Abstract
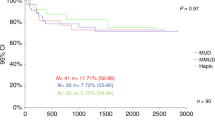
Donor selection may contribute to improve clinical outcomes of T cell-replete haploidentical stem cell transplantation (Haplo-SCT) with post-transplant cyclophosphamide (PT-Cy). Impact of second-degree related donor (SRD) was not fully elucidated in this platform. We retrospectively compared the outcome of patients receiving Haplo-SCT either from a SRD (n = 31) or a first-degree related donor (FRD, n = 957). Median time to neutrophil and platelet recovery did not differ between a SRD and a FRD transplant (p = 0.599 and 0.587). Cumulative incidence of grade II–IV acute graft-versus host disease (GVHD) and moderate-severe chronic GVHD was 13% and 19% after SRD vs 24% (p = 0.126) and 13% (p = 0.395) after FRD transplant. One-year cumulative incidence of non-relapse mortality (NRM) was 19% for SRD and 20% for FRD (p = 0.435) cohort. The 3-year probability of overall survival (OS) and progression-free survival (PFS) was 42% vs 55% (p = 0.273) and 49% vs 35% (p = 0.280) after SRD and FRD transplant, respectively. After propensity score adjustment or matched pair analysis, the outcome of patients receiving Haplo-SCT from a SRD or a FRD did not differ in terms of NRM, OS, PFS, acute and chronic GVHD. Our results suggest that a SRD is a viable option for Haplo-SCT with PT-Cy when a FRD is not available.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Luznik L, O’Donnell PV, Symons HJ, Chen AR, Leffell MS, Zahurak M, et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide. Biol Blood Marrow Transpl. 2008;14:641–50.
Chang YJ, Huang XJ. Haploidentical bone marrow transplantation without T-cell depletion. Semin Oncol. 2012;39:653–6.
Kasamon YL, Luznik L, Leffell MS, Kowalski J, Tsai H-L, Bolanos-Meade J, et al. Nonmyeloablative HLA-haploidentical bone marrow transplantation with high-dose posttransplantation cyclophosphamide: effect of HLA disparity on outcome. Biol Blood Marrow Transpl. 2010;16:482–9.
Ciurea SO, Al Malki MM, Kongtim P, Fuchs EJ, Luznik L, Huang XJ, et al. The European Society for Blood and Marrow Transplantation (EBMT) consensusrecommendations for donor selection in haploidentical hematopoietic cell transplantation. Bone Marrow Transpl. 2020;55:12–24.
Canaani J, Savani BN, Labopin M, Huang XJ, Ciceri F, Arcese W, et al. Impact of ABO incompatibility on patient outcome in haploidentical hematopoietic stem cell transplantation for acute myeloid leukemia - a report from the acute leukemia working party of the EBMT. Haematologica. 2017;102:1066–74.
Canaani J, Savani BN, Labopin M, Huang XJ, Ciceri F, Arcese W, et al. Donor age determines outcome in acute leukemia patients over 40 undergoing haploidentical hematopoietic cell transplantation. Am J Hematol. 2018;93:246–53.
Mariotti J, Raiola AM, Evangelista A, Carella AM, Martino M, Patriarca F, et al. Impact of donor age and kinship on clinical outcomes after T-cell-replete haploidentical transplantation with PT-Cy. Blood Adv. 2020;4:3900–12.
DeZern A, Franklin C, Tsai HL, Imus PH, Cooke KR, Varadhan R, et al. Relationship of donor age and relationship to outcomes of haploidentical transplantation with posttransplant cyclophosphamide. Blood Adv. 2021;5:1360–8.
McCurdy SR, Kanakry JA, Showel MM, Tsai HL, Bolaños-Meade J, Rosner GL. Outcomes of Nonmyeloablative HLA-Haploidentical Blood or Marrow Transplantation with High-dose post-transplantation cyclophosphamide in older adults. Blood. 2015;125:3024–31.
Solomon SR, Aubrey MT, Zhang X, Piluso A, Freed BM, Brown S, et al. Selecting the Best Donor for Haploidentical Transplant: Impact of HLA, Killer Cell Immunoglobulin-Like Receptor Genotyping, and Other Clinical Variables. Biol Blood Marrow Transpl. 2018;24:789–98.
Bazarbachi A, Boumendil A, Finel H, Mohty M, Castagna L, Blaise D, et al. Influence of donor type, stem cell source and conditioning on outcomes after haploidentical transplant for lymphoma - a LWP-EBMT study. Br J Haematol. 2020;188:745–56.
Garnier A, Guillaume T, Peterlin P, Bene MC, Le Bris Y, Dubruille V, et al. Second-degree relative donors for T-replete haploidentical allogeneic stem cell transplantation with high-dose post-transplant cyclophosphamide: toward crossing the major HLA barrier. Bone Marrow Transpl. 2017;52:1063–4.
Elmariah H, Kasamon YL, Zahurak M, Macfarlane KW, Tucker N, Rosner GL, et al. Haploidentical bone marrow transplantation with post-transplant cyclophosphamide using non-first-degree related donors. Biol Blood Marrow Transpl. 2018;24:1099–102.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transpl. 2009;15:1628–33.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transpl. 1995;15:825–8.
Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transpl. 2005;11:945–56.
Holtan SG, DeFor TE, Lazaryan A, Bejanyan N, Aroa M, Brunstein CG, et al. Composite end point of graft-versus-host disease-free, relapse-free survival after allogeneic hematopoietic cell transplantation. Blood 2015;125:1333–8.
Kaplan EL, Meier P. Non-parametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–81.
Gooley TA, Leisenring W, Crowley J, Storer BE. Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med. 1999;18:695–706.
Fine J, Gray R. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–7.
Zhang YY, Liu DH, Liu KY, Xu L-P, Chen H, Han W, et al. HLA-haploidentical hematopoietic SCT from collateral related donors without in vitro T-cell depletion for hematological malignancies. Bone Marrow Transpl. 2014;49:496–501.
Mo XD, Zhang YY, Zhang XH, Xu L-P, Wang Y, Yan C-H, et al. The role of collateral related donors in haploidentical hematopoietic stem cell transplantation. Sci Bull. 2018;63:1376–82.
Chang YJ, Luznik L, Fuchs EJ, Huang XJ. How do we choose the best donor for T-cell-replete, HLA-haploidentical transplantation? J Hematol Oncol. 2016;9:35.
McCurdy SR, Luznik L. How we perform haploidentical stem cell transplantation with posttransplant cyclophosphamide. Hematol Am Soc Hematol Educ Program. 2019;2019:513–21.
McCurdy SR, Zhang MJ, St Martin A, Al Malki MM, Bashey A, Gaballa S, et al. Effect of donor charac- teristics on haploidentical transplantation with post-transplantation cy- clophosphamide. Blood Adv. 2018;2:299–307.
Acknowledgements
This work was supported by the following funding: Fondazione Cariplo 2015/0603 (DM and BB) Associazione Italiana per la Ricerca sul Cancro IG21567, and Intramural Research Funding of Istituto Clinico Humanitas (5*1000 project) (DM and LC).
Author information
Authors and Affiliations
Contributions
JM performed research, analyzed and interpreted data, wrote the paper; AMR collected data and provided data interpretation; AE provided data analysis and interpretation; MAC, MM, FP, AR collected data and supervised the paper; AB, LG, LB, BL, GC, RF, AS, LM, SM, CF, SS, PC collected data; AB, DB, provided data interpretation and revised the paper; SB designed research; DM, EA, LC, and BB designed research, provided data interpretation, and supervised paper writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mariotti, J., Raiola, A.M., Evangelista, A. et al. Impact of second-degree related donor on the outcomes of T cell-replete haploidentical transplantation with post-transplant cyclophosphamide. Bone Marrow Transplant 57, 1758–1764 (2022). https://doi.org/10.1038/s41409-022-01565-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-022-01565-1