Abstract
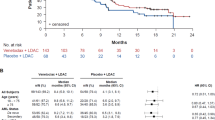
We retrospectively compared the outcomes of 20 patients receiving Venetoclax + low-dose Cytarabine + Actinomycin D (ACTIVE) with 29 patients receiving FLAG-Ida as salvage therapy for relapsed or refractory AML (R/R AML) after alloSCT. The groups were statistically balanced according to age, performance status, cytogenetics, and previous treatment. The overall response rate (CR + CRp + MLFS) of ACTIVE was 75% (15/20) in comparison to 66% (19/29) in the FLAG-Ida group (p = 0.542). The cumulative CR + CRp rate was significantly higher in the ACTIVE group compared to FLAG-Ida (70% (14/20) vs. 34% (10/29), respectively, p = 0.02). All three patients failing previous Venetoclax therapy and five out of seven patients with previous FLAG-Ida exposure achieved a CR/CRp after ACTIVE induction. ACTIVE patients survived longer compared to FLAG-Ida patients (13.1 vs. 5.1 months, respectively, p = 0.032). The treatment-related mortality was 0% in the ACTIVE group and 34% (10/29) in the FLAG-Ida patients (p = 0.003). The cumulative incidence of relapse did not differ between the two treatment groups. ACTIVE appears to have comparable antileukemic activity and lower toxicity compared to FLAG-Ida resulting in improved survival. Patients with Venetoclax or FLAG-Ida exposure responded to ACTIVE.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Büchner T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129:424–47.
Schmid C, de Wreede LC, van Biezen A, Finke J, Ehninger G, Ganser A, et al. Outcome after relapse of myelodysplastic syndrome and secondary acute myeloid leukemia following allogeneic stem cell transplantation: a retrospective registry analysis on 698 patients by the Chronic Malignancies Working Party of the European Society of Blood and Marrow Transplantation. Haematologica. 2018;103:237–45.
Christopeit M, Kuss O, Finke J, Bacher U, Beelen DW, Bornhäuser M, et al. Second allograft for hematologic relapse of acute leukemia after first allogeneic stem-cell transplantation from related and unrelated donors: the role of donor change. J Clin Oncol. 2013;31:3259–71.
Barrett AJ, Battiwalla M. Relapse after allogeneic stem cell transplantation. Expert Rev Hematol. 2010;3:429–41.
Levine JE, Braun T, Penza SL, Beatty P, Cornetta K, Martino R, et al. Prospective trial of chemotherapy and donor leukocyte infusions for relapse of advanced myeloid malignancies after allogeneic stem-cell transplantation. J Clin Oncol. 2002;20:405–12.
Martin MG, Augustin KM, Uy GL, Welch JS, Hladnik L, Goyal S, et al. Salvage therapy for acute myeloid leukemia with Fludarabine, Cytarabine, and Idarubicin with or without gemtuzumab ozogamicin and with concurrent or sequential G-CSF. Am J Hematol. 2009;84:733–7.
Sauer T, Silling G, Groth C, Rosenow F, Krug U, Görlich D, et al. Treatment strategies in patients with AML or high-risk myelodysplastic syndrome relapsed after Allo-SCT. Bone Marrow Transpl. 2015;50:485–92.
Bergua JM, Montesinos P, Martinez-Cuadrón D, Fernández-Abellán P, Serrano J, Sayas MJ, et al. A prognostic model for survival after salvage treatment with FLAG-Ida +/− gemtuzumab-ozogamicine in adult patients with refractory/relapsed acute myeloid leukaemia. Br J Haematol. 2016;174:700–10.
Motabi IH, Ghobadi A, Liu J, Schroeder M, Abboud CN, Cashen AF, et al. Chemotherapy versus hypomethylating agents for the treatment of relapsed acute myeloid leukemia and myelodysplastic syndrome after allogeneic stem cell transplant. Biol Blood Marrow Transplant. 2016;22:1324–9.
Shahswar R, Beutel G, Klement P, Rehberg A, Gabdoulline R, Koenecke C, et al. FLA-IDA salvage chemotherapy combined with a seven-day course of Venetoclax (FLAVIDA) in patients with relapsed/refractory acute leukaemia. Br J Haematol, 188:e11–5. https://doi.org/10.1111/bjh.16268
Perl AE, Martinelli G, Cortes JE, Neubauer A, Berman E, Paolini S, et al. Gilteritinib or chemotherapy for relapsed or refractory FLT3-mutated AML. N Engl J Med. 2019;381:1728–40.
DiNardo CD, Stein EM, de Botton S, Roboz GJ, Altman JK, Mims AS, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378:2386–98.
Stein EM, DiNardo CD, Pollyea DA, Fathi AT, Roboz GJ, Altman JK, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130:722–31.
Xu H, Krystal GW. Actinomycin D decreases Mcl-1 expression and acts synergistically with ABT-737 against small cell lung cancer cell lines. Clin Cancer Res. 2010;16:4392–400.
Tahir SK, Smith ML, Hessler P, Rapp LR, Idler KB, Park CH, et al. Potential mechanisms of resistance to Venetoclax and strategies to circumvent it. BMC Cancer. 2017;17:399.
Oppermann S, Ylanko J, Shi Y, Hariharan S, Oakes CC, Brauer PM, et al. High-content screening identifies kinase inhibitors that overcome Venetoclax resistance in activated CLL cells. Blood. 2016;128:934–47.
Luedtke DA, Niu X, Pan Y, Zhao J, Liu S, Edwards H, et al. Inhibition of Mcl-1 enhances cell death induced by the Bcl-2-selective inhibitor ABT-199 in acute myeloid leukemia cells. Signal Transduct Target Ther. 2017;2:17012.
Falini B, Brunetti L, Martelli MP. Dactinomycin in NPM1-mutated acute myeloid leukemia. N Engl J Med. 2015;373:1180–2.
Beziat G, Tavitian S, Bertoli S, Huguet F, Largeaud L, Luquet I, et al. Dactinomycin in acute myeloid leukemia with NPM1 mutations. Eur J Haematol. 2020;105:302–7. https://doi.org/10.1111/ejh.13438.
Zucenka A, Pileckyte R, Vaitekenaite V, Surauciute A, Griskevicius L. PF273 Venetoclax in combination with low dose Cytarabine and/or actinomycin D in real life relapsed/refractory acute myeloid leukemia patients. HemaSphere. 2019;3 S1:88.
Maiti A, DiNardo CD, Rausch CR, Cortes JE, Pemmaraju N, Daver NG, et al. Ten-day decitabine with Venetoclax (DEC10-VEN) in acute myeloid leukemia: updated results of a Phase II Trial. Blood. 2019;134:2637. Supplement_1
DiNardo CD, Rausch CR, Benton C, Kadia T, Jain N, Pemmaraju N, et al. Clinical experience with the BCL2-inhibitor Venetoclax in combination therapy for relapsed and refractory acute myeloid leukemia and related myeloid malignancies. Am J Hematol. 2018;93:401–7.
Goldberg AD, Horvat TZ, Hsu M, Devlin SM, Cuello BM, Daley RJ. et al. Venetoclax combined with either a hypomethylating agent or low-dose Cytarabine shows activity in relapsed and refractory myeloid malignancies. Blood. 2017;130 Supplement 1:1353.
Aldoss I, Yang D, Aribi A, Ali H, Sandhu K, Al Malki MM, et al. Efficacy of the combination of Venetoclax and hypomethylating agents in relapsed/refractory acute myeloid leukemia. Haematologica. 2018;103:e404–7.
Gaut D, Burkenroad A, Duong T, Feammelli J, Sasine J, Schiller G. Venetoclax combination therapy in relapsed/refractory acute myeloid leukemia: a single institution experience. Leuk Res. 2020;90:106314. https://doi.org/10.1016/j.leukres.2020.106314
Ganzel C, Ram R, Gural A, Wolach O, Gino-Moor S, Vainstein V, et al. Venetoclax is safe and efficacious in relapsed/refractory AML. Leuk Lymphoma. 2020;61:2221–5.
Asghari H. Outcomes of patients with relapsed or refractory acute myeloid leukemia receiving hypomethylating agent and Venetoclax. Blood (2019) 134 (Supplement_1): 1357. Available from: https://ash.confex.com/ash/2019/webprogram/Paper128958.html.
Bewersdorf JP, Giri S, Wang R, Williams RT, Tallman MS, Zeidan AM, et al. Venetoclax as monotherapy and in combination with hypomethylating agents or low dose Cytarabine in relapsed and treatment refractory acute myeloid leukemia: a systematic review and meta-analysis. Haematologica. 2020;105:2659–63. https://doi.org/10.3324/haematol.2019.242826
DiNardo CD, Lachowiez CA, Takahashi K, Loghavi S, Xiao L, Kadia T, et al. Venetoclax combined with FLAG-IDA induction and consolidation in newly diagnosed and relapsed or refractory acute myeloid leukemia. J Clin Oncol. 2021:JCO2003736. https://doi.org/10.1200/JCO.20.03736
de Weerdt I, Hofland T, Dobber J, Dubois J, Eldering E, Mobasher M. et al. First evidence of restoration of T and NK cell compartment after Venetoclax treatment. Blood. 2018;132 Supplement 1:1860 .
Elías EE, Almejún MB, Colado A, Cordini G, Vergara-Rubio M, Podaza E, et al. Autologous T-cell activation fosters ABT-199 resistance in chronic lymphocytic leukemia: rationale for a combined therapy with SYK inhibitors and anti-CD20 monoclonal antibodies. Haematologica. 2018;103:e458–61.
Mathew R, Haribhai D, Kohlhapp F, Duggan R, Ellis P, Riehm JJ. et al. The BCL-2-selective inhibitor Venetoclax spares activated T-cells during anti-tumor immunity. Blood. 2018;132 Supplement 1:3704.
Al-Sawaf O, Herling CD, Holtick U, Scheid C, Cramer P, Sasse S, et al. Venetoclax plus rituximab or obinutuzumab after allogeneic hematopoietic stem cell transplantation in chronic lymphocytic leukemia. Haematologica 2019;104:e224–6; https://doi.org/10.3324/haematol.2018.212837
Amit O, On YB, Perez G, Shargian-Alon L, Yeshurun M, Ram R. Venetoclax and donor lymphocyte infusion for early relapsed acute myeloid leukemia after allogeneic hematopoietic cell transplantation. A retrospective multicenter trial. Ann Hematol. 2021;100:817–24.
Bazarbachi A, Schmid C, Labopin M, Beelen D, Blau IW, Potter V, et al. Evaluation of trends and prognosis over time in patients with AML relapsing after allogeneic hematopoietic cell transplant reveals improved survival for young patients in recent years. Clin Cancer Res. 2020;26:6475–82.
Author information
Authors and Affiliations
Contributions
AZ: study design, data collection, statistical analysis, writing the manuscript LG: study design, writing the manuscript, study supervision. VV, KM, LD: data collection. RP, IT, VP: study design and implementation of the research. TZ, VS: statistical analysis. All authors provided critical feedback and helped shape the research, analysis and manuscript.
Corresponding author
Ethics declarations
Competing interests
Venetoclax was provided by Abbvie via patient early access program. AZ, RP, LG—Abbvie: Travel grants, honoraria; VV, KM, LD, IT, VP, TZ, VS—none.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zucenka, A., Vaitekenaite, V., Maneikis, K. et al. Venetoclax-based salvage therapy followed by Venetoclax and DLI maintenance vs. FLAG-Ida for relapsed or refractory acute myeloid leukemia after allogeneic stem cell transplantation. Bone Marrow Transplant 56, 2804–2812 (2021). https://doi.org/10.1038/s41409-021-01416-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-021-01416-5
This article is cited by
-
Venetoclax-based regimens in combination with trametinib for RAS-mutated relapsed or refractory myeloid malignancies
Bone Marrow Transplantation (2022)
-
Relapse after allogeneic hematopoietic stem cell transplantation in acute myeloid leukemia: an overview of prevention and treatment
International Journal of Hematology (2022)
-
Clinical experience with venetoclax in patients with newly diagnosed, relapsed, or refractory acute myeloid leukemia
Journal of Cancer Research and Clinical Oncology (2022)
-
Cytarabine/dactinomycin/venetoclax
Reactions Weekly (2022)
-
A retrospective single-center analysis of G-CSF-mobilized donor lymphocyte infusion in hematologic malignancies after unmanipulated allogenic PBSCT
International Journal of Hematology (2022)