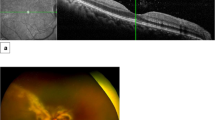
Abstract
Although cytomegalovirus (CMV) remains a leading cause of morbidity and mortality after allogeneic hematopoietic stem cell transplantation (HSCT), the incidence of CMV retinitis is considered to be lower than the incidence of CMV infection in other organs following allogeneic HSCT. In this study, the incidence and characteristics of CMV retinitis were retrospectively evaluated in recipients of allogeneic HSCT. Ophthalmological screening was performed at the development of ocular symptoms or positive CMV infection using peripheral blood evaluated by pp65 antigenemia or polymerase chain reaction. Of the 514 patients, 13 patients developed CMV retinitis. The median onset of CMV retinitis was day 34 (range, 21–118) post transplant, and the cumulative incidence was 2.5% (95% CI, 1.6–4.2) at 6 months after transplantation. Five patients presented ocular symptoms at the onset. In the remaining eight asymptomatic patients, the diagnosis of CMV retinitis was made by the screening guided by positive CMV infection. All evaluable patients responded to antiviral treatment but three showed incomplete improvement with ocular sequela. Our results suggest that the incidence of CMV retinitis after allogeneic HSCT is not negligible and active ophthalmological screening based not only on symptoms but also positive CMV infection monitoring contributes to the early diagnosis of CMV retinitis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Ljungman P, de la Camara R, Robin C, Crocchiolo R, Einsele H, Hill JA, et al. Guidelines for the management of cytomegalovirus infection in patients with haematological malignancies and after stem cell transplantation from the 2017 European Conference on Infections in Leukaemia (ECIL 7). Lancet Infect Dis. 2019;19:e260–e272.
Einsele H, Ljungman P, Boeckh M. How I treat CMV reactivation after allogeneic hematopoietic stem cell transplantation. Blood. 2020;135:1619–29.
Green ML, Leisenring W, Xie H, Mast TC, Cui Y, Sandmaier BM, et al. Cytomegalovirus viral load and mortality after haemopoietic stem cell transplantation in the era of pre-emptive therapy: a retrospective cohort study. Lancet Haematol. 2016;3:e119–127.
Marty FM, Ljungman P, Chemaly RF, Maertens J, Dadwal SS, Duarte RF, et al. Letermovir prophylaxis for cytomegalovirus in hematopoietic-cell transplantation. N Engl J Med. 2017;377:2433–44.
Mori T, Mori S, Kanda Y, Yakushiji K, Mineishi S, Takaue Y, et al. Clinical significance of cytomegalovirus (CMV) antigenemia in the prediction and diagnosis of CMV gastrointestinal disease after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2004;33:431–4.
Crippa F, Corey L, Chuang EL, Sale G, Boeckh M. Virological, clinical, and ophthalmologic features of cytomegalovirus retinitis after hematopoietic stem cell transplantation. Clin Infect Dis. 2001;32:214–9.
Xhaard A, Robin M, Scieux C, de Latour RP, Deplus S, Mazeron MC, et al. Increased incidence of cytomegalovirus retinitis after allogeneic hematopoietic stem cell transplantation. Transplantation. 2007;83:80–83.
Jeon S, Lee WK, Lee Y, Lee DG, Lee JW. Risk factors for cytomegalovirus retinitis in patients with cytomegalovirus viremia after hematopoietic stem cell transplantation. Ophthalmology. 2012;119:1892–8.
Mori T, Okamoto S, Matsuoka S, Yajima T, Wakui M, Watanabe R, et al. Risk-adapted pre-emptive therapy for cytomegalovirus disease in patients undergoing allogeneic bone marrow transplantation. Bone Marrow Transplant. 2000;25:765–9.
Mori T, Okamoto S, Watanabe R, Yajima T, Iwao Y, Yamazaki R, et al. Dose-adjusted preemptive therapy for cytomegalovirus disease based on real-time polymerase chain reaction after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2002;29:777–82.
Ljungman P, Boeckh M, Hirsch HH, Josephson F, Lundgren J, Nichols G, et al. Definitions of cytomegalovirus infection and disease in transplant patients for use in clinical trials. Clin Infect Dis. 2017;64:87–91.
Tomita Y, Kurihara T, Uchida A, Nagai N, Shinoda H, Tsubota K, et al. Wide-angle viewing system versus conventional indirect ophthalmoscopy for scleral buckling. Sci Rep. 2015;5:13256.
Ando Y, Terao K, Narita M, Oguchi Y, Sata T, Iwasaki T. Quantitative analyses of cytomegalovirus genome in aqueous humor of patients with cytomegalovirus retinitis. Jpn J Ophthalmol. 2002;46:254–60.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Larsson K, Lonnqvist B, Ringden O, Hedquist B, Ljungman P. CMV retinitis after allogeneic bone marrow transplantation: a report of five cases. Transpl Infect Dis. 2002;4:75–79.
Zollner SK, Herbruggen H, Kolve H, Mihailovic N, Schubert F, Reicherts C, et al. Cytomegalovirus retinitis in children and adolescents with acute leukemia following allogeneic hematopoietic stem cell transplantation. Transpl Infect Dis. 2019;21:e13089.
Heiden D, Ford N, Wilson D, Rodriguez WR, Margolis T, Janssens B, et al. Cytomegalovirus retinitis: the neglected disease of the AIDS pandemic. PLoS Med. 2007;4:e334.
Ausayakhun S, Yen M, Jirawison C, Ausayakhun S, Khunsongkiet P, Leenasirimakul P, et al. Visual acuity outcomes in cytomegalovirus retinitis: early versus late diagnosis. Br J Ophthalmol. 2018;102:1607–10.
Kim T, Lee YM, Lee SO, Choi SH, Kim YS, Woo JH, et al. Differences of cytomegalovirus diseases between kidney and hematopoietic stem cell transplant recipients during preemptive therapy. Korean J Intern Med. 2016;31:961–70.
Tsui E, Gonzales JA, Shantha JG, Acharya N, Doan T. Letermovir for the management of cytomegalovirus-associated uveitis. Ocul Immunol Inflamm. 2019:1–6. https://doi.org/10.1080/09273948.2019.1662062. [Epub ahead of print]
Turner N, Strand A, Grewal DS, Cox G, Arif S, Baker AW, et al. Use of letermovir as salvage therapy for drug-resistant cytomegalovirus retinitis. Antimicrob Agents Chemother. 2019;63:e02337–18.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
TM received research funding from MSD K.K., Novartis Pharma, LSI Medience, Medical & Biological Laboratories, Otsuka Pharmaceutical, Kyowa Kirin Co., Ltd., and Asahi Kasei Corporation, and personal fees from Pfizer Inc., MSD, Janssen Pharma, Sumitomo Dainippon Pharma, Novartis Pharma K.K., Kyowa Kirin Co., Ltd., Chugai Pharmaceutical, Shionogi & Co., Japan Blood Products Organization, Takeda Pharmaceutical, Ono Pharmaceutical, Shire, Eisai, and Astellas Pharma. TK received personal fees from Celgene, Bristol-Myers Squibb, Takeda Pharmaceutical, Otsuka Pharmaceutical, Eisai Pharmaceuticals, Sanofi K.K., and Janssen Pharma. MS received research funding from MSD K.K. and Nippon Shinyaku Co., Ltd., and personal fees from Nippon Shinyaku Co., Ltd., Takeda Pharmaceutical, Eisai Co., Ltd., and Otsuka Pharmaceutical. MS received personal fees from Bristol-Myers Squibb, Novartis Pharma, Sumitomo Dainippon Pharma, Chugai Pharma, Meiji Seika Parma, and Nippon Shinyaku.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mori, T., Kikuchi, T., Koh, M. et al. Cytomegalovirus retinitis after allogeneic hematopoietic stem cell transplantation under cytomegalovirus antigenemia-guided active screening. Bone Marrow Transplant 56, 1266–1271 (2021). https://doi.org/10.1038/s41409-020-01176-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-020-01176-8
This article is cited by
-
Clinical trials: design, endpoints and interpretation of outcomes
Bone Marrow Transplantation (2022)