Abstract
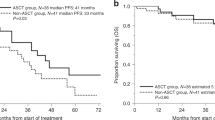
Supported by clinical trial proven survival benefit, clinical guidelines recommend upfront autologous stem cell transplantation (ASCT) for eligible MM patients. However, reported real-world utilisation is lower than expected (40–60%). We reviewed ASCT utilisation, demographics and outcomes for MM patients (≤70 years, ≥12-month follow-up) enroled onto the Australian/New Zealand MRDR from June 2012 to May 2020. In 982 patients (<65 years: 684, 65–70 years: 298), ASCT utilisation was 76% overall (<65 years: 83%, 65–70 years: 61%, front-line therapy: 67%). Non-ASCT recipients were older (median age: 65 years vs 60 years, p < 0.001), had more comorbidities (cardiac disease: 16.9% vs 5.4%, p < 0.001; diabetes: 19.1% vs 7.0%, p < 0.001; renal dysfunction: median eGFR(ml/min): 68 vs 80, p < 0.001), inferior performance status (ECOG ≥ 2: 26% vs 13%, p < 0.001) and higher-risk MM (ISS-3: 37% vs 26%, p = 0.009, R-ISS-3 18.6% vs 11.8%, p = 0.051) than ASCT recipients. ASCT survival benefit (median progression-free survival (PFS): 45.3 months vs 35.2 months, p < 0.001; overall survival (OS): NR vs 64.0 months, p < 0.001) was maintained irrespective of age (<65 years: median PFS: 45.3 months vs 37.7 months, p = 0.04, OS: NR vs 68.2 months, p = 0.002; 65–70 years: median PFS: 46.7 months vs 29.2 months, p < 0.001, OS: 76.9 months vs 55.6 months, p = 0.005). This large, real-world cohort reaffirms ASCT survival benefit, including in ‘older’ patients necessitating well-designed studies evaluating ASCT in ‘older’ MM to inform evidence-based patient selection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kyle RA, Steensma DP. History of multiple myeloma. Recent Results Cancer Res. 2011;183:3–23.
Kumar SK, Dispenzieri A, Lacy MQ, Gertz MA, Buadi FK, Pandey S, et al. Continued improvement in survival in multiple myeloma: changes in early mortality and outcomes in older patients. Leukemia. 2014;28:1122–8.
Naymagon L, Abdul-Hay M. Novel agents in the treatment of multiple myeloma: a review about the future. J Hematol Oncol. 2016;9:52.
Moreau P, Richardson PG, Cavo M, Orlowski RZ, San Miguel JF, Palumbo A, et al. Proteasome inhibitors in multiple myeloma: 10 years later. Blood. 2012;120:947–59.
Varga C, Laubach JP, Anderson KC, Richardson PG. Investigational agents in immunotherapy: a new horizon for the treatment of multiple myeloma. Br J Haematol. 2018;181:433–46.
Hipp S, Tai YT, Blanset D, Deegen P, Wahl J, Thomas O, et al. A novel BCMA/CD3 bispecific T-cell engager for the treatment of multiple myeloma induces selective lysis in vitro and in vivo. Leukemia. 2017;31:1743–51.
Zou J, Chen D, Zong Y, Ye S, Tang J, Meng H, et al. Immunotherapy based on bispecific T-cell engager with hIgG1 Fc sequence as a new therapeutic strategy in multiple myeloma. Cancer Sci. 2015;106:512–21.
D'Agostino M, Boccadoro M, Smith EL. Novel Immunotherapies for Multiple Myeloma. Curr Hematol Malig Rep. 2017;12:344–57.
Anderson KC. New insights into therapeutic targets in myeloma. Hematol Am Soc Hematol Educ Program. 2011;2011:184–90.
Attal M, Harousseau JL, Stoppa AM, Sotto JJ, Fuzibet JG, Rossi JF, et al. A prospective, randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. Intergroupe Francais du Myelome. N. Engl J Med. 1996;335:91–7.
Child JA, Morgan GJ, Davies FE, Owen RG, Bell SE, Hawkins K, et al. High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N. Engl J Med. 2003;348:1875–83.
Gay F, Oliva S, Petrucci MT, Conticello C, Catalano L, Corradini P, et al. Chemotherapy plus lenalidomide versus autologous transplantation, followed by lenalidomide plus prednisone versus lenalidomide maintenance, in patients with multiple myeloma: a randomised, multicentre, phase 3 trial. Lancet Oncol. 2015;16:1617–29.
Cavo M, Gay F, Beksac M, Pantani L, Petrucci MT, Dimopoulos MA, et al. Autologous haematopoietic stem-cell transplantation versus bortezomib-melphalan-prednisone, with or without bortezomib-lenalidomide-dexamethasone consolidation therapy, and lenalidomide maintenance for newly diagnosed multiple myeloma (EMN02/HO95): a multicentre, randomised, open-label, phase 3 study. Lancet Haematol. 2020;7:e456–68.
Attal M, Lauwers-Cances V, Hulin C, Leleu X, Caillot D, Escoffre M, et al. Lenalidomide, bortezomib, and dexamethasone with transplantation for myeloma. N. Engl J Med. 2017;376:1311–20.
Quach H, Joshua D, Ho J, Szer J, Spencer A, Harrison SJ, et al. Treatment of patients with multiple myeloma who are eligible for stem cell transplantation: position statement of the Myeloma Foundation of Australia Medical and Scientific Advisory Group. Intern Med J. 2015;45:94–105.
Moreau P, San Miguel J, Sonneveld P, Mateos MV, Zamagni E, Avet-Loiseau H, et al. Multiple myeloma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv52–iv61.
Mikhael J, Ismaila N, Cheung MC, Costello C, Dhodapkar MV, Kumar S, et al. Treatment of multiple myeloma: ASCO and CCO joint clinical practice guideline. J Clin Oncol. 2019;37:1228–63.
Wong Doo N, Coory M, White V, Harrison SJ, Prince HM, Millar JL, et al. Low uptake of upfront autologous transplantation for myeloma in a jurisdiction with universal health care coverage: a population-based patterns of care study in Australia. Clin Lymphoma Myeloma Leuk. 2014;14:61–7.
Nivison-Smith I, Bardy P, Dodds AJ, Ma DDF, Aarons D, Tran S, et al. A review of hematopoietic cell transplantation in Australia and New Zealand, 2005 to 2013. Biol Blood Marrow Transplant. 2016;22:284–91.
Morris TC, Velangi M, Jackson G, Marks DI, Ranaghan L. Northern Ireland Regional Haematology G, et al. Less than half of patients aged 65 years or under with myeloma proceed to transplantation: results of a two region population-based survey. Br J Haematol. 2005;128:510–2.
Bergin K, Moore E, McQuilten Z, Wood E, Augustson B, Blacklock H, et al. Design and development of the Australian and New Zealand (ANZ) myeloma and related diseases registry. BMC Med Res Methodol. 2016;16:151.
Palumbo A, Cavallo F, Gay F, Di Raimondo F, Ben Yehuda D, Petrucci MT, et al. Autologous transplantation and maintenance therapy in multiple myeloma. N. Engl J Med. 2014;371:895–905.
Merz M, Neben K, Raab MS, Sauer S, Egerer G, Hundemer M, et al. Autologous stem cell transplantation for elderly patients with newly diagnosed multiple myeloma in the era of novel agents. Ann Oncol. 2014;25:189–95.
Marini C, Maia T, Bergantim R, Pires J, Aguiar E, Guimarães JE, et al. Real-life data on safety and efficacy of autologous stem cell transplantation in elderly patients with multiple myeloma. Ann Hematol. 2019;98:369–79.
Cohen YC, Zuckerman T, Yeshurun M, Perez G, Magen H, Henig I, et al. Efficacy and safety of autologous hematopoietic cell transplantation in elderly patients with multiple myeloma: a retrospective national multi-site cohort study. Ann Hematol. 2017;96:271–8.
Garderet L, Beohou E, Caillot D, Stoppa AM, Touzeau C, Chretien ML, et al. Upfront autologous stem cell transplantation for newly diagnosed elderly multiple myeloma patients: a prospective multicenter study. Haematologica. 2016;101:1390–7.
Winn AN, Shah GL, Cohen JT, Lin P-J, Parsons SK. The real world effectiveness of hematopoietic transplant among elderly individuals with multiple myeloma. J Natl Cancer Inst. 2015;107:8.
Shah GL, Winn AN, Lin PJ, Klein A, Sprague KA, Smith HP, et al. Cost-effectiveness of autologous hematopoietic stem cell transplantation for elderly patients with multiple myeloma using the surveillance, epidemiology, and end results-medicare database. Biol Blood Marrow Transpl. 2015;21:1823–9.
Moreau P, Attal M. All transplantation-eligible patients with myeloma should receive ASCT in first response. Hematol Am Soc Hematol Educ Program. 2014;2014:250–4.
Nivison-Smith I, Simpson JM, Dodds AJ, Ma DD, Szer J, Bradstock KF. A population-based analysis of the effect of autologous hematopoietic cell transplant in the treatment of multiple myeloma. Leuk Lymphoma. 2013;54:1671–6.
Rosenberg AS, Brunson A, Jonas BA, Keegan THM, Wun T. Association between autologous stem cell transplant and survival among californians with multiple myeloma. J Natl Cancer Inst. 2019;111:78–85.
Costa LJ, Huang JX, Hari PN. Disparities in utilization of autologous hematopoietic cell transplantation for treatment of multiple myeloma. Biol Blood Marrow Transpl. 2015;21:701–6.
Hayden PJ, Eikema D-J, Koster L, Goldschmidt H, Blaise D, Potter M, et al. Trends in autologous transplantation for myeloma in EBMT centres between 1993 and 2017. Blood 2019;134:4575.
Gertz MA, Buadi FK, Hayman SR, Lacy MQ, Dispenzieri A, Dingli D, et al. Safety outcomes for autologous stem cell transplant in multiple myeloma. Mayo Clin Proc. 2018;93:56–8.
Sanchez L, Sylvester M, Parrondo R, Mariotti V, Eloy JA, Chang VT. In-hospital mortality and post-transplantation complications in elderly multiple myeloma patients undergoing autologous hematopoietic stem cell transplantation: a population-based study. Biol Blood Marrow Transpl. 2017;23:1203–7.
Spencer A, Prince HM, Roberts AW, Prosser IW, Bradstock KF, Coyle L, et al. Consolidation therapy with low-dose thalidomide and prednisolone prolongs the survival of multiple myeloma patients undergoing a single autologous stem-cell transplantation procedure. J Clin Oncol. 2009;27:1788–93.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Farina L, Bruno B, Patriarca F, Spina F, Sorasio R, Morelli M, et al. The hematopoietic cell transplantation comorbidity index (HCT-CI) predicts clinical outcomes in lymphoma and myeloma patients after reduced-intensity or non-myeloablative allogeneic stem cell transplantation. Leukemia. 2009;23:1131–8.
Palumbo A, Bringhen S, Mateos M-V, Larocca A, Facon T, Kumar SK, et al. Geriatric assessment predicts survival and toxicities in elderly myeloma patients: an International Myeloma Working Group report. Blood. 2015;125:2068–74.
Kaya H, Peressini B, Jawed I, Martincic D, Elaimy AL, Lamoreaux WT, et al. Impact of age, race and decade of treatment on overall survival in a critical population analysis of 40,000 multiple myeloma patients. Int J Hematol. 2012;95:64–70.
Rajkumar SV. Value and cost of myeloma therapy. Am Soc Clin Oncol Educ Book. 2018;38:662–6.
Rawstron AC, Child JA, de Tute RM, Davies FE, Gregory WM, Bell SE, et al. Minimal residual disease assessed by multiparameter flow cytometry in multiple myeloma: impact on outcome in the Medical Research Council Myeloma IX Study. J Clin Oncol. 2013;31:2540–7.
Paiva B, Vidriales MB, Cerveró J, Mateo G, Pérez JJ, Montalbán MA, et al. Multiparameter flow cytometric remission is the most relevant prognostic factor for multiple myeloma patients who undergo autologous stem cell transplantation. Blood. 2008;112:4017–23.
Munshi NC, Avet-Loiseau H, Rawstron AC, Owen RG, Child JA, Thakurta A, et al. Association of minimal residual disease with superior survival outcomes in patients with multiple myeloma: a meta-analysis. JAMA Oncol. 2017;3:28–35.
Barlogie B, Alexanian R, Dicke K, Zagars G, Spitzer G, Jagannath S, et al. High-dose chemoradiotherapy and autologous bone marrow transplantation for resistant multiple myeloma. Blood. 1987;70:869–72.
Barlogie B, Hall R, Zander A, Dicke K, Alexanian R. High-dose melphalan with autologous bone marrow transplantation for multiple myeloma. Blood. 1986;67:1298–301.
Muchtar E, Dingli D, Kumar S, Buadi FK, Dispenzieri A, Hayman SR, et al. Autologous stem cell transplant for multiple myeloma patients 70 years or older. Bone Marrow Transpl. 2016;51:1449–55.
Sharma M, Zhang MJ, Zhong X, Abidi MH, Akpek G, Bacher U, et al. Older patients with myeloma derive similar benefit from autologous transplantation. Biol Blood Marrow Transpl. 2014;20:1796–803.
Acknowledgements
The authors thank the patients, hospitals, clinicians, and research staff at participating institutions for their support of the MRDR. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors; however, the ANZ MRDR has received funding from Amgen, Bristol-Myers Squibb, Celgene, Gilead, Janssen, Novartis, Sanofi and Takeda.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Members of the Australian and New Zealand Myeloma and Related Diseases Registry are listed in Supplementary Information.
Rights and permissions
About this article
Cite this article
Bergin, K., Wellard, C., Augustson, B. et al. Real-world utilisation of ASCT in multiple myeloma (MM): a report from the Australian and New Zealand myeloma and related diseases registry (MRDR). Bone Marrow Transplant 56, 2533–2543 (2021). https://doi.org/10.1038/s41409-021-01308-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-021-01308-8