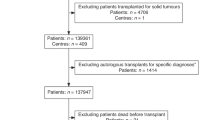
Abstract
The Web-based One Year Survival Outcomes Calculator developed by the Center for International Blood and Marrow Transplant Research (CIBMTR) applies large-scale registry data to generate individualized estimates of overall survival (OS) probability 1 year after first allogeneic hematopoietic cell transplant (HCT) and can therefore provide a data-driven foundation for personalized patient counseling. We assessed the calibration of the CIBMTR One Year Survival Outcomes Calculator when applied to retrospective data among adult recipients of first allogeneic HCT for acute myeloid leukemia (AML), acute lymphoblastic leukemia (ALL), or myelodysplastic syndrome (MDS) with peripheral blood stem cell transplant (PBSCT) from a 7/8- or 8/8-matched donor from 2000 through 2015 at a single center. Predicted 1 year OS was estimated for each patient using the CIBMTR Calculator. Corresponding observed 1 year OS was estimated for each group by the Kaplan-Meier method. A weighted Kaplan-Meier estimator was used to visually display the average of observed 1 year survival estimates over the continuous range of predicted OS. In the first analysis of its kind, we demonstrated that the CIBMTR One Year Survival Outcomes Calculator could be applied to larger patient cohorts and predicted 1 year prognosis with general agreement between predicted and observed survival.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated during and analyzed during the current study are not publicly available due to patient privacy considerations but are available in deidentified form from the corresponding author on reasonable request.
References
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the disease risk index for allogeneic stem cell transplantation. Blood. 2014;123:3664–71.
Gentleman R, Crowley J. Graphical methods for censored-data. J Am Stat Assoc. 1991;86:678–83.
Papadopoulos EB, Carabasi MH, Castro-Malaspina H, Childs BH, Mackinnon S, Boulad F, et al. T-cell-depleted allogeneic bone marrow transplantation as postremission therapy for acute myelogenous leukemia: freedom from relapse in the absence of graft-versus-host disease. Blood. 1998;91:1083–90.
Jakubowski AA, Small TN, Young JW, Kernan NA, Castro-Malaspina H, Hsu KC, et al. T cell depleted stem-cell transplantation for adults with hematologic malignancies: sustained engraftment of HLA-matched related donor grafts without the use of antithymocyte globulin. Blood. 2007;110:4552–9.
Castro-Malaspina H, Jabubowski AA, Papadopoulos EB, Boulad F, Young JW, Kernan NA, et al. Transplantation in remission improves the disease-free survival of patients with advanced myelodysplastic syndromes treated with myeloablative T cell-depleted stem cell transplants from HLA-identical siblings. Biol Blood Marrow Transpl. 2008;14:458–68.
Pasquini MC, Luznik L, Logan B, Soiffer R, Wu J, Devine S, et al., editors. Calcineurin inhibitor-free graft-versus-host disease (GVHD) prophylaxis in hematopoietic cell transplantation (HCT) with myeloablative conditioning regimens (MAC) and HLA-matched donors: results of the BMT CTN 1301 Progress II Trial. Oral presentation at: Transplant and Cellular Therapy Meetings of ASTCT and CIBMTR, February 2021; virtual meeting. https://tct.confex.com/tandem/2021/meetingapp.cgi/Paper/18048. Accessed 1 Nov 2021.
McDonald GB, Sandmaier BM, Mielcarek M, Sorror M, Pergam SA, Cheng GS, et al. Survival, nonrelapse mortality, and relapse-related mortality after allogeneic hematopoietic cell transplantation: comparing 2003-2007 versus 2013-2017 cohorts. Ann Intern Med. 2020;172:229–39.
Shouval R, Fein JA, Cho C, Avecilla S, Ruiz JD, Alarcon Tomas A, et al. The Simplified Comorbidity Index (SCI)—a new tool for prediction of non-relapse mortality in allogeneic HCT. Blood Adv. 2022;6:1525–35.
Shouval R, Fein JA, Labopin M, Cho C, Bazarbachi A, Baron F, et al. Development and validation of a disease risk stratification system for patients with haematological malignancies: a retrospective cohort study of the european society for blood and marrow transplantation registry. Lancet Haematol. 2021;8:e205–e15.
Shouval R, Labopin M, Bondi O, Mishan-Shamay H, Shimoni A, Ciceri F, et al. Prediction of allogeneic hematopoietic stem-cell transplantation mortality 100 days after transplantation using a machine learning algorithm: a european group for blood and marrow transplantation acute leukemia working party retrospective data mining study. J Clin Oncol. 2015;33:3144–51.
Holtan SG, Zhang L, DeFor TE, Bejanyan N, Arora M, Rashidi A, et al. Dynamic graft-versus-host disease-free, relapse-free survival: multistate modeling of the morbidity and mortality of allotransplantation. Biol Blood Marrow Transpl. 2019;25:1884–9.
Gerstung M, Papaemmanuil E, Martincorena I, Bullinger L, Gaidzik VI, Paschka P, et al. Precision oncology for acute myeloid leukemia using a knowledge bank approach. Nat Genet. 2017;49:332–40.
Acknowledgements
This work was supported in part by National Institutes of Health/National Cancer Institute grant P01 CA23766 and National Institutes of Health/National Cancer Institute Cancer Center Support grant P30 CA008748.
Author information
Authors and Affiliations
Contributions
CC, SD, SAG, and MAP designed the study. SD performed the biostatistical analyses. MM served as the data manager. CC curated the data. CC and SD wrote the manuscript. MMH, BL, JDR, BS, SAG, and MAP critically reviewed the manuscript and made substantive contributions to the text and interpretation of the data.
Corresponding author
Ethics declarations
Competing interests
CC, SD, MM, MMH, BL: None. JDR: Optum Stem Cell - Compensation as participant on expert panel to provide input/information about the center-specific survival analysis; compensation directed to Medical College of Wisconsin rather than personally. SAG: Amgen, Actinium, BMS, Celgene, GSK, Kite, Janssen, Jazz Pharma, Johnson & Johnson, Novartis, Pfizer, Sanofi, Spectrum Pharma, Takeda - Membership on board of directors or advisory committees. Amgen, Actinium, Celgene, Johnson & Johnson, Miltenyi, Omeros - Institutional research support. M-AP—Adicet, Allovir, Caribou Biosciences, Celgene, Bristol-Myers Squibb, Equilium, Exevir, Incyte, Karyopharm, Kite/Gilead, Merck, Miltenyi Biotec, MorphoSys, Nektar Therapeutics, Novartis, Omeros, OrcaBio, Syncopation, VectivBio AG, Vor Biopharma - Honoraria. Cidara Therapeutics, Medigene, Sellas Life Sciences - DSMB. NexImmune - Membership on advisory committee. NexImmune, Omeros, OrcaBio - Ownership interests. Other. Incyte, Kite/Gilead, Miltenyi Biotec, Nektar Therapeutics, Novartis - Institutional research support.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cho, C., Devlin, S., Maloy, M. et al. Application of the CIBMTR One Year Survival Outcomes Calculator as a tool for retrospective analysis. Bone Marrow Transplant 58, 1089–1095 (2023). https://doi.org/10.1038/s41409-023-02031-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-023-02031-2