Abstract
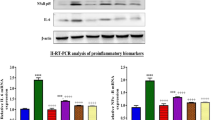
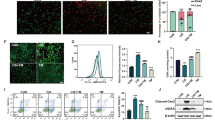
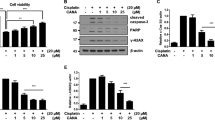
Acute kidney injury (AKI) caused by anti-tumor drugs, such as cisplatin, is a severe complication with no effective treatment currently, leading to the reduction or discontinuation of chemotherapy. Natural products or herbal medicines are gradually considered as promising agents against cisplatin-induced AKI with the advantages of multi-targeting, multi-effects, and less resistance. In this study, we investigated the effects of kaempferide, a natural flavonoid extracted from the rhizome of Kaempferia galanga, in experimental AKI models in vitro and in vivo. We first conducted pharmacokinetic study in mice and found a relative stable state of kaempferide with a small amount of conversion into kaempferol. We showed that both kaempferide (10 μM) and kaempferol (10 μM) significantly inhibited cisplatin-caused injuries in immortalized proximal tubule epithelial cell line HK-2. In AKI mice induced by injection of a single dose of cisplatin (15 mg/kg), oral administration of kaempferide (50 mg/kg) either before or after cisplatin injection markedly improved renal function, and ameliorated renal tissue damage. We demonstrated that kaempferide inhibited oxidative stress and induced autophagy in cisplatin-treated mice and HK-2 cells, thus increasing tubular cell viability and decreasing immune responses to attenuate the disease progression. In addition, treatment with kaempferide significantly ameliorated ischemia-reperfusion-induced renal injury in vitro and in vivo. We conclude that kaempferide is a promising natural product for treating various AKI. This study has great implications for promotion of its use in healthcare products, and help to break through the limited use of cisplatin in the clinic.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Dasari S, Tchounwou PB. Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharmacol. 2014;740:364–78.
Stehlik J, Armstrong B, Baran DA, Bridges ND, Chandraker A, Gordon R, et al. Early immune biomarkers and intermediate-term outcomes after heart transplantation: Results of Clinical Trials in Organ Transplantation-18. Am J Transpl. 2019;19:1518–28.
Pierson Marchandise M, Gras V, Moragny J, Micallef J, Gaboriau L, Picard S, et al. The drugs that mostly frequently induce acute kidney injury: a case - noncase study of a pharmacovigilance database. Br J Clin Pharmacol. 2017;83:1341–9.
Beyer J, Rick O, Weinknecht S, Kingreen D, Lenz K, Siegert W. Nephrotoxicity after high-dose carboplatin, etoposide and ifosfamide in germ-cell tumors: incidence and implications for hematologic recovery and clinical outcome. Bone Marrow Transpl. 1997;20:813–9.
Winston JA, Safirstein R. Reduced renal blood flow in early cisplatin-induced acute renal failure in the rat. Am J Physiol. 1985;249:F490–6.
Bonventre JV, Zuk A. Ischemic acute renal failure: an inflammatory disease? Kidney Int. 2004;66:480–5.
Yang YW, Yu XW, Zhang Y, Ding GX, Zhu CH, Huang SM, et al. Hypoxia-inducible factor prolyl hydroxylase inhibitor roxadustat (FG-4592) protects against cisplatin-induced acute kidney injury. Clin Sci. 2018;132:825–38.
Prasad SB, Rosangkima G, Kharbangar A. Structural and biochemical changes in mitochondria after cisplatin treatment of Dalton’s lymphoma-bearing mice. Mitochondrion. 2010;10:38–45.
Salei N, Rambichler S, Salvermoser J, Papaioannou NE, Schuchert R, Pakalniskyte D, et al. The kidney contains ontogenetically distinct dendritic cell and macrophage subtypes throughout development that differ in their inflammatory properties. J Am Soc Nephrol. 2020;31:257–78.
Yang C, Kaushal V, Shah SV, Kaushal GP. Autophagy is associated with apoptosis in cisplatin injury to renal tubular epithelial cells. Am J Physiol Ren Physiol. 2008;294:F777–87.
Jiang M, Wei Q, Dong G, Komatsu M, Su Y, Dong Z. Autophagy in proximal tubules protects against acute kidney injury. Kidney Int. 2012;82:1271–83.
Wang Y, Tang C, Cai J, Chen G, Zhang D, Zhang Z, et al. PINK1/Parkin-mediated mitophagy is activated in cisplatin nephrotoxicity to protect against kidney injury. Cell Death Dis. 2018;9:1113.
Ridzuan NRA, Rashid NA, Othman F, Budin SB, Hussan F, Teoh SL. Protective role of natural products in cisplatin-induced nephrotoxicity. Mini Rev Med Chem. 2019;19:1134–43.
Fang CY, Lou DY, Zhou LQ, Wang JC, Yang B, He QJ, et al. Natural products: potential treatments for cisplatin-induced nephrotoxicity. Acta Pharmacol Sin. 2021;42:1951–69.
Yue ME, Jiang TF, Shi YP. Fast determination of flavonoids in Hippophae rhamnoides and its medicinal preparation by capillary zone electrophoresis using dimethyl-beta-cyclodextrin as modifier. Talanta. 2004;62:695–9.
Zhang Q, Cui H. Simultaneous determination of quercetin, kaempferol, and isorhamnetin in phytopharmaceuticals of Hippophae rhamnoides L. by high-performance liquid chromatography with chemiluminescence detection. J Sep Sci. 2005;28:1171–8.
Pundir S, Garg P, Dviwedi A, Ali A, Kapoor VK, Kapoor D, et al. Ethnomedicinal uses, phytochemistry and dermatological effects of Hippophae rhamnoides L.: a review. J Ethnopharmacol. 2021;266:113434.
Yamaga M, Tani H, Nishikawa M, Fukaya K, Ikushiro SI, Murota K. Pharmacokinetics and metabolism of cinnamic acid derivatives and flavonoids after oral administration of Brazilian green propolis in humans. Food Funct. 2021;12:2520–30.
Otake Y, Walle T. Oxidation of the flavonoids galangin and kaempferide by human liver microsomes and CYP1A1, CYP1A2, and CYP2C9. Drug Metab Dispos. 2002;30:103–5.
Jiang ZT, Wang JC, Chen XF, Wang X, Wang TF, Zhu ZT, et al. Simultaneous determination of kaempferide, kaempferol and isorhamnetin in rat plasma by ultra-high performance liquid chromatography-tandem mass spectrometry and its application to a pharmacokinetic study. J Braz Chem Soc. 2018;29:535–42.
Tie F, Ding J, Hu N, Dong Q, Chen Z, Wang H. Kaempferol and kaempferide attenuate oleic acid-induced lipid accumulation and oxidative stress in HepG2 cells. Int J Mol Sci. 2021;22:8847.
Srinivasan E, Rajasekaran R. Comparative binding of kaempferol and kaempferide on inhibiting the aggregate formation of mutant (G85R) SOD1 protein in familial amyotrophic lateral sclerosis: a quantum chemical and molecular mechanics study. Biofactors. 2018;44:431–42.
Weng Q, Sun H, Fang C, Xia F, Liao H, Lee J, et al. Catalytic activity tunable ceria nanoparticles prevent chemotherapy-induced acute kidney injury without interference with chemotherapeutics. Nat Commun. 2021;12:1436.
Bejoy J, Qian ES, Woodard LE. Tissue culture models of AKI: from tubule cells to human kidney organoids. J Am Soc Nephrol. 2022;33:487–501.
Tanase DM, Gosav EM, Radu S, Costea CF, Ciocoiu M, Carauleanu A, et al. The predictive role of the biomarker kidney molecule-1 (KIM-1) in acute kidney injury (AKI) cisplatin-induced nephrotoxicity. Int J Mol Sci. 2019;20:5238.
Martensson J, Bellomo R. The rise and fall of NGAL in acute kidney injury. Blood Purif. 2014;37:304–10.
Niedernhofer LJ, Daniels JS, Rouzer CA, Greene RE, Marnett LJ. Malondialdehyde, a product of lipid peroxidation, is mutagenic in human cells. J Biol Chem. 2003;278:31426–33.
Kim D, Hwang HY, Kwon HJ. A natural small molecule induces MAPT clearance via mTOR-independent autophagy. Biochem Biophys Res Commun. 2021;568:30–6.
Kim D, Hwang HY, Ji ES, Kim JY, Yoo JS, Kwon HJ. Activation of mitochondrial TUFM ameliorates metabolic dysregulation through coordinating autophagy induction. Commun Biol. 2021;4:1.
Wang Y, Liu Z, Shu S, Cai J, Tang C, Dong Z. AMPK/mTOR signaling in autophagy regulation during cisplatin-induced acute kidney injury. Front Physiol. 2020;11:619730.
Zhao Y, Feng X, Li B, Sha J, Wang C, Yang T, et al. Dexmedetomidine protects against lipopolysaccharide-induced acute kidney injury by enhancing autophagy through inhibition of the PI3K/AKT/mTOR pathway. Front Pharmacol. 2020;11:128.
Zhu L, Yuan Y, Yuan L, Li L, Liu F, Liu J, et al. Activation of TFEB-mediated autophagy by trehalose attenuates mitochondrial dysfunction in cisplatin-induced acute kidney injury. Theranostics. 2020;10:5829–44.
Tang C, Livingston MJ, Liu Z, Dong Z. Autophagy in kidney homeostasis and disease. Nat Rev Nephrol. 2020;16:489–508.
Wu YT, Tan HL, Shui G, Bauvy C, Huang Q, Wenk MR, et al. Dual role of 3-methyladenine in modulation of autophagy via different temporal patterns of inhibition on class I and III phosphoinositide 3-kinase. J Biol Chem. 2010;285:10850–61.
Kanagasundaram NS. Pathophysiology of ischaemic acute kidney injury. Ann Clin Biochem. 2015;52:193–205.
Rao Y, Wan Q, Su H, Xiao X, Liao Z, Ji J, et al. ROS-induced HSP70 promotes cytoplasmic translocation of high-mobility group box 1b and stimulates antiviral autophagy in grass carp kidney cells. J Biol Chem. 2018;293:17387–401.
Su Y, Lu J, Gong P, Chen X, Liang C, Zhang J. Rapamycin induces autophagy to alleviate acute kidney injury following cerebral ischemia and reperfusion via the mTORC1/ATG13/ULK1 signaling pathway. Mol Med Rep. 2018;18:5445–54.
Yuan P, Sun X, Liu X, Hutterer G, Pummer K, Hager B, et al. Kaempferol alleviates calcium oxalate crystal-induced renal injury and crystal deposition via regulation of the AR/NOX2 signaling pathway. Phytomedicine. 2021;86:153555.
Ji X, Cao J, Zhang L, Zhang Z, Shuai W, Yin W. Kaempferol protects renal fibrosis through activating the BMP-7-Smad1/5 signaling pathway. Biol Pharm Bull. 2020;43:533–9.
Alshehri AS. Kaempferol attenuates diabetic nephropathy in streptozotocin-induced diabetic rats by a hypoglycaemic effect and concomitant activation of the Nrf-2/Ho-1/antioxidants axis. Arch Physiol Biochem. 2021:1–14. https://doi.org/10.1080/13813455.2021.1890129.
Martin MA, Ramos S. Impact of dietary flavanols on microbiota, immunity and inflammation in metabolic diseases. Nutrients. 2021;13:850.
Martins TF, Palomino OM, Alvarez-Cilleros D, Martin MA, Ramos S, Goya L. Cocoa flavanols protect human endothelial cells from oxidative stress. Plant Foods Hum Nutr. 2020;75:161–8.
Yan T, He B, Xu M, Wu B, Xiao F, Bi K, et al. Kaempferide prevents cognitive decline via attenuation of oxidative stress and enhancement of brain-derived neurotrophic factor/tropomyosin receptor kinase B/cAMP response element-binding signaling pathway. Phytother Res. 2019;33:1065–73.
Levine B, Kroemer G. Biological functions of autophagy genes: a disease perspective. Cell. 2019;176:11–42.
Peng X, Wang Y, Li H, Fan J, Shen J, Yu X, et al. ATG5-mediated autophagy suppresses NF-kappaB signaling to limit epithelial inflammatory response to kidney injury. Cell Death Dis. 2019;10:253.
Li H, Peng X, Wang Y, Cao S, Xiong L, Fan J, et al. Atg5-mediated autophagy deficiency in proximal tubules promotes cell cycle G2/M arrest and renal fibrosis. Autophagy. 2016;12:1472–86.
Filomeni G, De Zio D, Cecconi F. Oxidative stress and autophagy: the clash between damage and metabolic needs. Cell Death Differ. 2015;22:377–88.
Yun HR, Jo YH, Kim J, Shin Y, Kim SS, Choi TG. Roles of autophagy in oxidative stress. Int J Mol Sci. 2020;21:3289.
Tang H, Zeng Q, Ren N, Wei Y, He Q, Chen M, et al. Kaempferide improves oxidative stress and inflammation by inhibiting the TLR4/IkappaBalpha/NF-kappaB pathway in obese mice. Iran J Basic Med Sci. 2021;24:493–8.
Sayin VI, Ibrahim MX, Larsson E, Nilsson JA, Lindahl P, Bergo MO. Antioxidants accelerate lung cancer progression in mice. Sci Transl Med. 2014;6:221ra15.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 82003852), the Zhejiang Provincial Natural Science Foundation (No. LYY21H310010), Zhejiang Traditional Chinese Medicine Science and Technology Project (2022ZQ010), the Fundamental Research Funds for the Central Universities (No. 2022ZFJH003), and the Adjunct Talent Fund of Zhejiang Provincial People’s Hospital.
Author information
Authors and Affiliations
Contributions
QJH, JJW and YFS designed and interpreted experiments. YFS, BBT, YHD, CYF, LH performed experiments. CXS, ZXY, YPQ, JCW contributed to the data acquisition. QJH, JJW, YFS and TBB analyzed data and prepared the paper. BY and QJW critically reviewed the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shao, Yf., Tang, Bb., Ding, Yh. et al. Kaempferide ameliorates cisplatin-induced nephrotoxicity via inhibiting oxidative stress and inducing autophagy. Acta Pharmacol Sin 44, 1442–1454 (2023). https://doi.org/10.1038/s41401-023-01051-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41401-023-01051-4