Abstract
Mesothelioma is an aggressive cancer that is associated with exposure to asbestos. Although asbestos is banned in several countries, including the UK, an epidemic of mesothelioma is predicted to affect middle-income countries during this century owing to their heavy consumption of asbestos. The prognosis for patients with mesothelioma is poor, reflecting a failure of conventional chemotherapy that has ultimately resulted from an inadequate understanding of its biology. However, recent work has revolutionised the study of mesothelioma, identifying genetic and pathophysiological vulnerabilities, including the loss of tumour suppressors, epigenetic dysregulation and susceptibility to nutrient stress. We discuss how this knowledge, combined with advances in immunotherapy, is enabling the development of novel targeted therapies.
Similar content being viewed by others
Background
Mesothelioma is a malignancy primarily of the thoracic and abdominal linings that affects 2600 individuals in the UK annually with a median survival of about 1-year post-diagnosis [1]. It develops from cells of the mesothelium, a serous membrane that lines the coelomic cavities (Box 1), frequently as a result of asbestos exposure, although other potential triggers of mesothelioma include engineered long, straight, carbon nanotubes, genetic predisposition and radiation therapy. In the case of asbestos, inhaled microscopic fibres migrate through the lung to the pleural space where they can persist for decades, activating mitogenic and inflammatory pathways. Local generation of reactive oxygen species by the asbestos fibres appears to cause DNA damage, triggering malignant transformation.
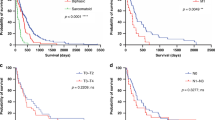
Malignant pleural mesothelioma accounts for around 80% of reported cases of mesothelioma, followed by peritoneal (∼20% cases) and tunica vaginalis (∼1% cases) mesotheliomas. This review focuses on the most common pleural mesothelioma. The three main histological subtypes are epithelioid (50–70% of cases), sarcomatoid (10–20% of cases) and mixed or biphasic (30% of cases) [2] [Fig. 1]. Epithelioid mesothelioma cells often resemble benign, reactive mesothelial cells—which exist as flat or cuboidal forms—with varying degrees of atypia. By contrast, sarcomatoid mesothelioma consists of spindle cells, while biphasic mesothelioma has both epithelioid and sarcomatoid elements. Typically, stroma is abundant and often comprises the majority of the tumour mass. The subtypes represent a spectrum of trans-differentiation with the sarcomatoid phenotype being driven by epithelial–mesenchymal transition (EMT); while epithelioid mesothelioma involves mesenchymal–epithelial transition (MET) [3]. Although EMT is not routinely used for diagnosis, it might be relevant to histological subtyping, with downregulation of EMT markers (e.g. cadherin, c-MYC and vascular endothelial growth factor receptor (VEGFR2)) and upregulation of EMT transcription factors (e.g. Slug, Twist, ZEB1 and ZEB2) observed in sarcomatoid mesothelioma [4, 5]. Accordingly, epithelial markers E-cadherin, β-catenin and cytokeratins 5/6 are abundantly expressed in epithelioid mesothelioma and progressively lost in biphasic and sarcomatoid subtypes [5].
Images of haematoxylin and eosin (H&E) stained normal pleura (×100), epithelioid (×100), sarcomatoid (×100) and biphasic (×100) mesothelioma subtypes, indicating the presence of flat, cuboidal cells in epithelioid mesothelioma as well as spindle cells and abundant stroma in sarcomatoid mesothelioma. Scale bar = 200 µm. The images were provided by Royal Papworth Hospital Research Tissue Bank.
The histological subtypes remain the main prognostic parameter for mesothelioma patients [6]: sarcomatoid mesothelioma has the worst prognosis, with a median survival of 4 months, compared with 13.1 months and 8.4 months for epithelioid and biphasic mesotheliomas, respectively [1, 5]. Histology can be combined with age, gender, probability of diagnosis and leukocyte count in the validated European Organisation for Research and Treatment of Cancer (EORTC) composite score to help predict survival [7, 8]. Other potential prognostic markers arising from studies of cohorts of patients with mesothelioma include the ER stress marker CHOP (C/EBP homologous protein) [8], monocarboxylate transporter 4 (MCT4) [9], CD31 (a stromal marker) [10], periostin and phosphatase and tensin homologue (PTEN) (EMT-related molecules) [11].
Owing to its insidious presentation, only a minority of patients are diagnosed with early stage 1A or 1B mesothelioma that is amenable to surgery. Although cytoreduction by pleurectomy-decortication or radical extra-pleural pneumonectomy (EPP) has been advocated [2], the results have been disappointing [12]. For inoperable cases, palliative chemotherapy is the current standard of care in Europe [13], with a combination of pemetrexed and cisplatin increasing median survival by 3 months [13]. The development of resistance to chemotherapy is a major problem, with almost 50% of mesotheliomas showing resistance to the treatment [13]. An important unmet need therefore exists for effective therapy for this cancer. Understanding the biology of mesothelioma could identify new therapeutic approaches.
Here, we discuss the genetic and cellular vulnerabilities identified in mesothelioma, and the tumour immune landscape, which together provide a rational for the development of novel treatments. We provide a comprehensive summary of previous and current clinical trials.
Hallmarks of mesothelioma of potential therapeutic relevance
Targeting intrinsic features of cancer, such as genetic instability, unchecked growth, aberrant energetics, enhanced angiogenesis, and immune evasion, suggest a variety of therapeutic options in mesothelioma. We therefore begin by assessing this biology.
Genetic alterations in mesothelioma
Mesothelioma has a modest mutational burden compared with other cancers [14,15,16]. Nevertheless, a number of key tumour suppressor genes are frequently affected, including those encoding cyclin-dependent kinase inhibitor 2A (CDKN2A), BRCA1-associated protein 1 (BAP1) and neurofibromin 2 (NF2) [17,18,19] [Fig. 2]. The importance of alterations in these genes is supported by whole-exome sequencing and somatic copy number alteration (SCNA) analyses [15, 16, 20, 21].
a In the endoplasmic reticulum (ER), BAP1 deubiquitinates and stabilises the type-3 inositol-1,4,5-trisphosphate receptor (IP3R3), controlling the Ca2+ flux into mitochondria and the subsequent release of cytochrome c and apoptosis. The nuclear BAP1 activity leads to BRCA1–BARD1-complex-dependent DNA repair as well as chromatin modification through histone deubiquitination. b At the plasma membrane, Merlin inhibits promigratory and prosurvival signalling cascades, including those mediated by focal adhesion kinase (FAK)–Src and phosphatidylinositol 3-kinase (PI3K)–AKT–mammalian target of rapamycin (mTOR)1 pathways. It also activates the mammalian Hippo pathway, which results in the phosphorylation of YAP/TAZ transcription co-activators, precluding their nuclear translocation and the TEA domain (TEAD)-dependent expression of numerous oncogenes. c p14ARF promotes cell-cycle arrest and apoptosis by preventing p53 degradation, while p16INK4a inhibits cell-cycle progression by binding and inactivating CDK4/6 protein and the downstream effector, retinoblastoma (Rb) protein.
BAP1
The importance of BAP1 genetics in the development and prognosis of mesothelioma has been extensively reviewed elsewhere [22,23,24]. In humans, germline mutations in BAP1 predispose to malignant mesothelioma [25], and together with other pathogenic mutations linked to cancer accounts for 12% of all cases [26]. Remarkably, however, patients with mesothelioma who carry germline mutations of BAP1 have a significantly better prognosis compared with those with sporadic disease, although the mechanism of this is unknown [26, 27]. BAP1 is an important regulator of interactions between genes and the environment [28], and loss of BAP1 enhances the susceptibility of fibroblasts and mesothelial cells to ionising or UV irradiation, and to asbestos [29], which contributes to the development of asbestos-induced mesothelioma in vivo [30, 31]. The BAP1 gene product is a deubiquitinating enzyme that plays a key role in the nucleus in the cell cycle, cell death and the DNA damage response [22, 23]; while in the cytoplasm, it triggers apoptosis by regulating the release of calcium ions from the endoplasmic reticulum through its interaction with the receptor for inositol 1,4,5-trisphosphate, IP3R3 [29]. BAP1 is also involved in the epigenetic regulation of many genes via polycomb repressor complex 2 (PRC2), which has potential therapeutic relevance [22], as the loss of BAP1 promotes sensitivity to PRC2 inhibitors, which block tumour growth and invasion [32]. One such drug, tazemetostat, is currently in phase II clinical trials for mesothelioma (NCT02860286).
NF2
NF2 encodes Merlin, which negatively regulates receptor-dependent mitogenic signalling, and downstream phosphatidylinositol 3-kinase (PI3K)–AKT activity, and activates Hippo pathway [33]. Although the loss of NF2 has been suggested to drive mesothelioma carcinogenesis [34], additional mutations are necessary as demonstrated by murine models that require combined deletions such as NF2;INK4a/ARF or NF2;p53 [35]. Merlin controls the expression of oncogenic genes by inhibiting the E3 ubiquitin ligase CRL4DCAF1, to stabilise large tumour suppressor kinase 1/2 (LATS1/2), which in turn inhibits the transcriptional co-activators YAP and TAZ, two downstream effectors of the Hippo pathway [36]. Mutations of Hippo pathway components, such as LATS1 and LATS2, have been observed in many mesothelioma specimens with aberrant YAP activation [16, 21]. Furthermore, co-inactivation of LATS2 and NF2 in mesothelioma cell lines triggers the loss of cell–cell contact inhibition, dysregulation of Hippo and mammalian target of rapamycin (mTOR) signalling, and correlates with higher sensitivity to inhibitors of the PI3K–AKT–mTOR pathway [37]. Targeting the activation of YAP commonly observed in mesothelioma [38] either by inhibiting Rho-associated kinase (ROCK), a downstream target of YAP, or by disrupting the interaction of YAP with TEA domain (TEAD) transcription factors using verteporfin—impedes mesothelioma cell proliferation and invasion in vitro [39]. Interestingly, Hippo signalling appears to stabilise growth arrest and DNA damage 34 (GADD34) protein, resulting in increased YAP phosphorylation and retention in the cytoplasm, as GADD34 directs protein phosphatase 1 (PP1) away from YAP [40]. This might be particularly relevant to mesothelioma, as GADD34 expression correlates with mesothelial cell differentiation and is lost in more aggressive subtypes [8].
CDKN2A
CDKN2A encodes two cell-cycle regulators, p16INK4a and p14ARF [17, 41]. p16 INK4a inhibits cyclin-dependent kinase (CDK)4 and CDK6-dependent phosphorylation of retinoblastoma protein (RB), whereas p14ARF prevents p53 degradation by Mdm2 [41]. Mutations in TP53 occur in less than 10% of mesotheliomas [15, 21] but correlate with worse survival [16]. Given that CDKN2A is deleted in approximately 45% of mesothelioma cases [15], palbociclib, a CDK4/6 kinase inhibitor, has been tested for its ability to induce cell-cycle arrest and senescence in mesothelioma [42]. Treatment with palbociclib led to AKT phosphorylation, but was shown to induce synergistic inhibition of cell proliferation when combined with PI3K/AKT/mTOR inhibitors.
Additional alterations
The majority of studies have focused on the more common epithelioid mesothelioma, with relatively few reports pertaining to the rarer, but more aggressive, sarcomatoid subtype. A report in 2020 showed that loss of PTEN or TP53 promotes the development of non-epithelioid mesotheliomas, with activation of PI3K and MEK–ERK/MAPK [43]. Accordingly, combined pharmacological inhibition of MEK using selumetinib and PI3K using AZD8186 inhibited tumour growth and increased survival in mice [43], pointing towards a potential novel targeted strategy for sarcomatoid mesothelioma. Other profiling studies have identified potential therapeutic vulnerabilities in this rare subset of mesotheliomas [44,45,46]. For instance, LOXL2 (an EMT marker) and VISTA (an immune checkpoint) were found to be overexpressed in tumours with sarcomatoid-like characteristics [16, 44], while differential expression of EMT-related genes identified a subgroup of mesothelioma patients with poor prognosis [45]. Another molecular subgroup displaying sarcomatoid features appears to be more sensitive to drugs inhibiting Wee1, a component of the G2/M cell-cycle checkpoint, and ROCK [44].
Epigenetic dysregulation in mesothelioma
Post-translational modification of histones and methylation of DNA are often altered in cancers [47] and, in mesothelioma, hypermethylation of tumour suppressor gene (CDK2NA, APC, CCND2 and RASSF1) promoters and upregulation of DNA methyltransferases (DNMTs) have been reported [48], while mutations in histone methyltransferases (SETDB1 and SETD2) are often found in patient samples [16, 21, 49]. These observations provided a rationale for targeting histone deacetylases (HDACs) using specific inhibitors (HDACi), which have multiple anticancer effects: they antagonise the cell-cycle and angiogenesis, promote apoptosis and exert anti-inflammatory activity [50, 51]. A phase I trial of vorinostat (an inhibitor of HDAC1,2,3 and 6) [52] in 13 patients with mesothelioma showed a partial response in two patients [53]. Unfortunately, a subsequent phase III trial in 650 patients with mesothelioma demonstrated no improvement in overall survival (OS) [54]. Newer HDACi, such as trichostatin A and its analogues, show promising activity in combination with the DNMT inhibitor decitabine plus immunotherapy [55]. In vitro studies suggest that the loss of BAP1 affects the sensitivity of mesothelioma cells to HDACi [56], indicating that stratification of patients might help to identify a responsive subgroup. Furthermore, decitabine might also prove anti-proliferative in mesothelioma through the upregulation of p21, independent of its effect on DNMTs [57].
Loss of tumour suppressive microRNAs that control cell growth, migration and apoptosis is often observed in mesothelioma [58]. Several preclinical studies have reported that restoring such microRNAs using synthetic mimics can confer anti-tumour activity; examples include miR-15/16 [59] and miR-34 [60] family members, as well as miR-1 [61], miR-31 [62] and miR-145 [63]. Moreover, miR-16-based mimics that target epidermal growth factor receptor (EGFR) have shown acceptable safety and preliminary activity in a phase I study in 26 mesothelioma patients [64].
Mesothelioma cell proliferation and motility
Focal adhesion kinase
Focal adhesion kinase (FAK) is a tyrosine kinase that regulates cancer cell survival, proliferation, migration and invasiveness [65]. In vitro studies have shown the upregulation of FAK in many mesothelioma cell lines, whereas FAK inhibition suppresses cell proliferation and the ability to form anchorage-independent colonies [66]. Accordingly, the FAK inhibitor BI853520 abrogates the growth of orthotopic tumours in vivo [67]. As mentioned above, the tumour suppressor Merlin is frequently inactivated in mesothelioma. Since Merlin can inhibit cell migration and invasiveness by attenuating FAK phosphorylation [68], low Merlin expression was predicted to increase sensitivity to FAK inhibitors [69] and, indeed, a phase I clinical trial of the FAK inhibitor GSK2256098 improved the median progression-free survival (PFS) of patients lacking detectable Merlin [70]. This result was supported by data from a phase Ib study of 34 patients treated with GSK2256098 plus the MEK inhibitor trametinib (to target probable mitogen-activated protein kinase pathway (MAPK) activation), in which longer PFS was reported for Merlin-negative tumours compared with Merlin-positive tumours [71]. Based on these data, a phase II trial of defactinib, an orally bioavailable FAK inhibitor, recruited 344 patients with mesothelioma and low levels of Merlin but, unfortunately, no improvement in disease outcome was observed [72]. Of note, GSK2256098 is selective for FAK while defactinib targets FAK and Pyk2, and so target specificity might have been a confounder.
c-MET
c-MET, a receptor tyrosine kinase important in cell proliferation and motility, is also overexpressed in mesothelioma [73]. In vitro and in vivo studies have revealed that targeting c-MET with tivantinib (ARQ 197) together with PI3K inhibition suppresses cell motility, growth and the development of tumours [74]. A phase I/II clinical trial of tivantinib in combination with pemetrexed and carboplatin/cisplatin is underway in patients with mesothelioma or non-small cell lung cancer (NCT02049060).
Tumour nutrient availability
Angiogenesis
Most solid cancers promote angiogenesis to support their growth [75]. Angiogenic signalling is important for mesothelioma growth [76], and high expression levels of pro-angiogenic signalling molecules, such as VEGF, fibroblast growth factor (FGF)-1, transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF) and PDGF receptor-β, have been reported in mesothelioma tissue and many cell lines [76,77,78]. Furthermore, a high density of tumour microvessels predicts poor survival in patients with malignant mesothelioma [79]. Consequently, inhibitors of angiogenesis have been investigated for potential treatment in patients with mesothelioma [80,81,82]. Thalidomide, a potent inhibitor of angiogenesis in other cancers [83, 84], unfortunately provided no benefit in a phase III trial in mesothelioma [85]. Cediranib, a tyrosine kinase inhibitor targeting VEGFR 1–3, c-Kit and PDGFR-β [86], seemed effective as a second-line agent in one phase II trial [82] and, when combined with chemotherapy, appeared to improve survival in treatment-naïve patients, but significant toxicity precluded its further development [87]. Another tyrosine kinase inhibitor, nintedanib, which targets VEGFR 1–3, FGFR 1–3, PDGFR α/β and Src-family members, increased PFS and OS when combined with pemetrexed/cisplatin in a phase II trial [81] but, unfortunately, this promising result failed to be validated in a subsequent phase III study [80]. More encouraging results have been achieved using bevacizumab (Avastin), a humanised anti-VEGFA monoclonal antibody, which demonstrated efficacy in combination with standard of care in many cancers [88, 89] including mesothelioma [90]. In a large phase III trial (Mesothelioma Avastin Cisplatin Pemetrexed Study, MAPS) of 448 patients, median OS was increased from 16.1 to 18.8 months [90], which resulted in bevacizumab being listed in the American National Comprehensive Cancer Network guidelines as a potential first-line treatment for unresectable mesothelioma [91]; however, at the time of writing, the manufacturer appears not to be pursuing its licensing for bevacizumab for the treatment of mesothelioma in Europe.
ER stress and nutrient stress
Mesothelial cells produce high quantities of cell-surface glycoproteins, most likely to help lubricate the pleural cavities, and therefore depend on the presence of a functional endoplasmic reticulum (ER) in which to fold these proteins. Increased protein misfolding, which might well occur in response to increased demands for protein secretion, is known to cause ER stress signalling, which has emerged as a key adaptive mechanism supporting cancer progression and resistance to treatment [92]. The expression of ER-stress-responsive GADD34 is lower in sarcomatoid mesothelioma than in epithelioid mesothelioma, while high levels of ER-stress-dependent transcription factor C/EBP homologous protein (CHOP) predict poor prognosis [8]. Increased expression of the ER chaperone BiP (GRP78) was reported in mesothelioma cell lines [93] and in patient samples [94]. Consequently, modulation of the ER stress response has been investigated in mesothelioma in preclinical models. For instance, bortezomib, a proteasome inhibitor, and epigallocatechin-3-gallate, a green tea polyphenol, trigger ER-stress-dependent cell death in mesothelioma cell lines [93, 95]. Moreover, HA15, an ER stress inducer that specifically targets BiP [96], exacerbates pre-existing high ER stress levels in mesothelioma cells to induce cell death and impairs mesothelioma cell growth in patient-derived xenografts in a CHOP-dependent manner [94].
Nutrient stress can also affect mesothelioma growth. For example, inhibition of a key component of the glycolytic pathway, 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3 (PFKFB3), results in anti-tumour activity in mesothelioma, with death occurring at least in part via the induction of ER stress [97]. Mesothelioma cells are often arginine auxotrophic owing to the loss of argininosuccinate synthetase 1 (ASS-1) [98], a phenomenon that led to a phase II trial of the arginine-degrading agent PEGylated arginine deiminase (ADI-PEG20). ADI-PEG20 treatment resulted in depletion of circulating arginine and improved PFS in mesothelioma patients [99]. Encouragingly, the TRAP phase I trial of ADI-PEG20 combined with pemetrexed/cisplatin chemotherapy revealed 94% disease control in biphasic and sarcomatoid subtypes [100]. The subsequent phase II/III ATOMIC-Meso global trial of 386 patients with non-epithelioid mesothelioma is recruiting, with a completion date of June 2022 (NCT02709512).
The immune landscape of mesothelioma
Tumour-infiltrating lymphocytes (TILs), macrophages and natural killer (NK) cells are detectable in mesothelioma tissue [101, 102], while mesothelioma can promote an immunosuppressive environment through regulatory T (TREG) cells [102] and M2 polarised macrophages [103]. Whereas infiltration with cytotoxic CD8+ lymphocytes correlates with better patient prognosis [101, 104], high levels of ‘tumour promoting’ M2 macrophages predict shorter patient survival in patients with pleural mesothelioma [105]. Interestingly, as reported for peritoneal mesothelioma, the histological subtypes might show distinct immune signatures, with TILs and Th-1 polarised T cells predominantly present in epithelioid mesothelioma, and myeloid cell infiltrating all subtypes [106]. In pleural mesothelioma, increased expression of immune checkpoint programmed death ligand-1 (PD-L1) and the presence of sarcomatoid components is associated with increased stromal TILs, which, if characterised by high CD8+ and low CD4+, predict poor prognosis [107]. Such knowledge is driving the development of immunomodulatory therapies for mesothelioma.
Immunotherapeutic strategies for mesothelioma
Rare cases of spontaneous regression of mesothelioma have been attributed to immune responses [108], suggesting that immunotherapy might be efficacious in this cancer type, similar to the case in several other cancers [109,110,111].
Immune checkpoint inhibitors
The tumour-killing immune response can be inhibited by cancer cells that express regulators of immune checkpoints such as cytotoxic T-lymphocyte antigen 4 (CTLA-4), T cell immunoglobulin mucin-3 (TIM-3) and PD-L1 [112]. PD-L1, for example, is expressed in up to 29% of mesotheliomas [113] and is associated with poor survival [114]. Inhibition of these checkpoints has proved useful in many cancers and trials have already investigated the use of immune checkpoint inhibitors in mesothelioma [Table 1]. Overall, the results have not been strongly successful, but several large studies have yet to report their data.
CTLA-4 appears not to be a useful target in mesothelioma. In an early trial, the anti-CTLA-4 monoclonal antibody (mAb) tremelimumab showed a limited response rate [115], whereas in a subsequent, more intensified, regimen, a partial response was seen in 1 of 29 patients enrolled in the study [116]. In a phase IIb trial, tremelimumab had no effect on OS [117]. Anti-PD-L1 approaches have yielded mixed results. In phase Ib and II trials, pembrolizumab, which targets the PD-1 receptor, appeared to improve overall response rate [118, 119], while increased PD-L1 expression was associated with a more durable PFS in the phase II study [119]. However, in a retrospective analysis, PD-L1 positivity was only a weak predictor of survival in pembrolizumab-treated cases [120]. In a phase III trial, pembrolizumab appears not to have improved overall survival (OS) compared with single-agent chemotherapy (ESMO 2019 conference, unpublished). Nivolumab, another anti-PD-1 mAb, slightly improved the overall response rate, with PD-L1 expression levels inconsistently predicting the response [121, 122]. A double-blind placebo-controlled trial (CONFIRM) to assess the effect of nivolumab on OS stopped recruitment last year but has yet to report results [123]. Other anti-PD-L1 agents including durvalumab and avelumab have also shown some promising activities [124, 125]. Theoretically, combining anti-CTLA4 with anti-PD-L1 treatment might be synergistic and avoid any potential emerging resistance. In a phase II trial, 40 mesothelioma patients given dual tremelimumab–durvalumab therapy showed a 28% partial response [126]. Likewise, combinations of ipilimumab plus nivolumab have been investigated in phase II trials; one trial failed to meet its primary endpoint [127], while another showed a 29% partial response [128]. In a randomised, open-label trial comparing nivolumab with nivolumab–ipilimumab dual therapy, the overall response rate was 19% with single and 29% with dual agents, but 5% of patients in the combination arm died from toxicity [129, 130]. The Checkmate743 phase III trial has now evaluated dual immunotherapy (nivolumab–ipilimumab) versus standard of care chemotherapy for patients with unresectable mesothelioma. Nivolumab plus ipilimumab significantly improved OS, predominantly in non-epithelioid disease (18.1 months versus 14.1 months for chemotherapy) [131]. The 2-year overall survival was 41% for nivolumab plus ipilimumab, but only 27% in the chemotherapy group.
Compared to other malignancies, mesothelioma is not highly immunogenic, which might explain its poor response to existing immune checkpoint inhibitors. Nevertheless, a number of proteins expressed by mesothelioma cells could serve as antigens for alternative immunotherapies, such as vaccines and CAR T cell therapy.
Cancer vaccines
In 1982, immunotherapy for mesothelioma using the Bacillus Calmette–Guérin (BCG) vaccine was described in 30 patients, and resulted in apparent increased survival in a group of patients with low tumour burden. [132]. The implication was that the vaccine might stimulate the immune system to destroy tumour cells. The BCG vaccine is currently used to treat bladder cancer; it induces multiple cell types and cytokines including interleukins (IL-2, IL-4, IL-5, IL-6, IL-10, IL-12 and IL-17) and interferon (IFN)-γ [133]. IFN-β has anti-proliferative effects on mesothelioma cell lines in vitro [134] and, in patients, recombinant IFN-α triggered a partial response in 12% of recipients [135], while intrapleural IFN-γ showed a 20–45% response rate [136]. Anecdotal evidence has suggested that lymphokine-activated killer cells and IL-2 might reduce the formation of pleural effusions [137], and an early trial with IL-2 suggested survival benefit [138], with a subsequent phase II trial reporting tumour responses in 22% of patients with malignant mesothelioma [139]. These results have encouraged efforts to develop cancer vaccines and other immunotherapies that are more targeted to mesothelioma antigens.
Mesothelin as a target
Mesothelin is a 40-kDa cell surface glycoprotein expressed by all epithelioid, but not by sarcomatoid or the spindle component of biphasic mesotheliomas [140]. High levels of soluble mesothelin predict a poor prognosis, perhaps as a marker of tumour load [141]. Mesothelin-targeted therapies are under development. For instance, amatuximab, a mAb targeting mesothelin, showed acceptable safety profiles in early phase I trials [142] and triggered a partial response of 40% when combined with chemotherapy, improving overall survival in a phase II study [143]. CRS-207, a live attenuated form of Listeria monocytogenes engineered to express human mesothelin, stimulated a tumour-specific CD8+ T cell response in 60% of subjects with advanced cancer, including two patients with mesothelioma [144]. An early phase Ib trial of CRS-207 in combination with chemotherapy suggested an increased overall response rate, a reduction in tumour size, and the expansion of TILs and circulating immune cells [145]. However, a phase II trial of combined CRS-207 and pembrolizumab failed to show patient benefit [146]. SS1P, a recombinant protein that contains bacterial Pseudomonas exotoxin A fused to a high-affinity anti-mesothelin antibody, showed limited efficacy in phase I trials, possibly due to the development of neutralising antibodies, which occurred in most patients after a single treatment cycle [147]. A subsequent trial using concomitant cyclophosphamide and pentostatin to reduce antibody formation resulted in a response in 3 out of 10 patients [148]. Another anti-mesothelin immunotoxin, LMB-100, showed in vitro activity when additionally carrying a paclitaxel payload but has yet to be tested in patients [149].
WT1 as a target
As overexpression of the Wilms’ tumour 1 (WT1) transcription factor occurs in several malignancies, including mesothelioma [150], WT1 peptide analogue vaccines have been developed to elicit CD4+ and CD8+ T cell responses [151]. A favourable safety profile and potential therapeutic effect has been reported in patients randomised to receive a WT1-peptide vaccine (galinpepimut-S) with immunologic adjuvants (montanide and GM-CSF) compared with adjuvants alone [152].
Using tumour lysates
Following the success of the approach in murine models [153], dendritic cells pulsed with autologous tumour lysate were administered by vaccination to patients with mesothelioma in human trials [154]. This therapeutic approach was well-tolerated and induced cytotoxic activity in a subset of patients. In a subsequent trial, tumour-pulsed dendritic cells were combined with cyclophosphamide to enhance the immune response by inhibiting TREG cells [155]. Only one patient achieved a complete response, but 7 of 10 patients survived for longer than 24 months. Autologous dendritic cells pulsed with allogeneic tumour lysate from 5 mesothelioma cell lines might also be effective [156]. A partial response was observed in 2 of 9 patients, although OS was unchanged [156]. A phase II/III trial (DENIM) using an allogeneic tumour-derived dendritic cell lysate vaccine (MesoPher) is currently recruiting adult subjects with mesothelioma (NCT03610360) [157].
CAR T cell therapies
The immune system can also be engaged by generating chimeric antigen receptor (CAR) T cells. In this approach, homologous T cells are collected from the individual, genetically engineered to express a tumour-specific antigen receptor and, following ex vivo expansion, re-administered to the patient. The CAR consists of an antibody single-chain variable fragment (scFvs) fused to a transmembrane domain, followed by an intracellular co-stimulatory domain (including CD28, 4-1BB, CD27 and CD134) and an intracellular T cell receptor CD3ζ chain [158]. Following the success of this approach in haematological malignancies, the efficacy of CAR T cell therapy has been assessed in mesothelioma [159]. Originally, T cells were engineered to transiently express anti-mesothelin CAR to avoid off-tumour on-target toxicity [160]. A clinical response was attained in only 2 of 4 patients, and one patient suffered a cardiac arrest due to an anaphylactic reaction [161]. Several phase I studies have since attempted to optimise the safety profile of second-generation anti-mesothelin CAR T cells [162]. Of note, PD-1 expression in the tumour reduced CAR T cell effector function, but this effect could be reversed by PD-1 checkpoint blockade with pembrolizumab [163, 164]. CAR T cells can also be engineered to target peritumoural components such as fibroblast activation protein (FAP), a transmembrane serine protease highly expressed in cancer-associated stromal cells. Preliminary data using intrapleural FAP-targeted CAR T cells in mesothelioma showed redirected T cell activity in vitro with no significant toxicity [165]; these data were later supported by a phase I clinical trial involving three mesothelioma patients [166]. Other potential CAR T cell targets for mesothelioma include members of the ErbB family [167], oncofetal cell surface glycoprotein 5T4 and chondroitin sulphate proteoglycan 4 (CSPG4) [162, 168, 169].
Antibody–drug conjugates
Antibody–drug conjugates (ADCs) use recombinant monoclonal antibodies that recognise tumour antigens to deliver cytotoxic payloads. Initially, as proof of concept, transferrin was used to deliver intracellular doxorubicin [170], but a number of ADCs that show activity in numerous cancers have subsequently been developed [171, 172]. Given its limited expression on normal tissues and high abundance on cancer cells, mesothelin is an attractive target [173]. So far, only one mesothelin-based ADC—BAY 94-9343 (anetumab ravtansine)—has been tested in mesothelioma. Anetumab ravtansine comprises a human anti-mesothelin antibody conjugated via a disulphide-containing linker to the maytansinoid DM4, which disrupts microtubule function and thus inhibits mitosis [174]. In vivo, anetumab ravtansine blocked mesothelioma growth in both subcutaneous and orthotopic xenograft models and was more effective than standard of care treatment [174]. Subsequently, three clinical trials have commenced for mesothelioma: a phase Ib trial of anetumab ravtansine in combination with pemetrexed and cisplatin (NCT02639091); a phase II study of anetumab ravtansine as a second-line treatment (NCT02610140); and a randomised phase I/II trial of anetumab ravtansine in combination with the anti-PD-1 mAb pembrolizumab (NCT03126630). However, anetumab ravtansine failed to increase PFS in relapsed mesothelioma in a phase II clinical trial (NCT02610140) when compared to vinorelbine, an anti-mitotic drug, [175].
Another ADC with potent anti-mesothelioma activity, αMSLN-MMAE, is a humanised anti-mesothelin mAb conjugated to the microtubule-disrupting drug monomethyl auristatin A (MMAE) with a lysosomal-protease-cleavable valine–citrulline linker [176]. A phase I clinical trial demonstrated that αMSLN–MMAE (also known as DMOT4039A) is well tolerated and exerts anti-tumour activity in patients with unresectable pancreatic or platinum-resistant ovarian cancer [177]; however, no clinical study of αMSLN-MMAE/DMOT4039A has yet been initiated for mesothelioma.
In vitro, epithelioid mesothelioma appears sensitive to brentuximab vedotin (BV), a recombinant chimeric mAb generated against CD30 and conjugated to MMAE via a protease-sensitive linker [178, 179]. Since CD30 is expressed to a greater degree in epithelioid than sarcomatoid mesotheliomas, studies will need to address if this agent shows subtype specificity. CD30 is a member of the tumour necrosis factor receptor (TNFR) superfamily that is involved in the regulation of apoptotic and inflammatory signalling pathways, and is a potential therapeutic target for various malignancies, including mesothelioma [180]. Brentuximab vedotin was initially tested for the treatment of anaplastic large-cell lymphomas and Hodgkin disease [181] but, following a successful safety assessment in solid tumours [182], a phase II trial is now underway to evaluate its efficacy in patients with unresectable CD30+ mesothelioma (NCT03007030).
A panel of ADCs that target trophoblast glycoprotein (also known as 5TA, an antigen expressed in several tumours) conjugated to a tubulin polymerisation inhibitor, yielded encouraging results in mesothelioma cells cultures expressing high levels of 5TA [183]. Another ADC was generated against the cell-surface glycoprotein CD26 [184], overexpressed in epithelioid and biphasic subtypes [185]. This ADC comprises the humanised anti-CD26 mAb YS110 and the TR-1 derivative of triptolide, a bioactive compound of Tripterygium wilfordii that shows a wide spectrum of anti-tumour activities [186]. It showed prominent cytotoxicity against mesothelioma and leukaemia cells in vitro and in vivo by impairing the RNA polymerase II activity through the TR-1-mediated inhibition of TFIIH, a transcription factor for RNA polymerase II [184].
Oncolytic viral therapies
Oncolytic viruses offer another promising therapeutic approach for several malignancies, including mesothelioma [187, 188], through their dual anti-tumour activity involving the direct killing by lysis of infected cells (alongside the release of viral progeny to propagate the effect in neighbouring cells) and indirect induction of immune responses. The interaction between pathogen and host surface receptors triggers the production of type I IFNs, which leads to viral clearance and the release of tumour-associated antigens, as well as danger signals of cellular and viral origin. These factors all stimulate the expression of major histocompatibility complex (MHC) class I proteins and the recruitment of lymphoid cells, such as dendritic cells, CD4+ and CD8+ T cells, as well as NK cells [189]. Oncolytic viruses show an intrinsic selectivity towards tumour cells, which is mostly due to malignant characteristics such as altered metabolism, defects in anti-viral responses, the loss of p53 or p16 tumour suppressors and the activation of aberrant oncogenic signalling pathways, including RAS–MEK–ERK/MAPK and Wnt [188, 190, 191].
Thanks to its localised nature, relative lack of metastasis and its physical accessibility, mesothelioma is an attractive candidate for viral therapy. The intrapleural or intraperitoneal administration of double-stranded RNA was reported in 1976 to prevent tumour growth [192] and the successful targeted delivery of a viral construct was later confirmed in vitro and in vivo [193]. To date, several viruses, including adenoviruses [194], herpes simplex virus type 1 (HSV-1) [195], measles virus [196], vaccinia virus [197], Newcastle disease virus [198], retrovirus [199] and reovirus [200], have been tested in mesothelioma. A number of genetic modifications have been introduced to enhance the anti-tumour specificity and therapeutic efficacy. For instance, a VEGF-promoter-based adenovirus was shown to selectively replicate in mesothelioma cell lines; in xenograft mouse models, this construct suppressed tumour growth and prolonged animal survival [201]. The intra-tumoural delivery of retrovirus expressing the yeast cytosine deaminase prodrug activator gene was demonstrated to kill mesothelioma cells in vitro and to abolish tumour growth in vivo upon administration of the prodrug 5-fluorocytosine [199]. Furthermore, oncolytic adenovirus armed with a CD46-binding sequence and a promoter of heparin-binding growth factor midkine (both are highly expressed in mesothelioma cell lines) has been reported to confer enhanced infectivity and mesothelioma-specific cytotoxicity [202].
Another promising oncolytic virus for mesothelioma therapy is a replication-competent HSV in which the viral genes encoding ICP6 and ICP34.5 have been deleted. The lack of ICP6 confers virus selectivity towards mitotic cells or cells with a p16INK4a deletion [203], suggesting a potential benefit in the treatment of mesothelioma with a loss of function of CDKN2A. Deletion of ICP34.5, a viral homologue of GADD34, markedly reduces the neurovirulence of the construct but inhibits viral protein synthesis [204], which consequently blunts viral replication and therapeutic efficacy. However, the homology of ICP34.5 to the DNA-damage-inducible GADD34 might enable viral replication to be complemented by the synergistic use of this construct with chemo- or radiotherapy [205, 206]; alternatively, defective viral replication could be overcome by introducing the GADD34 gene into the HSV genome [207]. The latter approach might be particularly relevant for more aggressive histological subtypes of mesothelioma, which are characterised by a loss of GADD34 [8].
Current clinical trials
Clinical trials investigating oncolytic viral therapy for mesothelioma are summarised in [Table 2]. The results from completed studies indicate that the adenovirus-mediated delivery of IFNα2b induced an anti-tumour immune response [208], which, when combined with chemotherapy as second-line treatment, significantly increased the OS [209]. When the clinical utility of vaccinia virus GL-ONC1 was assessed in patients with peritoneal cancers, including mesothelioma, tumour cell infection, virus replication and oncolysis were limited to the first cycle of treatment, possibly due to component-mediated virus inactivation as a result of the development of neutralising activities against GL-ONC1 [210]. The outcome of a phase II clinical trial investigating the effect of nivolumab in combination with MTG201, a replication-incompetent adenovirus containing the gene encoding REIC/Dkk-3, which confers anti-tumour activity, for mesothelioma is yet to be unveiled (NCT04013334).
Conclusions and future perspectives
Although studies of mesothelioma biology have revolutionised our understanding of this cancer, the prognosis for newly diagnosed patients nevertheless remains poor. The current epidemic of mesothelioma that is affecting industrialised countries will soon peak, but the number of cases is predicted to tail well into this century [211]. Unfortunately, the global consumption of asbestos remains undiminished owing to its growing use in middle-income nations, notably India and China [212]. Modern materials might also pose a future risk for mesothelioma: carbon nanotubes, for example, induce a mesothelioma-like pathology in preclinical models [213]. It is therefore important that clinical research focuses on mesothelioma.
Targeted therapies and immunotherapies that show promise in early trials frequently later fail in phase III trials. This might plausibly reflect a failure to pre-select those patients most likely to benefit from a particular treatment. Improved personalisation of therapies, through stratification of individuals by tumour type, antigen expression, or even genotype, might help overcome this [214]. There is cause for optimism in the treatment of mesothelioma. Innovative therapeutic approaches are now being directed towards mesothelioma [Fig. 3], while increased investment is enabling the development of better in vitro and in vivo model systems that should help to increase our chances of identifying effective therapies in the coming decade . It is crucial that this goal is achieved before the next wave of mesothelioma hits those countries that are currently repeating the mistakes of the West by consuming tonnes of asbestos to grow their economies.
Immune checkpoint inhibitors block signalling that suppresses immune-cell activity, such as PD-1–PD-L1 and B7-1/2–CTLA-4 interactions, which is upregulated by tumour cells. Tumour-associated antigens (TAAs) can be targeted using monoclonal antibodies against proteins such as mesothelin, by vaccine therapy to stimulate the immune response and by using antibody–drug conjugates (ADCs) that target proteins such as mesothelin, 5TA, CD26 and CD30. Autologous dendritic cells pulsed with autologous or allogeneic tumour cell lysate act to prime host immunity, while chimeric antigen receptor CAR T cells contain chimaeric receptors that have been generated to specifically bind to TAAs on the cell surface. Other therapeutic approaches include oncolytic viruses that directly kill cancer cells by lysis or indirectly by stimulating immune response. They have been engineered to increase viral specificity (by introducing TAAs), cytotoxicity (by introducing e.g. pro-apoptotic or immunostimulatory genes), and monitoring (by introducing reporter genes).
a Embryonic pleural development. (i) Representation of the trilaminar germ disc following gastrulation together with the amniotic cavity and definitive yolk sac. (ii) During gastrulation, the mesoderm layer forms and the lateral plate mesoderm subdivides into somatic and splanchnic mesoderm. (iii) Following lateral flexion, the intra-embryonic coelom is lined by somatic and splanchnic mesoderm. (iv) The pleuropericardial folds extend to the midline and fuse with the ventral surface of foregut mesoderm, forming the primitive pleural cavities. (v) The somatic and splanchnic mesoderm is lined by parietal and visceral pleura respectively, which are contiguous with each other at the level of the hilum. b Anatomy of the thorax showing the relationship between the lungs, thoracic ribcage and pleura. The pleural cavity is defined by the space between adjacent visceral and parietal pleura. The pleura can be subdivided into five layers: mesothelial cells, basal lamina, superficial elastic layer, connective tissue layer and the deep fibroelastic layer. The deep layer is tightly adhered to the underlying structures e.g. muscle, rib or lung parenchyme.
Data availability
Not applicable.
References
Beckett P, Edwards J, Fennell D, Hubbard R, Woolhouse I, Peake MD. Demographics, management and survival of patients with malignant pleural mesothelioma in the National Lung Cancer Audit in England and Wales. Lung Cancer. 2015;88:344–8.
van Zandwijk N, Clarke C, Henderson D, Musk AW, Fong K, Nowak A, et al. Guidelines for the diagnosis and treatment of malignant pleural mesothelioma. J Thorac Dis. 2013;5:E254–307.
Schramm A, Opitz I, Thies S, Seifert B, Moch H, Weder W, et al. Prognostic significance of epithelial-mesenchymal transition in malignant pleural mesothelioma. Eur J Cardiothorac Surg. 2010;37:566–72.
Bozzi F, Brich S, Dagrada GP, Negri T, Conca E, Cortelazzi B, et al. Epithelioid peritoneal mesothelioma: a hybrid phenotype within a mesenchymal-epithelial/epithelial-mesenchymal transition framework. Oncotarget. 2016;7:75503–17.
Fassina A, Cappellesso R, Guzzardo V, Dalla Via L, Piccolo S, Ventura L, et al. Epithelial-mesenchymal transition in malignant mesothelioma. Mod Pathol. 2012;25:86–99.
Ceresoli GL, Locati LD, Ferreri AJ, Cozzarini C, Passoni P, Melloni G, et al. Therapeutic outcome according to histologic subtype in 121 patients with malignant pleural mesothelioma. Lung Cancer. 2001;34:279–87.
Fennell DA, Parmar A, Shamash J, Evans MT, Sheaff MT, Sylvester R, et al. Statistical validation of the EORTC prognostic model for malignant pleural mesothelioma based on three consecutive phase II trials. J Clin Oncol. 2005;23:184–9.
Dalton LE, Clarke HJ, Knight J, Lawson MH, Wason J, Lomas DA, et al. The endoplasmic reticulum stress marker CHOP predicts survival in malignant mesothelioma. Br J Cancer. 2013;108:1340–7.
Dell’Anno I, Barone E, Mutti L, Rassl DM, Marciniak SJ, Silvestri R, et al. Tissue expression of lactate transporters (MCT1 and MCT4) and prognosis of malignant pleural mesothelioma (brief report). J Transl Med. 2020;18:341.
Chia PL, Russell P, Asadi K, Thapa B, Gebski V, Murone C, et al. Analysis of angiogenic and stromal biomarkers in a large malignant mesothelioma cohort. Lung Cancer. 2020;150:1–8.
Schramm A, Opitz I, Thies S, Seifert B, Moch H, Weder W, et al. Prognostic significance of epithelial–mesenchymal transition in malignant pleural mesothelioma. Eur J Cardiothorac Surg. 2010;37:566–72.
Milano MT, Zhang H. Malignant pleural mesothelioma: a population-based study of survival. J Thorac Oncol. 2010;5:1841–8.
Vogelzang NJ, Rusthoven JJ, Symanowski J, Denham C, Kaukel E, Ruffie P, et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J Clin Oncol. 2003;21:2636–44.
Ivanov SV, Miller J, Lucito R, Tang C, Ivanova AV, Pei J, et al. Genomic events associated with progression of pleural malignant mesothelioma. Int J Cancer. 2009;124:589–99.
Guo G, Chmielecki J, Goparaju C, Heguy A, Dolgalev I, Carbone M, et al. Whole-exome sequencing reveals frequent genetic alterations in BAP1, NF2, CDKN2A, and CUL1 in malignant pleural mesothelioma. Cancer Res. 2015;75:264–9.
Bueno R, Stawiski EW, Goldstein LD, Durinck S, De Rienzo A, Modrusan Z, et al. Comprehensive genomic analysis of malignant pleural mesothelioma identifies recurrent mutations, gene fusions and splicing alterations. Nat Genet. 2016;48:407–16.
Illei PB, Rusch VW, Zakowski MF, Ladanyi M. Homozygous deletion of CDKN2A and codeletion of the methylthioadenosine phosphorylase gene in the majority of pleural mesotheliomas. Clin Cancer Res. 2003;9:2108–13.
Thurneysen C, Opitz I, Kurtz S, Weder W, Stahel RA, Felley-Bosco E. Functional inactivation of NF2/merlin in human mesothelioma. Lung Cancer. 2009;64:140–7.
Bott M, Brevet M, Taylor BS, Shimizu S, Ito T, Wang L, et al. The nuclear deubiquitinase BAP1 is commonly inactivated by somatic mutations and 3p21.1 losses in malignant pleural mesothelioma. Nat Genet. 2011;43:668–72.
Kato S, Tomson BN, Buys TP, Elkin SK, Carter JL, Kurzrock R. Genomic landscape of malignant mesotheliomas. Mol Cancer Ther. 2016;15:2498–507.
Hmeljak J, Sanchez-Vega F, Hoadley KA, Shih J, Stewart C, Heiman D, et al. Integrative molecular characterization of malignant pleural mesothelioma. Cancer Discov. 2018;8:1548–65.
Carbone M, Yang H, Pass HI, Krausz T, Testa JR, Gaudino G. BAP1 and cancer. Nat Rev Cancer. 2013;13:153–9.
Carbone M, Harbour JW, Brugarolas J, Bononi A, Pagano I, Dey A, et al. Biological mechanisms and clinical significance of BAP1 mutations in human cancer. Cancer Discov. 2020;10:1103–20.
Carbone M, Adusumilli PS, Alexander HR Jr, Baas P, Bardelli F, Bononi A, et al. Mesothelioma: scientific clues for prevention, diagnosis, and therapy. CA Cancer J Clin. 2019;69:402–29.
Testa JR, Cheung M, Pei J, Below JE, Tan Y, Sementino E, et al. Germline BAP1 mutations predispose to malignant mesothelioma. Nat Genet. 2011;43:1022–5.
Pastorino S, Yoshikawa Y, Pass HI, Emi M, Nasu M, Pagano I, et al. A subset of mesotheliomas with improved survival occurring in carriers of BAP1 and other germline mutations. J Clin Oncol. 2018;36:JCO2018790352.
Baumann F, Flores E, Napolitano A, Kanodia S, Taioli E, Pass H, et al. Mesothelioma patients with germline BAP1 mutations have 7-fold improved long-term survival. Carcinogenesis. 2015;36:76–81.
Carbone M, Arron ST, Beutler B, Bononi A, Cavenee W, Cleaver JE, et al. Tumour predisposition and cancer syndromes as models to study gene-environment interactions. Nat Rev Cancer. 2020;20:533–49.
Bononi A, Giorgi C, Patergnani S, Larson D, Verbruggen K, Tanji M, et al. BAP1 regulates IP3R3-mediated Ca(2+) flux to mitochondria suppressing cell transformation. Nature. 2017;546:549–53.
Napolitano A, Pellegrini L, Dey A, Larson D, Tanji M, Flores EG, et al. Minimal asbestos exposure in germline BAP1 heterozygous mice is associated with deregulated inflammatory response and increased risk of mesothelioma. Oncogene. 2016;35:1996–2002.
Xu J, Kadariya Y, Cheung M, Pei J, Talarchek J, Sementino E, et al. Germline mutation of Bap1 accelerates development of asbestos-induced malignant mesothelioma. Cancer Res. 2014;74:4388–97.
LaFave LM, Beguelin W, Koche R, Teater M, Spitzer B, Chramiec A, et al. Loss of BAP1 function leads to EZH2-dependent transformation. Nat Med. 2015;21:1344–9.
Petrilli AM, Fernandez-Valle C. Role of Merlin/NF2 inactivation in tumor biology. Oncogene. 2016;35:537–48.
Fleury-Feith J, Lecomte C, Renier A, Matrat M, Kheuang L, Abramowski V, et al. Hemizygosity of Nf2 is associated with increased susceptibility to asbestos-induced peritoneal tumours. Oncogene. 2003;22:3799–805.
Jongsma J, van Montfort E, Vooijs M, Zevenhoven J, Krimpenfort P, van der Valk M, et al. A conditional mouse model for malignant mesothelioma. Cancer Cell. 2008;13:261–71.
Sato T, Sekido Y. NF2/Merlin inactivation and potential therapeutic targets in mesothelioma. Int J Mol Sci. 2018;19:988.
Tranchant R, Quetel L, Tallet A, Meiller C, Renier A, de Koning L, et al. Co-occurring mutations of tumor suppressor genes, LATS2 and NF2, in malignant pleural mesothelioma. Clin Cancer Res. 2017;23:3191–202.
Miyanaga A, Masuda M, Tsuta K, Kawasaki K, Nakamura Y, Sakuma T, et al. Hippo pathway gene mutations in malignant mesothelioma: revealed by RNA and targeted exon sequencing. J Thorac Oncol. 2015;10:844–51.
Zhang WQ, Dai YY, Hsu PC, Wang H, Cheng L, Yang YL, et al. Targeting YAP in malignant pleural mesothelioma. J Cell Mol Med. 2017;21:2663–76.
Wu H, Wei L, Fan F, Ji S, Zhang S, Geng J, et al. Integration of Hippo signalling and the unfolded protein response to restrain liver overgrowth and tumorigenesis. Nat Commun. 2015;6:6239.
Sherr CJ. The INK4a/ARF network in tumour suppression. Nat Rev Mol Cell Biol. 2001;2:731–7.
Bonelli MA, Digiacomo G, Fumarola C, Alfieri R, Quaini F, Falco A, et al. Combined inhibition of CDK4/6 and PI3K/AKT/mTOR pathways induces a synergistic anti-tumor effect in malignant pleural mesothelioma cells. Neoplasia. 2017;19:637–48.
Marques M, Tranchant R, Risa-Ebri B, Suarez-Solis ML, Fernandez LC, Carrillo-de-Santa-Pau E, et al. Combined MEK and PI3K/p110beta inhibition as a novel targeted therapy for malignant mesothelioma displaying sarcomatoid features. Cancer Res. 2020;80:843–56.
Blum Y, Meiller C, Quetel L, Elarouci N, Ayadi M, Tashtanbaeva D, et al. Dissecting heterogeneity in malignant pleural mesothelioma through histo-molecular gradients for clinical applications. Nat Commun. 2019;10:1333.
de Reynies A, Jaurand MC, Renier A, Couchy G, Hysi I, Elarouci N, et al. Molecular classification of malignant pleural mesothelioma: identification of a poor prognosis subgroup linked to the epithelial-to-mesenchymal transition. Clin Cancer Res. 2014;20:1323–34.
Galateau Salle F, Le Stang N, Tirode F, Courtiol P, Nicholson AG, Tsao MS, et al. Comprehensive molecular and pathologic evaluation of transitional mesothelioma assisted by deep learning approach: a multi-institutional study of the International Mesothelioma Panel from the MESOPATH Reference Center. J Thorac Oncol. 2020;15:1037–53.
Dawson MA, Kouzarides T. Cancer epigenetics: from mechanism to therapy. Cell. 2012;150:12–27.
Vandermeers F, Sriramareddy Neelature, Costa S, Hubaux C, Cosse R, Willems JP. L. The role of epigenetics in malignant pleural mesothelioma. Lung Cancer. 2013;81:311–8.
Kang HC, Kim HK, Lee S, Mendez P, Kim JW, Woodard G, et al. Whole exome and targeted deep sequencing identify genome-wide allelic loss and frequent SETDB1 mutations in malignant pleural mesotheliomas. Oncotarget. 2016;7:8321–31.
Paik PK, Krug LM. Histone deacetylase inhibitors in malignant pleural mesothelioma: preclinical rationale and clinical trials. J Thorac Oncol. 2010;5:275–9.
Tarasenko N, Nudelman A, Tarasenko I, Entin-Meer M, Hass-Kogan D, Inbal A, et al. Histone deacetylase inhibitors: the anticancer, antimetastatic and antiangiogenic activities of AN-7 are superior to those of the clinically tested AN-9 (Pivanex). Clin Exp Metastasis. 2008;25:703–16.
Martinez-Iglesias O, Ruiz-Llorente L, Sanchez-Martinez R, Garcia L, Zambrano A, Aranda A. Histone deacetylase inhibitors: mechanism of action and therapeutic use in cancer. Clin Transl Oncol. 2008;10:395–8.
Krug LM, Curley T, Schwartz L, Richardson S, Marks P, Chiao J, et al. Potential role of histone deacetylase inhibitors in mesothelioma: clinical experience with suberoylanilide hydroxamic acid. Clin Lung Cancer. 2006;7:257–61.
Krug LM, Kindler HL, Calvert H, Manegold C, Tsao AS, Fennell D, et al. Vorinostat in patients with advanced malignant pleural mesothelioma who have progressed on previous chemotherapy (VANTAGE-014): a phase 3, double-blind, randomised, placebo-controlled trial. Lancet Oncol. 2015;16:447–56.
Bensaid D, Blondy T, Deshayes S, Dehame V, Bertrand P, Gregoire M, et al. Assessment of new HDAC inhibitors for immunotherapy of malignant pleural mesothelioma. Clin Epigenetics. 2018;10:79.
Sacco JJ, Kenyani J, Butt Z, Carter R, Chew HY, Cheeseman LP, et al. Loss of the deubiquitylase BAP1 alters class I histone deacetylase expression and sensitivity of mesothelioma cells to HDAC inhibitors. Oncotarget. 2015;6:13757–71.
Amatori S, Papalini F, Lazzarini R, Donati B, Bagaloni I, Rippo MR, et al. Decitabine, differently from DNMT1 silencing, exerts its antiproliferative activity through p21 upregulation in malignant pleural mesothelioma (MPM) cells. Lung Cancer. 2009;66:184–90.
Balatti V, Maniero S, Ferracin M, Veronese A, Negrini M, Ferrocci G, et al. MicroRNAs dysregulation in human malignant pleural mesothelioma. J Thorac Oncol. 2011;6:844–51.
Reid G, Pel ME, Kirschner MB, Cheng YY, Mugridge N, Weiss J, et al. Restoring expression of miR-16: a novel approach to therapy for malignant pleural mesothelioma. Ann Oncol. 2013;24:3128–35.
Kubo T, Toyooka S, Tsukuda K, Sakaguchi M, Fukazawa T, Soh J, et al. Epigenetic silencing of microRNA-34b/c plays an important role in the pathogenesis of malignant pleural mesothelioma. Clin Cancer Res. 2011;17:4965–74.
Xu Y, Zheng M, Merritt RE, Shrager JB, Wakelee HA, Kratzke RA, et al. miR-1 induces growth arrest and apoptosis in malignant mesothelioma. Chest. 2013;144:1632–43.
Ivanov SV, Goparaju CM, Lopez P, Zavadil J, Toren-Haritan G, Rosenwald S, et al. Pro-tumorigenic effects of miR-31 loss in mesothelioma. J Biol Chem. 2010;285:22809–17.
Cioce M, Ganci F, Canu V, Sacconi A, Mori F, Canino C, et al. Protumorigenic effects of mir-145 loss in malignant pleural mesothelioma. Oncogene. 2014;33:5319–31.
van Zandwijk N, Pavlakis N, Kao SC, Linton A, Boyer MJ, Clarke S, et al. Safety and activity of microRNA-loaded minicells in patients with recurrent malignant pleural mesothelioma: a first-in-man, phase 1, open-label, dose-escalation study. Lancet Oncol. 2017;18:1386–96.
Lee BY, Timpson P, Horvath LG, Daly RJ. FAK signaling in human cancer as a target for therapeutics. Pharmacol Ther. 2015;146:132–49.
Kanteti R, Mirzapoiazova T, Riehm JJ, Dhanasingh I, Mambetsariev B, Wang J, et al. Focal adhesion kinase a potential therapeutic target for pancreatic cancer and malignant pleural mesothelioma. Cancer Biol Ther. 2018;19:316–27.
Laszlo V, Valko Z, Ozsvar J, Kovacs I, Garay T, Hoda MA, et al. The FAK inhibitor BI 853520 inhibits spheroid formation and orthotopic tumor growth in malignant pleural mesothelioma. J Mol Med (Berl). 2019;97:231–42.
Poulikakos PI, Xiao GH, Gallagher R, Jablonski S, Jhanwar SC, Testa JR. Re-expression of the tumor suppressor NF2/merlin inhibits invasiveness in mesothelioma cells and negatively regulates FAK. Oncogene. 2006;25:5960–8.
Shapiro IM, Kolev VN, Vidal CM, Kadariya Y, Ring JE, Wright Q, et al. Merlin deficiency predicts FAK inhibitor sensitivity: a synthetic lethal relationship. Sci Transl Med. 2014;6:237ra268.
Soria JC, Gan HK, Blagden SP, Plummer R, Arkenau HT, Ranson M, et al. A phase I, pharmacokinetic and pharmacodynamic study of GSK2256098, a focal adhesion kinase inhibitor, in patients with advanced solid tumors. Ann Oncol. 2016;27:2268–74.
Mak G, Soria JC, Blagden SP, Plummer R, Fleming RA, Nebot N, et al. A phase Ib dose-finding, pharmacokinetic study of the focal adhesion kinase inhibitor GSK2256098 and trametinib in patients with advanced solid tumours. Br J Cancer. 2019;120:975–81.
Fennell DA, Baas P, Taylor P, Nowak AK, Gilligan D, Nakano T, et al. Maintenance defactinib versus placebo after first-line chemotherapy in patients with merlin-stratified pleural mesothelioma: COMMAND-a double-blind, randomized, phase II study. J Clin Oncol. 2019;37:790–8.
Jagadeeswaran R, Ma PC, Seiwert TY, Jagadeeswaran S, Zumba O, Nallasura V, et al. Functional analysis of c-Met/hepatocyte growth factor pathway in malignant pleural mesothelioma. Cancer Res. 2006;66:352–61.
Kanteti R, Dhanasingh I, Kawada I, Lennon FE, Arif Q, Bueno R, et al. MET and PI3K/mTOR as a potential combinatorial therapeutic target in malignant pleural mesothelioma. PLoS ONE. 2014;9:e105919.
De Palma M, Biziato D, Petrova TV. Microenvironmental regulation of tumour angiogenesis. Nat Rev Cancer. 2017;17:457–74.
Strizzi L, Catalano A, Vianale G, Orecchia S, Casalini A, Tassi G, et al. Vascular endothelial growth factor is an autocrine growth factor in human malignant mesothelioma. J Pathol. 2001;193:468–75.
Kumar-Singh S, Weyler J, Martin MJ, Vermeulen PB, Van Marck E. Angiogenic cytokines in mesothelioma: a study of VEGF, FGF-1 and -2, and TGF beta expression. J Pathol. 1999;189:72–78.
Klominek J, Baskin B, Hauzenberger D. Platelet-derived growth factor (PDGF) BB acts as a chemoattractant for human malignant mesothelioma cells via PDGF receptor beta-integrin alpha3beta1 interaction. Clin Exp Metastasis. 1998;16:529–39.
Kumar-Singh S, Vermeulen PB, Weyler J, Segers K, Weyn B, Van Daele A, et al. Evaluation of tumour angiogenesis as a prognostic marker in malignant mesothelioma. J Pathol. 1997;182:211–6.
Scagliotti GV, Gaafar R, Nowak AK, Reck M, Tsao AS, van Meerbeeck J, et al. LUME-Meso: design and rationale of the phase III part of a placebo-controlled study of nintedanib and pemetrexed/cisplatin followed by maintenance nintedanib in patients with unresectable malignant pleural mesothelioma. Clin Lung Cancer. 2017;18:589–93.
Grosso F, Steele N, Novello S, Nowak AK, Popat S, Greillier L, et al. Nintedanib plus pemetrexed/cisplatin in patients with malignant pleural mesothelioma: phase II results from the randomized, placebo-controlled LUME-Meso trial. J Clin Oncol. 2017;35:3591–3600.
Garland LL, Chansky K, Wozniak AJ, Tsao AS, Gadgeel SM, Verschraegen CF, et al. Phase II study of cediranib in patients with malignant pleural mesothelioma: SWOG S0509. J Thorac Oncol. 2011;6:1938–45.
Figg WD, Dahut W, Duray P, Hamilton M, Tompkins A, Steinberg SM, et al. A randomized phase II trial of thalidomide, an angiogenesis inhibitor, in patients with androgen-independent prostate cancer. Clin Cancer Res. 2001;7:1888–93.
Fujii T, Tachibana M, Dhar DK, Ueda S, Kinugasa S, Yoshimura H, et al. Combination therapy with paclitaxel and thalidomide inhibits angiogenesis and growth of human colon cancer xenograft in mice. Anticancer Res. 2003;23:2405–11.
Buikhuisen WA, Burgers JA, Vincent AD, Korse CM, van Klaveren RJ, Schramel FM, et al. Thalidomide versus active supportive care for maintenance in patients with malignant mesothelioma after first-line chemotherapy (NVALT 5): an open-label, multicentre, randomised phase 3 study. Lancet Oncol. 2013;14:543–51.
Wedge SR, Kendrew J, Hennequin LF, Valentine PJ, Barry ST, Brave SR, et al. AZD2171: a highly potent, orally bioavailable, vascular endothelial growth factor receptor-2 tyrosine kinase inhibitor for the treatment of cancer. Cancer Res. 2005;65:4389–4400.
Tsao AS, Miao J, Wistuba II, Vogelzang NJ, Heymach JV, Fossella, et al. Phase II trial of cediranib in combination with cisplatin and pemetrexed in chemotherapy-naive patients with unresectable malignant pleural mesothelioma (SWOG S0905). J Clin Oncol. 2019;37:2537–47.
Chappell NP, Miller CR, Fielden AD, Barnett JC. Is FDA-approved bevacizumab cost-effective when included in the treatment of platinum-resistant recurrent ovarian cancer? J Oncol Pract. 2016;12:e775–783.
Murakami S, Saito H, Kondo T, Oshita F, Yamada K. Phase II study of bevacizumab, cisplatin, and pemetrexed in advanced non-squamous non-small cell lung cancer (NS-NSCLC) with EGFR wild-type. J Exp Ther Oncol. 2019;13:131–8.
Zalcman G, Mazieres J, Margery J, Greillier L, Audigier-Valette C, Moro-Sibilot D, et al. Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open-label, phase 3 trial. Lancet. 2016;387:1405–14.
Levin PA, Dowell JE. Spotlight on bevacizumab and its potential in the treatment of malignant pleural mesothelioma: the evidence to date. Onco Targets Ther. 2017;10:2057–66.
Urra H, Dufey E, Avril T, Chevet E, Hetz C. Endoplasmic reticulum stress and the hallmarks of cancer. Trends Cancer. 2016;2:252–62.
Martinotti S, Ranzato E, Burlando B. (-)- Epigallocatechin-3-gallate induces GRP78 accumulation in the ER and shifts mesothelioma constitutive UPR into proapoptotic ER stress. J Cell Physiol. 2018;233:7082–90.
Xu D, Yang H, Yang Z, Berezowska S, Gao Y, Liang SQ, et al. Endoplasmic reticulum stress signaling as a therapeutic target in malignant pleural mesothelioma. Cancers (Basel). 2019;11:1502.
Zhang L, Littlejohn JE, Cui Y, Cao X, Peddaboina C, Smythe WR. Characterization of bortezomib-adapted I-45 mesothelioma cells. Mol Cancer. 2010;9:110.
Cerezo M, Lehraiki A, Millet A, Rouaud F, Plaisant M, Jaune E, et al. Compounds triggering ER stress exert anti-melanoma effects and overcome BRAF inhibitor resistance. Cancer Cell. 2016;30:183.
Sarkar Bhattacharya S, Thirusangu P, Jin L, Roy D, Jung D, Xiao Y, et al. PFKFB3 inhibition reprograms malignant pleural mesothelioma to nutrient stress-induced macropinocytosis and ER stress as independent binary adaptive responses. Cell Death Dis. 2019;10:725.
Szlosarek PW, Klabatsa A, Pallaska A, Sheaff M, Smith P, Crook T, et al. In vivo loss of expression of argininosuccinate synthetase in malignant pleural mesothelioma is a biomarker for susceptibility to arginine depletion. Clin Cancer Res. 2006;12:7126–31.
Szlosarek PW, Steele JP, Nolan L, Gilligan D, Taylor P, Spicer J, et al. Arginine deprivation with pegylated arginine deiminase in patients with argininosuccinate synthetase 1-deficient malignant pleural mesothelioma: a randomized clinical trial. JAMA Oncol. 2017;3:58–66.
Phillips M, Szyszko T, Hall P, Cook GJR, Khadeir R, Steele JPC, et al. Expansion study of ADI-PEG 20, pemetrexed and cisplatin in patients with ASS1-deficient malignant pleural mesothelioma (TRAP). J Clin Oncol. 2017;35:8553–8553.
Yamada N, Oizumi S, Kikuchi E, Shinagawa N, Konishi-Sakakibara J, Ishimine A, et al. CD8+ tumor-infiltrating lymphocytes predict favorable prognosis in malignant pleural mesothelioma after resection. Cancer Immunol Immunother. 2010;59:1543–9.
Hegmans JP, Hemmes A, Hammad H, Boon L, Hoogsteden HC, Lambrecht BN. Mesothelioma environment comprises cytokines and T-regulatory cells that suppress immune responses. Eur Respir J. 2006;27:1086–95.
Burt BM, Rodig SJ, Tilleman TR, Elbardissi AW, Bueno R, Sugarbaker DJ. Circulating and tumor-infiltrating myeloid cells predict survival in human pleural mesothelioma. Cancer. 2011;117:5234–44.
Anraku M, Cunningham KS, Yun Z, Tsao MS, Zhang L, Keshavjee S, et al. Impact of tumor-infiltrating T cells on survival in patients with malignant pleural mesothelioma. J Thorac Cardiovasc Surg. 2008;135:823–9.
Cornelissen R, Lievense LA, Maat AP, Hendriks RW, Hoogsteden HC, Bogers AJ, et al. Ratio of intratumoral macrophage phenotypes is a prognostic factor in epithelioid malignant pleural mesothelioma. PLoS ONE. 2014;9:e106742.
Tazzari M, Brich S, Tuccitto A, Bozzi F, Beretta V, Spagnuolo RD, et al. Complex immune contextures characterise malignant peritoneal mesothelioma: loss of adaptive immunological signature in the more aggressive histological types. J Immunol Res. 2018;2018:5804230.
Fusco N, Vaira V, Righi I, Sajjadi E, Venetis K, Lopez G, et al. Characterization of the immune microenvironment in malignant pleural mesothelioma reveals prognostic subgroups of patients. Lung Cancer. 2020;150:53–61.
Robinson BW, Robinson C, Lake RA. Localised spontaneous regression in mesothelioma - possible immunological mechanism. Lung Cancer. 2001;32:197–201.
Luke JJ, Flaherty KT, Ribas A, Long GV. Targeted agents and immunotherapies: optimizing outcomes in melanoma. Nat Rev Clin Oncol. 2017;14:463–82.
Teachey DT, Hunger SP. Acute lymphoblastic leukaemia in 2017: immunotherapy for ALL takes the world by storm. Nat Rev Clin Oncol. 2018;15:69–70.
Assi HI, Kamphorst AO, Moukalled NM, Ramalingam SS. Immune checkpoint inhibitors in advanced non-small cell lung cancer. Cancer. 2018;124:248–61.
Marcq E, Siozopoulou V, De Waele J, van Audenaerde J, Zwaenepoel K, Santermans E, et al. Prognostic and predictive aspects of the tumor immune microenvironment and immune checkpoints in malignant pleural mesothelioma. Oncoimmunology. 2017;6:e1261241.
Cedres S, Ponce-Aix S, Zugazagoitia J, Sansano I, Enguita A, Navarro-Mendivil A, et al. Analysis of expression of programmed cell death 1 ligand 1 (PD-L1) in malignant pleural mesothelioma (MPM). PLoS ONE. 2015;10:e0121071.
Mansfield AS, Roden AC, Peikert T, Sheinin YM, Harrington SM, Krco CJ, et al. B7-H1 expression in malignant pleural mesothelioma is associated with sarcomatoid histology and poor prognosis. J Thorac Oncol. 2014;9:1036–40.
Calabro L, Morra A, Fonsatti E, Cutaia O, Amato G, Giannarelli D, et al. Tremelimumab for patients with chemotherapy-resistant advanced malignant mesothelioma: an open-label, single-arm, phase 2 trial. Lancet Oncol. 2013;14:1104–11.
Calabro L, Morra A, Fonsatti E, Cutaia O, Fazio C, Annesi D, et al. Efficacy and safety of an intensified schedule of tremelimumab for chemotherapy-resistant malignant mesothelioma: an open-label, single-arm, phase 2 study. Lancet Respir Med. 2015;3:301–9.
Maio M, Scherpereel A, Calabro L, Aerts J, Cedres Perez S, Bearz A, et al. Tremelimumab as second-line or third-line treatment in relapsed malignant mesothelioma (DETERMINE): a multicentre, international, randomised, double-blind, placebo-controlled phase 2b trial. Lancet Oncol. 2017;18:1261–73.
Alley EW, Lopez J, Santoro A, Morosky A, Saraf S, Piperdi B, et al. Clinical safety and activity of pembrolizumab in patients with malignant pleural mesothelioma (KEYNOTE-028): preliminary results from a non-randomised, open-label, phase 1b trial. Lancet Oncol. 2017;18:623–30.
Desai A, Karrison T, Rose B, Tan Y, Hill B, Pemberton E, et al. OA08.03 phase II trial of pembrolizumab (NCT02399371) in previously-treated malignant mesothelioma (MM): final analysis. J Thorac Oncol. 2018;13:S339.
Rivalland G, Kao SC-H, Pavlakis N, Hughes BGM, Thapa B, Pal A, et al. Outcomes of anti-PD-1 therapy in mesothelioma and correlation with PD-L1 expression. J Clin Oncol. 2017;35:8514.
Goto Y, Okada M, Kijima T, Aoe K, Kato T, Fujimoto N, et al. MA 19.01 a phase II study of nivolumab: a multicenter, open-label, single arm study in malignant pleural mesothelioma (MERIT). J Thorac Oncol. 2017;12:S1883.
Quispel-Janssen J, van der Noort V, de Vries JF, Zimmerman M, Lalezari F, Thunnissen E, et al. Programmed death 1 blockade with nivolumab in patients with recurrent malignant pleural mesothelioma. J Thorac Oncol. 2018;13:1569–76.
Fennell DA, Kirkpatrick E, Cozens K, Nye M, Lester J, Hanna G, et al. CONFIRM: a double-blind, placebo-controlled phase III clinical trial investigating the effect of nivolumab in patients with relapsed mesothelioma: study protocol for a randomised controlled trial. Trials. 2018;19:233.
Nowak A, Kok P, Lesterhuis W, Hughes B, Brown C, Kao S, et al. OA08.02 DREAM - a phase 2 trial of durvalumab with first line chemotherapy in mesothelioma: final result. J Thorac Oncol. 2018;13:S338–S339.
Hassan R, Thomas A, Nemunaitis JJ, Patel MR, Bennouna J, Chen FL, et al. Efficacy and safety of avelumab treatment in patients with advanced unresectable mesothelioma: phase 1b results from the JAVELIN Solid Tumor Trial. JAMA Oncol. 2019;5:351–7.
Calabro L, Morra A, Giannarelli D, Amato G, D’Incecco A, Covre A, et al. Tremelimumab combined with durvalumab in patients with mesothelioma (NIBIT-MESO-1): an open-label, non-randomised, phase 2 study. Lancet Respir Med. 2018;6:451–60.
Venkatraman D, Anderson A, Digumarthy S, Lizotte PH, Awad MM. Phase 2 study of tremelimumab plus durvalumab for previously-treated malignant pleural mesothelioma (MPM). J Clin Oncol. 2019;37:8549–8549.
Disselhorst MJ, Quispel-Janssen J, Lalezari F, Monkhorst K, de Vries JF, van der Noort V, et al. Ipilimumab and nivolumab in the treatment of recurrent malignant pleural mesothelioma (INITIATE): results of a prospective, single-arm, phase 2 trial. Lancet Respir Med. 2019;7:260–70.
Scherpereel A, Mazieres J, Greillier L, Lantuejoul S, Do P, Bylicki O, et al. Nivolumab or nivolumab plus ipilimumab in patients with relapsed malignant pleural mesothelioma (IFCT-1501 MAPS2): a multicentre, open-label, randomised, non-comparative, phase 2 trial. Lancet Oncol. 2019;20:239–53.
Zalcman G, Mazieres J, Greillier L, Lantuejoul S, Dô P, Bylicki O, et al. LBA58_PRSecond or 3rd line nivolumab (Nivo) versus nivo plus ipilimumab (Ipi) in malignant pleural mesothelioma (MPM) patients: updated results of the IFCT-1501 MAPS2 randomized phase 2 trial. Ann Oncol. 2017;28:v605–49.
Baas P, Scherpereel A, Nowak AK, Fujimoto N, Peters S, Tsao AS, et al. First-line nivolumab plus ipilimumab in unresectable malignant pleural mesothelioma (CheckMate 743): a multicentre, randomised, open-label, phase 3 trial. Lancet. 2021;397:375–86.
Webster I, Cochrane JW, Burkhardt KR. Immunotherapy with BCG vaccine in 30 cases of mesothelioma. S Afr Med J. 1982;61:277–8.
Fuge O, Vasdev N, Allchorne P, Green JS. Immunotherapy for bladder cancer. Res Rep Urol. 2015;7:65–79.
Li Q, Kawamura K, Yang S, Okamoto S, Kobayashi H, Tada Y, et al. Interferon-beta produces synergistic combinatory anti-tumor effects with cisplatin or pemetrexed on mesothelioma cells. PLoS ONE. 2013;8:e72709.
Christmas TI, Manning LS, Garlepp MJ, Musk AW, Robinson BW. Effect of interferon-alpha 2a on malignant mesothelioma. J Interferon Res. 1993;13:9–12.
Boutin C, Nussbaum E, Monnet I, Bignon J, Vanderschueren R, Guerin JC, et al. Intrapleural treatment with recombinant gamma-interferon in early stage malignant pleural mesothelioma. Cancer. 1994;74:2460–7.
Yanagawa H, Sone S, Fukuta K, Nishioka Y, Ogura T. Local adoptive immunotherapy using lymphokine-activated killer cells and interleukin-2 against malignant pleural mesothelioma: report of two cases. Jpn J Clin Oncol. 1991;21:377–83.
Astoul P, Picat-Joossen D, Viallat JR, Boutin C. Intrapleural administration of interleukin-2 for the treatment of patients with malignant pleural mesothelioma: a phase II study. Cancer. 1998;83:2099–104.
Castagneto B, Zai S, Mutti L, Lazzaro A, Ridolfi R, Piccolini E, et al. Palliative and therapeutic activity of IL-2 immunotherapy in unresectable malignant pleural mesothelioma with pleural effusion: Results of a phase II study on 31 consecutive patients. Lung Cancer. 2001;31:303–10.
Ordonez NG. Value of mesothelin immunostaining in the diagnosis of mesothelioma. Mod Pathol. 2003;16:192–7.
Tian L, Zeng R, Wang X, Shen C, Lai Y, Wang M, et al. Prognostic significance of soluble mesothelin in malignant pleural mesothelioma: a meta-analysis. Oncotarget. 2017;8:46425–35.
Hassan R, Cohen SJ, Phillips M, Pastan I, Sharon E, Kelly RJ, et al. Phase I clinical trial of the chimeric anti-mesothelin monoclonal antibody MORAb-009 in patients with mesothelin-expressing cancers. Clin Cancer Res. 2010;16:6132–8.
Hassan R, Kindler HL, Jahan T, Bazhenova L, Reck M, Thomas A, et al. Phase II clinical trial of amatuximab, a chimeric antimesothelin antibody with pemetrexed and cisplatin in advanced unresectable pleural mesothelioma. Clin Cancer Res. 2014;20:5927–36.
Le DT, Brockstedt DG, Nir-Paz R, Hampl J, Mathur S, Nemunaitis J, et al. A live-attenuated Listeria vaccine (ANZ-100) and a live-attenuated Listeria vaccine expressing mesothelin (CRS-207) for advanced cancers: phase I studies of safety and immune induction. Clin Cancer Res. 2012;18:858–68.
Hassan R, Alley E, Kindler H, Antonia S, Jahan T, Honarmand S, et al. Clinical response of live-attenuated, Listeria monocytogenes expressing mesothelin (CRS-207) with chemotherapy in patients with malignant pleural mesothelioma. Clin Cancer Res. 2019;25:5787–98.
Alley EW, Tanvetyanon T, Jahan TM, Gandhi L, Peikert T, Stevenson J, et al. A phase II single-arm study of CRS-207 with pembrolizumab (pembro) in previously treated malignant pleural mesothelioma (MPM). J Clin Oncol. 2019;37:29–29.
Hassan R, Bullock S, Premkumar A, Kreitman RJ, Kindler H, Willingham MC, et al. Phase I study of SS1P, a recombinant anti-mesothelin immunotoxin given as a bolus I.V. infusion to patients with mesothelin-expressing mesothelioma, ovarian, and pancreatic cancers. Clin Cancer Res. 2007;13:5144–9.
Hassan R, Miller AC, Sharon E, Thomas A, Reynolds JC, Ling A, et al. Major cancer regressions in mesothelioma after treatment with an anti-mesothelin immunotoxin and immune suppression. Sci Transl Med. 2013;5:208ra147.
Zhang J, Khanna S, Jiang Q, Alewine C, Miettinen M, Pastan I, et al. Efficacy of anti-mesothelin immunotoxin RG7787 plus Nab-paclitaxel against mesothelioma patient-derived xenografts and mesothelin as a biomarker of tumor response. Clin Cancer Res. 2017;23:1564–74.
Amin KM, Litzky LA, Smythe WR, Mooney AM, Morris JM, Mews DJ, et al. Wilms’ tumor 1 susceptibility (WT1) gene products are selectively expressed in malignant mesothelioma. Am J Pathol. 1995;146:344–56.
Krug LM, Dao T, Brown AB, Maslak P, Travis W, Bekele S, et al. WT1 peptide vaccinations induce CD4 and CD8 T cell immune responses in patients with mesothelioma and non-small cell lung cancer. Cancer Immunol Immunother. 2010;59:1467–79.
Okada M, Kijima T, Aoe K, Kato T, Fujimoto N, Nakagawa K, et al. Clinical efficacy and safety of nivolumab: results of a multicenter, open-label, single-arm, Japanese phase II study in malignant pleural mesothelioma (MERIT). Clin Cancer Res. 2019;25:5485–92.
Hegmans JP, Hemmes A, Aerts JG, Hoogsteden HC, Lambrecht BN. Immunotherapy of murine malignant mesothelioma using tumor lysate-pulsed dendritic cells. Am J Respir Crit Care Med. 2005;171:1168–77.
Hegmans JP, Veltman JD, Lambers ME, de Vries IJ, Figdor CG, Hendriks RW, et al. Consolidative dendritic cell-based immunotherapy elicits cytotoxicity against malignant mesothelioma. Am J Respir Crit Care Med. 2010;181:1383–90.
Cornelissen R, Hegmans JP, Maat AP, Kaijen-Lambers ME, Bezemer K, Hendriks RW, et al. Extended tumor control after dendritic cell vaccination with low-dose cyclophosphamide as adjuvant treatment in patients with malignant pleural mesothelioma. Am J Respir Crit Care Med. 2016;193:1023–31.
Aerts J, de Goeje PL, Cornelissen R, Kaijen-Lambers MEH, Bezemer K, van der Leest CH, et al. Autologous dendritic cells pulsed with allogeneic tumor cell lysate in mesothelioma: from mouse to human. Clin Cancer Res. 2018;24:766–76.
Belderbos RA, Baas P, Berardi R, Cornelissen R, Fennell DA, van Meerbeeck JP, et al. A multicenter, randomized, phase II/III study of dendritic cells loaded with allogeneic tumor cell lysate (MesoPher) in subjects with mesothelioma as maintenance therapy after chemotherapy: DENdritic cell Immunotherapy for Mesothelioma (DENIM) trial. Transl Lung Cancer Res. 2019;8:280–5.
Fesnak AD, June CH, Levine BL. Engineered T cells: the promise and challenges of cancer immunotherapy. Nat Rev Cancer. 2016;16:566–81.
Park JH, Geyer MB, Brentjens RJ. CD19-targeted CAR T-cell therapeutics for hematologic malignancies: interpreting clinical outcomes to date. Blood. 2016;127:3312–20.
Beatty GL, Haas AR, Maus MV, Torigian DA, Soulen MC, Plesa G, et al. Mesothelin-specific chimeric antigen receptor mRNA-engineered T cells induce anti-tumor activity in solid malignancies. Cancer Immunol Res. 2014;2:112–20.
Maus MV, Haas AR, Beatty GL, Albelda SM, Levine BL, Liu X, et al. T cells expressing chimeric antigen receptors can cause anaphylaxis in humans. Cancer Immunol Res. 2013;1:26–31.
Klampatsa A, Haas AR, Moon EK, Albelda SM. Chimeric antigen receptor (CAR) T cell therapy for malignant pleural mesothelioma (MPM). Cancers (Basel). 2017;9:115.
Cherkassky L, Morello A, Villena-Vargas J, Feng Y, Dimitrov DS, Jones DR, et al. Human CAR T cells with cell-intrinsic PD-1 checkpoint blockade resist tumor-mediated inhibition. J Clin Invest. 2016;126:3130–44.
Adusumilli PS, Zauderer MG, Rusch VW, O’Cearbhaill R, Zhu A, Ngai D, et al. Regional delivery of mesothelin-targeted CAR T cells for pleural cancers: safety and preliminary efficacy in combination with anti-PD-1 agent. J Clin Oncol. 2019;37:2511–2511.
Curioni A, Britschgi C, Hiltbrunner S, Bankel L, Gulati P, Weder W, et al. 1226P A phase I clinical trial of malignant pleural mesothelioma treated with locally delivered autologous anti-FAP-targeted CAR T-cells. Ann Oncol. 2019;30:mdz253.
Hiltbrunner S, Britschgi C, Schuberth P, Bankel L, Nguyen-Kim TDL, Gulati P, et al. Local delivery of CAR T cells targeting fibroblast activation protein is safe in patients with pleural mesothelioma: first report of FAPME, a phase I clinical trial. Ann Oncol. 2021;32:120–1.
Klampatsa A, Achkova DY, Davies DM, Parente-Pereira AC, Woodman N, Rosekilly J, et al. Intracavitary ‘T4 immunotherapy’ of malignant mesothelioma using pan-ErbB re-targeted CAR T-cells. Cancer Lett. 2017;393:52–59.
Beard RE, Zheng Z, Lagisetty KH, Burns WR, Tran E, Hewitt SM, et al. Multiple chimeric antigen receptors successfully target chondroitin sulfate proteoglycan 4 in several different cancer histologies and cancer stem cells. J Immunother Cancer. 2014;2:25.
Rivera Z, Ferrone S, Wang X, Jube S, Yang H, Pass HI, et al. CSPG4 as a target of antibody-based immunotherapy for malignant mesothelioma. Clin Cancer Res. 2012;18:5352–63.
Barabas K, Sizensky JA, Faulk WP. Evidence in support of the plasma membrane as the target for transferrin-adriamycin conjugates in K562 cells. Am J Reprod Immunol. 1991;25:120–3.
Thomas A, Teicher BA, Hassan R. Antibody-drug conjugates for cancer therapy. Lancet Oncol. 2016;17:e254–e262.
Xie H, Adjei AA. Antibody-drug conjugates for the therapy of thoracic malignancies. J Thorac Oncol. 2019;14:358–76.
Pastan I, Hassan R. Discovery of mesothelin and exploiting it as a target for immunotherapy. Cancer Res. 2014;74:2907–12.
Golfier S, Kopitz C, Kahnert A, Heisler I, Schatz CA, Stelte-Ludwig B, et al. Anetumab ravtansine: a novel mesothelin-targeting antibody-drug conjugate cures tumors with heterogeneous target expression favored by bystander effect. Mol Cancer Ther. 2014;13:1537–48.
Kindler HL, Novello S, Fennell D, Blumenschein G, Bearz A, Ceresoli G, et al. OA 02.01 randomized phase II study of anetumab ravtansine or vinorelbine in patients with metastatic pleural mesothelioma. J Thorac Oncol. 2017;12:S1746.
Scales SJ, Gupta N, Pacheco G, Firestein R, French DM, Koeppen H, et al. An antimesothelin-monomethyl auristatin e conjugate with potent antitumor activity in ovarian, pancreatic, and mesothelioma models. Mol Cancer Ther. 2014;13:2630–40.
Weekes CD, Lamberts LE, Borad MJ, Voortman J, McWilliams RR, Diamond JR, et al. Phase I study of DMOT4039A, an antibody-drug conjugate targeting mesothelin, in patients with unresectable pancreatic or platinum-resistant ovarian cancer. Mol Cancer Ther. 2016;15:439–47.
Francisco JA, Cerveny CG, Meyer DL, Mixan BJ, Klussman K, Chace DF, et al. cAC10-vcMMAE, an anti-CD30-monomethyl auristatin E conjugate with potent and selective antitumor activity. Blood. 2003;102:1458–65.
Dabir S, Kresak A, Yang M, Fu P, Wildey G, Dowlati A. CD30 is a potential therapeutic target in malignant mesothelioma. Mol Cancer Ther. 2015;14:740–6.
van der Weyden CA, Pileri SA, Feldman AL, Whisstock J, Prince HM. Understanding CD30 biology and therapeutic targeting: a historical perspective providing insight into future directions. Blood Cancer J. 2017;7:e603.
Yi JH, Kim SJ, Kim WS. Brentuximab vedotin: clinical updates and practical guidance. Blood Res. 2017;52:243–53.
Sharman JP, Wheler JJ, Einhorn L, Dowlati A, Shapiro GI, Hilton J, et al. A phase 2, open-label study of brentuximab vedotin in patients with CD30-expressing solid tumors. Invest New Drugs. 2019;37:738–47.
Schunselaar LM, Monkhorst K, van der Noort V, Wijdeven R, Peters D, Zwart W, et al. Trophoblast glycoprotein is associated with a favorable outcome for mesothelioma and a target for antibody drug conjugates. J Thorac Oncol. 2018;13:1577–87.
Hayashi M, Madokoro H, Yamada K, Nishida H, Morimoto C, Sakamoto M, et al. Novel antibody-drug conjugate with anti-CD26 humanized monoclonal antibody and transcription factor IIH (TFIIH) inhibitor, triptolide, inhibits tumor growth via impairing mRNA synthesis. Cancers (Basel). 2019;11:1138.
Aoe K, Amatya VJ, Fujimoto N, Ohnuma K, Hosono O, Hiraki A, et al. CD26 overexpression is associated with prolonged survival and enhanced chemosensitivity in malignant pleural mesothelioma. Clin Cancer Res. 2012;18:1447–56.
Zhou ZL, Yang YX, Ding J, Li YC, Miao ZH. Triptolide: structural modifications, structure-activity relationships, bioactivities, clinical development and mechanisms. Nat Prod Rep. 2012;29:457–75.
Pease DF, Kratzke RA. Oncolytic viral therapy for mesothelioma. Front Oncol. 2017;7:179.
Kaufman HL, Kohlhapp FJ, Zloza A. Oncolytic viruses: a new class of immunotherapy drugs. Nat Rev Drug Discov. 2015;14:642–62.
Bommareddy PK, Shettigar M, Kaufman HL. Integrating oncolytic viruses in combination cancer immunotherapy. Nat Rev Immunol. 2018;18:498–513.
Seymour LW, Fisher KD. Oncolytic viruses: finally delivering. Br J Cancer. 2016;114:357–61.
Harrington K, Freeman DJ, Kelly B, Harper J, Soria JC. Optimizing oncolytic virotherapy in cancer treatment. Nat Rev Drug Discov. 2019;18:689–706.
Pimm MV, Baldwin RW. Treatment of transplanted rat tumours with double-stranded RNA(BRL 5907). II. Treatment of pleural and peritoneal growths. Br J Cancer. 1976;33:166–71.
Smythe WR, Kaiser LR, Hwang HC, Amin KM, Pilewski JM, Eck SJ, et al. Successful adenovirus-mediated gene transfer in an in vivo model of human malignant mesothelioma. Ann Thorac Surg. 1994;57:1395–401.
Kuryk L, Haavisto E, Garofalo M, Capasso C, Hirvinen M, Pesonen S, et al. Synergistic anti-tumor efficacy of immunogenic adenovirus ONCOS-102 (Ad5/3-D24-GM-CSF) and standard of care chemotherapy in preclinical mesothelioma model. Int J Cancer. 2016;139:1883–93.
Adusumilli PS, Stiles BM, Chan MK, Mullerad M, Eisenberg DP, Ben-Porat L, et al. Imaging and therapy of malignant pleural mesothelioma using replication-competent herpes simplex viruses. J Gene Med. 2006;8:603–15.
Li H, Peng KW, Dingli D, Kratzke RA, Russell SJ. Oncolytic measles viruses encoding interferon beta and the thyroidal sodium iodide symporter gene for mesothelioma virotherapy. Cancer Gene Ther. 2010;17:550–8.
Kelly KJ, Woo Y, Brader P, Yu Z, Riedl C, Lin SF, et al. Novel oncolytic agent GLV-1h68 is effective against malignant pleural mesothelioma. Hum Gene Ther. 2008;19:774–82.
Silberhumer GR, Brader P, Wong J, Serganova IS, Gonen M, Gonzalez SJ, et al. Genetically engineered oncolytic Newcastle disease virus effectively induces sustained remission of malignant pleural mesothelioma. Mol Cancer Ther. 2010;9:2761–9.
Kawasaki Y, Tamamoto A, Takagi-Kimura M, Maeyama Y, Yamaoka N, Terada N, et al. Replication-competent retrovirus vector-mediated prodrug activator gene therapy in experimental models of human malignant mesothelioma. Cancer Gene Ther. 2011;18:571–8.
Comins C, Spicer J, Protheroe A, Roulstone V, Twigger K, White CM, et al. REO-10: a phase I study of intravenous reovirus and docetaxel in patients with advanced cancer. Clin Cancer Res. 2010;16:5564–72.
Harada A, Uchino J, Harada T, Nakagaki N, Hisasue J, Fujita M, et al. Vascular endothelial growth factor promoter-based conditionally replicative adenoviruses effectively suppress growth of malignant pleural mesothelioma. Cancer Sci. 2017;108:116–23.
Takagi-Kimura M, Yamano T, Tamamoto A, Okamura N, Okamura H, Hashimoto-Tamaoki T, et al. Enhanced antitumor efficacy of fiber-modified, midkine promoter-regulated oncolytic adenovirus in human malignant mesothelioma. Cancer Sci. 2013;104:1433–9.
Aghi M, Visted T, Depinho RA, Chiocca EA. Oncolytic herpes virus with defective ICP6 specifically replicates in quiescent cells with homozygous genetic mutations in p16. Oncogene. 2008;27:4249–54.
Li Y, Zhang C, Chen X, Yu J, Wang Y, Yang Y, et al. ICP34.5 protein of herpes simplex virus facilitates the initiation of protein translation by bridging eukaryotic initiation factor 2alpha (eIF2alpha) and protein phosphatase 1. J Biol Chem. 2011;286:24785–92.
Bennett JJ, Adusumilli P, Petrowsky H, Burt BM, Roberts G, Delman KA, et al. Up-regulation of GADD34 mediates the synergistic anticancer activity of mitomycin C and a gamma134.5 deleted oncolytic herpes virus (G207). FASEB J. 2004;18:1001–3.
Adusumilli PS, Stiles BM, Chan MK, Chou TC, Wong RJ, Rusch VW, et al. Radiation therapy potentiates effective oncolytic viral therapy in the treatment of lung cancer. Ann Thorac Surg. 2005;80:409–16.
Nakashima H, Nguyen T, Kasai K, Passaro C, Ito H, Goins WF, et al. Toxicity and efficacy of a novel GADD34-expressing oncolytic HSV-1 for the treatment of experimental glioblastoma. Clin Cancer Res. 2018;24:2574–84.
Sterman DH, Haas A, Moon E, Recio A, Schwed D, Vachani A, et al. A trial of intrapleural adenoviral-mediated Interferon-alpha2b gene transfer for malignant pleural mesothelioma. Am J Respir Crit Care Med. 2011;184:1395–9.
Sterman DH, Alley E, Stevenson JP, Friedberg J, Metzger S, Recio A, et al. Pilot and feasibility trial evaluating immuno-gene therapy of malignant mesothelioma using intrapleural delivery of adenovirus-IFNalpha combined with chemotherapy. Clin Cancer Res. 2016;22:3791–3800.
Lauer UM, Schell M, Beil J, Berchtold S, Koppenhofer U, Glatzle J, et al. Phase I study of oncolytic vaccinia virus GL-ONC1 in patients with peritoneal carcinomatosis. Clin Cancer Res. 2018;24:4388–98.
Gilham C, Rake C, Hodgson J, Darnton A, Burdett G, Peto Wild J, et al. Past and current asbestos exposure and future mesothelioma risks in Britain: The Inhaled Particles Study (TIPS). Int J Epidemiol. 2018;47:1745–56.
Frank AL, Joshi TK. The global spread of asbestos. Ann Glob Health. 2014;80:257–62.
Chernova T, Murphy FA, Galavotti S, Sun XM, Powley IR, Grosso S, et al. Long-fiber carbon nanotubes replicate asbestos-induced mesothelioma with disruption of the tumor suppressor gene Cdkn2a (Ink4a/Arf). Curr Biol. 2017;27:3302.e6–14.e6.
Shamseddin M, Obacz J, Garnett MJ, Rintoul RC, Francies HE, Marciniak SJ. Use of preclinical models for malignant pleural mesothelioma. Thorax. 2021. https://doi.org/10.1136/thoraxjnl-2020-216602.
Winters NI, Williams AM, Bader DM. Resident progenitors, not exogenous migratory cells, generate the majority of visceral mesothelium in organogenesis. Dev Biol. 2014;391:125–32.
Dixit R, Ai X, Fine A. Derivation of lung mesenchymal lineages from the fetal mesothelium requires hedgehog signaling for mesothelial cell entry. Development. 2013;140:4398–406.
Winters N, Bader D. Development of the serosal mesothelium. J Dev Biol. 2013;1:64–81.
Karki S, Surolia R, Hock TD, Guroji P, Zolak JS, Duggal R, et al. Wilms’ tumor 1 (Wt1) regulates pleural mesothelial cell plasticity and transition into myofibroblasts in idiopathic pulmonary fibrosis. FASEB J. 2014;28:1122–31.
Rump A, Morikawa Y, Tanaka M, Minami S, Umesaki N, Takeuchi M, et al. Binding of ovarian cancer antigen CA125/MUC16 to mesothelin mediates cell adhesion. J Biol Chem. 2004;279:9190–8.
Bera TK, Pastan I. Mesothelin is not required for normal mouse development or reproduction. Mol Cell Biol. 2000;20:2902–6.
Funding
JO and MS are British Lung Foundation-Victor Dahdaleh Charitable Foundation-Royal Papworth Hospital Research Fellows. HY is supported by a Scadding-Morriston-Davies fellowship, XL by the British Lung Foundation and the China Scholarship Council (CSC) and DF-J by the Royal Society funding through a University Research Fellowship. RCR is supported by the NIHR Cambridge Biomedical Research Centre (BRC-1215-20014), Cancer Research UK Cambridge Centre, British Lung Foundation (VPDCF17-18, MKMRFPG18-6) and Royal Papworth Hospital. SJM is supported by the Medical Research Council (MR/R009120/1, MR/V028669/1), Engineering and Physical Sciences Research Council (EP/R03558X/1), British Lung Foundation (MKMRFPG18-6), June Hancock Mesothelioma Research Fund (JH-18-07), the NIHR Cambridge Biomedical Research Centre (BRC-1215-20014), Royal Papworth Hospital and the Alpha1-Foundation. DMR is supported by the NIHR Cambridge Biomedical Research Centre (BRC-1215-20014) and British Lung Foundation (VPDCF17-18). MZN acknowledges funding from the Rutherford Fund Fellowship allocated by the MRC as part of the UK Regenerative Medicine Platform 2 (MR/5005579/1). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care.
Author information
Authors and Affiliations
Contributions
JO contributed to work design, wrote and revised the manuscript. HY, MS, EL, XL and AA wrote parts of the manuscript and contributed to figures’ design. DMR revised the manuscript and provided the images of the H&E staining of mesothelioma. DF-J, RCR and MZN extensively revised the manuscript. SJM designed the work, revised and finalised the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Obacz, J., Yung, H., Shamseddin, M. et al. Biological basis for novel mesothelioma therapies. Br J Cancer 125, 1039–1055 (2021). https://doi.org/10.1038/s41416-021-01462-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01462-2
This article is cited by
-
Clonal gene signatures predict prognosis in mesothelioma and lung adenocarcinoma
npj Precision Oncology (2024)
-
Genomic and transcriptomic analysis of a diffuse pleural mesothelioma patient-derived xenograft library
Genome Medicine (2022)