Abstract
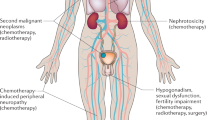
Male-specific late effects after hematopoietic cell transplantation (HCT) include genital chronic graft-versus-host disease (GvHD), hypogonadism, sexual dysfunction, infertility, and subsequent malignancies. They may be closely intertwined and cause prolonged morbidity and decreased quality of life after HCT. We provide a systematic review of male-specific late effects in a collaboration between transplant physicians, endocrinologists, urologists, dermatologists, and sexual health professionals through the Late Effects and Quality of Life Working Committee of the Center for International Blood and Marrow Transplant Research, and the Transplant Complications Working Party of the European Society of Blood and Marrow Transplantation. The systematic review summarizes incidence, risk factors, screening, prevention and treatment of these complications and provides consensus evidence-based recommendations for clinical practice and future research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
CIBMTR supports accessibility of research in accord with the National Institutes of Health (NIH) Data Sharing Policy and the National Cancer Institute (NCI) Cancer Moonshot Public Access and Data Sharing Policy. The CIBMTR only releases de-identified datasets that comply with all relevant global regulations regarding privacy and confidentiality.
References
Copelan EA. Hematopoietic stem-cell transplantation. N Engl J Med. 2006;354:1813–26. https://doi.org/10.1056/NEJMra052638.
Singh AK, McGuirk JP. Allogeneic stem cell transplantation: a historical and scientific overview. Cancer Res. 2016;76:6445–51. https://doi.org/10.1158/0008-5472.CAN-16-1311.
Bhatia S, Armenian SH, Landier W. How I monitor long-term and late effects after blood or marrow transplantation. Blood. 2017;130:1302–14. https://doi.org/10.1182/blood-2017-03-725671
Inamoto Y, Lee SJ. Late effects of blood and marrow transplantation. Haematologica. 2017;102:614–25. https://doi.org/10.3324/haematol.2016.150250
Tichelli A, Rovo A, Passweg J, Schwarze CP, Van Lint MT, Arat M, et al. Late complications after hematopoietic stem cell transplantation. Expert Rev Hematol. 2009;2:583–601. https://doi.org/10.1586/ehm.09.48
Hamilton BK, Goje O, Savani BN, Majhail NS, Stratton P. Clinical management of genital chronic GvHD. Bone Marrow Transpl. 2017;52:803–10. https://doi.org/10.1038/bmt.2016.315
Murphy J, McKenna M, Abdelazim S, Battiwalla M, Stratton P. A practical guide to gynecologic and reproductive health in women undergoing hematopoietic stem cell transplant. Blood Marrow Transplant. 2019;25:e331–e343. https://doi.org/10.1016/j.bbmt.2019.07.038.
Stratton P. Gynecologic care after hematopoietic cell transplantation: a call to action to include 646 gynecologists in the transplant team. Bone Marrow Transplant. 2015;50:1–2. https://doi.org/10.1038/bmt.2014.247.
Chow EJ, Anderson L, Baker KS, Bhatia S, Guilcher GM, Huang JT, et al. Late effects surveillance recommendations among survivors of childhood hematopoietic cell transplantation: a Children’s Oncology Group Report. Biol Blood Marrow Transplant. 2016;22:782–95. https://doi.org/10.1016/j.bbmt.2016.01.023.
Sharma A, Badawy S, Suelzer E, Eissa H, Murthy H, Prasad P, et al. Male-specific late effects after hematopoietic cell transplantation. In: PROSPERO 2020 CRD42020147640; 2020.
Sharma A, Badawy S, Suelzer E, Murphy E, Prasad P, Eissa H, et al. Systematic reviews in hematopoietic cell transplantation and cellular therapy: considerations and guidance from the American Society for Transplantation and Cellular Therapy, European Society for Blood and Marrow Transplantation, and Center for International Blood and Marrow Transplant Research Late Effects and Quality of Life Working Committee. Transplant Cell Ther. 2021;27:380–8. https://doi.org/10.1016/j.jtct.2020.12.002.
Sharma A, Badawy SM, Suelzer EM, Murthy HS, Prasad P, Eissa H, et al. Systematic reviews in hematopoietic cell transplantation and cellular therapy: considerations and guidance from the American Society for Transplantation and Cellular Therapy, European Society for Blood and Marrow Transplantation, and the Center for International Blood and Marrow Transplant Research late effects and quality of life working committee. Bone Marrow Transplant 2022. https://doi.org/10.1038/s41409-020-01199-1.
Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924–6. https://doi.org/10.1136/bmj.39489.470347.AD.
Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev. 2015;4:1 https://doi.org/10.1186/2046-4053-4-1.
Au WY, Yeung CK, Cheung MC, Trendell-Smith NJ. Penile lichen sclerosus after allogeneic stem cell transplantation. Br J Dermatol. 2008;159:470–2. https://doi.org/10.1111/j.1365-2133.2008.08645.x
Grigg AP, Underhill C, Russell J, Sale G. Peyronie’s disease as a complication of chronic graft versus host disease. Hematology. 2002;7:165–8. https://doi.org/10.1080/1024533021000008182.
Jain NA, Venkatesan K, Anandi P, Ito S, Kumar D, Lu K, et al. A rare consequence of chronic graft versus host disease-Peyronie’s disease. Arch Cancer Res. 2015;3:18. https://doi.org/10.21767/2254-6081.100018.
Kami M, Kanda Y, Sasaki M, Takeda N, Tanaka Y, Saito T, et al. Phimosis as a manifestation of chronic graft-versus-host disease after allogeneic bone marrow transplantation. Bone Marrow Transplant. 1998;21:721–3. https://doi.org/10.1038/sj.bmt.1701159.
Lara LA, De Andrade JM, Mauad LM, Ferrarese SR, Marana HR, Tiezzi DG, et al. Genital manifestation of graft-vs.-host disease: a series of case reports. J Sex Med. 2010;7:3216–25. https://doi.org/10.1111/j.1743-6109.2010.01885.x.
Nylander E, Wahlin YB, Lundskog B, Wahlin A. Genital graft-versus-host disease in a male following allogeneic stem cell transplantation. Acta Derm Venereol. 2007;87:367–8. https://doi.org/10.2340/00015555-0226.
Odorici G, Baraldi C, Loi C, Bardazzi F. Chronic graft-versus-host disease of the male genitalia: an underrecognized manifestation. J Dtsch Dermatol Ges. 2017;15:746–8. https://doi.org/10.1111/ddg.13281.
Tauchmanova L, Alviggi C, Foresta C, Strina I, Garolla A, Colao A, et al. Cryptozoospermia with normal testicular function after allogeneic stem cell transplantation: a case report. Hum Reprod. 2007;22:495–9. https://doi.org/10.1093/humrep/del376.
Thomas LJ, Shim TN, Borysiewicz C, Dinneen M, Fawcett H, Roy A, et al. Male genital lichen sclerosus in recipients of bone marrow transplants. Clin Exp Dermatol. 2016;41:495–7. https://doi.org/10.1111/ced.12812
Yared J, Gojo I, Akpek G. Glans penis involvement: an under-recognized manifestation of chronic GVHD. Bone Marrow Transpl. 2012;47:1006–7. https://doi.org/10.1038/bmt.2011.209.
Andreini A, Zampieri N, Costantini C, Malerba G, Bruno C, Salvagno G, et al. Chronic graft versus host disease is associated with erectile dysfunction in allogeneic hematopoietic stem cell transplant patients: a single-center experience. Leuk Lymphoma. 2018;59:2719–22. https://doi.org/10.1080/10428194.2018.1443338.
Dyer G, Gilroy N, Bradford J, Brice L, Kabir M, Greenwood M, et al. A survey of fertility and sexual health following allogeneic haematopoietic stem cell transplantation in New South Wales, Australia. Br J Haematol. 2016;172:592–601. https://doi.org/10.1111/bjh.13872.
Mueller SM, Haeusermann P, Rovo A, Halter JP, Passweg J, Itin P, et al. Genital chronic GVHD in men after hematopoietic stem cell transplantation: a single-center cross-sectional analysis of 155 patients. Biol Blood Marrow Transplant. 2013;19:1574–80. https://doi.org/10.1016/j.bbmt.2013.07.010
Wong FL, Francisco L, Togawa K, Kim H, Bosworth A, Atencio L, et al. Longitudinal trajectory of sexual functioning after hematopoietic cell transplantation: impact of chronic graft-versus-host disease and total body irradiation. Blood. 2013;122:3973–81. https://doi.org/10.1182/blood-2013-05-499806.
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National institutes of health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 Diagnosis and Staging Working Group Report. Biol Blood Marrow Transplant. 2015;21:389–401. https://doi.org/10.1016/j.bbmt.2014.12.001
Eeltink CM, Witte BI, Stringer J, Liptrott S, Babic A, Greenfield DM, et al. Health-care professionals’ perspective on discussing sexual issues in adult patients after haematopoietic cell transplantation. Bone Marrow Transpl. 2018;53:235–45. https://doi.org/10.1038/s41409-017-0027-y.
Majhail NS, Rizzo JD, Lee SJ, Aljurf M, Atsuta Y, Bonfim C, et al. Recommended screening and preventive practices for long-term survivors after hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2012;18:348–71. https://doi.org/10.1016/j.bbmt.2011.12.519
Greenfield DM, Walters SJ, Coleman RE, Hancock BW, Eastell R, Davies HA, et al. Prevalence and consequences of androgen deficiency in young male cancer survivors in a controlled cross- sectional study. J Clin Endocrinol Metab. 2007;92:3476–82. https://doi.org/10.1210/jc.2006-2744.
Gebauer J, Higham C, Langer T, Denzer C, Brabant G. Long-term endocrine and metabolic consequences of cancer treatment: a systematic review. Endocr Rev. 2019;40:711–67. https://doi.org/10.1210/er.2018-00092.
Chatterjee R, Kottaridis PD, McGarrigle HH, Eliahoo J, McKeag N, Mackinnon S, et al. Patterns of Leydig cell insufficiency in adult males following bone marrow transplantation for haematological malignancies. Bone Marrow Transplant. 2001;28:497–502. https://doi.org/10.1038/sj.bmt.1703160.
Chatterjee R, Andrews HO, McGarrigle HH, Kottaridis PD, Lees WR, Mackinnon S, et al. Cavernosal arterial insufficiency is a major component of erectile dysfunction in some recipients of high-dose chemotherapy/chemo-radiotherapy for haematological malignancies. Bone Marrow Transplant. 2000;25:1185–9. https://doi.org/10.1038/sj.bmt.1702391.
Greenfield DM, Boland E, Ezaydi Y, Ross RJ, Ahmedzai SH, Snowden JA, et al. Endocrine, metabolic, nutritional and body composition abnormalities are common in advanced intensively-treated (transplanted) multiple myeloma. Bone Marrow Transplant. 2014;49:907–12. https://doi.org/10.1038/bmt.2014.63.
Gundgurthi A, Garg MK, Nair V, Pakhetra R, Das S, Sharma S, et al. Endocrine complications after busulphan and cyclophosphamide based hematopoietic stem cell transplant: a single tertiary care centre experience. Indian J Endocrinol Metab. 2013;17:855–63. https://doi.org/10.4103/2230-8210.117248.
Schimmer AD, Ali V, Stewart AK, Imrie K, Keating A. Male sexual function after autologous blood or marrow transplantation. Biol Blood Marrow Transplant. 2001;7:279–83. https://doi.org/10.1053/bbmt.2001.v7.pm11400950.
Schneidewind L, Neumann T, Probst KA, Schmidt CA, Kruger W. Recovery from hypogonadism and male health in adult allogeneic stem cell transplantation. Eur J Haematol. 2018;100:584–91. https://doi.org/10.1111/ejh.13052
Sirmatel P, Yilmaz H, Gündüz E. Non-malignant late effects in lymphoma patients treated with autologous hematopoietic stem cell transplantation. Eur J Ther. 2017;23:106–10.
Tauchmanova L, Selleri C, De Rosa G, Esposito M, Di Somma C, Orio F, et al. Endocrine disorders during the first year after autologous stem-cell transplant. Am J Med. 2005;118:664–70. https://doi.org/10.1016/j.amjmed.2005.02.009.
Tauchmanova L, Selleri C, Rosa GD, Pagano L, Orio F, Lombardi G, et al. High prevalence of endocrine dysfunction in long-term survivors after allogeneic bone marrow transplantation for hematologic diseases. Cancer. 2002;95:1076–84. https://doi.org/10.1002/cncr.10773.
Vaezi M, Gharib C, Souri M, Ghavamzadeh A. Late complications in acute Leukemia patients following HSCT: a single center experience. Int J Hematol Oncol Stem Cell Res. 2016;10:1–6.
Bhasin S, Brito JP, Cunningham GR, Hayes FJ, Hodis HN, Matsumoto AM, et al. Testosterone therapy in men with hypogonadism: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2018;103:1715–44. https://doi.org/10.1210/jc.2018-00229.
Taskinen M, Kananen K, Valimaki M, Loyttyniemi E, Hovi L, Saarinen-Pihkala U, et al. Risk factors for reduced areal bone mineral density in young adults with stem cell transplantation in childhood. Pediatr Transplant. 2006;10:90–97. https://doi.org/10.1111/j.1399-3046.2005.00405.x.
Anandi P, Jain NA, Tian X, Wu CO, Pophali PA, Koklanaris E, et al. Factors influencing the late phase of recovery after bone mineral density loss in allogeneic stem cell transplantation survivors. Bone Marrow Transplant. 2016;51:1101–6. https://doi.org/10.1038/bmt.2016.85.
Orio F, Muscogiuri G, Palomba S, Serio B, Sessa M, Giudice V, et al. Endocrinopathies after allogeneic and autologous transplantation of hematopoietic stem cells. ScientificWorldJournal. 2014;2014:282147 https://doi.org/10.1155/2014/282147.
Li Z, Mewawalla P, Stratton P, Yong AS, Shaw BE, Hashmi S, et al. Sexual health in hematopoietic stem cell transplant recipients. Cancer. 2015;121:4124–31. https://doi.org/10.1002/cncr.29675
Ponce OJ, Spencer-Bonilla G, Alvarez-Villalobos N, Serrano V, Singh-Ospina N, Rodriguez- Gutierrez R, et al. The efficacy and adverse events of testosterone replacement therapy in hypogonadal men: a systematic review and meta-analysis of randomized, placebo-controlled trials. J Clin Endocrinol Metab. 2018. https://doi.org/10.1210/jc.2018-00404
Mulhall JP, Trost LW, Brannigan RE, Kurtz EG, Redmon JB, Chiles KA, et al. Evaluation and management of testosterone deficiency: AUA guideline. J Urol. 2018;200:423–32. https://doi.org/10.1016/j.juro.2018.03.115.
Walsh JS, Marshall H, Smith IL, Greenfield DM, Swain J, Best E, et al. Testosterone replacement in young male cancer survivors: a 6-month double-blind randomised placebo-controlled trial. PLoS Med. 2019;16:e1002960 https://doi.org/10.1371/journal.pmed.1002960.
Howell SJ, Radford JA, Adams JE, Smets EM, Warburton R, Shalet SM. Randomized placebo- controlled trial of testosterone replacement in men with mild Leydig cell insufficiency following cytotoxic chemotherapy. Clin Endocrinol. 2001;55:315–24. https://doi.org/10.1046/j.1365-2265.2001.01297.x.
Chatterjee R, Kottaridis PD, McGarrigle HH, Linch DC. Management of erectile dysfunction by combination therapy with testosterone and sildenafil in recipients of high-dose therapy for haematological malignancies. Bone Marrow Transplant. 2002;29:607–10. https://doi.org/10.1038/sj.bmt.1703421
Kananen K, Volin L, Laitinen K, Alfthan H, Ruutu T, Valimaki MJ. Prevention of bone loss after allogeneic stem cell transplantation by calcium, vitamin D, and sex hormone replacement with or without pamidronate. J Clin Endocrinol Metab. 2005;90:3877–85. https://doi.org/10.1210/jc.2004-2161.
Bar M, Ott SM, Lewiecki EM, Sarafoglou K, Wu JY, Thompson MJ, et al. Bone health management after hematopoietic cell transplantation: an expert panel opinion from the American Society for Transplantation and Cellular Therapy. Biol Blood Marrow Transplant. 2020;26:1784–802. https://doi.org/10.1016/j.bbmt.2020.07.001.
Boquiren VM, Esplen MJ, Wong J, Toner B, Warner E, Malik N. Sexual functioning in breast cancer survivors experiencing body image disturbance. Psychooncology. 2016;25:66–76. https://doi.org/10.1002/pon.3819.
Knoepp LR, Shippey SH, Chen CC, Cundiff GW, Derogatis LR, Handa VL. Sexual complaints, pelvic floor symptoms, and sexual distress in women over forty. J Sex Med. 2010;7:3675–82. https://doi.org/10.1111/j.1743-6109.2010.01955.x.
Thygesen KH, Schjodt I, Jarden M. The impact of hematopoietic stem cell transplantation on sexuality: a systematic review of the literature. Bone Marrow Transpl. 2012;47:716–24. https://doi.org/10.1038/bmt.2011.169.
Syrjala KL, Langer SL, Abrams JR, Storer BE, Martin PJ. Late effects of hematopoietic cell transplantation among 10-year adult survivors compared with case-matched controls. J Clin Oncol. 2005;23:6596–606. https://doi.org/10.1200/JCO.2005.12.674.
Watson M, Buck G, Wheatley K, Homewood JR, Goldstone AH, Rees JK, et al. Adverse impact of bone marrow transplantation on quality of life in acute myeloid leukaemia patients; analysis of the UK Medical Research Council AML 10 Trial. Eur J Cancer. 2004;40:971–8. https://doi.org/10.1016/S0959-8049(03)00628-2.
Messerer D, Engel J, Hasford J, Schaich M, Ehninger G, Sauerland C, et al. Impact of different post-remission strategies on quality of life in patients with acute myeloid leukemia. Haematologica. 2008;93:826–33. https://doi.org/10.3324/haematol.11987.
Lee SJ, Fairclough D, Parsons SK, Soiffer RJ, Fisher DC, Schlossman RL, et al. Recovery after stem-cell transplantation for hematologic diseases. J Clin Oncol. 2001;19:242–52. https://doi.org/10.1200/JCO.2001.19.1.242.
Hjermstad MJ, Evensen SA, Kvaloy SO, Loge JH, Fayers PM, Kaasa S. The CARES-SF used for prospective assessment of health-related quality of life after stem cell transplantation. Psychooncology. 2003;12:803–13. https://doi.org/10.1002/pon.708.
Gruber U, Fegg M, Buchmann M, Kolb HJ, Hiddemann W. The long-term psychosocial effects of haematopoetic stem cell transplantation. Eur J Cancer Care. 2003;12:249–56.
Lee HG, Park EY, Kim HM, Kim K, Kim WS, Yoon SS, et al. Sexuality and quality of life after hematopoietic stem cell transplantation. Korean J Intern Med. 2002;17:19–23. https://doi.org/10.3904/kjim.2002.17.1.19.
Gifford G, Gilroy N, Dyer G, Brice L, Kabir M, Greenwood M, et al. The experience of survival following allogeneic haematopoietic stem cell transplantation in New South Wales, Australia. Bone Marrow Transplant. 2016;51:1361–8. https://doi.org/10.1038/bmt.2016.135.
Bersvendsen HS, Haugnes HS, Dahl AA, Fagerli UM, Fluge O, Holte H, et al. Sexual function in long-term male lymphoma survivors after high-dose therapy with autologous stem-cell transplantation. Bone Marrow Transplant. 2020;55:891–905. https://doi.org/10.1038/s41409-019-0745-4.
Syrjala KL, Schoemans H, Yi JC, Langer SL, Mukherjee A, Onstad L, et al. Sexual functioning in long-term survivors of hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2020. https://doi.org/10.1016/j.bbmt.2020.09.027.
Socie G, Mary JY, Esperou H, Robert DV, Aractingi S, Ribaud P, et al. Health and functional status of adult recipients 1 year after allogeneic haematopoietic stem cell transplantation. Br J Haematol. 2001;113:194–201. https://doi.org/10.1046/j.1365-2141.2001.02678.x.
Heinonen H, Volin L, Uutela A, Zevon M, Barrick C, Ruutu T. Gender-associated differences in the quality of life after allogeneic BMT. Bone Marrow Transpl. 2001;28:503–9. https://doi.org/10.1038/sj.bmt.1703158.
Noerskov KH, Schjodt I, Syrjala KL, Jarden M. Sexual function 1-year after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transpl. 2016;51:833–40. https://doi.org/10.1038/bmt.2015.342.
Chiodi S, Spinelli S, Ravera G, Petti AR, Van Lint MT, Lamparelli T, et al. Quality of life in 244 recipients of allogeneic bone marrow transplantation. Br J Haematol. 2000;110:614–9. https://doi.org/10.1046/j.1365-2141.2000.02053.x.
Humphreys CT, Tallman B, Altmaier EM, Barnette V. Sexual functioning in patients undergoing bone marrow transplantation: a longitudinal study. Bone Marrow Transplant. 2007;39:491–6. https://doi.org/10.1038/sj.bmt.1705613.
Mosher CE, DuHamel KN, Rini C, Corner G, Lam J, Redd WH. Quality of life concerns and depression among hematopoietic stem cell transplant survivors. Support Care Cancer. 2011;19:1357–65. https://doi.org/10.1007/s00520-010-0958-y.
Syrjala KL, Kurland BF, Abrams JR, Sanders JE, Heiman JR. Sexual function changes during the 5 years after high-dose treatment and hematopoietic cell transplantation for malignancy, with case-matched controls at 5 years. Blood. 2008;111:989–96. https://doi.org/10.1182/blood-2007-06-096594
Norskov KH, Schmidt M, Jarden M. Patients’ experience of sexuality 1-year after allogeneic Haematopoietic Stem Cell Transplantation. Eur J Oncol Nurs. 2015;19:419–26. https://doi.org/10.1016/j.ejon.2014.12.005.
Yoo KH, Kang D, Kim IR, Choi EK, Kim JS, Yoon SS, et al. Satisfaction with sexual activity and sexual dysfunction in hematopoietic stem cell transplantation survivors and their partners: a couple study. Bone Marrow Transpl. 2018;53:967–76. https://doi.org/10.1038/s41409-018-0097-5.
Case P. The impact of gender role on recovery from bone marrow transplantation. Illn Crisis Loss. 2002;10:344–55.
Polomeni A, Lapusan S, Bompoint C, Rubio MT, Mohty M. The impact of allogeneic- hematopoietic stem cell transplantation on patients’ and close relatives’ quality of life and relationships. Eur J Oncol Nurs. 2016;21:248–56. https://doi.org/10.1016/j.ejon.2015.10.011.
Bishop MM, Beaumont JL, Hahn EA, Cella D, Andrykowski MA, Brady MJ, et al. Late effects of cancer and hematopoietic stem-cell transplantation on spouses or partners compared with survivors and survivor-matched controls. J Clin Oncol. 2007;25:1403–11. https://doi.org/10.1200/JCO.2006.07.5705.
Langer S, Abrams J, Syrjala K. Caregiver and patient marital satisfaction and affect following hematopoietic stem cell transplantation: a prospective, longitudinal investigation. Psychooncology. 2003;12:239–53. https://doi.org/10.1002/pon.633.
Booker R, Walker L, Raffin Bouchal S. Sexuality after hematopoietic stem cell transplantation: a mixed methods study. Eur J Oncol Nurs. 2019;39:10–20. https://doi.org/10.1016/j.ejon.2019.01.001.
Kim IR, Jang SY, Shin HS, Choi HJ, Jung CW, Yoon SS, et al. Association between sexuality knowledge and sexual dysfunction in hematopoietic stem cell transplantation patients and their partners. Patient Educ Couns. 2020;103:1630–6. https://doi.org/10.1016/j.pec.2020.03.007.
Russell C, Harcourt D, Henderson L, Marks DI. Patients’ experiences of appearance changes following allogeneic bone marrow transplantation. Cancer Nurs. 2011;34:315–21. https://doi.org/10.1097/NCC.0b013e3181f8f884.
Carter J, Lacchetti C, Andersen BL, Barton DL, Bolte S, Damast S, et al. Interventions to Address Sexual Problems in People With Cancer: American Society of Clinical Oncology Clinical Practice Guideline Adaptation of Cancer Care Ontario Guideline. J Clin Oncol. 2018;36:492–511. https://doi.org/10.1200/JCO.2017.75.8995.
El-Jawahri A, Fishman SR, Vanderklish J, Dizon DS, Pensak N, Traeger L, et al. Pilot study of a multimodal intervention to enhance sexual function in survivors of hematopoietic stem cell transplantation. Cancer. 2018;124:2438–46. https://doi.org/10.1002/cncr.31333.
Burnett AL, Nehra A, Breau RH, Culkin DJ, Faraday MM, Hakim LS, et al. Erectile dysfunction: AUA guideline. J Urol. 2018;200:633–41. https://doi.org/10.1016/j.juro.2018.05.004.
Tichelli A, Rovo A. Fertility issues following hematopoietic stem cell transplantation. Expert Rev Hematol. 2013;6:375–88. https://doi.org/10.1586/17474086.2013.816507
Anserini P, Chiodi S, Spinelli S, Costa M, Conte N, Copello F, et al. Semen analysis following allogeneic bone marrow transplantation. Additional data for evidence-based counselling. Bone Marrow Transplant. 2002;30:447–51. https://doi.org/10.1038/sj.bmt.1703651.
Rovo A, Aljurf M, Chiodi S, Spinelli S, Salooja N, Sucak G, et al. Ongoing graft-versus-host disease is a risk factor for azoospermia after allogeneic hematopoietic stem cell transplantation: a survey of the Late Effects Working Party of the European Group for Blood and Marrow Transplantation. Haematologica. 2013;98:339–45. https://doi.org/10.3324/haematol.2012.071944
Rovo A, Tichelli A, Passweg JR, Heim D, Meyer-Monard S, Holzgreve W, et al. Spermatogenesis in long-term survivors after allogeneic hematopoietic stem cell transplantation is associated with age, time interval since transplantation, and apparently absence of chronic GvHD. Blood. 2006;108:1100–5. https://doi.org/10.1182/blood-2006-01-0176
Joshi S, Savani BN, Chow EJ, Gilleece MH, Halter J, Jacobsohn DA, et al. Clinical guide to fertility preservation in hematopoietic cell transplant recipients. Bone Marrow Transplant. 2014;49:477–84. https://doi.org/10.1038/bmt.2013.211
Logan S, Perz J, Ussher JM, Peate M, Anazodo A. Systematic review of fertility-related psychological distress in cancer patients: Informing on an improved model of care. Psychooncology. 2019;28:22–30. https://doi.org/10.1002/pon.4927.
Ussher JM, Perz J, Australian C. Threat of biographical disruption: the gendered construction and experience of infertility following cancer for women and men. BMC Cancer. 2018;18:250 https://doi.org/10.1186/s12885-018-4172-5.
Borgmann-Staudt A, Rendtorff R, Reinmuth S, Hohmann C, Keil T, Schuster FR, et al. Fertility after allogeneic haematopoietic stem cell transplantation in childhood and adolescence. Bone Marrow Transplant. 2012;47:271–6. https://doi.org/10.1038/bmt.2011.78.
Grigg AP, McLachlan R, Zaja J, Szer J. Reproductive status in long-term bone marrow transplant survivors receiving busulfan-cyclophosphamide (120 mg/kg). Bone Marrow Transplant. 2000;26:1089–95. https://doi.org/10.1038/sj.bmt.1702695.
Lukusa AK, Vermylen C, Vanabelle B, Curaba M, Brichard B, Chantrain C, et al. Bone marrow transplantation or hydroxyurea for sickle cell anemia: long-term effects on semen variables and hormone profiles. Pediatr Hematol Oncol. 2009;26:186–94. https://doi.org/10.1080/07357900902892780.
Panasiuk A, Nussey S, Veys P, Amrolia P, Rao K, Krawczuk-Rybak M, et al. Gonadal function and fertility after stem cell transplantation in childhood: comparison of a reduced intensity conditioning regimen containing melphalan with a myeloablative regimen containing busulfan. Br J Haematol. 2015;170:719–26. https://doi.org/10.1111/bjh.13497.
Savani BN, Kozanas E, Shenoy A, Barrett AJ. Recovery of spermatogenesis after total-body irradiation. Blood. 2006;108:4292–3. https://doi.org/10.1182/blood-2006-08-044289.
Wilhelmsson M, Vatanen A, Borgstrom B, Gustafsson B, Taskinen M, Saarinen-Pihkala UM, et al. Adult testicular volume predicts spermatogenetic recovery after allogeneic HSCT in childhood and adolescence. Pediatr Blood Cancer. 2014;61:1094–1100. https://doi.org/10.1002/pbc.24970.
Mathiesen S, Sorensen K, Nielsen MM, Suominen A, Ifversen M, Grell K, et al. Male gonadal function after allogeneic hematopoietic stem cell transplantation in childhood: a cross-sectional, population-based study. Biol Blood Marrow Transpl. 2020;26:1635–45. https://doi.org/10.1016/j.bbmt.2020.05.009.
Kerbauy MN, Mariano L, Seber A, Rocha V. The impact of low dose busulfan on gonodal function after allogeneic hematopoietic stem cell transplantation for aplastic anemia. Bone Marrow Transplant. 2020;55:1169–71. https://doi.org/10.1038/s41409-019-0639-5.
Wagner AM, Beier K, Christen E, Hollander GA, Krenger W. Leydig cell injury as a consequence of an acute graft-versus-host reaction. Blood. 2005;105:2988–90. https://doi.org/10.1182/blood-2004-07-2646.
Babb A, Farah N, Lyons C, Lindsay K, Reddy N, Goldman J, et al. Uptake and outcome of assisted reproductive techniques in long-term survivors of SCT. Bone Marrow Transplant. 2012;47:568–73. https://doi.org/10.1038/bmt.2011.134.
Branvall E, Derolf AR, Johansson E, Hultcrantz M, Bergmark K, Bjorkholm M. Self-reported fertility in long-term survivors of acute myeloid leukemia. Ann Hematol. 2014;93:1491–8. https://doi.org/10.1007/s00277-014-2088-y.
Carter A, Robison LL, Francisco L, Smith D, Grant M, Baker KS, et al. Prevalence of conception and pregnancy outcomes after hematopoietic cell transplantation: report from the Bone Marrow Transplant Survivor Study. Bone Marrow Transpl. 2006;37:1023–9. https://doi.org/10.1038/sj.bmt.1705364.
Hammond C, Abrams JR, Syrjala KL. Fertility and risk factors for elevated infertility concern in 10-year hematopoietic cell transplant survivors and case-matched controls. J Clin Oncol. 2007;25:3511–7. https://doi.org/10.1200/JCO.2007.10.8993
Hayden PJ, Keogh F, Ni Conghaile M, Carroll M, Crowley M, Fitzsimon N, et al. A single-centre assessment of long-term quality-of-life status after sibling allogeneic stem cell transplantation for chronic myeloid leukaemia in first chronic phase. Bone Marrow Transplant. 2004;34:545–56. https://doi.org/10.1038/sj.bmt.1704638.
Ishiguro H, Yasuda Y, Tomita Y, Shinagawa T, Shimizu T, Morimoto T, et al. Gonadal shielding to irradiation is effective in protecting testicular growth and function in long-term survivors of bone marrow transplantation during childhood or adolescence. Bone Marrow Transplant. 2007;39:483–90. https://doi.org/10.1038/sj.bmt.1705612.
Loren AW, Chow E, Jacobsohn DA, Gilleece M, Halter J, Joshi S, et al. Pregnancy after hematopoietic cell transplantation: a report from the late effects working committee of the Center for International Blood and Marrow Transplant Research (CIBMTR). Biol Blood Marrow Transplant. 2011;17:157–66. https://doi.org/10.1016/j.bbmt.2010.07.009.
Rahal I, Galambrun C, Bertrand Y, Garnier N, Paillard C, Frange P, et al. Late effects after hematopoietic stem cell transplantation for beta-thalassemia major: the French national experience. Haematologica. 2018;103:1143–9. https://doi.org/10.3324/haematol.2017.183467
Salooja N, Szydlo RM, Socie G, Rio B, Chatterjee R, Ljungman P, et al. Pregnancy outcomes after peripheral blood or bone marrow transplantation: a retrospective survey. Lancet. 2001;358:271–6. https://doi.org/10.1016/s0140-6736(01)05482-4.
Wilhelmsson M, Glosli H, Ifversen M, Abrahamsson J, Winiarski J, Jahnukainen K, et al. Long- term health outcomes in survivors of childhood AML treated with allogeneic HSCT: a NOPHO- AML Study. Bone Marrow Transpl. 2019;54:726–36. https://doi.org/10.1038/s41409-018-0337-8.
Rovo A, Aljurf M, Chiodi S, Spinelli S, Salooja N, Sucak G, et al. Paternity wishes in long-term survivors after allogeneic hematopoietic SCT. A study of the late effects working party of the EBMT. Bone Marrow Transplant. 2013;48:878–9. https://doi.org/10.1038/bmt.2012.255
Balduzzi A, Dalle JH, Jahnukainen K, von Wolff M, Lucchini G, Ifversen M, et al. Fertility preservation issues in pediatric hematopoietic stem cell transplantation: practical approaches from the consensus of the Pediatric Diseases Working Party of the EBMT and the International BFM Study Group. Bone Marrow Transplant. 2017;52:1406–15. https://doi.org/10.1038/bmt.2017.147
Nahata L, Cohen LE, Lehmann LE, Yu RN. Semen analysis in adolescent cancer patients prior to bone marrow transplantation: when is it too late for fertility preservation? Pediatr Blood Cancer. 2013;60:129–32. https://doi.org/10.1002/pbc.24172.
Schlegel PN, Sigman M, Collura B, De Jonge CJ, Eisenberg ML, Lamb DJ, et al. Diagnosis and treatment of infertility in men: AUA/ASRM guideline part II. Fertil Steril. 2020. https://doi.org/10.1016/j.fertnstert.2020.11.016.
Borgstrom B, Fridstrom M, Gustafsson B, Ljungman P, Rodriguez-Wallberg KA. A prospective study on the long-term outcome of prepubertal and pubertal boys undergoing testicular biopsy for fertility preservation prior to hematologic stem cell transplantation. Pediatr Blood Cancer. 2020;67:e28507 https://doi.org/10.1002/pbc.28507.
Bernaudin F, Dalle JH, Bories D, de Latour RP, Robin M, Bertrand Y, et al. Long-term event-free survival, chimerism and fertility outcomes in 234 patients with sickle-cell anemia younger than 30 years after myeloablative conditioning and matched-sibling transplantation in France. Haematologica. 2020;105:91–101. https://doi.org/10.3324/haematol.2018.213207.
Abdelaal O, Barber H, Atala A, Sadri-Ardekani H. Purging of malignant cell contamination prior to spermatogonia stem cell autotransplantation to preserve fertility: progress & prospects. Curr Opin Endocrinol Diabetes Obes. 2019;26:166–74. https://doi.org/10.1097/MED.0000000000000481.
Syrjala KL, Martin PJ, Lee SJ. Delivering care to long-term adult survivors of hematopoietic cell transplantation. J Clin Oncol. 2012;30:3746–51. https://doi.org/10.1200/JCO.2012.42.3038
Bilmon IA, Ashton LJ, Le Marsney RE, Dodds AJ, O’Brien TA, Wilcox L, et al. Second cancer risk in adults receiving autologous haematopoietic SCT for cancer: a population-based cohort study. Bone Marrow Transpl. 2014;49:691–8. https://doi.org/10.1038/bmt.2014.13.
Gallagher G, Forrest DL. Second solid cancers after allogeneic hematopoietic stem cell transplantation. Cancer. 2007;109:84–92. https://doi.org/10.1002/cncr.22375.
Rizzo JD, Curtis RE, Socie G, Sobocinski KA, Gilbert E, Landgren O, et al. Solid cancers after allogeneic hematopoietic cell transplantation. Blood. 2009;113:1175–83. https://doi.org/10.1182/blood-2008-05-158782
Mahindra A, Raval G, Mehta P, Brazauskas R, Zhang MJ, Zhong X, et al. New cancers after autotransplantations for multiple myeloma. Biol Blood Marrow Transplant. 2015;21:738–45. https://doi.org/10.1016/j.bbmt.2014.12.028.
Baker KS, Leisenring WM, Goodman PJ, Ermoian RP, Flowers ME, Schoch G, et al. Total body irradiation dose and risk of subsequent neoplasms following allogeneic hematopoietic cell transplantation. Blood. 2019;133:2790–9. https://doi.org/10.1182/blood.2018874115.
Wu PA, Stern RS, Huang V, Liu KX, Chen CA, Tzachanis D, et al. Reduced-Intensity conditioning regimens, prior chronic lymphocytic leukemia, and graft-versus-host disease are associated with higher rates of skin cancer after allogeneic hematopoietic stem cell transplantation. J Investig Dermatol. 2019;139:591–9. https://doi.org/10.1016/j.jid.2018.08.025.
Inamoto Y, Matsuda T, Tabuchi K, Kurosawa S, Nakasone H, Nishimori H, et al. Outcomes of patients who developed subsequent solid cancer after hematopoietic cell transplantation. Blood Adv. 2018;2:1901–13. https://doi.org/10.1182/bloodadvances.2018020966
Bhatia S, Louie AD, Bhatia R, O’Donnell MR, Fung H, Kashyap A, et al. Solid cancers after bone marrow transplantation. J Clin Oncol. 2001;19:464–71. https://doi.org/10.1200/JCO.2001.19.2.464.
Leisenring W, Friedman DL, Flowers ME, Schwartz JL, Deeg HJ. Nonmelanoma skin and mucosal cancers after hematopoietic cell transplantation. J Clin Oncol. 2006;24:1119–26. https://doi.org/10.1200/JCO.2005.02.7052
Tichelli A, Beohou E, Labopin M, Socie G, Rovo A, Badoglio M, et al. Evaluation of second solid cancers after hematopoietic stem cell transplantation in European patients. JAMA Oncol. 2019;5:229–35. https://doi.org/10.1001/jamaoncol.2018.4934.
Atsuta Y, Suzuki R, Yamashita T, Fukuda T, Miyamura K, Taniguchi S, et al. Continuing increased risk of oral/esophageal cancer after allogeneic hematopoietic stem cell transplantation in adults in association with chronic graft-versus-host disease. Ann Oncol. 2014;25:435–41. https://doi.org/10.1093/annonc/mdt558.
Baker KS, DeFor TE, Burns LJ, Ramsay NK, Neglia JP, Robison LL. New malignancies after blood or marrow stem-cell transplantation in children and adults: incidence and risk factors. J Clin Oncol. 2003;21:1352–8. https://doi.org/10.1200/JCO.2003.05.108
Majhail NS, Brazauskas R, Rizzo JD, Sobecks RM, Wang Z, Horowitz MM, et al. Secondary solid cancers after allogeneic hematopoietic cell transplantation using busulfan-cyclophosphamide conditioning. Blood. 2011;117:316–22. https://doi.org/10.1182/blood-2010-07-294629
Ringden O, Brazauskas R, Wang Z, Ahmed I, Atsuta Y, Buchbinder D, et al. Second solid cancers after allogeneic hematopoietic cell transplantation using reduced-intensity conditioning. Biol Blood Marrow Transplant. 2014;20:1777–84. https://doi.org/10.1016/j.bbmt.2014.07.009
Vajdic CM, Mayson E, Dodds AJ, O’Brien T, Wilcox L, Nivison-Smith I, et al. Second cancer risk and late mortality in adult Australians receiving allogeneic hematopoietic stem cell transplantation: a population-based cohort study. Biol Blood Marrow Transplant. 2016;22:949–56. https://doi.org/10.1016/j.bbmt.2016.01.027.
Yokota A, Ozawa S, Masanori T, Akiyama H, Ohshima K, Kanda Y, et al. Secondary solid tumors after allogeneic hematopoietic SCT in Japan. Bone Marrow Transplant. 2012;47:95–100. https://doi.org/10.1038/bmt.2011.23.
Danner-Koptik KE, Majhail NS, Brazauskas R, Wang Z, Buchbinder D, Cahn JY, et al. Second malignancies after autologous hematopoietic cell transplantation in children. Bone Marrow Transplant. 2013;48:363–8. https://doi.org/10.1038/bmt.2012.166.
Shimoni A, Shem-Tov N, Chetrit A, Volchek Y, Tallis E, Avigdor A, et al. Secondary malignancies after allogeneic stem-cell transplantation in the era of reduced-intensity conditioning; the incidence is not reduced. Leukemia. 2013;27:829–35. https://doi.org/10.1038/leu.2012.299
Gutierrez-Pascual M, Vicente-Martin FJ, Lopez-Estebaranz JL. Lichen sclerosus and squamous cell carcinoma. Actas Dermosifiliogr. 2012;103:21–28. https://doi.org/10.1016/j.adengl.2011.05.004.
Snowden JA, McGrath E, Duarte RF, Saccardi R, Orchard K, Worel N, et al. JACIE accreditation for blood and marrow transplantation: past, present and future directions of an international model for healthcare quality improvement. Bone Marrow Transplant. 2017;52:1367–71. https://doi.org/10.1038/bmt.2017.54.
Acknowledgements
The CIBMTR is supported primarily by Public Health Service U24CA076518 from the National Cancer Institute (NCI), the National Heart, Lung and Blood Institute (NHLBI) and the National Institute of Allergy and Infectious Diseases (NIAID); HHSH250201700006C from the Health Resources and Services Administration (HRSA); and N00014-20-1-2705 and N00014-20-1-2832 from the Office of Naval Research; Support is also provided by Be the Match Foundation, the Medical College of Wisconsin, the National Marrow Donor Program, and from the following commercial entities: AbbVie; Accenture; Actinium Pharmaceuticals, Inc.; Adaptive Biotechnologies Corporation; Adienne SA; Allovir, Inc.; Amgen, Inc.; Astellas Pharma US; bluebird bio, inc.; Bristol Myers Squibb Co.; CareDx; CSL Behring; CytoSen Therapeutics, Inc.; Daiichi SankyoCo., Ltd; Eurofins Viracor; ExcellThera; Fate Therapeutics; Gamida-Cell, Ltd; Genentech Inc; Gilead; GlaxoSmithKline; Incyte Corporation; Janssen/Johnson & Johnson; Jasper Therapeutics; Jazz Pharmaceuticals, Inc.; Karyopharm Therapeutics; Kiadis Pharma; Kite, a Gilead Company; Kyowa Kirin; Magenta Therapeutics; Medac GmbH; Merck & Co.; Millennium, the Takeda Oncology Co.; Miltenyi Biotec, Inc.; MorphoSys; Novartis Pharmaceuticals Corporation; Omeros Corporation; Oncopeptides, Inc.; Orca Biosystems, Inc.; Pfizer, Inc.; Pharmacyclics, LLC; Sanofi Genzyme; Seagen, Inc.; Stemcyte; Takeda Pharmaceuticals; Tscan; Vertex; Vor Biopharma; Xenikos BV.
Author information
Authors and Affiliations
Contributions
RP and HS. designed and directed the study plan and contributed to manuscript writing and revision. AT, AI, and RLH served as study co-leaders and contributed to manuscript writing and revision. YI, DB, MTL-S, SP, AR, and LB served as section subgroup leaders and contributed to manuscript writing and revision; ES performed the literature search and contributed to manuscript writing and revision. SMB, HE, HSM, PP, AS, MA, KB, GWB, ZD, LDG, AD, HE, MLE, NF, AF, RPG, HG, BKH, SH, PH, KJ, DM, JM, SN, SN, SP, ZP, DP, RR, Amir S, ISO, BNS, JAS, Akshay S, DT, SCV, LW, DW, JAY, VA, NE, AJS, and SMS contributed to manuscript writing and revision. All authors critically reviewed the data and approved the final manuscript before it was submitted.
Corresponding author
Ethics declarations
Competing interests
HE reports grants and other from BMS/Celgene, grants and other from Janssen, grants and other from Amgen, other from Takeda, grants and other from Sanofi, grants and other from GSK, during the conduct of the study. SH reports other from Pfizer, other from Novartis, other from Therakos, other from Janssen, other from MSD, outside the submitted work. YI reports personal fees from Novartis, personal fees from Janssen, personal fees from Meiji Seika Pharma, outside the submitted work. RP reports research funding from Amgen. RR reports other from Diurnal Plc, during the conduct of the study. HS reports participation in advisory boards for Janssen & Novartis; speaker’s fees from Incyte, Jazz Pharmaceuticals, Takeda, Novartis and the BHS (Belgian Hematological Society); travel grants from EBMT, CIBMTR, Incyte & Gilead and research funding from Novartis & the BHS, outside of the submitted work. AR reports grants from Novartis, grants from CSL Behring, grants from Alexion, personal fees from Novartis, personal fees from BMS, personal fees from OrPha Swiss GmbH, other from Amgen, other from AstraZeneca, other from Sanofi, other from Celgene, outside the submitted work. ASharma reports clinical trial salary support from Vertex Pharmaceuticals, CRISPR Therapeutics, Novartis paid to his institution, and personal consultancy fees from Spotlight Therapeutics, outside the submitted work. JAS reports personal fees from MEDAC, personal fees from KIADIS, personal fees from GILEAD, personal fees from JANSSEN, personal fees from MALLINCKRODT, personal fees from JAZZ, personal fees from ACTELION, outside the submitted work. DW reports grants and personal fees from Novartis, personal fees from Mallinckrodt, personal fees from Behring, outside the submitted work. PG is a consultant to Ascenage Pharma, BeiGene Ltd, Kite Pharma Inc., Fusion Pharma LLC, LaJolla NanoMedical Inc., Mingsight Parmaceuticals Inc. and CStone Pharmaceuticals; Medical Director, FFF Enterprises Inc.; Partner, AZCA Inc.; Board of Directors, RakFond Foundation for Cancer Research Support; Scientific Advisory Board, Antegene Biotech LLC and StemRad Ltd.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is co-published in the journals Bone Marrow Transplantation and Transplantation and Cellular Therapy https://doi.org/10.1038/s41409-022-01591-z and https://doi.org/10.1016/j.jtct.2021.10.013
Supplementary information
Rights and permissions
About this article
Cite this article
Phelan, R., Im, A., Hunter, R.L. et al. Male-specific late effects in adult hematopoietic cell transplantation recipients: a systematic review from the Late Effects and Quality of Life Working Committee of the Center for International Blood and Marrow Transplant Research and Transplant Complications Working Party of the European Society of Blood and Marrow Transplantation. Bone Marrow Transplant 57, 1150–1163 (2022). https://doi.org/10.1038/s41409-022-01591-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-022-01591-z
This article is cited by
-
Prospective Association of Psychological Distress and Sexual Quality of Life Among Hematopoietic Stem Cell Transplant Survivors
Journal of Clinical Psychology in Medical Settings (2024)
-
Practice harmonization workshops of EBMT: an expert-based approach to generate practical and contemporary guidelines within the arena of hematopoietic cell transplantation and cellular therapy
Bone Marrow Transplantation (2023)
-
Progress and Challenges in Survivorship After Acute Myeloid Leukemia in Adults
Current Hematologic Malignancy Reports (2022)