Abstract
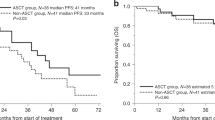
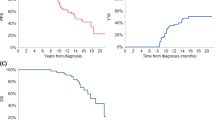
We evaluated the outcomes of 168 patients undergoing delayed or second autologous stem cell transplant (ASCT) for relapsed multiple myeloma (MM) from 2010 to 2019. Overall, 21% (n = 35) patients had received a prior transplant and 69% (n = 116) underwent transplant at first relapse. Overall, 27% patients had high-risk cytogenetics and 15% had ISS stage III disease. Stem cell collection was performed after relapse in 72% and 35% of patients received maintenance therapy. Median PFS from salvage treatment and transplant were 28 and 19 months, respectively. Median OS from salvage treatment and transplant was 69 and 55 months. Multivariate analysis revealed that ASCT in first relapse was associated with superior PFS (HR 0.63, p = 0.03) and OS (HR 0.59, p = 0.04) compared to later lines of therapy. In addition, PFS of ≥36 months with prior therapy was associated with improved PFS (HR 0.62, p = 0.04) and OS (HR 0.41, p = 0.01). Ninety-five patients underwent delayed transplant at first relapse, median PFS and OS from start of therapy was 30 and 69 months, and median OS from diagnosis was 106 months. These data may serve as a guide when counseling patients undergoing ASCT for relapsed MM and provide a benchmark in designing clinical trials of transplantation/comparative treatments for relapsed MM.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Palumbo A, Bringhen S, Ludwig H, Dimopoulos MA, Blade J, Mateos MV, et al. Personalized therapy in multiple myeloma according to patient age and vulnerability: a report of the European Myeloma Network (EMN). Blood. 2011;118:4519–29. https://doi.org/10.1182/blood-2011-06-358812.
Smith BD, Smith GL, Hurria A, Hortobagyi GN, Buchholz TA. Future of cancer incidence in the United States: burdens upon an aging, changing nation. J Clin Oncol. 2009;27:2758–65. https://doi.org/10.1200/JCO.2008.20.8983.
Attal M, Harousseau JL, Stoppa AM, Sotto JJ, Fuzibet JG, Rossi JF, et al. A prospective, randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. Intergroupe Francais du Myelome. N. Engl J Med. 1996;335:91–7. https://doi.org/10.1056/NEJM199607113350204.
Child JA, Morgan GJ, Davies FE, Owen RG, Bell SE, Hawkins K, et al. High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N. Engl J Med. 2003;348:1875–83. https://doi.org/10.1056/NEJMoa022340.
Rajkumar SV. Multiple myeloma: 2020 update on diagnosis, risk-stratification and management. Am J Hematol. 2020;95:548–67. https://doi.org/10.1002/ajh.25791.
Attal M, Lauwers-Cances V, Hulin C, Leleu X, Caillot D, Escoffre M, et al. Lenalidomide, bortezomib, and dexamethasone with transplantation for myeloma. N. Engl J Med. 2017;376:1311–20. https://doi.org/10.1056/NEJMoa1611750.
Bhutani D, Zonder J, Valent J, Tageja N, Ayash L, Deol A, et al. Evaluating the effects of lenalidomide induction therapy on peripheral stem cells collection in patients undergoing autologous stem cell transplant for multiple myeloma. Support Care Cancer. 2013;21:2437–42. https://doi.org/10.1007/s00520-013-1808-5. e-pub ahead of print 2013/04/18.
Goldschmidt H, Hegenbart U, Wallmeier M, Hohaus S, Haas R. Factors influencing collection of peripheral blood progenitor cells following high-dose cyclophosphamide and granulocyte colony-stimulating factor in patients with multiple myeloma. Br J Haematol. 1997;98:736–44. https://doi.org/10.1046/j.1365-2141.1997.2783095.x.
Working G. Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol. 2003;121:749–57.
Greipp PR, San Miguel J, Durie BG, Crowley JJ, Barlogie B, Blade J, et al. International staging system for multiple myeloma. J Clin Oncol: Off J Am Soc Clin Oncol. 2005;23:3412–20. https://doi.org/10.1200/JCO.2005.04.242.
Saad A, Mahindra A, Zhang MJ, Zhong X, Costa LJ, Dispenzieri A, et al. Hematopoietic cell transplant comorbidity index is predictive of survival after autologous hematopoietic cell transplantation in multiple myeloma. Biol Blood Marrow Transplant. 2014;20:402–8 e401. https://doi.org/10.1016/j.bbmt.2013.12.557.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9. https://doi.org/10.1182/blood-2005-05-2004.
Kumar S, Paiva B, Anderson KC, Durie B, Landgren O, Moreau P, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 2016;17:e328–e346. https://doi.org/10.1016/S1470-2045(16)30206-6.
Child JA, Morgan GJ, Davies FE, Owen RG, Bell SE, Hawkins K, et al. High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N. Engl J Med. 2003;348:1875–83. https://doi.org/10.1056/NEJMoa022340.
Cook G, Ashcroft AJ, Cairns DA, Williams CD, Brown JM, Cavenagh JD, et al. The effect of salvage autologous stem-cell transplantation on overall survival in patients with relapsed multiple myeloma (final results from BSBMT/UKMF Myeloma X Relapse [Intensive]): a randomised, open-label, phase 3 trial. Lancet Haematol. 2016;3:e340–351. https://doi.org/10.1016/s2352-3026(16)30049-7.
Goldschmidt H, Baertsch MA, Schlenzka J, Becker N, Habermehl C, Hielscher T, et al. Salvage autologous transplant and lenalidomide maintenance vs. lenalidomide/dexamethasone for relapsed multiple myeloma: the randomized GMMG phase III trial ReLApsE. Leukemia. 2020. https://doi.org/10.1038/s41375-020-0948-0.
Abdallah AO, Mohyuddin GR, Mahmoudjafari Z, Atrash S, Kawsar H, Sigle M, et al. Outcomes of daratumumab, pomalidomide, and dexamethasone, followed by high-dose chemotherapy and autologous stem cell transplantation, in patients with relapsed/refractory multiple myeloma. Clin Lymphoma Myeloma Leuk. 2020. https://doi.org/10.1016/j.clml.2020.08.026.
Biran N, Rowley SD, Vesole DH, Zhang S, Donato ML, Richter J, et al. A phase I/II study of escalating doses of bortezomib in conjunction with high-dose melphalan as a conditioning regimen for salvage autologous peripheral blood stem cell transplantation in patients with multiple myeloma. Biol Blood Marrow Transplant. 2016;22:2165–71. https://doi.org/10.1016/j.bbmt.2016.08.017.
Costa LJ, Landau HJ, Chhabra S, Hari P, Innis-Shelton R, Godby KN, et al. Phase 1/2 trial of carfilzomib plus high-dose melphalan preparative regimen for salvage autologous hematopoietic cell transplantation followed by maintenance carfilzomib in patients with relapsed/refractory multiple myeloma. Biol Blood Marrow Transplant. 2018;24:1379–85. https://doi.org/10.1016/j.bbmt.2018.01.036.
Dhakal B, D’Souza A, Kleman A, Chhabra S, Mohan M, Hari P. Salvage second transplantation in relapsed multiple myeloma. Leukemia. 2020. https://doi.org/10.1038/s41375-020-1005-8.
Gonsalves WI, Kansagra A. Second autologous hematopoietic stem cell transplant as salvage therapy for relapsed multiple myeloma: a global treatment option for eligible patients. Acta Haematol. 2018;139:45–6. https://doi.org/10.1159/000486272.
Hagen PA, Stiff P. The role of salvage second autologous hematopoietic cell transplantation in relapsed multiple myeloma. Biol Blood Marrow Transplant. 2019;25(3):e98–e107. https://doi.org/10.1016/j.bbmt.2018.12.002.
Holstein SA, McCarthy PL. Improved survival with salvage autologous stem-cell transplantation in myeloma. Lancet Haematol. 2016;3:e306–307. https://doi.org/10.1016/s2352-3026(16)30058-8.
Manjappa S, Fiala MA, King J, Kohnen DA, Vij R. The efficacy of salvage autologous stem cell transplant among patients with multiple myeloma who received maintenance therapy post initial transplant. Bone marrow Transplant. 2018;53:1483–6. https://doi.org/10.1038/s41409-018-0216-3.
Miller KC, Gertz MA, Buadi FK, Hayman SR, Lacy MQ, Dispenzieri AA, et al. The impact of re-induction prior to salvage autologous stem cell transplantation in multiple myeloma. Bone marrow Transplant. 2019;54:2039–50. https://doi.org/10.1038/s41409-019-0590-5.
Nieto Y, Valdez BC, Pingali SR, Bassett R, Delgado R, Nguyen J, et al. High-dose gemcitabine, busulfan, and melphalan for autologous stem-cell transplant in patients with relapsed or refractory myeloma: a phase 2 trial and matched-pair comparison with melphalan. Lancet Haematol. 2017;4:e283–e292. https://doi.org/10.1016/s2352-3026(17)30080-7.
Singh Abbi KK, Zheng J, Devlin SM, Giralt S, Landau H. Second autologous stem cell transplant: an effective therapy for relapsed multiple myeloma. Biol Blood Marrow Transplant. 2015;21:468–72. https://doi.org/10.1016/j.bbmt.2014.11.677.
McCarthy PL, Owzar K, Hofmeister CC, Hurd DD, Hassoun H, Richardson PG, et al. Lenalidomide after stem-cell transplantation for multiple myeloma. N. Engl J Med. 2012;366:1770–81. https://doi.org/10.1056/NEJMoa1114083.
Palumbo A, Cavallo F, Gay F, Di Raimondo F, Ben Yehuda D, Petrucci MT, et al. Autologous transplantation and maintenance therapy in multiple myeloma. N. Engl J Med. 2014;371:895–905. https://doi.org/10.1056/NEJMoa1402888.
Holstein SA, Jung SH, Richardson PG, Hofmeister CC, Hurd DD, Hassoun H, et al. Updated analysis of CALGB (Alliance) 100104 assessing lenalidomide versus placebo maintenance after single autologous stem-cell transplantation for multiple myeloma: a randomised, double-blind, phase 3 trial. Lancet Haematol. 2017;4:e431–e442. https://doi.org/10.1016/s2352-3026(17)30140-0.
McCarthy PL, Holstein SA, Petrucci MT, Richardson PG, Hulin C, Tosi P, et al. Lenalidomide maintenance after autologous stem-cell transplantation in newly diagnosed multiple myeloma: a meta-analysis. J Clin Oncol. 2017;35:3279–89. https://doi.org/10.1200/JCO.2017.72.6679.
Fiala MA, Vosuri V, Goldsmith S, Schroeder MA, Ghobadi A, Wildes TM, et al. Maintenance therapy following salvage autologous stem cell transplant in patients with multiple myeloma. Bone marrow Transplant. 2020;55:1188–90. https://doi.org/10.1038/s41409-019-0663-5.
Gertz MA, Ansell SM, Dingli D, Dispenzieri A, Buadi FK, Elliott MA, et al. Autologous stem cell transplant in 716 patients with multiple myeloma: low treatment-related mortality, feasibility of outpatient transplant, and effect of a multidisciplinary quality initiative. Mayo Clin Proc. 2008;83:1131–8. https://doi.org/10.4065/83.10.1131.
Auner HW, Iacobelli S, Sbianchi G, Knol-Bout C, Blaise D, Russell NH, et al. Melphalan 140 mg/m(2) or 200 mg/m(2) for autologous transplantation in myeloma: results from the Collaboration to Collect Autologous Transplant Outcomes in Lymphoma and Myeloma (CALM) study. A report by the EBMT Chronic Malignancies Working Party. Haematologica. 2018;103:514–21. https://doi.org/10.3324/haematol.2017.181339.
Abdi J, Chen G, Chang H. Drug resistance in multiple myeloma: latest findings and new concepts on molecular mechanisms. Oncotarget. 2013;4:2186–207. https://doi.org/10.18632/oncotarget.1497.
Michaelis LC, Saad A, Zhong X, Le-Rademacher J, Freytes CO, Marks DI, et al. Salvage second hematopoietic cell transplantation in myeloma. Biol Blood Marrow Transplant. 2013;19:760–6. https://doi.org/10.1016/j.bbmt.2013.01.004.
Olin RL, Vogl DT, Porter DL, Luger SM, Schuster SJ, Tsai DE, et al. Second auto-SCT is safe and effective salvage therapy for relapsed multiple myeloma. Bone marrow Transplant. 2009;43:417–22. https://doi.org/10.1038/bmt.2008.334.
Lee SE, Yoon JH, Shin SH, Cho BS, Eom KS, Kim YJ, et al. Impact of failed response to novel agent induction in autologous stem cell transplantation for multiple myeloma. Ann Hematol. 2014;93:627–34. https://doi.org/10.1007/s00277-013-1911-1.
Vij R, Kumar S, Zhang MJ, Zhong X, Huang J, Dispenzieri A, et al. Impact of pretransplant therapy and depth of disease response before autologous transplantation for multiple myeloma. Biol Blood Marrow Transplant. 2015;21:335–41. https://doi.org/10.1016/j.bbmt.2014.10.023.
Tandon N, Sidana S, Rajkumar SV, Gertz MA, Buadi FK, Lacy MQ, et al. Outcomes with early response to first-line treatment in patients with newly diagnosed multiple myeloma. Blood Adv. 2019;3:744–50. https://doi.org/10.1182/bloodadvances.2018022806.
de la Rubia J, Blade J, Lahuerta JJ, Ribera JM, Martinez R, Alegre A, et al. Effect of chemotherapy with alkylating agents on the yield of CD34+ cells in patients with multiple myeloma. Results of the Spanish Myeloma Group (GEM) Study. Haematologica. 2006;91:621–7.
Baertsch MA, Schlenzka J, Lisenko K, Krzykalla J, Becker N, Weisel K, et al. Cyclophosphamide-based stem cell mobilization in relapsed multiple myeloma patients: A subgroup analysis from the phase III trial ReLApsE. Eur J Haematol. 2017;99:42–50. https://doi.org/10.1111/ejh.12888.
Gay F, Musto P, Rota Scalabrini D, Galli M, Belotti A, Zamagni E, et al. Survival analysis of newly diagnosed transplant-eligible multiple myeloma patients in the randomized forte trial. Blood. 2020;136:35–7. https://doi.org/10.1182/blood-2020-136907.
Cavo M, Gay F, Beksac M, Dimopoulos MA, Pantani L, Petrucci MT, et al. Upfront autologous hematopoietic stem-cell transplantation improves overall survival in comparison with bortezomib-based intensification therapy in newly diagnosed multiple myeloma: long-term follow-up analysis of the randomized phase 3 EMN02/HO95 study. Blood. 2020;136:37–8. https://doi.org/10.1182/blood-2020-137575.
Perrot A, Lauwers-Cances V, Cazaubiel T, Facon T, Caillot D, Clement-Filliatre L, et al. Early versus late autologous stem cell transplant in newly diagnosed multiple myeloma: long-term follow-up analysis of the IFM 2009 trial. Blood. 2020;136:39–39. https://doi.org/10.1182/blood-2020-134538.
Acknowledgements
Stanford BMT Database Management team. SS was supported by: KL2TR003143, KL2 Mentored Career Development Program, Stanford Clinical Translational Science Award Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
SS: consultancy: Janssen; Research support: Janssen, Allogene, and Magenta Therapeutics. AR: Research Support, Pharmacyclics; one-time advisory boards for Nohla and Kaleido; expert witness, US Department of Justice; brother employed by Johnson & Johnson.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lemieux, C., Muffly, L.S., Iberri, D.J. et al. Outcomes after delayed and second autologous stem cell transplant in patients with relapsed multiple myeloma. Bone Marrow Transplant 56, 2664–2671 (2021). https://doi.org/10.1038/s41409-021-01371-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-021-01371-1
This article is cited by
-
Clinical factors associated with autologous stem cell transplantation outcomes in multiple myeloma: upfront transplant with MEL200 remains the standard of care
Annals of Hematology (2024)
-
Bridging advanced myeloma patients to subsequent treatments and clinical trials with classical chemotherapy and stem cell support
Bone Marrow Transplantation (2023)
-
Cellular Immunotherapies for Multiple Myeloma: Current Status, Challenges, and Future Directions
Oncology and Therapy (2022)