Abstract
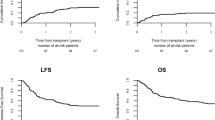
Withdrawal of radiotherapy in patients with brain tumors under four years decreases chance for cure. AutoHSCT in a series of pilot studies demonstrated a potential to improve outcomes in these patients. The study included 50 patients with median age of 39 months (7–53). Medulloblastoma (n = 28, 56%), ETMR (n = 9, 18%) and other histological types (n = 13, 26%) were most commonly diagnosed. Forty two patients (84%) received tandem autoHSCT by HIT-MED protocol, and single autoHSCT was performed in eight children (16%). Adjuvant radiotherapy was administered in 25 (50%) children and treatment of relapse included radiotherapy in 6 (12%). Median follow-up was 39.6 months (6–121). Long-term CIR was 37%, and TRM – 6%. Five-year OS was 71% in medulloblastoma, 37% in ETMR and in other tumors – 51% (p = 0.07). Irradiation-free OS at 5 years for children with medulloblastoma was 24%. For the whole cohort of CNS tumors, independently of histology, OS and PFS at five years were 60% and 46%, respectively Young children with medulloblastoma, following tandem autoHSCT, demonstrate OS comparable to older children. Patients with other histological types demonstrate suboptimal long-term survival rates after autoHSCT and one should assess whether these patients benefit from autoHSCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others

References
Ottensmeier H, Schlegel PG, Eyrich M, Wolff JE, Juhnke B-O, von Hoff K, et al. Treatment of children under four years of age with medulloblastoma and ependymoma in the HIT2000/HIT-REZ 2005 trials: Neuropsychological outcome 5 years after treatment. PLoS ONE. 2020;15:e0227693.
Donahue B. Short- and long-term complications of radiation therapy for pediatric brain tumors. PediatrNeurosurg. 1992;18:207–17.
Fouladi M, Gilger E, Kocak M, Wallace D, Buchanan G, Reeves C, et al. Intellectual and functional outcome of children three years old or younger who have CNS malignancies. J ClinOncol. 2005;23:7152–60.
Mynarek M, von Hoff K, Pietsch T, Ottensmeier H, Warmuth-Metz M, Bison B, et al. Nonmetastatic medulloblastoma of early childhood: results from the prospective clinical trial HIT-2000 and an extended validation cohort. J ClinOncol 2020;38:2028–40. https://doi.org/10.1200/JCO.19.03057.
Thomas A, Noël G. Medulloblastoma: optimizing care with a multidisciplinary approach. J MultidiscipHealthc. 2019;12:335–47.
Kool M, Jones DT, Jäger N, Northcott PA, Pugh TJ, Hovestadt V, et al. ICGC PedBrain Tumor Project. Genome sequencing of SHH medulloblastoma predicts genotype-related response to smoothened inhibition. Cancer cell. 2014;25:393–405.
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, et al. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. ActaNeuropathol 2016;131:803–20. https://doi.org/10.1007/s00401-016-1545-1.
Dhall G, O’Neil SH, Ji L, Haley K, Whitaker AM, Nelson MD, et al. Excellent outcome of young children with nodular desmoplastic medulloblastoma treated on “Head Start” III: a multi-institutional, prospective clinical trial. Neuro-Oncol. 2020;22:1862–72.
Robinson GW, Rudneva VA, Buchhalter I, Billups CA, Waszak SM, Smith KS, et al. Risk-adapted therapy for young children with medulloblastoma (SJYC07): therapeutic and molecular outcomes from a multicentre, phase 2 trial. Lancet Oncol 2018;19:768–84. https://doi.org/10.1016/S1470-2045(18)30204-3.
Perez-Martinez A, Lassaletta A, Gonzalez-Vicent M, Sevilla J, Diaz MA, Madero L. High-dose chemotherapy with autologous stem cell rescue for children with high risk and recurrent medulloblastoma and supratentorial primitive neuroectodermal tumors. JNeurooncol. 2005;71:33–8.
Dinikina YV, Smirnova AY, Egorov AE, Chernova SI, Toshina YK, Belogurova MB. High dose chemotherapy and autologous stem cell rescue in children with pediatric malignancies. Cell Ther Transpl. 2019;8:50–51.
Lee SH, Son MH, Sung KW, Choi YB, Lee NH, Yoo KH, et al. Toxicity of tandem high-dose chemotherapy and autologous stem cell transplantation using carboplatin-thiotepa-etoposide and cyclophosphamide-melphalan regimens for malignant brain tumors in children and young adults. J Neurooncol. 2014;120:507–13.
Sung KW, Lim DH, Lee SH, Yoo KH, Koo HH, Kim JH, et al. Tandem high-dose chemotherapy and auto-SCT for malignant brain tumors in children under 3 years of age. BoneMarrowTransplant. 2013;48(Jul):932–8.
Guerra JA, Dhall G, Marachelian A, Castillo E, Malvar J, Wong K, et al. Marrow-ablative chemotherapy followed by tandem autologous hematopoietic cell transplantation in pediatric patients with malignant brain tumors. BoneMarrowTransplant. 2017;52:1543–8.
Ladenstein R, Glogova E, Lanza F Solid Tumours. In: Carreras E, Dufour C, Mohty M, Kröger N (eds). The EBMT handbook: hematopoietic stem cell transplantation and cellular therapies. 7th ed. Cham (CH): Springer; 2019, pp 685–98.
Dhall G, Grodman H, Ji L, Sands S, Gardner S, Dunkel IJ, et al. Outcome of children less than three years old at diagnosis with non-metastatic medulloblastoma treated with chemotherapy on the «Head Start» I and II protocols. Pediatr Blood Cancer. 2008;50:1169–75.
Juhnke BO, Gessi M, Gerber NU, Friedrich C, Haberler C, Mynarek M, et al. ETMR-14. Treatment of embryonal tumors with multilayered rosettes (ETMR) with carboplatin-etoposide induction and tandem high-dose chemotherapy within the prospective HIT-trials and registries. NeuroOncol 2020;22:iii325–iii326. https://doi.org/10.1093/neuonc/noaa222.218.
Curran EK, Le GM, Sainani KL, Propp JM, Fisher PG. Do children and adults differ in survival from medulloblastoma? A study from the SEER registry. J Neurooncol. 2009;95:81–85.
Jaramillo S, Grosshans DR, Philip N, Varan A, Akyüz C, McAleer MF, et al. Radiation for ETMR: Literature review and case series of patients treated with proton therapy. ClinTranslRadiatOncol. 2018;15:31–37. 7
Stensvold E, Krossnes BK, Lundar T, Due-Tønnessen BJ, Fric R, Due-Tønnessen P, et al. Outcome for children treated for medulloblastoma and supratentorial primitive neuroectodermal tumor (CNS-PNET) – a retrospective analysis spanning 40 years treatment. ActaOncol. 2017;56:698–705.
Raleigh DR, Tomlin B, Buono BD, Roddy E, Sear K, Byer L, et al. Survival after chemotherapy and stem cell transplant followed by delayed craniospinal irradiation is comparable to upfront craniospinal irradiation in pediatric embryonal brain tumor patients. J Neurooncol. 2017;131(Jan):359–68.
Lafay-Cousin L, Smith A, Chi SN, Wells E, Madden J, Margol A, et al. Clinical, Pathological, and Molecular Characterization of Infant Medulloblastomas Treated with Sequential High-Dose Chemotherapy. PediatrBloodCancer. 2016;63:1527–34. https://doi.org/10.1002/pbc.26042.
Johnston DL, Keene D, Bartels U, Bruce Crooks A-SC, Eisenstat D-D, Fryer C, et al. Medulloblastoma in children under the age of three years: a retrospective Canadian review. J Neurooncol. 2009;94:51–56.
Pompe RS, von Bueren AO, Mynarek M, von Hoff K, Friedrich C, et al. Intraventricular methotrexate as part of primary therapy for children with infant and/or metastatic medulloblastoma: Feasibility, acute toxicity and evidence for efficacy. Eur J Cancer. 2015;51:2634–42.
Berthold F, Boos J, Burdach S, Erttmann R, Henze G, Hermann J, et al. Myeloablative megatherapy with autologous stem-cell rescue versus oral maintenance chemotherapy as consolidation treatment in patients with high-risk neuroblastoma: a randomised controlled trial. Lancet Oncol. 2005;6:649–58.
Gajjar A, Chintagumpala M, Ashley D, Kellie S, Kun LE, Merchant TE, Gilbertson RJ. et al. Risk-adapted craniospinal radiotherapy followed by high-dose chemotherapy and stem-cell rescue in children with newly diagnosed medulloblastoma (St Jude Medulloblastoma-96): long-term results from a prospective, multicentre trial. Lancet Oncol. 2006;7:813–20. https://doi.org/10.1016/S1470-2045(06)70867-1.
Gyurkocza B, Sandmaier BM. Conditioning regimens for hematopoietic cell transplantation: one size does not fit all. Blood. 2014;124:344–53. https://doi.org/10.1182/blood-2014-02-514778.
Altshuler C, Haley K, Dhall G, Vasquez L, Gardner SL, Stanek J, et al. Decreased morbidity and mortality of autologous hematopoietic transplants for children with malignant central nervous system tumors: the ‘Head Start’. trials, 1991–2009 BoneMarrowTransplant. 2016;51:945–8. https://doi.org/10.1038/bmt.2016.45.
Nazemi KJ, Shen V, Finlay JL, Boyett J, Kocak M, Lafond D, et al. S. high incidence of veno-occlusive disease with myeloablative chemotherapy following craniospinal irradiation in children with newly diagnosed high-risk cns embryonal tumors: a report from the children’s oncology group (CCG-99702). Pediatr Blood Cancer. 2016;63:1563–70.
Cohen BH, Geyer JR, Miller DC, Curran JG, Zhou T, Holmes E, et al. Children’s Oncology Group. Pilot Study of Intensive Chemotherapy With Peripheral Hematopoietic Cell Support for Children Less Than 3 Years of Age With Malignant Brain Tumors, the CCG-99703 Phase I/II Study. A Report From the Children’s Oncology Group. Pediatr Neurol. 2015;53:31–46.
Author information
Authors and Affiliations
Contributions
Conceptualization, AGG and LSZ; methodology, OGZ; resources, ADK; software, PST; formal analysis, IVK; investigation, TVY; data curation, EVM; writing—original draft preparation, AVK and AGG; writing—review and editing, YAP and OGZ; supervision, LSZ.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gevorgian, A.G., Kozlov, A.V., Tolkunova, P.S. et al. Tandem autologous hematopoietic stem cell transplantation for embryonal brain tumors in infants and very young children. Bone Marrow Transplant 57, 607–612 (2022). https://doi.org/10.1038/s41409-022-01593-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-022-01593-x