disparity of race reporting and representation in clinical trials leading to cancer drug approvals from 2008 to 2018. JAMA Oncol. 2019;5:e191870.
Mol L, Koopman M, Gils CWM, van, Ottevanger PB, Punt CJA. Comparison of treatment outcome in metastatic colorectal cancer patients included in a clinical trial versus daily practice in The Netherlands. Acta Oncol. 2013;52:950–5.
Templeton AJ, Vera-Badillo FE, Wang L, Attalla M, Gouveia PD, Leibowitz-Amit R, et al. Translating clinical trials to clinical practice: outcomes of men with metastatic castration resistant prostate cancer treated with docetaxel and prednisone in and out of clinical trials. Ann Oncol. 2013;24:2972–7.
Green AK, Curry M, Trivedi N, Bach PB, Mailankody S. Assessment of outcomes associated with the use of newly approved oncology drugs in Medicare beneficiaries. JAMA Netw Open. 2021;4:e210030.
Wallach JD, Sullivan PG, Trepanowski JF, Sainani KL, Steyerberg EW, Ioannidis JPA. Evaluation of evidence of statistical support and corroboration of subgroup claims in randomized clinical trials. JAMA Intern Med. 2017;177:554–7.
Sun X, Briel M, Busse JW, You JJ, Akl EA, Mejza F, et al. Credibility of claims of subgroup effects in randomised controlled trials: systematic review. BMJ. 2012;344:e1553–e1553.
Templeton AJ, Amir E, Tannock IF. Informative censoring—a neglected cause of bias in oncology trials. Nat Rev Clin Oncol. 2020;17:327–8.
Tannock IF, Templeton AJ. Flawed trials for cancer. Ann Oncol. 2020;31:331–3.
Raghav KPS, Mahajan S, Yao JC, Hobbs BP, Berry DA, Pentz RD, et al. From protocols to publications: a study in selective reporting of outcomes in randomized trials in oncology. J Clin Oncol. 2015;33:3583–90.
Vera-Badillo FE, Napoleone M, Krzyzanowska MK, Alibhai SMH, Chan A-W, Ocana A, et al. Bias in reporting of randomised clinical trials in oncology. Eur J Cancer. 2016;61:29–35.
Vera-Badillo FE, Shapiro R, Ocana A, Amir E, Tannock IF. Bias in reporting of end points of efficacy and toxicity in randomized, clinical trials for women with breast cancer. Ann Oncol. 2013;24:1238–44.
Altwairgi AK, Booth CM, Hopman WM, Baetz TD. Discordance between conclusions stated in the abstract and conclusions in the article: analysis of published randomized controlled trials of systemic therapy in lung cancer. J Clin Oncol. 2012;30:3552–7.
Boutron I, Altman DG, Hopewell S, Vera-Badillo F, Tannock I, Ravaud P. Impact of spin in the abstracts of articles reporting results of randomized controlled trials in the field of cancer: the SPIIN randomized controlled trial. J Clin Oncol. 2014;32:4120–6.
Yavchitz A, Boutron I, Bafeta A, Marroun I, Charles P, Mantz J, et al. Misrepresentation of randomized controlled trials in press releases and news coverage: a cohort study. PLoS Med. 2012;9:e1001308–11.
Tam VC, Tannock IF, Massey C, Rauw J, Krzyzanowska MK. Compendium of unpublished phase III trials in oncology: characteristics and impact on clinical practice. J Clin Oncol. 2011;29:3133–9.
Wells JC, Sharma S, Paggio JCD, Hopman WM, Gyawali B, Mukherji D, et al. An analysis of contemporary oncology randomized clinical trials from low/middle-income vs high-income countries. JAMA Oncol. 2021;7:379–85.
Herrmann D, Sinnett P, Holmes J, Khan S, Koller C, Vassar M. Statistical controversies in clinical research: publication bias evaluations are not routinely conducted in clinical oncology systematic reviews. Ann Oncol. 2017;28:931–7.
Seruga B, Templeton AJ, Badillo FE, Ocana A, Amir E, Tannock IF. Under-reporting of harm in clinical trials. Lancet Oncol. 2016;17:e209–19.
Flier JS. Academia and industry: allocating credit for discovery and development of new therapies. J Clin Invest. 2019;129:2172–4.
Kaestner V, Prasad V. Financial conflicts of interest among editorialists in high-impact journals. Blood Cancer J. 2017;7:e611.
Tannock IF, Joshua AM. Purchasing silence. Ann Oncol. 2018;29:1339–40.
Wayant C, Turner E, Meyer C, Sinnett P, Vassar M. Financial conflicts of interest among oncologist authors of reports of clinical drug trials. JAMA Oncol. 2018;4:1426–8.
Tannock IF. Disclosure—in the blink of an eye. Ann Oncol. 2019;30:169–70.
Hayes MJ, Prasad V. Financial conflicts of interest at FDA drug advisory committee meetings. Hastings Cent Rep. 2018;2:10–13.
Tibau A, Bedard PL, Srikanthan A, Ethier J-L, Vera-Badillo FE, Templeton AJ, et al. Author financial conflicts of interest, industry funding, and clinical practice guidelines for anticancer drugs. J Clin Oncol. 2015;33:100–6.
Wagner J, Marquart J, Ruby J, Lammers A, Mailankody S, Kaestner V, et al. Frequency and level of evidence used in recommendations by the National Comprehensive Cancer Network guidelines beyond approvals of the US Food and Drug Administration: retrospective observational study. BMJ. 2018;360:k668.
Lu E, Shatzel J, Shin F, Prasad V. What constitutes an “unmet medical need” in oncology? An empirical evaluation of author usage in the biomedical literature. Semin Oncol. 2017;44:8–12.
Hwang TJ, Franklin JM, Chen CT, Lauffenburger JC, Gyawali B, Kesselheim AS, et al. Efficacy, safety, and regulatory approval of Food and Drug Administration–designated breakthrough and nonbreakthrough cancer medicines. J Clin Oncol. 2018;36:1805–12.
Zhou J, Vallejo J, Kluetz P, Pazdur R, Kim T, Keegan P, et al. Overview of oncology and hematology drug approvals at US Food and Drug Administration between 2008 and 2016. J Natl Cancer Inst. 2019;111:449–58.
FDA approves margetuximab for metastatic HER2-positive breast cancer. 2020. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-margetuximab-metastatic-her2-positive-breast-cancer. Accessed date 6 June 2021.
Rugo HS, Im S-A, Cardoso F, Cortes J, Curigliano G, Musolino A, et al. Efficacy of margetuximab vs trastuzumab in patients with pretreated ERBB2-positive advanced breast cancer: a phase 3 randomized clinical trial. JAMA Oncol. 2021. https://doi.org/10.1001/jamaoncol.2020.7932.
Vreman RA, Leufkens HGM, Kesselheim AS. Getting the right evidence after drug approval. Front Pharmacol. 2020;11:569535.
Pease AM, Krumholz HM, Downing NS, Aminawung JA, Shah ND, Ross JS. Postapproval studies of drugs initially approved by the FDA on the basis of limited evidence: systematic review. BMJ. 2017;357:j1680.
Salcher-Konrad M, Naci H, Davis C. Approval of cancer drugs with uncertain therapeutic value: a comparison of regulatory decisions in Europe and the United States. Milbank Q. 2020;98:1219–56.
Cipriani A, Ioannidis JPA, Rothwell PM, Glasziou P, Li T, Hernandez AF, et al. Generating comparative evidence on new drugs and devices after approval. Lancet. 2020;395:998–1010.
Ratain MJ, Moslehi JJ, Lichter AS. Ibrutinib’s cardiotoxicity-an opportunity for postmarketing regulation. JAMA Oncol. 2020. https://doi.org/10.1001/jamaoncol.2020.5742.
Morice P-M, Leary A, Dolladille C, Chrétien B, Poulain L, González-Martín A, et al. Myelodysplastic syndrome and acute myeloid leukaemia in patients treated with PARP inhibitors: a safety meta-analysis of randomised controlled trials and a retrospective study of the WHO pharmacovigilance database. Lancet Haematol. 2021;8:e122–e134.
FDA approves niraparib for first-line maintenance of advanced ovarian cancer. 2020. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-niraparib-first-line-maintenance-advanced-ovarian-cancer. Accessed date 6 June 2021.
González-Martín A, Pothuri B, Vergote I, Christensen RD, Graybill W, Mirza MR, et al. Niraparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 2019;381:2391–402.
FDA approves enzalutamide for castration-resistant prostate cancer. 2018. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-enzalutamide-castration-resistant-prostate-cancer. Accessed date 6 June 2021.
FDA approves apalutamide for non-metastatic castration-resistant prostate cancer. 2018. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-apalutamide-non-metastatic-castration-resistant-prostate-cancer. Accessed date 6 June 2021.
Hussain M, Fizazi K, Saad F, Rathenborg P, Shore N, Ferreira U, et al. Enzalutamide in men with nonmetastatic, castration-resistant prostate cancer. N Engl J Med. 2018;378:2465–74.
Smith MR, Saad F, Chowdhury S, Oudard S, Hadaschik BA, Graff JN, et al. Apalutamide treatment and metastasis-free survival in prostate Cancer. N Engl J Med. 2018;378:1408–18.
Parikh RB, Prasad V. Metastasis-free survival in prostate cancer: faster drug approvals, better drugs? J Clin Oncol. 2019;37:266–8.
Sumi E, Asada R, Lu Y, Ito-Ihara T, Grimes KV. A qualitative study on the differences between trial populations and the approved therapeutic indications of antineoplastic agents by 3 regulatory agencies from 2010 to 2018. Clin Ther. 2020;42:305–20.
Cohen D. Cancer drugs: high price, uncertain value. BMJ. 2017;359:j4543.
Tay-Teo K, Ilbawi A, Hill SR. Comparison of sales income and research and development costs for FDA-approved cancer drugs sold by originator drug companies. JAMA Netw Open. 2019;2:e186875.
Lathyris DN, Patsopoulos NA, Salanti G, Ioannidis JPA. Industry sponsorship and selection of comparators in randomized clinical trials. Eur J Clin Invest. 2010;40:172–82.
Polite BN, Ratain MJ, Lichter AS. Oncology’s “hockey stick” moment for the cost of cancer drugs—the climate is about to change. JAMA Oncol. 2021;7:25–26.
DeMartino PC, Miljković MD, Prasad V. Potential cost implications for all US Food and Drug Administration oncology drug approvals in 2018. JAMA Intern Med. 2021;181:162–7.
Hoffmann TC, Del, Mar C. Clinicians’ expectations of the benefits and harms of treatments, screening, and tests: a systematic review. JAMA Intern Med. 2017;177:407–19.
Berry LL, Keiningham T, Aksoy L, Deming KA. When cancer centers mislead prospective patients. JCO Oncol Pract. 2020;16:219–22.
Ozdemir S, Finkelstein EA. Cognitive Bias: The downside of shared decision making. JCO Clin Cancer Inform. 2018;2:1–10.
Boudewyns V, Southwell BG, DeFrank JT, Ferriola-Bruckenstein K, Halpern MT, ODonoghue AC, et al. Patients understanding of oncology clinical endpoints: a literature review. Patient Educ Couns. 2018;103:1724–35.
Matsuyama R, Reddy S, Smith TJ. Why do patients choose chemotherapy near the end of life? A review of the perspective of those facing death from cancer. J Clin Oncol. 2006;24:3490–6.
Prigerson HG, Bao Y, Shah MA, Paulk ME, LeBlanc TW, Schneider BJ, et al. Chemotherapy use, performance status, and quality of life at the end of life. JAMA Oncol. 2015;1:778–84.
Wright AA, Keating NL, Balboni TA, Matulonis UA, Block SD, Prigerson HG. Place of death: correlations with quality of life of patients with cancer and predictors of bereaved caregivers’ mental health. J Clin Oncol. 2010;28:4457–64.
Currow DC, Agar MR, Phillips JL. Role of hospice care at the end of life for people with cancer. J Clin Oncol. 2020;38:937–43.
Hamers PAH, Elferink MAG, Stellato RK, Punt CJA, May AM, Koopman M, et al. Informing metastatic colorectal cancer patients by quantifying multiple scenarios for survival time based on real-life data. Int J Cancer. 2021;148:296–306.
Enzinger AC, Zhang B, Schrag D, Prigerson HG. Outcomes of prognostic disclosure: associations with prognostic understanding, distress, and relationship with physician among patients with advanced cancer. J Clin Oncol. 2015;33:3809–16.
Gharzai LA, Jiang R, Wallington D, Jones G, Birer S, Jairath N, et al. Intermediate clinical endpoints for surrogacy in localised prostate cancer: an aggregate meta-analysis. Lancet Oncol. 2021;22:402–10.
Ioannidis JPA. The proposal to lower P value thresholds to .005. JAMA. 2018;319:1429–1430.
Benjamin DJ, Berger JO, Johannesson M, Nosek BA, Wagenmakers EJ, Berk R, et al. Redefine statistical significance. Nat Hum Behav. 2018;2:6–10.
Hwang TJ, Vokinger KN, Sachs RE. Evaluating new rules on transparency in cancer research and drug development. JAMA Oncol. 2019;5:461–2.
Elliott MH, Skydel JJ, Dhruva SS, Ross JS, Wallach JD. Characteristics and reporting of number needed to treat, number needed to harm, and absolute risk reduction in controlled clinical trials, 2001-19. JAMA Intern Med. 2021;181:282–4.
Mok TSK, Wu YL, Kudaba I, Kowalski DM, Cho BC, Turna HZ, et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet. 2019;393:1819–30.
Gomez-Peralta F, Dunn T, Landuyt K, Xu Y, Merino-Torres JF. Flash glucose monitoring reduces glycemic variability and hypoglycemia: real-world data from Spain. BMJ Open Diabetes Res Care. 2020;1:e001052.
Ratain MJ, Goldstein DA, Lichter AS. Interventional pharmacoeconomics—a new discipline for a cost-constrained environment. JAMA Oncol. 2019. https://doi.org/10.1001/jamaoncol.2019.1341.
Saletti P, Sanna P, Gabutti L, Ghielmini M. Choosing wisely in oncology: necessity and obstacles. ESMO Open. 2018;3:e000382.
Acknowledgements
Not applicable.
Funding
The authors received no specific funding for this work.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the preparation of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent to publish
By all authors.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schnog, JJ.B., Samson, M.J., Gans, R.O.B. et al. An urgent call to raise the bar in oncology. Br J Cancer 125, 1477–1485 (2021). https://doi.org/10.1038/s41416-021-01495-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01495-7
This article is cited by
-
Analyses of quality of life in cancer drug trials - a review of measurements and analytical choices in post-reimbursement studies
BMC Cancer (2024)
-
Design issues with lutetium-177 PSMA-617 registration studies that bias the outcome of the experimental arm reflect an increasing misalignment of contemporary oncology trials with true patient benefit
BJC Reports (2024)
-
The NeuroPoint alliance SRS & tumor QOD registries
Journal of Neuro-Oncology (2024)
-
Cancer Drugs Reimbursed with Limited Evidence on Overall Survival and Quality of Life: Do Follow-Up Studies Confirm Patient Benefits?
Clinical Drug Investigation (2023)
-
30-day mortality in patients treated for brain metastases: extracranial causes dominate
Radiation Oncology (2022)