Abstract
Adenosine, its interacting A1 and A2A receptors, and particularly the variant rs5751876 in the A2A gene ADORA2A have been shown to modulate anxiety, arousal, and sleep. In a pilot positron emission tomography (PET) study in healthy male subjects, we suggested an effect of rs5751876 on in vivo brain A1 receptor (A1AR) availability. As female sex and adenosinergic/dopaminergic interaction partners might have an impact on this rs5751876 effect on A1AR availability, we aimed to (1) further investigate the pilot male-based findings in an independent, newly recruited cohort including women and (2) analyze potential modulation of this rs5751876 effect by additional adenosinergic/dopaminergic gene variation. Healthy volunteers (32/11 males/females) underwent phenotypic characterization including self-reported sleep and A1AR-specific quantitative PET. Rs5751876 and 31 gene variants of adenosine A1, A2A, A2B, and A3 receptors, adenosine deaminase, and dopamine D2 receptor were genotyped. Multivariate analysis revealed an rs5751876 effect on A1AR availability (P = 0.047), post hoc confirmed in 30 of 31 brain regions (false discovery rate (FDR) corrected P values < 0.05), but statistically stronger in anxiety-related regions (e.g., amygdala, hippocampus). Additional effects of ADORA1 rs1874142 were identified; under its influence rs5751876 and rs5751876 × sleep had strengthened effects on A1AR availability (Pboth < 0.02; post hoc FDR-corrected Ps < 0.05 for 29/30 regions, respectively). Our results support the relationship between rs5751876 and A1AR availability. Additional impact of rs1874142, together with rs5751876 and sleep, might be involved in regulating arousal and thus the development of mental disorders like anxiety disorders. The interplay of further detected suggestive ADORA2A × DRD2 interaction, however, necessitates larger future samples more comparable to magnetic resonance imaging (MRI)-based samples.
Similar content being viewed by others
Introduction
Anxiety is a fundamental emotion eliciting considerable reactions like increased attention, heart rate, and energy metabolism1. In case of pathological anxiety/anxiety disorders, dysregulation in brain regions of the “fear network” occurs, e.g., hyperactivation of amygdala, hippocampus, thalamus, and cingulum, as well as decreased interconnectivity between these regions2,3. Anxiety and arousal are tightly connected, both counteract sleep, and both are strongly linked to the adenosinergic system4,5,6,7. The purine nucleoside adenosine is ubiquitous in body and brain and involved in the regulation of e.g., heart rate, energetic homeostasis, sleep, arousal, and anxiety4,5,8. It acts through the four G-protein coupled adenosine receptors (AR) A1AR, A2AAR, A2BAR, and A3AR, of which particularly the first is widely distributed in the brain and has a high affinity to adenosine4,5,6,9. An increase in A1AR function was reported to elicit anxiolytic effects10,11 and quantification of A1AR availability via positron emission tomography (PET) demonstrated an A1AR increase after sleep loss12,13. A further PET investigation revealed a modulation of A1AR availability by caffeine14, which antagonistically binds to both A1AR and A2AAR15. Both receptors are coexpressed, form heteromers, and interact to regulate adenosinergic and glutamatergic neurotransmission in the brain, which, apart from various physiological functions, have been implicated in pathological alterations related to psychiatric and anxiety disorders (e.g.,4,5,6,16,17,18). Genetic loss of A1AR or A2AAR enhanced anxiety-related behavior in knockout mice19,20 and variants in the corresponding human genes ADORA1 and ADORA2A were reported to be associated with, for example, sympathetic arousal-related blood-injury phobia21, panic disorder22,23, and anxiety-related autism spectrum disorder24. Further associations were identified in healthy subjects with caffeine-induced anxiety25,26,27 and caffeine effects on sleep (e.g.,28). In all these studies ADORA2A was reported to have the stronger impact compared with ADORA1. Especially the ADORA2A single-nucleotide variant rs5751876 (1976C/T, formerly 1083 C/T) revealed strongest and most consistent associations, and its rare T-allele repeatedly turned out at risk for elevated or pathological anxiety (e.g.,21,22,23,25,26,27). Further studies support the notion that there is a relation between anxiety and caffeine challenge in female rs5751876 TT homozygotes, indicating also a female sex influence in increased anxiety29,30.
Among this direct ADORA2A rs5751876 effect on anxiety, a previous pilot study reported the T-allele to be associated with increased brain A1AR availability in healthy participants as measured by PET31, suggesting indirect effects on anxiety via A1AR modulation. Although this finding highlights the potential putative role of an interaction between both receptors, the pilot study was restricted to a small sample of 28 male individuals only, allowing only exploratory analyses of few ADORA2A and ADORA1 variants. However, there is evidence to suggest that female sex and additional adenosinergic or dopaminergic genetic variants might as well have a modulatory impact on the rs5751876 effect on A1AR availability. Female sex is associated with a higher risk of anxiety/anxiety disorders as well as with a higher risk of sleep- (disruption-) related disorders like insomnia or hypertension (e.g.,29,32). A2BAR, A3AR, and the adenosine deaminase as members of the adenosinergic system mediate adenosine actions via receptor-receptor interactions33 and were linked to anxiety- or arousal-related behavior in mice9,34,35. The dopamine D2 receptor (D2R) functions as major interaction partner of A2AAR as demonstrated by their colocalization, heteromerization, and cross-desensitization36,37. Genetic interaction of DRD2-ADORA2A variants has been shown to affect caffeine-induced anxiety26, whereas DRD2 variants alone, notably the DRD2/ANKK1 variant rs1800497 (Taq1A polymorphism), were reported to be associated with anxiety symptoms/disorders (e.g.,38,39,40,41).
Taken together, previous studies suggest that female sex and additional genetic variations might further modulate the ADORA2A rs5751876 effect on brain A1AR availability. Therefore, the present study aimed to expand on the original pilot study findings by analyzing a larger set of potentially interacting genetic variants in an independent newly recruited male/female mixed cohort. We hypothesized firstly, that the pilot male-based rs5751876 effect on in vivo brain A1AR availability31 can be observed also in an independent male-female based sample of healthy humans. Second, we hypothesized that genetic variants of A1AR, A2BAR, A3AR, ADA, and D2R contribute to the modulation of the rs5751876 effect on in vivo brain A1AR availability.
Subjects and methods
Subjects
All subjects were newly recruited via advertisement and a subsequent interview by phone. Forty-three healthy volunteers were finally included in the study (mean age 34.2 ± 14.1 y; 32 males, 11 females; for further details see supplemental material) after obtaining written informed consent for imaging procedure, blood sampling, and genetic testing. Exclusion criteria were neurological or psychiatric disorders, head trauma, systemic diseases, or substance/drug abuse interfering with ARs. All these subjects were phenotypically characterized by a further standardized interview (subjects demographics/characteristics in Table 1) and underwent medical examination before PET imaging at the PET laboratory of the Institute of Neuroscience and Medicine (Forschungszentrum Jülich). All subjects had a history free of psychiatric and neurological diseases. Self-reported habitual sleep duration, current medication, presence of allergies, and consumption of cigarettes, alcohol, coffee and caffeine-containing drinks were obtained by self-rating on a standardized questionnaire routinely used in the PET laboratory (for answers/results see Table 1), which all subjects filled-in before PET imaging. Average sleep duration per night was given in hours and coffee consumption in cups/day (0.15 mL). Whole amount of caffeine-containing drinks per day was assessed in liters. Prior to PET scanning participants were asked to avoid caffeine consumption for at least 24 h. All procedures were approved by the Ethics Committee of the Medical Faculty of the University of Duesseldorf and the German Federal Office for Radiation Protection.
Imaging procedures
PET acquisitions and high-resolution three-dimensional T1-weighted magnetic resonance (MR) images were performed as previously reported31 while postprocessing for calculations of 18F-8-cyclopentyl-3-(3-fluoropropyl)-1-propylxanthine ([18F]CPFPX42) binding in cerebral tissues differed slightly. In brief, realignment, coregistrations, segmentation, and normalization of three-dimensional PET and corresponding MRI data were done with PMOD software (version 3.408, PMOD Group, Zürich, Switzerland). For definition of regions of interest, the AAL-template43 implemented in the PMOD software was used. Regional time-activity curves (TACs) were corrected for decay and the contribution of intracerebral blood volume. Corrected TACs were used to estimate the A1AR availability in terms of the [18F]CPFPX binding potential (BPND44) via the Logan Plot (t* = 20 min, fixed k2’45) with the cerebellar gray matter as a reference region46. Imaging procedures are further detailed in Table 1 and in the supplements. Investigated regions are given in Table 2.
Genotyping
DNA from blood samples was isolated and used for genotyping of selected variants as described previously31 and in detail in the supplements (text and Tabs. S1 and S2). In brief, ADORA2A and ADORA1 variants were the same as used for the pilot sample based on the initially described selection process31 (using tagging information of the international HapMap Project, functional potential information of the UCSC Genome browser and association information of previous studies21,22,23,24,25,26,27,28,29,30). Additional pairwise linkage disequilibrium (LD) analyses performed in the pilot study sample provided LD information and redundancy of several variants31. These were dropped, leaving five ADORA2A variants (rs5751862, rs5760405, rs2236624, rs5751876, rs4822492) and seven ADORA1 variants (rs1874142, rs10920568, rs12135643, rs3766566, rs17511192, rs6677137, rs3753472) for the present study (for all see Suppl. Tab S1). All other adenosinergic/dopaminergic variants were selected based on association studies revealing significant variant effects on anxiety or psychiatric disorder-related context, or selected based on functional potential, tagging and frequency information (study references and variant details in Suppl. Tab. S1): rs758857, rs2535609 (A2BAR gene ADORA2B); rs1890245, rs35254520, rs2786995, rs10776727, rs1544224, rs2229155 (A3AR gene ADORA3); rs73598374, rs427483 (ADA gene ADA); rs4648317, rs7131056, rs4936272, rs4245146, rs17529477, rs55900980, rs1076560, rs6275, rs6277, rs1800497 (D2R gene DRD2). Of these additional ten adenosinergic and ten dopaminergic gene variants no LD analyses existed so far. Genotypes were determined and controlled for genotyping errors by standard PCR-based methods blind with respect to the phenotypic characteristics of the subjects (for description, primers, and assay conditions see supplements (text and Tab. S2)).
Statistical analyses
Hardy-Weinberg equilibrium and pairwise LD were assessed using Haploview v.4.1 as reported before31. Genotypes were grouped separately for each variant into carriers of both common alleles (common homozygotes) vs. carriers of at least one rare allele (heterozygotes + rare homozygotes); see supplements (Tab. S1). All other analyses were conducted two-sided using SPSS v.25 (IBM, Chicago, IL, USA).
According to our first hypothesis the impact of ADORA2A rs5751876 (CC homozygotes vs. T-allele carrier; as previously reported31) on A1AR availability was investigated in our male-female-based sample in a first analysis model. We started with testing the A1AR availability data for mutual dependence (intercorrelation) between brain regions and with testing the participants’ demographics/characteristics for potential confounding, both as reported before31 and as detailed in Table 1 as well as in the supplements. Based on the detected intercorrelation and confounders (see results section below) we then utilized general linear model (GLM) multivariate procedure for simultaneous analysis of multiple (intercorrelated) dependent variables47 (i.e., the brain region specific A1AR availabilities) by genotype group effect with additional confounders in a single model. Thereby the multivariate ADORA2A rs5751876 genotype group effect on global A1AR brain availability was analyzed and, if significant, followed by post hoc ANCOVAs to identify single brain regions contributing to the global effect as detailed in the supplements. Significance level alpha was set to 0.05 for all pretests and the global multivariate analysis. The ANCOVAs were further corrected for testing 31 brain regions by controlling the false discovery rate (FDR) following the Benjamini-Hochberg procedure48.
According to our second hypothesis the ADORA2A rs5751876 effect on A1AR brain availability was investigated under the assumption of additional modulatory effects of adenosinergic or dopaminergic gene variation. In analogy to the first model, GLM multivariate procedures were used to analyze the rs5751876 genotype group effect on A1AR availability (same confounders and model composition), but now including one by one each genetic variant as main factor and in interaction with rs5751876. Significance level was adjusted to 0.025 because of the high similarity to the first model. In case of a significant global effect of rs5751876 on A1AR availability, again FDR-corrected post hoc ANCOVAs followed to identify single brain regions contributing to this effect. Testing two to seven genetic variants per gene (ADORA1: 6, ADORA2B: 2, ADORA3: 6, ADA: 2, DRD2: 7) remained uncorrected in the second model approach.
Results
All genotype distributions were in Hardy-Weinberg equilibrium (P > 0.05). Several variants in close/complete LD (D’ = 1.0 and r2 > 0.8: rs4822492, rs3766566, rs1076560, rs55900980, rs4936272; supplemental Tab. S3) were excluded from further analysis to avoid redundancy. All ADORA2A variants revealed high pairwise LD (D’ ≥ 0.78), thus potential confounding across individual genotype groups was considered relevant for the whole ADORA2A gene and included as confounder for our rs5751876-based analyses.
ADORA2A rs5751876 effect on A1AR availability in mixed male/female sample
High intercorrelation of A1AR availability between brain regions was detected (all P < 0.004, pairwise correlation coefficients: mean = 0.832, range = 0.431–0.983). Analysis of participants’ demographics/characteristics revealed the following statistically relevant confounders: sex, age, self-reported habitual sleep, self-reported presence of allergies, self-reported caffeine and alcohol consumption, application mode of radiotracer, injected activity of radiotracer, and total amount of injected substance (Pall < 0.05; Table 1). Since sex and sleep had effects on both, A1AR availability and across ADORA2A genotype groups, additional interaction terms were formed (rs5751876 × sex; rs5751876 × sleep) and further included as confounders.
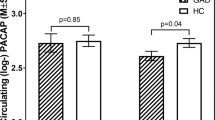
Our first analysis model comprising these confounders and interaction terms revealed a significant global multivariate effect of rs5751876 on A1AR availability (P = 0.047). Post hoc ANCOVAs confirmed this effect for nearly all (30 of 31) investigated brain regions with nominal P values even robust to Benjamini-Hochberg FDR correction for testing multiple regions (corrected Pall < 0.05 (except pallidum); Table 2). Moreover, anxiety-related regions such as amygdala, hippocampus–parahippocampus, cingulate cortex, calcarine fissures, temporal lobe, and thalamus were found strongly associated with nominal P values < 0.01 (Fig. 1; Table 2).
Mean in vivo MR image illustrates the strongly rs5751876 associated brain regions (post hoc P values < 0.01), which are overlaid based on the utilized regional atlas in transversal (left), sagittal (middle), and coronal (right) direction. Red labeling indicates anxiety-related regions and blue labeling the regions without strong relationship to anxiety or anxiety disorders.
Modulatory influence of ADORA1 rs1874142 on rs5751876 and rs5751876 × sleep effects
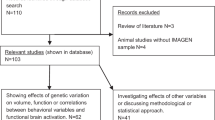
Second model analyses revealed ADORA1 rs1874142 to statistically strengthen the model, i.e. under the influence of rs1874142 stronger significances were detected for the effects of rs5751876 (global P = 0.018; Table 2) and the interaction rs5751876 × sleep (global P = 0.020) on A1AR availability. Both these rs5751876-related terms revealed P values ranging below the adjusted significance level of 0.025. Further, both findings could be confirmed post hoc for nearly all investigated brain regions (Table 2; FDR-corrected P values < 0.05 (except pallidum and anterior cingulum for rs5751876; except pallidum for rs5751876 × sleep)). Again, anxiety-related regions such as amygdala, hippocampus/Parahippocampus, calcarine fissures, and temporal lobe were found strongly associated with nominal P values < 0.01 (Fig. 2; Table 2).
GLM multivariate bar plots based on second model post hoc analyses illustrate significant rs5751876 effect on A1AR availability in the anxiety-related regions amygdala (P = 0.007; FDR Pcorr = 0.024), hippocampus/Parahippocampus (P = 0.005; FDR Pcorr = 0.029), temporal lobe (P = 0.008; FDR Pcorr = 0.023), and calcarine fissures (P = 0.0003; FDR Pcorr = 0.009) under the modulatory influence of the ADORA1 variant rs1874142. Plots represent adjusted means according to sleep (7.37 h), age (33.4 y), injected activity (240.06 MBq), and amount of substance (2.91 nmol) with standard error bars (SEM).
In addition, ADORA2B rs2535609 seemed to statistically strengthen the model. Under its influence, stronger significances were detected for the multivariate effects of rs5751876 (global P = 0.005) and rs5751876-related interactions (rs5751876 × sleep, global P = 0.005; rs5751876 × rs2535609, global P = 0.012) on A1AR availability (Table 3). However, none of these multivariate findings could be confirmed post hoc for individual brain regions, as none of them survived correction for multiple brain region testing (all FDR-corrected P values > 0.05).
All other adenosinergic or dopaminergic gene variants seemed to statistically weaken second model analyses (for all: multivariate global P values > 0.05, see Table 3).
Discussion
Our present study revealed two main findings. First, we were able to corroborate the ADORA2A rs5751876 effect on A1AR availability, originally detected in our pilot sample of males only31, in an independent male/female sample. This allowed a more-generalized interpretation compared with the pilot model and thus further underlined the important role of ADORA2A interacting with A1AR as detailed in the introduction (e.g.,4,5,6,16,17,18). Second, our findings showed modulation by ADORA1 rs1874142, resulting in statistically strengthened effects of rs5751876 and rs5751876 × sleep on A1AR availability. In these findings, anxiety-related brain regions were particularly involved, underscoring the close link between ADORA2A rs5751876, anxiety, and anxiety disorders. This is in line with the variety of anxiety-relevant investigations reported in the introduction (e.g.,21,22,23,24,25,26,27). The recruitment of slightly more individuals compared with the pilot study appears to have increased the power of the rs5751876 effect enough (28 subjects in pilot vs. 43 in present sample), to have resulted in now statistically significant multivariate results in contrast to the pilot study31. Furthermore, the identified interactive effect of rs5751876 × sleep on A1AR availability under the ADORA1 rs1874142 modulation underscores the close interrelationship between the adenosine system, particularly the interplay of A1 and A2A receptors, and sleep as mentioned before5,8,12,13,28.
However, though the rs5751876 effect on A1AR availability was distinctly stronger in the present sample compared with the pilot study, the direction of the rs5751876 effect disclosed discrepancies. In the pilot study the rs5751876 T-allele increased A1AR availability31, whereas in the present sample the T-allele predominantly decreased A1AR availability (in 31 regions in the first model; in 23 regions in the second model under modulation of ADORA1 rs1874142). Analysis of the underlying genotype effects (rs5751876 CC vs. CT vs. TT) uncovered consistent CT to TT (lower to higher A1AR availability, respectively) gene-dose effects in both samples. In contrast, the CC carriers scored lowest (with respect to A1AR availability) in the pilot, thereby completing the gene-dose distribution, but high in the present sample (in 23 of 31 brain regions even higher than TTs), thereby forming a U-shaped distribution (Suppl. Fig. S1). Though sex differences exist between both samples (males only vs. mixed males/females), female sex was not related to the detected discrepancy in the direction of the rs5751876 genotype effect between present and pilot sample (present sample males alone: similar rs5751876 T-allele coupled decrease in A1AR availability in 30 of 31 brain regions). Besides intended sex differences, both samples were similarly recruited with regard to demographic data like age, body mass index, history of diseases, and habits like sleep, coffee, and nicotine consumption.
To probe alternative explanations for this discrepancy in the direction of the rs5751876 genotype effect and based on the strong interaction of rs5751876 × sleep, we checked if sleep habit might contribute to the rs5751876 genotype effect discrepancies between present and pilot study31. As both samples show comparable sleep habits (mean ± SD self-reported habitual sleep duration of present sample 7.35 ± 0.76 vs. pilot sample 7.32 ± 1.01; P = 0.811). Thus, we dichotomized the variant in the subgroup categories “less sleep than mean” (less sleep) and “more sleep than mean” (more sleep) with comparable subgroup sizes in both samples (N (less/more sleep) in present sample: 21/22; in pilot sample: 14/13). In these subgroups, we qualitatively rechecked the respective rs5751876 genotype dependent distribution of A1AR availability values with promising findings. In the less sleep group of the pilot sample most regions showed a linear gene-dose relationship, but however 7 of 19 brain regions (hippocampus, thalamus, caudate, putamen, parietal cortex, occipital cortex, sensorimotor cortex; Suppl. Tab. S4) revealed indeed U-shaped distribution, which resembles the U-shaped distribution in 31 brain regions in our present study (Suppl. Tab. S5). In contrast, the more sleep group of the pilot sample revealed the above mentioned gene-dose distribution in almost all (17 of 19) regions (Suppl. Tab. S4; only posterior cingulate gyrus and putamen with inverted U-shaped distributions). This again resembles the more sleep group of our present study revealing as well gene-dose genotype distribution in 13 of 31 brain regions, including particularly the anxiety-relevant regions amygdala, hippocampus/Parahippocampus, cingulate cortex (anterior, middle), thalamus, temporal lobe, calcarine fissures, and occipital lobe; Suppl. Tab. S5). Taken together, though self-reported and not measured by objective methods like actigraphy or EEG, sleep might explain at least several of the observed discrepancies. This is in line with previous findings of a tight connection between ADORA2A and sleep as well as between sleep and A1AR availabilities. ADORA2A variants (including rs5751876) modulate psychomotor vigilance in a rested state and after sleep deprivation28, as well as A1AR availability increases after prolonged wakefulness12,13. Slight A1AR availability increases might also occur in our less sleep group participants owing to their shorter habitual sleep duration and therefore slightly prolonged wakefulness. Such slightly increased A1AR availability might then have obscured or masked the presumably small effect size of the ADORA2A rs5751876 T-allele effect. In contrast, our more sleep group probably would not have slightly prolonged wakefulness and therefore presumably would have overall lower A1AR availabilities. In such cases, the small effect of the rs5751876 T-allele and the resulting slightly upregulated A1AR availability might be visible. Such slight but possibly permanent upregulation of A1ARs by genotype might provide protection against overly increased arousal or anxiety, or even anxiety disorders. Indeed a protective role of upregulated A1AR was reported toward hyperarousal associated with restless legs syndrome49. In rodents, upregulation or positive allosteric modulation of A1AR caused robust anxiolytic effects10,11, whereas lack of A1AR enhanced anxiety and arousal20.
Finally, we checked all 31 additional adenosinergic and dopaminergic gene variants for potentially divergent genotype group distributions in pilot vs. present sample by nonparametric statistics (Fisher’s exact test; see also Suppl. Tab. S6 with footnotes), which might further explain the rs5751876 genotype effect discrepancies between both samples. Strikingly, DRD2 variants rs4245146 and rs1800497 were distributed differently between both samples (both P < 0.05; Suppl. Tab. S6), which might contribute to the gene-dose vs. U-shaped rs5751876 effects. Although 15 rs4245146 CC homozygotes existed in the present sample, only one was present in the pilot (Suppl. Tab. S6), and this rs4245146 CC-homozygosity seemed to further modulate ADORA2A rs5751876 effects. The 15 rs4245146 CC homozygotes had increased A1AR availabilities in all 31 brain regions in rs5751876 CC carriers (11 regions P < 0.05; 10 regions P < 0.1) but had decreased A1AR availabilities in all 31 brain regions in rs5751876 CT-/TT-carriers (one region P < 0.05; one region P < 0.1; Suppl. Tab. S7). Thus, rs4245146 CC-dependent modulation might interplay with the rs5751876 effect on A1AR availability in the present sample (Suppl. Fig. S2) but not the pilot sample with only one rs4245146 CC-subject. As well, carriers of the rare rs1800497 A-allele are four times more frequent in the present sample (N = 16) than in the pilot (N = 4; Suppl. Tab. S6). This higher number of A-allele carriers might have lowered individual D2R levels, in line with a reduced striatal D2R binding already shown in rs1800497 A-allele carriers50. As A2A and D2 receptors have a tight spatial/functional interplay and interact antagonistically36,37, lower D2R levels could lead to increased A2AR levels in our 16 rs1800497 A-allele carriers. Via the similarly tight spatial/functional antagonistic interplay of A2A and A1 receptors (c.f. introduction), such putatively increased A2AR levels in the 16 subjects (=37% of present sample) might have decreased A1AR levels and thereby covered or obscured the presumably mild ADORA2A rs5751876 effect on A1AR availability. In comparison, Eisenstein et al.50 reported normal striatal D2R specific binding in rs1800497 GG-homozygotes, which might be the precondition to clearly identify the small effect size ADORA2A rs5751876 genotype group effects in our present study (63% of subjects vs. 86% of pilot subjects). Interestingly, both DRD2 variants, rs1800497 and rs4245146, were described to play prominent roles in anxiety before (e.g.,39,40,41), further supporting our more distinctive findings in anxiety-related brain regions. Moreover, dopaminergic genes including DRD2 have been shown to be involved in inverted U-shaped relationships between dopamine signaling and prefrontal cortical function (e.g.,51,52), supporting our assumption of DRD2 variants presumably contributing to our sample ADORA2A rs5751876 differences. Unfortunately, the size of our present sample did not allow inclusion of additional genetic variants like rs4245146 or rs1800497 together with rs5751876 and rs1874142, as the model is underpowered then, warranting distinctly larger samples in subsequent investigations.
A next step could be to compare individuals with and without anxiety symptoms and heart rate measurements with regard to their objectively measured sleep duration and genotypes. Such multidimensional approach would demand higher statistical power, which underscores the need to extend future sample size. In our present study it was modest, albeit in the range of comparable recent PET-based studies (e.g.,13,53,54) and well-powered enough for our ADORA2A rs5751876-focused analyses to detect the hypothesized effects. It did unfortunately not allow for additional detailed investigation of for example additional interacting effects of DRD2 variants together with ADORA2A rs5751876 and ADORA1 rs1874142 in the same multivariate models as mentioned above. Further, our study provided only cross-sectional data, whereas a longitudinal study might allow prospective insights and maybe the potential for interventional approaches.
Conclusion
Our present findings support the role of ADORA2A rs5751876 on in vivo A1AR availability in the human brain. Further analyses suggested additional modulatory effects of ADORA1 rs1874142 on the effects of rs5751876 and rs5751876 × sleep on A1AR availability. Together, these effects might contribute to the regulation of the sleep-arousal system and thus might be involved in the development of particularly arousal-related mental disorders such as anxiety disorders. However, transferability of our results between present and pilot samples was difficult due to small effect sizes and complex gene × gene (ADORA2A × ADORA1, ADORA2A × DRD2) and gene × environment (ADORA2A × sleep) interactions, which modulated the direction of the biological outcome, i.e., A1AR availability in the brain. This might complicate also comparisons with future samples and therefore necessitates increased sample sizes, more comparable to current MRI-based samples, to allow the detection of such complex underlying and modifying relationships. In the next steps, more in-depth analyses of the ADORA2A rs5751876 × sleep relation are required based on objectively measured individual sleep parameters ideally complemented by more-detailed information of individual anxiety sensitivity, life styles, (night shift) workload, and past or present stressful life events. Further, investigation of samples preferably including also individuals with anxiety symptoms and in a longitudinal design are needed.
References
Hohoff, C. Anxiety in mice and men: a comparison. J. Neural Transm. 116, 679–687 (2009).
Brooks, S. J. & Stein, D. J. A systematic review of the neural bases of psychotherapy for anxiety and related disorders. Dialog. Clin. Neurosci. 17, 261–279 (2015).
Lai, C. H. & Wu, Y. T. The explorative analysis to revise fear network model for panic disorder: functional connectome statistics. Med. (Baltim.) 95, e3597 (2016).
Deckert, J. & Gleiter, C. H. Adenosine-an endogenous neuroprotective metabolite and neuromodulator. J. Neur. Transm. Suppl. 43, 23–31 (1994).
Ribeiro, J. A., Sebastião, A. M. & de Mendonça, A. Adenosine receptors in the nervous system: pathophysiological implications. Prog. Neurobiol. 68, 377–392 (2002).
Gomes, C. V., Kaster, M. P., Tomé, A. R., Agostinho, P. M. & Cunha, R. A. Adenosine receptors and brain diseases: neuroprotection and neurodegeneration. Biochim. Biophys. Acta 1808, 1380–1399 (2011).
Goldstein, A. N. & Walker, M. P. The role of sleep in emotional brain function. Annu. Rev. Clin. Psychol. 10, 679–708 (2014).
Headrick, J. P., Peart, J. N., Reichelt, M. E. & Haseler, L. J. Adenosine and its receptors in the heart: regulation, retaliation and adaptation. Biochim. Biophys. Acta 1808, 1413–1428 (2011).
Wei, C. J., Li, W. & Chen, J. F. Normal and abnormal functions of adenosine receptors in the central nervous system revealed by genetic knockout studies. Biochim. Biophys. Acta 1808, 1358–1379 (2011).
Serchov, T. et al. Increased signaling via adenosine A1 receptors, sleep deprivation, imipramine, and ketamine inhibit depressive-like behavior via induction of Homer1a. Neuron 87, 549–562 (2015).
Vincenzi, F. et al. Positive allosteric modulation of A1 adenosine receptors as a novel and promising therapeutic strategy for anxiety. Neuropharmacology 111, 283–292 (2016).
Elmenhorst, D. et al. Sleep deprivation increases A1 adenosine receptor binding in the human brain: a positron emission tomography study. J. Neurosci. 27, 2410–2415 (2007).
Elmenhorst, D. et al. Recovery sleep after extended wakefulness restores elevated A1 adenosine receptor availability in the human brain. Proc. Natl Acad. Sci. USA 114, 4243–4248 (2017).
Elmenhorst, D., Meyer, P. T., Matusch, A., Winz, O. H. & Bauer, A. Caffeine occupancy of human cerebral A1 adenosine receptors: in vivo quantification with 18F-CPFPX and PET. J. Nucl. Med. 53, 1723–1729 (2012).
Fredholm, B. B., Bättig, K., Holmén, J., Nehlig, A. & Zvartau, E. E. Actions of caffeine in the brain with special reference to factors that contribute to its widespread use. Pharmacol. Rev. 51, 83–133 (1999).
Ferré, S. et al. Adenosine receptor heteromers and their integrative role in striatal function. Sci. World J. 7, 74–85 (2007).
Casadó, V. et al. Gi protein coupling to adenosine A1-A2A receptor heteromers in human brain caudate nucleus. J. Neurochem. 114, 972–980 (2010).
Kashfi, S., Ghaedi, K., Baharvand, H., Nasr-Esfahani, M. H. & Javan, M. A1 adenosine receptor activation modulates central nervous system development and repair. Mol. Neurobiol. 54, 8128–8139 (2017).
Ledent, C. et al. Aggressiveness, hypoalgesia and high blood pressure in mice lacking the adenosine A2a receptor. Nature 388, 674–678 (1997).
Johansson, B. et al. Hyperalgesia, anxiety, and decreased hypoxic neuroprotection in mice lacking the adenosine A1 receptor. Proc. Natl. Acad. Sci. USA 98, 9407–9412 (2001).
Hohoff, C. et al. Sympathetic activity relates to adenosine A(2A) receptor gene variation in blood-injury phobia. J. Neural Transm. 116, 659–662 (2009).
Deckert, J. et al. Systematic mutation screening and association study of the A1 and A2a adenosine receptor genes in panic disorder suggest a contribution of the A2a gene to the development of disease. Mol. Psychiatry 3, 81–85 (1998).
Hohoff, C. et al. Adenosine A(2A) receptor gene: evidence for association of risk variants with panic disorder and anxious personality. J. Psychiatr. Res. 44, 930–937 (2010).
Freitag, C. M. et al. Adenosine A(2A) receptor gene (ADORA2A) variants may increase autistic symptoms and anxiety in autism spectrum disorder. Eur. Child Adolesc. Psychiatry 19, 67–74 (2010).
Alsene, K., Deckert, J., Sand, P. & de Wit, H. Association between A2a receptor gene polymorphisms and caffeine-induced anxiety. Neuropsychopharmacology 28, 1694–1702 (2003).
Childs, E. et al. Association between ADORA2A and DRD2 polymorphisms and caffeine-induced anxiety. Neuropsychopharmacology 30, 2791–2800 (2008).
Rogers, P. J. et al. Association of the anxiogenic and alerting effects of caffeine with ADORA2A and ADORA1 polymorphisms and habitual level of caffeine consumption. Neuropsychopharmacology 35, 1973–1983 (2010).
Bodenmann, S. et al. Polymorphisms of ADORA2A modulate psychomotor vigilance and the effects of caffeine on neurobehavioural performance and sleep EEG after sleep deprivation. Br. J. Pharmacol. 165, 1904–1913 (2012).
Domschke, K. et al. ADORA2A Gene variation, caffeine, and emotional processing: a multi-level interaction on startle reflex. Neuropsychopharmacology 37, 759–769 (2012).
Gajewska, A. et al. Effects of ADORA2A gene variation and caffeine on prepulse inhibition: a multi-level risk model of anxiety. Prog. Neuropsychopharmacol. Biol. Psychiatry 40, 115–121 (2013).
Hohoff, C. et al. Association of adenosine receptor gene polymorphisms and in vivo adenosine A1 receptor binding in the human brain. Neuropsychopharmacology 39, 2989–2999 (2014).
Bailey, M. & Silver, R. Sex differences in circadian timing systems: implications for disease. Front. Neuroendocrinol. 35, 111–139 (2014).
Sebastião, A. M. & Ribeiro, J. A. Fine-tuning neuromodulation by adenosine. Trends Pharmacol. Sci. 21, 341–346 (2000).
Gonçalves, F. Q. et al. Adenosine A2b receptors control A1 receptor-mediated inhibition of synaptic transmission in the mouse hippocampus. Eur. J. Neurosci. 41, 878–888 (2015).
Sauer, A. V. et al. Alterations in the brain adenosine metabolism cause behavioral and neurological impairment in ADA-deficient mice and patients. Sci. Rep. 7, 40136 (2017).
Hillion, J. et al. Coaggregation, cointernalization, and codesensitization of adenosine A2A receptors and dopamine D2 receptors. J. Biol. Chem. 277, 18091–18097 (2002).
Fuxe, K. et al. Adenosine A2A and dopamine D2 heteromeric receptor complexes and their function. J. Mol. Neurosci. 26, 209–220 (2005).
Hayden, E. P. et al. The dopamine D2 receptor gene and depressive and anxious symptoms in childhood: associations and evidence for gene-environment correlation and gene-environment interaction. Psychiatr. Genet. 20, 304–310 (2010).
Sipilä, T. et al. An association analysis of circadian genes in anxiety disorders. Biol. Psychiatry 67, 1163–1170 (2010).
Thomas, S. A. et al. Allelic variation of risk for anxiety symptoms moderates the relation between adolescent safety behaviors and social anxiety symptoms. J. Psychopathol. Behav. Assess. 37, 597–610 (2015).
Li, L. et al. The association between genetic variants in the dopaminergic system and posttraumatic stress disorder: a meta-analysis. Medicine (Baltim.) 95, e3074 (2016).
Holschbach, M. H. et al. Synthesis and evaluation of no-carrier-added 8-cyclopentyl-3-(3-[(18)F]fluoropropyl)-1-propylxanthine ([(18)F]CPFPX): a potent and selective A(1)-adenosine receptor antagonist for in vivo imaging. J. Med. Chem. 45, 5150–5156 (2002).
Tzourio-Mazoyer, N. et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15, 273–289 (2002).
Innis, R. B. et al. Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J. Cereb. Blood Flow. Metab. 27, 1533–1539 (2007).
Logan, J. et al. Distribution volume ratios without blood sampling from graphical analysis of PET data. J. Cereb. Blood Flow. Metab. 16, 834–840 (1996).
Meyer, P. T. et al. Effect of aging on cerebral A1 adenosine receptors: A [18F]CPFPX PET study in humans. Neurobiol. Aging 28, 1914–1924 (2007).
Field A. (ed). Discovering Statistics Using IBM SPSS Statistics (SAGE Publications, London, 2009).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B 57, 298–300 (1995).
Quiroz, C. et al. Adenosine receptors as markers of brain iron deficiency: implications for restless legs syndrome. Neuropharmacology 111, 160–168 (2016).
Eisenstein, S. A. et al. Prediction of striatal D2 receptor binding by DRD2/ANKK1 TaqIA allele status. Synapse 70, 418–431 (2016).
Schacht, J. P. COMT val158met moderation of dopaminergic drug effects on cognitive function: a critical review. Pharmacogenomics J. 16, 430–438 (2016).
Holst, S. C. et al. Functional polymorphisms in dopaminergic genes modulate neurobehavioral and neurophysiological consequences of sleep deprivation. Sci. Rep. 7, 45982 (2017).
Sigurdardottir, H. L. et al. Effects of norepinephrine transporter gene variants on NET binding in ADHD and healthy controls investigated by PET. Hum. Brain Mapp. 37, 884–895 (2016).
Elmenhorst, E. M. et al. Cognitive impairments by alcohol and sleep deprivation indicate trait characteristics and a potential role for adenosine A1 receptors. Proc. Natl Acad. Sci. USA 115, 8009–8014 (2018).
Acknowledgements
The excellent technical assistance of Mona-Larissa Ziegler (University of Münster) as well as of Magdalene Vögeling, Sylvia Köhler-Dibowski, Stephanie Krause, Dorothe Krug, Annette von Wächter, Barbara Elghahwagi, Susanne Schaden, Silke Frensch, Elisabeth Theelen, and Lutz Tellmann (all of the Forschungszentrum Jülich) is gratefully acknowledged. Further, we thank the department of radiochemistry (INM-5) of Forschungszentrum Jülich for the supply of the radioligand. Support for our study was provided by the Deutsche Forschungsgemeinschaft (SFB TRR58 TP A08 and Z02).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
J. Deckert reports joint projects with P1Vital and BioVariance funded by the EU and the Bavarian secretary of commerce, respectively, and funding of his research by DFG, BMBF, and Vogel Foundation. D. Elmenhorst received funding by ERA-NET NEURON project SleepLess supported by BMBF (01EW1808), FWO and FRQS under the frame of Neuron Cofund. All other authors declare no potential conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hohoff, C., Kroll, T., Zhao, B. et al. ADORA2A variation and adenosine A1 receptor availability in the human brain with a focus on anxiety-related brain regions: modulation by ADORA1 variation. Transl Psychiatry 10, 406 (2020). https://doi.org/10.1038/s41398-020-01085-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-020-01085-w
This article is cited by
-
Repeated caffeine intake suppresses cerebral grey matter responses to chronic sleep restriction in an A1 adenosine receptor-dependent manner: a double-blind randomized controlled study with PET-MRI
Scientific Reports (2024)
-
Adenosine protects D-galactose induced alterations in rat model of aging via attenuating neurochemical profile and redox status
Metabolic Brain Disease (2022)