Abstract
Accurate prediction of suicide risk among children and adolescents within an actionable time frame is an important but challenging task. Very few studies have comprehensively considered the clinical risk factors available to produce quantifiable risk scores for estimation of short- and long-term suicide risk for pediatric population. In this paper, we built machine learning models for predicting suicidal behavior among children and adolescents based on their longitudinal clinical records, and determining short- and long-term risk factors. This retrospective study used deidentified structured electronic health records (EHR) from the Connecticut Children’s Medical Center covering the period from 1 October 2011 to 30 September 2016. Clinical records of 41,721 young patients (10–18 years old) were included for analysis. Candidate predictors included demographics, diagnosis, laboratory tests, and medications. Different prediction windows ranging from 0 to 365 days were adopted. For each prediction window, candidate predictors were first screened by univariate statistical tests, and then a predictive model was built via a sequential forward feature selection procedure. We grouped the selected predictors and estimated their contributions to risk prediction at different prediction window lengths. The developed predictive models predicted suicidal behavior across all prediction windows with AUCs varying from 0.81 to 0.86. For all prediction windows, the models detected 53–62% of suicide-positive subjects with 90% specificity. The models performed better with shorter prediction windows and predictor importance varied across prediction windows, illustrating short- and long-term risks. Our findings demonstrated that routinely collected EHRs can be used to create accurate predictive models for suicide risk among children and adolescents.
Similar content being viewed by others
Introduction
Suicide among children and adolescents is one of the most critical public health concerns1,2,3,4,5. As the second leading cause of death among children, adolescents and young adults between ages 10–24 years, suicide claims over 6000 young lives every year in the US alone6. There has been an alarming increase in suicide rates among those 10–24 years of age7. Compared to 2000, suicide rates were 2–3 times higher in 2016 for this population8. The burden of suicide attempts is many folds higher than suicide deaths. In 2017, about 1 in 6 adolescents and young adults seriously considered attempting suicide and 1 in 13 attempted suicide9. Every year approximately 700,000 adolescents seek healthcare after an attempted suicide10.
In clinical practice, predicting the risk of suicide accurately within an actionable time frame is critical11,12,13,14. However, most of the clinical risk assessment tools available to clinicians are not sufficiently accurate to identify high-risk patients14,15,16,17. While the Columbia Suicide Severity rating scale presents an effective alternative for older clinical assessment tools, it may not be possible to screen every patient with this tool during the clinical encounter. Hence, it is evident that clinical practitioners need more than just clinical assessment tools to identify patients at risk of suicide. Recent efforts to apply machine learning with electronic health record (EHR) data to predict suicide risk in adult populations18,19,20,21,22,23 have not only confirmed the importance of prominent risk factors for suicidal behavior identified in prior research24,25,26,27 but also identified other characteristics leading to improved accuracy in suicide prediction compared to previous efforts19,28,29. However, there is scarcity of advanced risk prediction models that can be applied in clinical practice to the general population of children, adolescents, and young adults. Walsh et al.30 constructed a predictive model for adolescents who required no in-person assessment using EHR data with good prediction performance in a limited population in the Southern US. However, there is a need for a more comprehensive risk predictive models that consider a wider range of clinical factors beyond mental health comorbidities and medications and generate quantifiable risk scores that can be applied to determine the patient’s risk level for suicidal behavior.
In this retrospective study, we examined a range of demographic, diagnostic, laboratory, and medication-related factors derived from EHRs to identify significant predictors for suicidal behavior among children and adolescents receiving inpatient and outpatient care at a major children’s hospital to compare and contrast predictors across different time windows to examine the potential for variability in the factors associated with short- and longer-term risk. This is one of the few analyses in the suicide risk modeling literature to explicitly differentiate models predicting near term vs. longer term risk.
Methods
Ethics statement
Data analyzed in this study were deidentified by removing protected personal health information. Our study was approved by the University of Connecticut Health Center Institutional Review Board and Weill Cornell Medical College Institutional Review Board.
Cohort
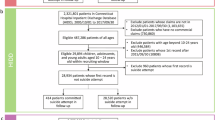
We analyzed data from the Connecticut Children’s Medical Center (CCMC) EHR database, which contains information on 641,708 visits among 129,485 patients from 1 October 2011, to 30 September 2016. We included in the analysis all encounters related to outpatient, emergency room, or inpatient care among patients between 10 and 18 years of age at the time of the first suicide diagnosis (for whom has the suicide attempts) or the last visit (for whom has no suicide attempt). Patients who have no encounter related to outpatient, emergency room, or inpatient care were considered as missing longitudinal data and hence were excluded for analysis. In addition, patients whose first recorded visits due to suicide attempts were excluded since the lack of longitudinal information. To identify suicide attempts, we used an algorithm including both the external cause of injury codes of International Classification of Diseases, Ninth Revision (ICD-9) and other ICD-9 code combinations that are indicative of suicidal behaviors (as shown in Supplementary Table 1)31,32. Since our data included diagnostic codes in ICD-10 format, we converted the ICD-9 codes from the algorithm to ICD-10, using a public toolkit, AHRQ MapIT33. Figure 1 summarizes the data screening process to define the study population and suicide-positive and -negative groups for this study.
Measures and outcomes
To gain insight on both short- and long-term suicide risk and how the risk factors evolve through different periods of observation, we developed a series of models predicting the risk of future suicide attempts with varying prediction windows. We constructed models for prediction windows, including 0, 7, 14, 30, 60, 90, 180, 270, and 365 days. The model for each prediction window only uses data equal to or more distant in time than the length of the window. For example, the model for a prediction window of 60 days aims to predict the occurrence of a future suicide attempt that happens 60 days or longer following the encounter. Consequently, the model is trained using patient’s data that are captured at least 60 days prior to their end points, where the end point for a suicide-positive subject is defined as the time of his/her first suicide attempt, and the end point of a negative subject is defined as the time of his/her last recorded clinical visit. The model trained with the 0 day prediction window is expected to predict the suicide risk any time after the latest encounter. See Supplementary Fig. 1 and Table 2 for an illustration.
We utilized a broad range of variables as candidate predictors, including patients’ demographic characteristics, diagnosis codes, prescribed medications, and laboratory test data. Demographic characteristics include age, sex, race, and ethnicity. All diagnosis information was presented in ICD-10 format. For medication data, we used a publicly available toolkit MedEx34 based on RxNorm35 to standardize the prescribed medications. Laboratory data include the name of the laboratory test and its results. The predictors were constructed as binary variables, each of which indicates the absence or presence of a particular factor related to diagnoses, medications, or laboratory tests. We also included all the two-way interactions among these binary variables as candidate predictors.
Predictor screening
Predictor screening was performed based on testing the marginal correlation of each predictor and the occurrence of suicide attempts. Specifically, each predictor and the occurrence of suicide attempts produced a 2 × 2 contingency table. If all the cells were bigger than 5, then a Chi-square test was used to test the association, else Fisher’s exact test was used. To adjust for multiple testing, we implemented p value correction using the Benjamini–Hochberg procedure36 to control the false discovery rate (FDR) at 10%. As such, all the predictors with an adjusted p value smaller than 0.1 were used as candidate predictors to build the predictive models. We also applied the marginal screening procedure on the interaction variables (see Supplementary Appendix 1 for more details).
Model development and predictor selection
For each prediction window, we randomly divided the data into 90% training and 10% testing sets. As shown in Supplementary Fig. 2, we first performed the predictor screening to select the informative candidate predictors. Next, we developed the predictive model using the training set. A logistic regression classifier was built via a sequential forward selection procedure37 for selecting predictors to minimize the prediction error. The use of the sequential forward selection procedure allows us to watch the order in which predictors are added, and hence can provide valuable information about the quality of the candidate predictors. We concluded the selection procedure when predictive performance, as judged by a fivefold cross-validation, reached the peak. We repeated all the above procedures 10 times to validate the effectiveness of our predictive model and identify the predictors with the strongest association with suicide attempts. The level of importance of each predictor was measured by its frequency of being selected among the 10 predictive models, based on which the final set of predictors was determined. Supplementary Appendix 2 provides more details about the method for constructing predictive model for each prediction window.
Model evaluation
In order to estimate the proposed predictive models, we implemented logistic regression with L1 regularization for comparisons. The L1 logistic regression models were trained based on candidate predictors passed the univariate screening. To evaluate the predictive performance of the models, we examined out-of-sample performance metrics, including area under the receiver operator characteristic curve (AUC), sensitivity, specificity, and positive predictive value (PPV). AUC is a broad metric of discrimination performance in the machine learning community that ranges from 0.5 (random guessing) to 1.0 (perfect prediction). Since our dataset is highly imbalanced, i.e., 180 suicide-positive subjects vs. 41,541 suicide-negative subjects, we calculated sensitivities when setting specificities to 90% and 95%, respectively. We also calculated PPV, which is the probability that predicted high-risk patients have actual suicide attempts. For each prediction window, performance metrics were calculated based on prediction results over the testing set.
To further evaluate the usefulness of our predictive models, we grouped the selected predictors into seven categories: demographics, depression-related factors (including both diagnoses and medications), other mental health-related factors, routine tests, drug tests, pregnancy-related factors (among females), and other factors. We implemented logistic regression analysis on female and male cohorts, respectively, to estimate the contribution coefficient for each category.
Results
Following application of our inclusion and exclusion criteria, 180 (0.43%) patients with suicide attempts were labeled as positive subjects, and 41,541 patients without suicide attempts were labeled as negative subjects. Table 1 illustrates the demographic characteristics of the study population.
Model performance
Patients with no clinical records before the prediction window were excluded for training the model. Hence as the prediction window increases, the number of patients eligible for analysis decrease (see Table 2). Though we analyzed the effects of two-way interactions among the predictors on the risk of suicidal behavior, we ultimately excluded them for building predictive models due to poor predictive performance when they were included. This was mainly due to the general sparseness of the data which was exacerbated by noise introduced by the interaction terms.
Statistics summarizing the ability of our models to predict the suicide risk, including receiver operating characteristic (ROC) curves, AUC, sensitivity at predefined specificity levels, and PPV, are presented in Table 2, and Supplementary Figs. 3 and 4. The proposed models predicted suicidal behavior with an overall AUC > 0.80 across all prediction time windows. The model performed similarly in terms of AUC for 0- to 270-day prediction windows (AUC = 0.84–0.86). The predictive performance declines for the one-year prediction window (AUC = 0.81, 95% confidence interval [CI] 0.76–0.86) since fewer patients had clinical records 1 year before the observation point. For all prediction windows, the model detected 53–62% of suicide cases with 90% specificity. Consistent with the low prevalence (from 0.43 to 0.95%) of suicidal behavior in the studied cohort, the PPVs across all prediction windows ranged from 3 to 6% for 90% specificity, and from 4 to 8% for 95% specificity (see Table 2). Overall, predictive performances of the proposed models were higher than those of the baseline L1 penalized logistic regression models (see Table 2 and Supplementary Fig. 4).
Predictor importance
Figure 2 depicts predictor importance, as measured by the frequency a specific predictor was enrolled by the sequential forward selection procedure. Characteristics of predictors are listed in Supplementary Tables 3–11. The importance of predictors varied across the prediction windows. ICD-10 code R45 (symptoms and signs involving emotional state) and F32 (depressive episode), and gender are the most common risk factors across all prediction windows. Age is also a significant factor, with patients between 10 and 12 years old less likely to attempt suicide than older patients. In addition, antidepressant medications, including sertraline and escitalopram, and urine culture tests are risk factors that show relatively high importance across most prediction windows.
Importance of predictors of which summation of frequencies over all prediction windows is no less than 0.5. Predictor importance is measured by frequency that specific predictor is enrolled by the sequential forward selection procedure. Each predictor is shown with its associated type in square brackets embedded as D demographics, I International Classification of Diseases, Tenth Revision (ICD-10) diagnostic codes, M medication, T Lab Test. An asterisk concatenating two variables indicates interaction predictor. In addition, each Lab Test is shown with its associated result in braces embedded as U unspecified, H high, A abnormal.
To aid in the interpretation of factors having different impacts across short- and longer-term time windows, we grouped the selected predictors into seven categories and calculated the contribution coefficient for each category in identifying suicide risk among both female and male patients (see Fig. 3). Details of each predictor category are listed in Supplementary Table 12. The radar charts presented in Fig. 3 show that, in general, demographics, depression-related factors, and other mental health-related factors were important predictors across all prediction windows. However, as the prediction window lengthens to greater than 180 days several diagnostic factors and laboratory tests are much less useful in predicting suicide risk. In particular, when the prediction window is larger than 270 days, the effects of depression-related and other mental health-related factors became much smaller. For female patients, the effects of female-specific predictors, i.e., pregnancy-related factors, vanish when prediction window is larger than 180 days. Supplementary Figure 5 reveals that the information available for estimating suicide risk, as reflected in the percent of patients in each predictor category having a particular characteristic, declines substantially over time.
The selected predictors were grouped into seven categories: demographics, depression-related factors (including both diagnoses and medications), other mental health-related factors, routine tests, drug tests, pregnancy-related factors (among females), and other factors. For each prediction window, the value of each predictor category was the normalized cumulative predictor contribution coefficient derived by logistic regression analysis on female patients (red) or male patients (blue), respectively.
Discussion
Accurate risk prediction plays an important role in the intervention of suicide attempts among children and adolescents. The traditional clinical risk assessment tools have been demonstrated to be not sufficiently accurate to identify high-risk patients14,15,16,17. Recently machine learning approaches have been applied to EHR data for the prediction of suicide risk in either adult populations18,19,20,21,22 or children and adolescent population30. Although the previous studies have archived advanced predictive performances compared to the traditional methods, there remains a need for a more comprehensive risk prediction models that utilize clinical data available to produce quantifiable risk scores that can be applied to estimate the patient’s risk level.
In this analysis we have shown that a combination of patient demographic characteristics, diagnoses, procedures, medications, and laboratory tests can be used to construct accurate machine learning models predicting the risk of suicide attempts among pediatric patients receiving inpatient and outpatient care in a children’s medical center. This distinguishes our analysis from virtually all other efforts at suicide risk prediction among both children and adolescents, which have typically had risk horizons of several years to maximize the cases available for analysis11. Notably, our models demonstrated good performance over very short time windows, indicating that the detection of short-term risk of suicidal behavior in this population is attainable. In addition, the proposed models accounted for improvements in both short- and long-term prediction, compared to the traditional logistic regression with L1 regularization. The PPVs observed from the proposed models constitute a 5- to 10-fold improvement in suicide attempt prediction compared to the base rate (see Supplementary Table 13), which is superior to that reported in a similar study by Barak-Corren et al.19.
Another major finding in this analysis (see Supplementary Figs. 3 and 4, and Table 2) is that time matters how well the models perform. The models performed well in time windows ranging between 7 and 180 days, with declining performance observed 9 months and 1 year prior to the attempt. While this is likely due to the fact that, major identified risk factors, such as depression24,38,39, other mental disorders27,40,41,42, and substance abuse25,43,44,45 are proximate risks for suicidal behavior; on the other hand, it is also due to the volume and content of the information available in varying time windows. Supplementary Figure 5 shows that percent of patients whose medical records contained evidence of the identified risk factors falls precipitously over time, indicating that there is a dearth of information with which to construct an accurate suicide risk model among pediatric patients as the time from the previous medical encounter increases beyond 6 months.
In addition, time also matters the attendant risk factors (see Figs. 2 and 3), i.e., contributions of the identified predictors to risk detection vary across prediction windows. In particular, as shown in Figs. 2 and 3, except for demographics, depression-, and other mental disorder-related factors, most predictors shift their importance in risk prediction across prediction windows. This validated the previous finding that majority of the risk factors don’t continuously contribute to the suicide risk30,46. Even though identification of the concrete role of an individual risk factor over time is difficult, we did detect predictor importance pattern reflecting short- and long-term risk of suicide behavior. Our produced risk factor panels, as shown in Fig. 3, leads to the potential of assisting the clinical practitioners in assessing risk levels of the patients. First, female’s short-term risk factors (0- to 90-day prediction windows) come from a broader spectrum, as with a higher area within the curve of each radar chart. The important short-term risk factors of female include demographics, depression-, and other mental disorder-related factors. In contrast, the most important risk factors of male are mental disorder-related factors. The findings suggest that, when estimating short-term suicide risk, risk factors of female and male populations could be emphasized differently. In addition, for both female and male, impacts of the non-demographic risk factors for long-term risk estimation (270- and 365-day prediction windows) decrease due to the dearth of information (Supplementary Fig. 5). Besides, depression- and other mental disorder-related factors are also important in predicting long-term risk.
Strengths and limitations
This study provides a number of critical insights to inform clinical practice. First, we have shown that information that is routinely collected in clinical encounters and maintained in structured clinical records can be used to create accurate predictive models of the risk of suicidal behavior among children and adolescents. Nothing drastically novel was observed among the factors emerging as significant predictors of suicide risk, which is a good thing: it means that the information needed to identify at risk patients is readily available and just requires a mechanism to incorporate it into clinical care. Second, not only do we find that short-term risk of suicidal behavior can be detected, but that longer periods between clinical encounters results in less accurate prediction of suicide risk. This indicates that high-risk patients, whether identified through risk algorithms or by clinical history (e.g., a prior attempt), would benefit from ongoing clinical monitoring.
The present study has several limitations. First, the data are restricted to a single clinical setting with a limited number of suicidal events, potentially limiting both its power and generalizability. Among the 41,721 patients eligible for analysis, only 180 (0.43%) are cases with records of suicide attempt. We did not introduce specific strategies to address the positive-negative imbalance of suicide attempt. One possible consequence of this limitation is that certain risk factors, which are associated with suicide risk but appear infrequently in this patient population, would not be identified. This highlights the need of methods addressing imbalanced clinical data analysis47,48. Second, data for this analysis were collected from a single institution, i.e., CCMC EHR database. This may lead to the possibility that patients identified as negative subjects due to an absence of suicide records actually have been treated for suicide attempts at other institutions. In addition, drawing training and testing data from the same dataset also downgrades the power and generalizability of our models. Third, mining the text of clinical notes has been demonstrated to enrich the predictive models of suicide attempts21,22,49, while in this analysis we did not have access to patients’ clinical notes. Future work may incorporate clinical notes to combine with structured EHR data to enhance our predictive models, but it should be noted that de-identification in clinical text is more complex than that in the structured EHR data50,51 and hence needs more attention. Finally, among the 41,721 eligible patients, 19,941 (47.80%) received health insurance through Medicaid, which is slightly higher than the national average for Medicaid coverage among children (38%)52. This may lead to bias and potentially limit the generalizability of our findings for commercially insured patients.
Conclusions
Our study demonstrated the feasibility of creating predictive models of suicide risk of children and adolescents by using demographics, comorbidity diagnosis codes, laboratory test results, and medications from clinical records. Such models showed good predictive performances for estimation of short-term and long-term risks and identified significant predictors which may assist in clinical practices.
References
Nock, M. K. et al. Suicide and suicidal behavior. Epidemiol. Rev. 30, 133–154 (2008).
Nock, M. K. et al. Prevalence, correlates, and treatment of lifetime suicidal behavior among adolescents: results from the national comorbidity survey replication adolescent supplement. JAMA Psychiatry 70, 300–310 (2013).
Kessler, R. C., Borges, G. & Walters, E. E. Prevalence of and risk factors for lifetime suicide attempts in the national comorbidity survey. Arch. Gen. Psychiatry 56, 617–626 (1999).
Voss, C. et al. Prevalence, onset, and course of suicidal behavior among adolescents and young adults in Germany. JAMA Netw. Open 2, e1914386–e1914386 (2019).
Doshi, R. P. et al. Identifying risk factors for mortality among patients previously hospitalized for a suicide attempt. Sci. Rep. 10, 15223 (2020).
Web-based injury statistics query and reporting system (WISQARS) (Centers for Disease Control and Prevention, 2019).
Stone, D. M. et al. Vital signs: trends in state suicide rates—United States, 1999–2016 and circumstances contributing to suicide—27 states, 2015. Morbidity Mortal. Wkly Rep. 67, 617 (2018).
Hedegaard H., Curtin S. C. & Warner M. Suicide Rates in the United States Continue to Increase. NCHS Data Brief (Centers for Disease Control and Prevention, National Center for Health Statistics, 2018).
Kann, L. et al. Youth risk behavior surveillance—United States, 2017. MMWR Surveill. Summ. 67, 1–114 (2018).
Shaffer, D. & Pfeffer, C. R. Practice parameter for the assessment and treatment of children and adolescents with suicidal behavior. J. Am. Acad. Child Adolesc. Psychiatry 40, 24S–51S (2001).
Belsher, B. E. et al. Prediction models for suicide attempts and deaths: a systematic review and simulation. JAMA Psychiatry 76, 642–651 (2019).
Walsh, C. G., Ribeiro, J. D. & Franklin, J. C. Predicting risk of suicide attempts over time through machine learning. Clin. Psychol. Sci. 5, 457–469 (2017).
Burke, T. A., Ammerman, B. A. & Jacobucci, R. The use of machine learning in the study of suicidal and non-suicidal self-injurious thoughts and behaviors: a systematic review. J. Affect. Disord. 245, 869–884 (2019).
Franklin, J. C. et al. Risk factors for suicidal thoughts and behaviors: a meta-analysis of 50 years of research. Psychol. Bull. 143, 187 (2017).
Carter, G. et al. Predicting suicidal behaviours using clinical instruments: systematic review and meta-analysis of positive predictive values for risk scales. Br. J. Psychiatry 210, 387–395 (2017).
Bentley, K. H. et al. Anxiety and its disorders as risk factors for suicidal thoughts and behaviors: a meta-analytic review. Clin. Psychol. Rev. 43, 30–46 (2016).
Ribeiro, J. D. et al. Self-injurious thoughts and behaviors as risk factors for future suicide ideation, attempts, and death: a meta-analysis of longitudinal studies. Psychol. Med. 46, 225–236 (2016).
Kessler, R. C. et al. Predicting suicides after psychiatric hospitalization in US Army soldiers: the Army Study to Assess Risk and Resilience in Servicemembers (Army STARRS). JAMA Psychiatry 72, 49–57 (2015).
Barak-Corren, Y. et al. Predicting suicidal behavior from longitudinal electronic health records. Am. J. Psychiatry 174, 154–162 (2017).
Simon, G. E. et al. Predicting suicide attempts and suicide deaths following outpatient visits using electronic health records. Am. J. Psychiatry 175, 951–960 (2018).
Poulin, C. et al. Predicting the risk of suicide by analyzing the text of clinical notes. PLoS ONE 9, e85733 (2014).
Ben-Ari, A. & Hammond, K. Text mining the EMR for modeling and predicting suicidal behavior among US veterans of the 1991 Persian Gulf War. In Proc. 2015 48th Hawaii International Conference on System Sciences, pp 3168–3175 (Kauai, HI, USA, 2015).
Su, C., Xu, Z., Pathak, J. & Wang, F. Deep learning in mental health outcome research: a scoping review. Transl. Psychiatry 10, 116 (2020).
Ribeiro, J. D., Huang, X., Fox, K. R. & Franklin, J. C. Depression and hopelessness as risk factors for suicide ideation, attempts and death: meta-analysis of longitudinal studies. Br. J. Psychiatry 212, 279–286 (2018).
Harford, T. C., Yi, H.-y, Chen, C. M. & Grant, B. F. Substance use disorders and self-and other-directed violence among adults: results from the National Survey on Drug Use and Health. J. Affect. Disord. 225, 365–373 (2018).
Felitti, V. J. et al. Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults: The Adverse Childhood Experiences (ACE) Study. Am. J. Prev. Med. 56, 774–786 (2019).
Ahmedani, B. K. et al. Major physical health conditions and risk of suicide. Am. J. Prev. Med. 53, 308–315 (2017).
Walkup, J. T., Townsend, L., Crystal, S. & Olfson, M. A systematic review of validated methods for identifying suicide or suicidal ideation using administrative or claims data. Pharmacoepidemiol. Drug Saf. 21(S1), 174–182 (2012).
Platt, R. & Carnahan, R. The U.S. Food and Drug Administration’s Mini-Sentinel Program. Pharmacoepidemiol. Drug Saf. 21(S1), 1–303 (2012).
Walsh, C. G., Ribeiro, J. D. & Franklin, J. C. Predicting suicide attempts in adolescents with longitudinal clinical data and machine learning. J. Child Psychol. Psychiatry 59, 1261–1270 (2018).
Patrick, A. R. et al. Identification of hospitalizations for intentional self-harm when E-codes are incompletely recorded. Pharmacoepidemiol. Drug Saf. 19, 1263–1275 (2010).
Chen, K. & Aseltine, R. H. Using hospitalization and mortality data to identify areas at risk for adolescent suicide. J. Adolesc. Health 61, 192–197 (2017).
MapIT. AHRQ: Agency for Healthcare Research and Quality. https://www.qualityindicators.ahrq.gov/Resources/Toolkits.aspx (2018).
Xu, H. et al. MedEx: a medication information extraction system for clinical narratives. J. Am. Med. Inform. Assoc. 17, 19–24 (2010).
RxNorm. U.S. National Library of Medicine. https://www.nlm.nih.gov/research/umls/rxnorm/index.html (2019).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B 57, 289–300 (1995).
Ferri, F., Pudil, P., Hatef, M. & Kittler, J. Comparative study of techniques for large-scale feature selection. Mach. Intell. Pattern Recognit. 16, 403–413 (1994).
Kazdin, A. E., French, N. H., Unis, A. S., Esveldt-Dawson, K. & Sherick, R. B. Hopelessness, depression, and suicidal intent among psychiatrically disturbed inpatient children. J. Consult. Clin. Psychol. 51, 504 (1983).
Beck, A. T., Brown, G. & Steer, R. A. Prediction of eventual suicide in psychiatric inpatients by clinical ratings of hopelessness. J. Consult. Clin. Psychol. 57, 309 (1989).
Tardiff, K. & Sweillam, A. Assault, suicide, and mental illness. Arch. Gen. Psychiatry 37, 164–169 (1980).
Mortensen, P. B., Agerbo, E., Erikson, T., Qin, P. & Westergaard-Nielsen, N. Psychiatric illness and risk factors for suicide in Denmark. Lancet 355, 9–12 (2000).
Bolton, J. M., Gunnell, D. & Turecki, G. Suicide risk assessment and intervention in people with mental illness. BMJ 351, h4978 (2015).
Brent, D. A. Risk factors for adolescent suicide and suicidal behavior: mental and substance abuse disorders, family environmental factors, and life stress. Suicide Life Threat. Behav. 25, 52–63 (1995).
Crumley, F. E. Substance abuse and adolescent suicidal behavior. JAMA 263, 3051–3056 (1990).
Bronisch, T. & Wittchen, H. U. Suicidal ideation and suicide attempts: Comorbidity with depression, anxiety disorders, and substance abuse disorder. Eur. Arch. Psychiatry Clin. Neurosci. 244, 93–98 (1994).
Rudd, M. D. et al. Warning signs for suicide: theory, research, and clinical applications. Suicide Life Threat Behav. 36, 255–262 (2006).
Khalilia, M., Chakraborty, S. & Popescu, M. Predicting disease risks from highly imbalanced data using random forest. BMC Med. Inform. Decis. Mak. 11, 51 (2011).
Mazurowski, M. A. et al. Training neural network classifiers for medical decision making: the effects of imbalanced datasets on classification performance. Neural Netw. 21, 427–436 (2008).
Demner-Fushman, D., Chapman, W. W. & McDonald, C. J. What can natural language processing do for clinical decision support? J. Biomed. Informatics 42, 760–772 (2009).
Neamatullah, I. et al. Automated de-identification of free-text medical records. BMC Med. Inform. Decis. Mak. 8, 32 (2008).
Meystre, S. M., Friedlin, F. J., South, B. R., Shen, S. & Samore, M. H. Automatic de-identification of textual documents in the electronic health record: a review of recent research. BMC Med. Res. Methodol. 10, 70 (2010).
Foundation K. F. Health Insurance Coverage of Children 0–18. https://www.kff.org/other/state-indicator/children-0-18/?currentTimeframe=0&sortModel=%7B%22colId%22:%22Location%22,%22sort%22:%22asc%22%7D (2019).
Acknowledgements
The research is supported by NIH R01MH124740 and R01MH112148. The work of F.W. is also partially supported by NSF 1750326 and ONR N00014-18-1-2585.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Su, C., Aseltine, R., Doshi, R. et al. Machine learning for suicide risk prediction in children and adolescents with electronic health records. Transl Psychiatry 10, 413 (2020). https://doi.org/10.1038/s41398-020-01100-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-020-01100-0
This article is cited by
-
Role of machine learning algorithms in suicide risk prediction: a systematic review-meta analysis of clinical studies
BMC Medical Informatics and Decision Making (2024)
-
Accuracy and transportability of machine learning models for adolescent suicide prediction with longitudinal clinical records
Translational Psychiatry (2024)
-
Analysis and evaluation of explainable artificial intelligence on suicide risk assessment
Scientific Reports (2024)
-
The Intersectionality of Factors Predicting Co-occurring Disorders: A Decision Tree Model
International Journal of Mental Health and Addiction (2024)
-
The key artificial intelligence technologies in early childhood education: a review
Artificial Intelligence Review (2024)