Abstract
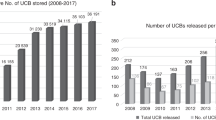
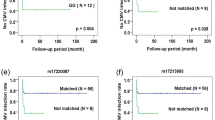
The possibility to use CCR5-∆32 umbilical cord blood to cure HIV infection in patients in need of a hematopoietic transplant has been suggested. The less stringent HLA compatibility needed in this type of transplant facilitates the search of a suitable donor having the CCR5-∆32 mutation. To achieve an inventory of CCR5-∆32 cord blood units, the 20,236 best cell quality units of the Spanish Registry were genotyped. Furthermore, their CD34+ and total nucleated cells counts, blood type, gender, HLA and donor’s geographical and ancestral origin were analyzed. The results showed 130 (0.64%) units homozygous for the deletion, 2,646 (13.08%) heterozygous and 17,460 (86.28%) did not present the mutation. Interestingly, a significant lower amount of CD34+ cells was found in the CCR5-∆32 homozygous units. In addition, a significant association was found among donor’s ancestral origin and the mutation, with a higher percentage of CCR5-∆32 units with a European ancestry. In summary, identification of a relatively high number of CCR5-∆32 units is feasible and will facilitate the development of clinical trials for HIV cure in patients requiring hematopoietic transplantation. Further studies are required to understand the significance of lower cell counts within the CCR5-∆32 homozygous group and its clinical impact.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Deng H, Liu R, Ellmeier W, Choe S, Unutmaz D, Burkhart M, et al. Identification of a major co-receptor for primary isolates of HIV-1. Nature. 1996;381:661–6. https://doi.org/10.1038/381661a0
Liu R, Paxton WA, Choe S, Ceradini D, Martin SR, Horuk R, et al. Homozygous defect in HIV-1 coreceptor accounts for resistance of some multiply-exposed individuals to HIV-1 infection. Cell. 1996;86:367–77.
Lucotte G. Frequencies of 32 base pair deletion of the (Delta 32) allele of the CCR5 HIV-1 co-receptor gene in Caucasians: a comparative analysis. Infect, Genet Evol: J Mol Epidemiol Evolut Genet Infect Dis. 2002;1:201–5.
Martinson JJ, Chapman NH, Rees DC, Liu YT, Clegg JB. Global distribution of the CCR5 gene 32-basepair deletion. Nat Genet. 1997;16:100–3. https://doi.org/10.1038/ng0597-100
Gonzalez G, Park S, Chen D, Armitage S, Shpall E, Behringer R. Identification and frequency of CCR5Delta32/Delta32 HIV-resistant cord blood units from Houston area hospitals. HIV Med. 2011;12:481–6. https://doi.org/10.1111/j.1468-1293.2010.00911.x
Dean M, Carrington M, Winkler C, Huttley GA, Smith MW, Allikmets R, et al. Genetic restriction of HIV-1 infection and progression to AIDS by a deletion allele of the CKR5 structural gene. Hemophilia Growth and Development Study, Multicenter AIDS Cohort Study, Multicenter Hemophilia Cohort Study, San Francisco City Cohort, ALIVE Study. Science. 1996;273:1856–62.
Mulherin SA, O’Brien TR, Ioannidis JP, Goedert JJ, Buchbinder SP, Coutinho RA, et al. Effects of CCR5-Delta32 and CCR2-64I alleles on HIV-1 disease progression: the protection varies with duration of infection. Aids. 2003;17:377–87. https://doi.org/10.1097/01.aids.0000050783.28043.3e
Samson M, Libert F, Doranz BJ, Rucker J, Liesnard C, Farber CM, et al. Resistance to HIV-1 infection in caucasian individuals bearing mutant alleles of the CCR-5 chemokine receptor gene. Nature. 1996;382:722–5. https://doi.org/10.1038/382722a0
Hutter G, Nowak D, Mossner M, Ganepola S, Mussig A, Allers K, et al. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. N Engl J Med. 2009;360:692–8. https://doi.org/10.1056/NEJMoa0802905
Allers K, Hutter G, Hofmann J, Loddenkemper C, Rieger K, Thiel E, et al. Evidence for the cure of HIV infection by CCR5Delta32/Delta32 stem cell transplantation. Blood. 2011;117:2791–9. https://doi.org/10.1182/blood-2010-09-309591
Hutter G, Thiel E. Allogeneic transplantation of CCR5-deficient progenitor cells in a patient with HIV infection: an update after 3 years and the search for patient no. 2. Aids. 2011;25:273–4. https://doi.org/10.1097/QAD.0b013e328340fe28
Hutter G. HIV+ patients and HIV eradication - allogeneic transplantation. Expert Rev Hematol. 2016;9:615–6. https://doi.org/10.1080/17474086.2016.1183478
Petz L. Cord blood transplantation for cure of HIV infections. Stem Cells Transl Med. 2013;2:635–7. https://doi.org/10.5966/sctm.2012-0089
Smith AR, Wagner JE. Alternative haematopoietic stem cell sources for transplantation: place of umbilical cord blood. Br J Haematol. 2009;147:246–61. https://doi.org/10.1111/j.1365-2141.2009.07828.x
Petz LD, Redei I, Bryson Y, Regan D, Kurtzberg J, Shpall E, et al. Hematopoietic cell transplantation with cord blood for cure of HIV infections. Biol Blood Marrow Transplant. 2013;19:393–7. https://doi.org/10.1016/j.bbmt.2012.10.017
Gonzalez G, Shpall EJ, Gathe JC, Behringer RR. Cord blood stem cell therapy for acquired immune deficiency syndrome. Stem Cells Dev. 2009;18:1–2. https://doi.org/10.1089/scd.2008.0304
Duarte RF, Salgado M, Sanchez-Ortega I, Arnan M, Canals C, Domingo-Domenech E, et al. CCR5 Delta32 homozygous cord blood allogeneic transplantation in a patient with HIV: a case report. Lancet HIV. 2015;2:e236–242. https://doi.org/10.1016/S2352-3018(15)00083-1
Petz LD, Burnett JC, Li H, Li S, Tonai R, Bakalinskaya M, et al. Progress toward curing HIV infection with hematopoietic cell transplantation. Stem Cells Cloning: Adv Appl. 2015;8:109–16. https://doi.org/10.2147/SCCAA.S56050
Bachelerie F, Ben-Baruch A, Burkhardt AM, Combadiere C, Farber JM, Graham GJ, et al. International Union of Basic and Clinical Pharmacology. [corrected]. LXXXIX. Update on the extended family of chemokine receptors and introducing a new nomenclature for atypical chemokine receptors. Pharmacol Rev. 2014;66:1–79. https://doi.org/10.1124/pr.113.007724
Ruiz ME, Cicala C, Arthos J, Kinter A, Catanzaro AT, Adelsberger J, et al. Peripheral blood-derived CD34+ progenitor cells: CXC chemokine receptor 4 and CC chemokine receptor 5 expression and infection by HIV. J Immunol. 1998;161:4169–76.
Clements JM, Craig S, Gearing AJ, Hunter MG, Heyworth CM, Dexter TM, et al. Biological and structural properties of MIP-1 alpha expressed in yeast. Cytokine. 1992;4:76–82.
Mayani H, Little MT, Dragowska W, Thornbury G, Lansdorp PM. Differential effects of the hematopoietic inhibitors MIP-1 alpha, TGF-beta, and TNF-alpha on cytokine-induced proliferation of subpopulations of CD34+ cells purified from cord blood and fetal liver. Exp Hematol. 1995;23:422–7.
de Wynter EA, Durig J, Cross MA, Heyworth CM, Testa NG. Differential response of CD34+ cells isolated from cord blood and bone marrow to MIP-1 alpha and the expression of MIP-1 alpha receptors on these immature cells. Stem Cells. 1998;16:349–56. https://doi.org/10.1002/stem.160349
Capmany G, Querol S, Cancelas JA, Garcia J. Short-term, serum-free, static culture of cord blood-derived CD34+ cells: effects of FLT3-L and MIP-1alpha on in vitro expansion of hematopoietic progenitor cells. Haematologica. 1999;84:675–82.
Ghorban K, Dadmanesh M, Hassanshahi G, Momeni M, Zare-Bidaki M, Arababadi MK, et al. Is the CCR5 Delta 32 mutation associated with immune system-related diseases? Inflammation. 2013;36:633–42. https://doi.org/10.1007/s10753-012-9585-8
Slominski B, Lawrynowicz U, Mysliwska J, Ryba-Stanislawowska M, Skrzypkowska M, Brandt A. CCR5-Delta32 gene polymorphism is associated with retinopathy in patients with type 1 diabetes. Mol Cell Endocrinol. 2017;439:256–60. https://doi.org/10.1016/j.mce.2016.09.009
Ungvari I, Tolgyesi G, Semsei AF, Nagy A, Radosits K, Keszei M, et al. CCR5 Delta 32 mutation, mycoplasma pneumoniae infection, and asthma. J Allergy Clin Immunol. 2007;119:1545–7. https://doi.org/10.1016/j.jaci.2007.02.038
MacDonald KS, Fowke KR, Kimani J, Dunand VA, Nagelkerke NJ, Ball TB, et al. Influence of HLA supertypes on susceptibility and resistance to human immunodeficiency virus type 1 infection. J Infect Dis. 2000;181:1581–9. https://doi.org/10.1086/315472
Balas A, Garcia-Sanchez F, Vicario JL. Allelic and haplotypic HLA frequency distribution in Spanish hematopoietic patients. Implications for unrelated donor searching. Tissue Antigens. 2011;77:45–53. https://doi.org/10.1111/j.1399-0039.2010.01578.x
Acknowledgements
The authors would like to thank the collaboration from the following institutions and colleagues: Anthony Nolan Research Institute (Alejandro Madrigal), Banco de Cordón del País Vasco (Clara Rodriguez-Ayerbe, Silvia Santos), Banco de Cordón de Valencia (María D. Planelles), Banc de Sang i Teixits de Catalunya (Nina Borràs, José L. Caro, Nerea Castillo, María Piron, Sílvia Saludeda, Mar Sánchez, Laura Mongay), Centro de Transfusión de la Comunidad de Madrid (José L. Vicario), IrsiCaixa (Javier Martínez-Picado). The statistical analysis was performed by the Unidad de Estadística y Bioinformática (UEB) of the Vall d’Hebron Hospital Research Institute (VHIR).
Funding
This study was funded by a grant from the Spanish Organización Nacional de Trasplante.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Enrich, E., Vidal, F., Sánchez-Gordo, F. et al. Analysis of the Spanish CCR5-∆32 inventory of cord blood units: lower cell counts in homozygous donors. Bone Marrow Transplant 53, 741–748 (2018). https://doi.org/10.1038/s41409-018-0114-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-018-0114-8