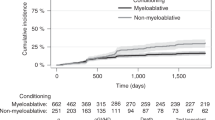
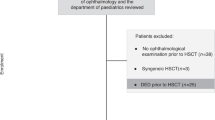
Abstract
Non-graft-vs.-host disease (non-GVHD) ocular complications are generally uncommon after hematopoietic cell transplantation (HCT), but can cause prolonged morbidity affecting activities of daily living and quality of life. Here we provide an expert review of non-GVHD ocular complications in a collaboration between transplant physicians and ophthalmologists through the Late Effects and Quality of Life Working Committee of the Center for International Blood and Marrow Transplant Research and the Transplant Complications Working Party of the European Society of Blood and Marrow Transplantation. Complications discussed in this review include cataracts, glaucoma, ocular infections, ocular involvement with malignancy, ischemic microvascular retinopathy, central retinal vein occlusion, retinal hemorrhage, retinal detachment, and ocular toxicities associated with medications. We have summarized incidence, risk factors, screening, prevention and treatment of individual complicastions and generated evidence-based recommendations. Baseline ocular evaluation before HCT should be considered in all patients who undergo HCT. Follow-up evaluations should be considered according to clinical symptoms, signs and risk factors. Better preventive strategies and treatments remain to be investigated for individual ocular complications after HCT. Both transplant physicians and ophthalmologists should be knowledgeable of non-GVHD ocular complications and provide comprehensive collaborative team care.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Change history
27 February 2019
The original version of this Article was updated to correct author name ‘Ahmad A’ to ‘Aisha Al-Khinji’.
27 February 2019
In the original version of this article, author ‘Aisha Al-Khinji’ was incorrectly listed as ‘Aisha Ahmed’. This has now been corrected in both the PDF and HTML versions of the article to ‘Aisha Al-Khinji’.
References
Socie G, Salooja N, Cohen A, Rovelli A, Carreras E, Locasciulli A, et al. Nonmalignant late effects after allogeneic stem cell transplantation. Blood. 2003;101:3373–85.
Inamoto Y, Lee SJ. Late effects of blood and marrow transplantation. Haematologica. 2017;102:614–25.
Battiwalla M, Hashmi S, Majhail N, Pavletic S, Savani BN, Shelburne N. National Institutes of Health Hematopoietic Cell Transplantation Late Effects Initiative: Developing Recommendations to Improve Survivorship and Long-Term Outcomes. Biol Blood Marrow Transplant. 2017;23:6–9.
Couriel D, Carpenter PA, Cutler C, Bolanos-Meade J, Treister NS, Gea-Banacloche J, et al. Ancillary therapy and supportive care of chronic graft-versus-host disease: national institutes of health consensus development project on criteria for clinical trials in chronic Graft-versus-host disease: V. Ancillary Therapy and Supportive Care Working Group Report. Biol Blood Marrow Transplant. 2006;12:375–96.
Dunn JP, Jabs DA, Wingard J, Enger C, Vogelsang G, Santos G. Bone marrow transplantation and cataract development. Arch Ophthalmol. 1993;111:1367–73.
Tichelli A, Gratwohl A, Egger T, Roth J, Prunte A, Nissen C, et al. Cataract formation after bone marrow transplantation. Ann Intern Med. 1993;119:1175–80.
Kinori M, Bielorai B, Souroujon D, Hutt D, Ben-Bassat Mizrachi I, Huna-Baron R. Ocular complications in children after hematopoietic stem cell transplantation without total body irradiation. Graefes Arch Clin Exp Ophthalmol. 2015;253:1397–402.
Frisk P, Hagberg H, Mandahl A, Soderberg P, Lonnerholm G. Cataracts after autologous bone marrow transplantation in children. Acta Paediatr. 2000;89:814–9.
Benyunes MC, Sullivan KM, Deeg HJ, Mori M, Meyer W, Fisher L, et al. Cataracts after bone marrow transplantation: long-term follow-up of adults treated with fractionated total body irradiation. Int J Radiat Oncol Biol Phys. 1995;32:661–70.
Belkacemi Y, Labopin M, Vernant JP, Prentice HG, Tichelli A, Schattenberg A, et al. Cataracts after total body irradiation and bone marrow transplantation in patients with acute leukemia in complete remission: a study of the European Group for Blood and Marrow Transplantation. Int J Radiat Oncol Biol Phys. 1998;41:659–68.
van Kempen-Harteveld ML, van Weel-Sipman MH, Emmens C, Noordijk EM, van der Tweel I, Revesz T, et al. Eye shielding during total body irradiation for bone marrow transplantation in children transplanted for a hematological disorder: risks and benefits. Bone Marrow Transplant. 2003;31:1151–6.
Fahnehjelm KT, Tornquist AL, Olsson M, Winiarski J. Visual outcome and cataract development after allogeneic stem-cell transplantation in children. Acta Ophthalmol Scand. 2007;85:724–33.
van Kempen-Harteveld ML, Struikmans H, Kal HB, van der Tweel I, Mourits MP, Verdonck LF, et al. Cataract-free interval and severity of cataract after total body irradiation and bone marrow transplantation: influence of treatment parameters. Int J Radiat Oncol Biol Phys. 2000;48:807–15.
Najima Y, Kakihana K, Ohashi K, Yamamoto N, Kobayashi T, Yamashita T, et al. Incidence, risk factors, and clinical outcomes of cataracts following hematopoietic stem cell transplantation. Am J Hematol. 2011;86:508–10.
Cumming RG, Mitchell P, Leeder SR. Use of inhaled corticosteroids and the risk of cataracts. N Engl J Med. 1997;337:8–14.
Bernard F, Auquier P, Herrmann I, Contet A, Poiree M, Demeocq F, et al. Health status of childhood leukemia survivors who received hematopoietic cell transplantation after BU or TBI: an LEA study. Bone Marrow Transplant. 2014;49:709–16.
Tear Fahnehjelm K, Tornquist AL, Olsson M, Backstrom I, Andersson Gronlund M, Winiarski J. Cataract after allogeneic hematopoietic stem cell transplantation in childhood. Acta Paediatr. 2016;105:82–89.
de Melo Franco R, Kron-Gray MM, De la Parra-Colin P, He Y, Musch DC, Mian SI, et al. Outcomes of cataract surgery in graft-versus-host disease. Cornea. 2015;34:506–11.
Tomblyn M, Chiller T, Einsele H, Gress R, Sepkowitz K, Storek J, et al. Guidelines for preventing infectious complications among hematopoietic cell transplantation recipients: a global perspective. Biol Blood Marrow Transplant. 2009;15:1143–238.
Ullmann AJ, Schmidt-Hieber M, Bertz H, Heinz WJ, Kiehl M, Kruger W, et al. Infectious diseases in allogeneic haematopoietic stem cell transplantation: prevention and prophylaxis strategy guidelines 2016. Ann Hematol. 2016;95:1435–55.
Tabbara KF, Al-Ghamdi A, Al-Mohareb F, Ayas M, Chaudhri N, Al-Sharif F, et al. Ocular findings after allogeneic hematopoietic stem cell transplantation. Ophthalmology. 2009;116:1624–9.
Coskuncan NM, Jabs DA, Dunn JP, Haller JA, Green WR, Vogelsang GB, et al. The eye in bone marrow transplantation. VI. Retinal complications. Arch Ophthalmol. 1994;112:372–9.
Sumi M, Aosai F, Norose K, Takeda W, Kirihara T, Sato K, et al. Acute exacerbation of Toxoplasma gondii infection after hematopoietic stem cell transplantation: five case reports among 279 recipients. Int J Hematol. 2013;98:214–22.
Decembrino N, Comelli A, Genco F, Vitullo A, Recupero S, Zecca M, et al. Toxoplasmosis disease in paediatric hematopoietic stem cell transplantation: do not forget it still exists. Bone Marrow Transplant. 2017;52:1326–9.
Crippa F, Corey L, Chuang EL, Sale G, Boeckh M. Virological, clinical, and ophthalmologic features of cytomegalovirus retinitis after hematopoietic stem cell transplantation. Clin Infect Dis. 2001;32:214–9.
Jeon S, Lee WK, Lee Y, Lee DG, Lee JW. Risk factors for cytomegalovirus retinitis in patients with cytomegalovirus viremia after hematopoietic stem cell transplantation. Ophthalmology. 2012;119:1892–8.
Hiwarkar P, Gajdosova E, Qasim W, Worth A, Breuer J, Chiesa R, et al. Frequent occurrence of cytomegalovirus retinitis during immune reconstitution warrants regular ophthalmic screening in high-risk pediatric allogeneic hematopoietic stem cell transplant recipients. Clin Infect Dis. 2014;58:1700–6.
Larochelle MB, Phan R, Craddock J, Abzug MJ, Curtis D, Robinson CC, et al. Cytomegalovirus Retinitis in Pediatric Stem Cell Transplants: Report of a Recent Cluster and the Development of a Screening Protocol. Am J Ophthalmol. 2017;175:8–15.
Chen Y, Scieux C, Garrait V, Socie G, Rocha V, Molina JM, et al. Resistant herpes simplex virus type 1 infection: an emerging concern after allogeneic stem cell transplantation. Clin Infect Dis. 2000;31:927–35.
Koc Y, Miller KB, Schenkein DP, Griffith J, Akhtar M, DesJardin J, et al. Varicella zoster virus infections following allogeneic bone marrow transplantation: frequency, risk factors, and clinical outcome. Biol Blood Marrow Transplant. 2000;6:44–49.
Walton RC, Reed KL. Herpes zoster ophthalmicus following bone marrow transplantation in children. Bone Marrow Transplant. 1999;23:1317–20.
Castleton A, Kottaridis PD. A case of ‘red eye’ post allogeneic stem cell transplantation. Bone Marrow Transplant. 2007;39:241–2.
Donahue SP, Greven CM, Zuravleff JJ, Eller AW, Nguyen MH, Peacock JE Jr., et al. Intraocular candidiasis in patients with candidemia. Clin Implic Deriv a Prospect Multicent Study Ophthalmol. 1994;101:1302–9.
Shah VM, Tandon R, Satpathy G, Nayak N, Chawla B, Agarwal T, et al. Randomized clinical study for comparative evaluation of fourth-generation fluoroquinolones with the combination of fortified antibiotics in the treatment of bacterial corneal ulcers. Cornea. 2010;29:751–7.
Kramer M, Kramer MR, Blau H, Bishara J, Axer-Siegel R, Weinberger D. Intravitreal voriconazole for the treatment of endogenous Aspergillus endophthalmitis. Ophthalmology. 2006;113:1184–6.
Prajna NV, Krishnan T, Mascarenhas J, Rajaraman R, Prajna L, Srinivasan M, et al. The mycotic ulcer treatment trial: a randomized trial comparing natamycin vs voriconazole. JAMA Ophthalmol. 2013;131:422–9.
Martino R, Bretagne S, Einsele H, Maertens J, Ullmann AJ, Parody R, et al. Early detection of Toxoplasma infection by molecular monitoring of Toxoplasma gondii in peripheral blood samples after allogeneic stem cell transplantation. Clin Infect Dis. 2005;40:67–78.
Bosch-Driessen LH, Verbraak FD, Suttorp-Schulten MS, van Ruyven RL, Klok AM, Hoyng CB, et al. A prospective, randomized trial of pyrimethamine and azithromycin vs pyrimethamine and sulfadiazine for the treatment of ocular toxoplasmosis. Am J Ophthalmol. 2002;134:34–40.
Leen AM, Bollard CM, Mendizabal AM, Shpall EJ, Szabolcs P, Antin JH, et al. Multicenter study of banked third-party virus-specific T cells to treat severe viral infections after hematopoietic stem cell transplantation. Blood. 2013;121:5113–23.
Saral R, Burns WH, Laskin OL, Santos GW, Lietman PS. Acyclovir prophylaxis of herpes-simplex-virus infections. N Engl J Med. 1981;305:63–67.
Matthes-Martin S, Feuchtinger T, Shaw PJ, Engelhard D, Hirsch HH, Cordonnier C, et al. European guidelines for diagnosis and treatment of adenovirus infection in leukemia and stem cell transplantation: summary of ECIL-4. Transpl Infect Dis. 2011;2012:555–63.
Green W, Rao PK, Harocopos GJ. Extramedullary relapse of acute myelogenous leukemia presenting as a large serous retinal detachment. Ocul Oncol Pathol. 2017;3:95–100.
Matsuo T, Tanaka T, Ichimura K, Meguri Y. Intraocular relapse with hypopyon and retinal infiltrates after chemotherapy and peripheral blood stem cell transplantation for extranodal NK/T-cell lymphoma. J Clin Exp Hematop. 2015;55:157–61.
Rothova A, Ooijman F, Kerkhoff F, Van Der Lelij A, Lokhorst HM. Uveitis masquerade syndromes. Ophthalmology. 2001;108:386–99.
Cohen VM, Dinakaran S, Parsons MA, Rennie IG. Transvitreal fine needle aspiration biopsy: the influence of intraocular lesion size on diagnostic biopsy result. Eye. 2001;15:143–7.
Hormigo A, Abrey L, Heinemann MH, DeAngelis LM. Ocular presentation of primary central nervous system lymphoma: diagnosis and treatment. Br J Haematol. 2004;126:202–8.
Smith JR, Rosenbaum JT, Wilson DJ, Doolittle ND, Siegal T, Neuwelt EA, et al. Role of intravitreal methotrexate in the management of primary central nervous system lymphoma with ocular involvement. Ophthalmology. 2002;109:1709–16.
Wiegel T, Bottke D, Kreusel KM, Schmidt S, Bornfeld N, Foerster MH, et al. External beam radiotherapy of choroidal metastases--final results of a prospective study of the German Cancer Society (ARO 95-08). Radiother Oncol. 2002;64:13–18.
Kaliki S, Shields CL, Al-Dahmash SA, Mashayekhi A, Shields JA. Photodynamic therapy for choroidal metastasis in 8 cases. Ophthalmology. 2012;119:1218–22.
Gratwohl A, Gloor B, Hahn H, Speck B. Retinal cotton-wool patches in bone-marrow-transplant recipients. N Engl J Med. 1983;308:1101.
Hirst LW, Jabs DA, Tutschka PJ, Green WR, Santos GW. The eye in bone marrow transplantation. I. Clinical study. Arch Ophthalmol. 1983;101:580–4.
Bernauer W, Gratwohl A, Keller A, Daicker B. Microvasculopathy in the ocular fundus after bone marrow transplantation. Ann Intern Med. 1991;115:925–30.
Johnson DW, Cagnoni PJ, Schossau TM, Stemmer SM, Grayeb DE, Baron AE, et al. Optic disc and retinal microvasculopathy after high-dose chemotherapy and autologous hematopoietic progenitor cell support. Bone Marrow Transplant. 1999;24:785–92.
Bylsma GW, Hall AJ, Szer J, West R. Atypical retinal microvasculopathy after bone marrow transplantation. Clin Exp Ophthalmol. 2001;29:225–9.
Westeneng AC, Hettinga Y, Lokhorst H, Verdonck L, van Dorp S, Rothova A. Ocular graft-versus-host disease after allogeneic stem cell transplantation. Cornea. 2010;29:758–63.
Ivanir Y, Shimoni A, Ezra-Nimni O, Barequet IS. Prevalence of dry eye syndrome after allogeneic hematopoietic stem cell transplantation. Cornea. 2013;32:e97–101.
Aissopou EK, Papathanassiou M, Nasothimiou EG, Konstantonis GD, Tentolouris N, Theodossiadis PG, et al. The Keith-Wagener-Barker and Mitchell-Wong grading systems for hypertensive retinopathy: association with target organ damage in individuals below 55 years. J Hypertens. 2015;33:2303–9.
Kaushik M, Pulido JS, Schild SE, Stafford S. Risk of radiation retinopathy in patients with orbital and ocular lymphoma. Int J Radiat Oncol Biol Phys. 2012;84:1145–50.
Cooper AE, Cho JH, Menges S, Masood S, Xie J, Yang J, et al. Immunosuppressive treatment can alter visual performance in the Royal College of surgeons rat. J Ocul Pharmacol Ther. 2016;32:296–303.
Avery R, Jabs DA, Wingard JR, Vogelsang G, Saral R, Santos G. Optic disc edema after bone marrow transplantation. Possib- role cyclosporine Toxic Ophthalmol. 1991;98:1294–301.
Lopez PF, Sternberg P Jr., Dabbs CK, Vogler WR, Crocker I, Kalin NS. Bone marrow transplant retinopathy. Am J Ophthalmol. 1991;112:635–46.
Pahl DA, Green NS, Bhatia M, Lee MT, Chang JS, Licursi M, et al. Optical coherence tomography angiography and ultra-widefield fluorescein angiography for early detection of adolescent sickle retinopathy. Am J Ophthalmol. 2017;183:91–98.
Joltikov KA, de Castro VM, Davila JR, Anand R, Khan SM, Farbman N, et al. Multidimensional functional and structural evaluation reveals neuroretinal impairment in early diabetic retinopathy. Invest Ophthalmol Vis Sci. 2017;58:BIO277–BIO290.
Hillier RJ, Ojaimi E, Wong DT, Mak MYK, Berger AR, Kohly RP, et al. Aqueous humor cytokine levels and anatomic response to intravitreal Ranibizumab in diabetic macular edema. JAMA Ophthalmol. 2018;136:382–8.
Baker KS, Ness KK, Weisdorf D, Francisco L, Sun CL, Forman S, et al. Late effects in survivors of acute leukemia treated with hematopoietic cell transplantation: a report from the Bone Marrow Transplant Survivor Study. Leukemia. 2010;24:2039–47.
Saboo US, Amparo F, Shikari H, Dana R. Prevalence of ocular hypertension and glaucoma in patients with chronic ocular graft-versus-host disease. Graefes Arch Clin Exp Ophthalmol. 2016;254:923–8.
Takeda A, Shigematsu N, Suzuki S, Fujii M, Kawata T, Kawaguchi O, et al. Late retinal complications of radiation therapy for nasal and paranasal malignancies: relationship between irradiated-dose area and severity. Int J Radiat Oncol Biol Phys. 1999;44:599–605.
Tatematsu Y, Ogawa Y, Abe T, Kamoi M, Uchino M, Saijo-Ban Y, et al. Grading criteria for chronic ocular graft-versus-host disease: comparing the NIH eye score, Japanese dry eye score, and DEWS 2007 score. Sci Rep. 2014;4:6680.
Tripathi RC, Parapuram SK, Tripathi BJ, Zhong Y, Chalam KV. Corticosteroids and glaucoma risk. Drugs Aging. 1999;15:439–50.
Kwok AK, Lam DS, Ng JS, Fan DS, Chew SJ, Tso MO. Ocular-hypertensive response to topical steroids in children. Ophthalmology. 1997;104:2112–6.
Katz G, Dubiner H, Samples J, Vold S, Sall K. Three-month randomized trial of fixed-combination brinzolamide, 1%, and brimonidine, 0.2%. JAMA Ophthalmol. 2013;131:724–30.
Krupin T, Liebmann JM, Greenfield DS, Ritch R, Gardiner S. Low-Pressure Glaucoma Study G. A randomized trial of brimonidine versus timolol in preserving visual function: results from the Low-Pressure Glaucoma Treatment Study. Am J Ophthalmol. 2011;151:671–81.
Im L, Allingham RR, Singh I, Stinnett S, Fekrat S. A prospective study of early intraocular pressure changes after a single intravitreal triamcinolone injection. J Glaucoma. 2008;17:128–32.
Rubin B, Taglienti A, Rothman RF, Marcus CH, Serle JB. The effect of selective laser trabeculoplasty on intraocular pressure in patients with intravitreal steroid-induced elevated intraocular pressure. J Glaucoma. 2008;17:287–92.
Zhou JQ, Xu L, Wang S, Wang YX, You QS, Tu Y, et al. The 10-year incidence and risk factors of retinal vein occlusion: the Beijing eye study. Ophthalmology. 2013;120:803–8.
Yoo YS, Na KS, Shin JA, Park YH, Lee JW. Posterior eye segment complications related to allogeneic hematopoietic stem cell transplantation. Retina. 2017;37:135–43.
Campa C, Alivernini G, Bolletta E, Parodi MB, Perri P. Anti-VEGF Therapy for Retinal Vein Occlusions. Curr Drug Targets. 2016;17:328–36.
Epstein DL, Algvere PV, von Wendt G, Seregard S, Kvanta A. Benefit from bevacizumab for macular edema in central retinal vein occlusion: twelve-month results of a prospective, randomized study. Ophthalmology. 2012;119:2587–91.
Heier JS, Campochiaro PA, Yau L, Li Z, Saroj N, Rubio RG, et al. Ranibizumab for macular edema due to retinal vein occlusions: long-term follow-up in the HORIZON trial. Ophthalmology. 2012;119:802–9.
Haller JA, Bandello F, Belfort R Jr., Blumenkranz MS, Gillies M, Heier J, et al. Dexamethasone intravitreal implant in patients with macular edema related to branch or central retinal vein occlusion twelve-month study results. Ophthalmology. 2011;118:2453–60.
Pikkel YY, Sharabi-Nov A, Beiran I, Pikkel J. Comparison of anti-vascular endothelial growth factors, laser treatments and a combination of the both for treatment of central retinal vein occlusion. Int J Ophthalmol. 2016;9:431–3.
Eid AJ, Bakri SJ, Kijpittayarit S, Razonable RR. Clinical features and outcomes of cytomegalovirus retinitis after transplantation. Transpl Infect Dis. 2008;10:13–18.
Peng KL, Chen SJ, Lin PY, Hsu WM, Yang MH, Tzeng CH, et al. Exudative bullous retinal detachment after peripheral blood stem cell transplantation. Eye. 2005;19:603–6.
Wiznia RA, Rose A, Levy AL. Occlusive microvascular retinopathy with optic disc and retinal neovascularization in acute lymphocytic leukemia. Retina. 1994;14:253–5.
Alloin AL, Barlogis V, Auquier P, Contet A, Poiree M, Demeocq F, et al. Prevalence and risk factors of cataract after chemotherapy with or without central nervous system irradiation for childhood acute lymphoblastic leukaemia: an LEA study. Br J Haematol. 2014;164:94–100.
Abud TB, Amparo F, Saboo US, Di Zazzo A, Dohlman TH, Ciolino JB, et al. A clinical trial comparing the safety and efficacy of topical tacrolimus versus methylprednisolone in ocular graft-versus-host disease. Ophthalmology. 2016;123:1449–57.
Dastjerdi MH, Hamrah P, Dana R. High-frequency topical cyclosporine 0.05% in the treatment of severe dry eye refractory to twice-daily regimen. Cornea. 2009;28:1091–6.
Dietrich-Ntoukas T, Cursiefen C, Westekemper H, Eberwein P, Reinhard T, Bertz, et al. Diagnosis and treatment of ocular chronic graft-versus-host disease: report from the German-Austrian-Swiss Consensus Conference on Clinical Practice in chronic GVHD. Cornea. 2012;31:299–310.
Boucher HW, Groll AH, Chiou CC, Walsh TJ. Newer systemic antifungal agents: pharmacokinetics, safety and efficacy. Drugs. 2004;64:1997–2020.
Slattery A, Liebelt E, Gaines LA. Common ocular effects reported to a poison control center after systemic absorption of drugs in therapeutic and toxic doses. Curr Opin Ophthalmol. 2014;25:519–23.
Santaella RM, Fraunfelder FW. Ocular adverse effects associated with systemic medications: recognition and management. Drugs. 2007;67:75–93.
Jaanus SD. Ocular side effects of selected systemic drugs. Optom Clin. 1992;2:73–96.
Richa S, Yazbek JC. Ocular adverse effects of common psychotropic agents: a review. CNS Drugs. 2010;24:501–26.
Li J, Tripathi RC, Tripathi BJ. Drug-induced ocular disorders. Drug Saf. 2008;31:127–41.
Laties AM, Fraunfelder FT. Ocular safety of Viagra, (sildenafil citrate). Trans Am Ophthalmol Soc. 1999;97:115–25. discussion125-118
Gaynes BI, Fiscella R. Topical nonsteroidal anti-inflammatory drugs for ophthalmic use: a safety review. Drug Saf. 2002;25:233–50.
Mynarek M, Ganzenmueller T, Mueller-Heine A, Mielke C, Gonnermann A, Beier R, et al. Patient, virus, and treatment-related risk factors in pediatric adenovirus infection after stem cell transplantation: results of a routine monitoring program. Biol Blood Marrow Transplant. 2014;20:250–6.
Lass JH, Lazarus HM, Reed MD, Herzig RH. Topical corticosteroid therapy for corneal toxicity from systemically administered cytarabine. Am J Ophthalmol. 1982;94:617–21.
Fraunfelder FW, Solomon J, Druker BJ, Esmaeli B, Kuyl J. Ocular side-effects associated with imatinib mesylate (Gleevec). J Ocul Pharmacol Ther. 2003;19:371–5.
Acknowledgements
The CIBMTR is supported primarily by Public Health Service Grant/Cooperative Agreement 5U24CA076518 from the National Cancer Institute (NCI), the National Heart, Lung and Blood Institute (NHLBI) and the National Institute of Allergy and Infectious Diseases (NIAID); a Grant/Cooperative Agreement 4U10HL069294 from NHLBI and NCI; a contract HHSH250201200016C with Health Resources and Services Administration (HRSA/DHHS); two Grants N00014-17-1-2388 and N0014-17-1-2850 from the Office of Naval Research; and grants from *Actinium Pharmaceuticals, Inc.; *Amgen, Inc.; *Amneal Biosciences; *Angiocrine Bioscience, Inc.; Anonymous donation to the Medical College of Wisconsin; Astellas Pharma US; Atara Biotherapeutics, Inc.; Be the Match Foundation; *bluebird bio, Inc.; *Bristol Myers Squibb Oncology; *Celgene Corporation; Cerus Corporation; *Chimerix, Inc.; Fred Hutchinson Cancer Research Center; Gamida Cell Ltd.; Gilead Sciences, Inc.; HistoGenetics, Inc.; Immucor; *Incyte Corporation; Janssen Scientific Affairs, LLC; *Jazz Pharmaceuticals, Inc.; Juno Therapeutics; Karyopharm Therapeutics, Inc.; Kite Pharma, Inc.; Medac, GmbH; MedImmune; The Medical College of Wisconsin; *Mediware; *Merck & Co, Inc.; *Mesoblast; MesoScale Diagnostics, Inc.; Millennium, the Takeda Oncology Co.; *Miltenyi Biotec, Inc.; National Marrow Donor Program; *Neovii Biotech NA, Inc.; Novartis Pharmaceuticals Corporation; Otsuka Pharmaceutical Co, Ltd. – Japan; PCORI; *Pfizer, Inc; *Pharmacyclics, LLC; PIRCHE AG; *Sanofi Genzyme; *Seattle Genetics; Shire; Spectrum Pharmaceuticals, Inc.; St. Baldrick’s Foundation; *Sunesis Pharmaceuticals, Inc.; Swedish Orphan Biovitrum, Inc.; Takeda Oncology; Telomere Diagnostics, Inc.; and University of Minnesota. The views expressed in this article do not reflect the official policy or position of the National Institute of Health, the Department of the Navy, the Department of Defense, Health Resources and Services Administration (HRSA) or any other agency of the U.S. Government. *Corporate Members
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Inamoto, Y., Petriček, I., Burns, L. et al. Non-GVHD ocular complications after hematopoietic cell transplantation: expert review from the Late Effects and Quality of Life Working Committee of the CIBMTR and Transplant Complications Working Party of the EBMT. Bone Marrow Transplant 54, 648–661 (2019). https://doi.org/10.1038/s41409-018-0339-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-018-0339-6
This article is cited by
-
International recommendations for screening and preventative practices for long-term survivors of transplantation and cellular therapy: a 2023 update
Bone Marrow Transplantation (2024)
-
Okuläre Graft-Versus-Host Erkrankung
Die Ophthalmologie (2023)
-
Makulopathie bei Sichelzellerkrankung
Der Ophthalmologe (2021)
-
Ocular surface system alterations in ocular graft-versus-host disease: all the pieces of the complex puzzle
Graefe's Archive for Clinical and Experimental Ophthalmology (2019)