Abstract
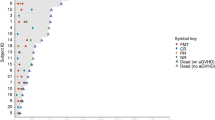
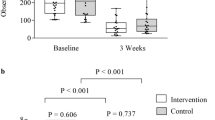
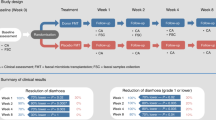
Autologous stem cell transplant (ASCT) for multiple myeloma (MM) is associated with diarrhea during the peri-transplant period. We aimed to appraise mechanisms of peri-ASCT diarrhea in a prospective, longitudinal study of patients with MM. We compared by repeated measures (RM)-ANOVA daily bowel movements (BMs) and consistency [7-point Bristol Stool Form Scale (BSFS)], fecal calprotectin (intestinal inflammation), 13C-mannitol excretion in urine 0–2 h (small intestinal permeability), fasting serum C4 (bile acid synthesis) and total and primary bile acid in stool samples during baseline, peri-transplant period (Days 5–7 after stem cell infusion), and after hematological recovery post-ASCT. The 12 (5F, 7M) patients’ median age was 61 y (IQR 54.8–63.3). All participants reported increased BMs (increase of 2 and 1 per day with and without engraftment syndrome, respectively). There were no significant increases in serum C4, total fecal bile acids, or intestinal permeability. Relative to patients without engraftment syndrome, four participants with engraftment syndrome had looser stool consistency (mean 2.6 points higher BSFS compared to without engraftment syndrome), increased primary fecal bile acids relative to baseline (>33 µmol/L vs. 6 µmol/L without engraftment syndrome), and increased fecal calprotectin compared to baseline (313 μg/mL vs. 35.6 μg/mL without engraftment syndrome; p = 0.06). Engraftment syndrome post-ASCT is associated with increased fecal primary bile acids.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
D’Souza A, Lee S, Zhu X, Pasquini M. Current use and trends in hematopoietic cell transplantation in the United States. Biol Blood Marrow Transpl. 2017;23:1417–21.
Gay F, Oliva S, Petrucci MT, Conticello C, Catalano L, Corradini P, et al. Chemotherapy plus lenalidomide versus autologous transplantation, followed by lenalidomide plus prednisone versus lenalidomide maintenance, in patients with multiple myeloma: a randomised, multicentre, phase 3 trial. Lancet Oncol. 2015;16:1617–29.
Attal M, Lauwers-Cances V, Hulin C, Leleu X, Caillot D, Escoffre M, et al. Lenalidomide, bortezomib, and dexamethasone with transplantation for myeloma. N Engl J Med. 2017;376:1311–20.
McCarthy PL Jr., Hahn T, Hassebroek A, Bredeson C, Gajewski J, Hale G, et al. Trends in use of and survival after autologous hematopoietic cell transplantation in North America, 1995–2005: significant improvement in survival for lymphoma and myeloma during a period of increasing recipient age. Biol Blood Marrow Transpl. 2013;19:1116–23.
McCarthy PL Jr, La W. Stem cell transplantation. Past, present, and future. In Buchsel PC, editor. Stem Cell Transplantation: A Clinical Textbook. 1st edn. Pittsburgh, PA, USA: Oncology Nursing Society; 2000, pp. 22–36.
Tomblyn M, Gordon L, Singhal S, Tallman M, Williams S, Winter J, et al. Rarity of toxigenic Clostridium difficile infections after hematopoietic stem cell transplantation: implications for symptomatic management of diarrhea. Bone Marrow Transpl. 2002;30:517–9.
Spitzer TR. Engraftment syndrome: double-edged sword of hematopoietic cell transplants. Bone Marrow Transpl. 2015;50:469–75.
Jo JC, Kang BW, Jang G, Sym SJ, Lee SS, Koo JE, et al. BEAC or BEAM high-dose chemotherapy followed by autologous stem cell transplantation in non-Hodgkin’s lymphoma patients: comparative analysis of efficacy and toxicity. Ann Hematol. 2008;87:43–8.
Sharma A, Kayal S, Iqbal S, Malik PS, Raina V. Comparison of BEAM vs. LEAM regimen in autologous transplant for lymphoma at AIIMS. SpringerPlus. 2013;2:489.
Joshi NM, Hassan S, Jasani P, Dixon S, Cavenagh JD, Oakervee HE, et al. Bile acid malabsorption in patients with graft-versus-host disease of the gastrointestinal tract. Br J Haematol. 2012;157:403–7.
Gupta A, Muls AC, Lalji A, Thomas K, Watson L, Shaw C, et al. Outcomes from treating bile acid malabsorption using a multidisciplinary approach. Support Care Cancer. 2015;23:2881–90.
Hofmann AF, Small DM. Detergent properties of bile salts: correlation with physiological function. Ann Rev Med. 1967;18:333–76.
Camilleri M. Physiological underpinnings of irritable bowel syndrome: neurohormonal mechanisms. J Physiol. 2014;592:2967–80.
Hofmann AF. The syndrome of ileal disease and the broken enterohepatic circulation: cholerheic enteropathy. Gastroenterology. 1967;52:752–7.
Wedlake L, A’Hern R, Russell D, Thomas K, Walters JR, Andreyev HJ. Systematic review: the prevalence of idiopathic bile acid malabsorption as diagnosed by SeHCAT scanning in patients with diarrhoea-predominant irritable bowel syndrome. Aliment Pharm Ther. 2009;30:707–17.
Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014;15:e538–48.
Lewis SJ, Heaton KW. Stool form scale as a useful guide to intestinal transit time. Scand J Gastroenterol. 1997;32:920–4.
Walters JR, Johnston IM, Nolan JD, Vassie C, Pruzanski ME, Shapiro DA. The response of patients with bile acid diarrhoea to the farnesoid X receptor agonist obeticholic acid. Aliment Pharm Ther. 2015;41:54–64.
Grover M, Camilleri M, Hines J, Burton D, Ryks M, Wadhwa A, et al. 13C mannitol as a novel biomarker for measurement of intestinal permeability. Neurogastroenterol Motil. 2016;28:1114–9.
Lostia AM, Lionetto L, Principessa L, Evangelisti M, Gamba A, Villa MP, et al. A liquid chromatography/mass spectrometry method for the evaluation of intestinal permeability. Clin Biochem. 2008;41:887–92.
Rao AS, Camilleri M, Eckert DJ, Busciglio I, Burton DD, Ryks M, et al. Urine sugars for in vivo gut permeability: validation and comparisons in irritable bowel syndrome-diarrhea and controls. Am J Physiol. 2011;301:G919–28.
Metafuni E, Giammarco S, De Ritis DG, Rossi M, De Michele T, Zuppi C, et al. Fecal but not serum calprotectin is a potential marker of GVHD after stem cell transplantation. Ann Hematol. 2017;96:929–33.
Hod K, Sperber AD, Ron Y, Boaz M, Dickman R, Berliner S, et al. A double-blind, placebo-controlled study to assess the effect of a probiotic mixture on symptoms and inflammatory markers in women with diarrhea-predominant IBS. Neurogastroenterol Motil. 2017;29. https://doi.org/10.1111/nmo.13037.
Galman C, Arvidsson I, Angelin B, Rudling M. Monitoring hepatic cholesterol 7alpha-hydroxylase activity by assay of the stable bile acid intermediate 7alpha-hydroxy-4-cholesten-3-one in peripheral blood. J Lipid Res. 2003;44:859–66.
Camilleri M, Nadeau A, Tremaine WJ, Lamsam J, Burton D, Odunsi S, et al. Measurement of serum 7alpha-hydroxy-4-cholesten-3-one (or 7alphaC4), a surrogate test for bile acid malabsorption in health, ileal disease and irritable bowel syndrome using liquid chromatography-tandem mass spectrometry. Neurogastroenterol Motil. 2009;21:734–e43.
Donato LJ, Lueke A, Kenyon SM, Meeusen JW, Camilleri M. Description of analytical method and clinical utility of measuring serum 7-alpha-hydroxy-4-cholesten-3-one (7aC4) by mass spectrometry. Clin Biochem. 2018;52:106–11.
Shin A, Camilleri M, Vijayvargiya P, Busciglio I, Burton D, Ryks M, et al. Bowel functions, fecal unconjugated primary and secondary bile acids, and colonic transit in patients with irritable bowel syndrome. Clin Gastroenterol Hepatol. 2013;11:1270–5. e1
Camilleri M, Acosta A, Busciglio I, Boldingh A, Dyer RB, Zinsmeister AR, et al. Effect of colesevelam on fecal bile acids and bowel functions in diarrhea-predominant irritable bowel syndrome. Aliment Pharm Ther. 2015;41:438–48.
Gordillo C, Parmar S, Blanco M, Delille EM, Assal A, Mapara MY, et al. Gastrointestinal toxicity of high-dose melphalan in autologous stem-cell transplantation: identification of risk factors and a benchmark for experimental therapies. Blood. 2017;130(Suppl 1):5464.
Hammami MB, Talkin R, Al-Taee AM, Schoen MW, Goyal SD, Lai JP. Autologous graft-versus-host disease of the gastrointestinal tract in patients with multiple myeloma and hematopoietic stem cell transplantation. Gastroenterol Res. 2018;11:52–57.
Rapoport AP, Stadtmauer EA, Aqui N, Vogl D, Chew A, Fang HB, et al. Rapid immune recovery and GVHD-like engraftment syndrome following adoptive transfer of costimulated autologous T cells. Clin Cancer Res. 2009;15:4499–507.
Shidham VB, Chang C-C, Shidham G, Ghazala F, Lindholm PF, Kampalath B, et al. Colon biopsies for evaluation of acute graft-versus-host disease (A-GVHD) in allogeneic bone marrow transplant patients. BMC Gastroenterol. 2003;3:5.
Hall AG, Tilby MJ. Mechanisms of action of, and modes of resistance to, alkylating agents used in the treatment of haematological malignancies. Blood Rev. 1992;6:163–73.
Weber D, Frauenschlager K, Ghimire S, Peter K, Panzer I, Hiergeist A, et al. The association between acute graft-versus-host disease and antimicrobial peptide expression in the gastrointestinal tract after allogeneic stem cell transplantation. PloS ONE. 2017;12:e0185265.
Pawlyn C, Khan MS, Muls A, Sriskandarajah P, Kaiser MF, Davies FE, et al. Lenalidomide-induced diarrhea in patients with myeloma is caused by bile acid malabsorption that responds to treatment. Blood. 2014;124:2467–8.
Acknowledgements
The authors thank Mrs. Cindy Stanislav for excellent secretarial assistance.
Funding
This study was supported by a grant to Dr. MC from Fondation Dr. Corinne Schuler.
Author contributions
WG and MC—co-principal investigators, patient care, authorship of manuscript. PV—authorship of manuscript. DB—database management. WR, AT—study coordination, patient recruitment. AL—lab measurements of serum C4 and total and individual fecal bile acids LD—laboratory supervision, authorship of manuscript. WJH and TM—protocol development.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Vijayvargiya, P., Gonsalves, W., Burton, D. et al. Increased fecal primary bile acids in multiple myeloma with engraftment syndrome diarrhea after stem cell transplant. Bone Marrow Transplant 54, 1898–1907 (2019). https://doi.org/10.1038/s41409-019-0581-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-019-0581-6