Abstract
Alzheimer’s disease (AD) is the most common cause of age-related dementia and is currently incurable. The failures of current clinical trials and the establishment of modifiable risk factors have shifted the AD intervention from treatment to prevention in the at-risk population. Previous studies suggest that there is a geographic overlap between AD incidence and spicy food consumption. We previously reported that capsaicin-rich diet consumption was associated with better cognition and lower serum Amyloid-beta (Aβ) levels in people aged 40 years and over. In the present study, we found that intake of capsaicin, the pungent ingredient in chili peppers, reduced brain Aβ burden and rescued cognitive decline in APP/PS1 mice. Our in vivo and in vitro studies revealed that capsaicin shifted Amyloid precursor protein (APP) processing towards α-cleavage and precluded Aβ generation by promoting the maturation of a disintegrin and metalloproteinase 10 (ADAM10). We also found that capsaicin alleviated other AD-type pathologies, such as tau hyperphosphorylation, neuroinflammation and neurodegeneration. The present study suggests that capsaicin is a potential therapeutic candidate for AD and warrants clinical trials on chili peppers or capsaicin as dietary supplementation for the prevention and treatment of AD.
Similar content being viewed by others
Introduction
Alzheimer’s disease (AD), the most common cause of age-related dementia, causes heavy social and economic burdens. There is no effective therapy to cure AD or even to halt the course of the disease. With the failure of a series of clinical trials, the strategy of AD treatment has recently shifted to disease prevention, with clinical testing carried out in at-risk populations1,2,3,4,5. In fact, our analysis suggests that one-third of AD cases worldwide are attributable to several common modifiable risk factors6,7,8, most of which can be controlled by healthy dietary and lifestyle, making dietary and lifestyle intervention research hotspots9,10.
Chili pepper is a basic element of culinary culture consumed worldwide as vegetable and spice. Capsaicin is the major component in chili pepper, accounting for the spicy/pungent flavour. Previous studies suggest that there is an interesting geographic overlap between AD incidence and spicy food consumption in China. The incidence of AD in west China (3.99/1000 person-years) is lower than that in the east (5.58/1000 person-years)11, and in the west, the proportion of dishes with chili is higher and the pungency degree is greater than in the east12,13. Previously, we reported that capsaicin-rich diet consumption was associated with better cognition and lower serum amyloid-β (Aβ) levels in people aged 40 years and over14. These findings imply that capsaicin may be protective against AD.
In the present study, we aimed to further investigate whether capsaicin plays protective roles in AD pathogenesis. We investigated the effects of capsaicin on brain Aβ burden and cognition in APPswe/PS1dE9 (APP/PS1) mice, and explored the underlying mechanisms in human neuroblastoma SH-SY5Y-APP695 cells and APP/PS1 mice. Additionally, we observed the effects of capsaicin on other AD-type pathologies, including tau hyperphosphorylation, neuroinflammation, and neurodegeneration in APP/PS1 mice.
Methods
Cell culture and treatment
Human neuroblastoma SH-SY5Y-APP695 cells were stably transfected with the human wild-type APP69515. The cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 4 mM l-glutamine and 4500 mg/L glucose (HyClone, USA) at 37°C and 5% CO2. The medium was supplemented with 10% foetal bovine serum (FBS, HyClone, USA). ZeocinTM selection antibiotic (Invitrogen, USA) were added (100 μg/ml) to avoid bacterial contamination and mycoplasma contamination tests were conducted before analysis. To observe the dose-effect relationship between capsaicin and Aβ generation, the cells were incubated without (control) or with various concentrations of capsaicin (0.1, 1, 5, 10, 50 μM) (Sigma-Aldrich, USA) for 24 h. The culture medium did not contain FBS when the cells were treated with capsaicin. The cell culture medium of each group was subjected to ELISA for Aβ40 and Aβ42 concentrations, while the cell lysates were subjected to Western blot for APP metabolites and ELISA for Aβ40 and Aβ42 concentrations in triplicate.
MTT assay
To evaluate the toxicity of capsaicin, the cells were seeded in 96-well plates (5 × 104 cells/ml) and incubated without (control) or with various concentrations of capsaicin (5, 10, 25, 50, 75, 100, 125, 150 μM) for 24 h. The cell culture medium was changed immediately before the MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] assay to avoid an interaction between MTT and capsaicin. Then 10 μl MTT (5 mg/ml, Sigma-Aldrich, USA) solution was added for 4 h. The supernatant was then removed, and 100 μl DMSO was added to dissolve the dye crystals. Absorbance at 490 nm was read. Capsaicin impaired cell viability at concentrations >75 μM. At the concentration of 75 μM, cell viability presented impaired tendency (Supplemental Fig. 1).
Animals and treatment
All mouse husbandry procedures and operations in the present study were approved by the Third Military Medical University Animal Welfare Committee. The APP/PS1 transgenic (Tg) mice on C57BL/6 background and C57BL/6 wild-type (Wt) mice were obtained from Jackson Laboratory and bred in the animal facility of Daping Hospital. The mice were housed under a 12 h light/dark cycle with free access to food and water. Tg mice were given the normal standard chow (Tg control groups) or normal chow plus with 0.01% capsaicin16,17 (Tg cap group) at randomization from 3 months old to 9 months old (n = 8 per group, half female and half male). Based on the amount of consumed food per day and the weight of mice, we approximately estimated that the daily intake of capsaicin was about 30 mg/kg. Age- and sex-matched WT mice were used as baseline controls.
Behavioural tests
A battery of behavioural tests comprising the Morris water maze (MWM), Y-maze and open-field tests were conducted to assess behavioural performance of participant mice as previously described18,19. In brief, the MWM test was conducted in a circular pool with 120 cm in diameter, which was filled with opaque water stained with milk and surrounded by a set of spatial cues. The tank was imaginarily divided into four quadrants. A platform with 9 cm diameter was submerged 1 cm under water surface in a quadrant. The MWM test consisted of three platform trials per day for 5 consecutive days, followed by a probe trial. In the platform trial, the mouse navigated in the pool to locate the platform and was then able to escape. If the mouse failed to locate the platform within 60 s, it was directed to the platform. The mouse was allowed to remain on the platform for 60 s once it escaped onto the platform. The distance of the path taken and the escape latency were measured to test spatial learning ability. In the probe trial, the platform was withdrawn. The mouse navigated in the pool freely for 1 min. The time spent in each quadrant and the number of annulus crossings were recorded to assess memory consolidation. The Y-maze tests were comprised of the spontaneous alternation test and novel arm exploration test. The Y-maze apparatus consists of three enclosed yellow Plexiglas arms (31 cm long, 8 cm wide and 31 cm high) at 120° angles to each other, radiating out from the central point. In the spontaneous alternation test, the mouse was placed at the end of one arm facing the wall and allowed to walk freely in the maze for a 5 min session. Alternation, aiming to test spatial working memory, was defined as consecutive entries into the three arms on overlapping triplet sets without repetition. In the novel arm exploration test, the mouse was placed at the end of home arm and allowed to explore the maze for 5 min with one arm (novel arm) closed in the training trial. The mouse was returned to their home cage until the retrieval trial, during which the mouse was allowed to explore all three arms freely within the maze for 5 min. The percentage of novel arm entry and time spent in the novel arm were analyzed to measure spatial recognition memory. In the open-field test, the mouse was placed in the centre of the open-field apparatus (50 cm length, 50 cm wide and 45 cm high), and allowed to explore freely for 5 min. The total travelling distance was recorded to measure the spontaneous locomotor activity, whereas the ratio of time in the central zone to the peripheral zone and the numbers of rearings, groomings, defecations, and urinations were recorded to determine anxiety-like behaviour. All performances were video-recorded and analyzed with image-analyzing software (ANY-maze, Stoelting, Wood Dale, IL, USA).
Brain sampling
The mice were sacrificed at 9 months old. There was no significant difference in body weight of mice among each group. Blood was drawn from the eyes, followed by intracardial perfusion with 100 ml of 0.1% NaNO2 in normal saline under anaesthesia. Brains were sampled as described previously20. Briefly, the left hemisphere was fixed with 4% paraformaldehyde, and coronal sections were cut at 35-μm thickness for histological analysis, while the right hemisphere was snap-frozen and ground into powder in liquid nitrogen, divided into three vials, weighed, and stored at −80 °C for biochemical analysis. Brain sampling and histological/biochemical analysis were conducted by different investigators for blinding.
Histology and quantification
Aβ plaques and cerebral amyloid angiopathy (CAA)
For Aβ plaques in parenchyma, brain tissue sections were stained with Congo red for compact Aβ plaques or with 6E10 antibody using a free-floating immunohistochemistry (IHC) method for total Aβ plaques containing compact and diffuse plaques20. The area fraction and plaque number of Congo red- or 6E10-positive staining in neocortex and hippocampus were quantified with ImageJ software. CAA was visualized with Congo red staining and manually selected from two slices of the hippocampus21, CAA number per slice were quantified.
In histology and quantification, a series of five equally spaced tissue sections (~1.3 mm apart) spanning the entire brain of each mouse were used for immunohistochemistry staining (Supplemental Fig. 2). All the sections were stained and photos were taken under the same conditions at the same time. Images were analyzed with ImageJ software under the same conditions in a blinded manner to the group information of the sections.
Tau pathology, neuroinflammation, and neurodegeneration
Immunohistochemistry was used to detect phosphorylated tau with anti-pS396-tau antibody (Signalway, USA), microgliosis with anti-CD68 antibody (Abcam, UK), and astrocytosis with anti-glial fibrillary acidic protein (GFAP) antibody (Abcam, UK). Apoptosis of neurons was measured by double-immunofluorescence staining for NeuN (Abcam, UK) and caspase-3 (Millipore, USA). Neuronal loss and neurite degeneration were detected by double-immunofluorescence staining for NeuN and microtubule-associated protein (MAP)-2 (Millipore, USA). The fraction of positive staining as a proportion of total area and integrated fluorescence intensity were quantified with ImageJ software.
ELISA
One vial of brain powder was used for sequential protein extraction in Tris buffer solution (TBS), 2% sodium dodecyl sulfonate (SDS) and 70% formic acid (FA). The levels of human Aβ42 and Aβ40 in the TBS, SDS, and FA were measured using ELISA kits (Invitrogen, USA) according to the manufacturer’s instructions. Inflammatory factors, including mouse tumour necrosis factor-alpha (TNF-α), interferon-γ (IFN-γ), interleukin-1β (IL-1β) and IL-6, in brain homogenates were measured using corresponding ELISA kits (R&D Systems, USA).
RNA extraction and quantitative real-time PCR
The total RNA was extracted from brain powder using TRIzol reagent (Life Technologies, USA) according to the manufacturer’s instructions. For each RNA sample, an equal amount of total RNA (1 μg) was reverse-transcribed into cDNA using iScriptTM cDNA synthesis kit (BIO-RAD, USA). qRT-PCR was performed on a CFX96TM Real-Time System (BIO-RAD, USA) with cDNA (equivalent to 50 ng RNA per 20 μl PCR assay).
Western blot
For Western blot analysis, one vial of brain powder was suspended in RIPA buffer, and proteins were extracted. Identical amounts of RIPA-extracted protein were loaded and separated by 4-20% PAGE Gels (KeyGEN BioTECH, China) and transferred to nitrocellulose membranes. The blots were probed with the following primary antibodies: anti-APP C-terminal antibody (Millipore, USA) to detect C-terminal fragment (CTF)-α and CTF-β, 6E10 (BioLegend, USA) to detect Aβ, full-length APP (APPfl), and secreted APP (sAPP)-α (sAPPα); anti-a disintegrin and metalloproteinase 10 (ADAM10) antibody (Abcam, UK); anti-β-secretase 1 (BACE-1) antibody (Abcam, UK); anti-Presenilin-1 (PS-1) antibody (Abcam, UK); anti-transient receptor potential vanilloid 1 (TRPV1) antibody (Millipore, USA); anti-peroxisome proliferator-activated receptor α (PPARα) antibody (Abcam, UK); anti-insulin-degrading enzyme (IDE) antibody (Millipore, USA); anti-neprilysin (NEP) antibody (Millipore, USA); anti-receptor for advanced glycation end products (RAGE) antibody (Millipore, USA); anti-lipoprotein receptor-related protein 1 (LRP-1) antibody (Abcam, UK); anti-pS199-tau, anti-pS396-tau, anti-pT231-tau, Tau5 antibodies (Signalway, USA); anti-Synapsin-1 (SYN1) antibody (Millipore, USA); anti-postsynaptic density protein 95 (PSD95) antibody (Millipore, USA); anti-synaptosomal associated protein 25 (SNAP25) antibody (Millipore, USA); anti-vesicle-associated membrane protein 1 (VAMP1) antibody (Abcam, UK); anti-β-actin antibody (Sigma, USA). The membranes were incubated with IRDye 800 CW secondary antibodies (Li-COR, USA) and scanned using the Odyssey fluorescent scanner. The band density was normalized to β-actin for analysis.
Statistics
The results are presented as the mean ± SEM unless otherwise stated. Statistical comparisons between two groups were made using Student’s t test or the Mann–Whitney U test, as applicable. One-way ANOVA and Tukey’s test were used to compare three groups, and two-way ANOVA was used to compare two groups at multiple timepoints. P values less than 0.05 (two-sided) were considered significant. All analyses were performed with GraphPad Prism software, version 7.0, or SPSS software, version 20.0.
Results
Dietary capsaicin rescues cognition impairment in APP/PS1 mice
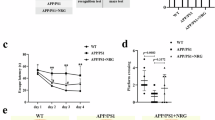
To investigate the effects of dietary capsaicin on cognition impairment in AD, we conducted capsaicin prevention experiments in APP/PS1 mice. The mice were feed with 0.01% capsaicin-rich diet or standard diet from 3 months of age when Aβ pathologies are not formed, and were subjected to analysis at 9 months old, when extensive Aβ pathologies and obvious cognitive impairment occur. APP/PS1 control mice displayed an impaired learning ability and spatial reference memory compared with Wt mice, while the mice received capsaicin diet presented improved spatial learning ability, as reflected by a significant reduction in the escape latency, and better memory consolidation, reflected by a higher number of platform area crossings and more time spent in the target quadrant compared with APP/PS1 controls in the MWM test (Fig. 1a, b). There was no difference in swimming speed during platform trials or path length to the platform during probe trials among each groups (Supplemental Fig. 3a, b). APP/PS1 mice received the capsaicin diet also showed more entries into and more time spent in the novel arm in the Y-maze test, a reflection of better spatial recognition memory (Fig. 1c). However, the mice did not display different performance in the spontaneous exploration test of Y-maze (Supplemental Fig. 3c, d). In open-field tests, the capsaicin-treated mice showed a longer travelling distance, a higher number of rearing and a reduced ratio of time spent in the central zone to the peripheral zone, suggesting an enhanced locomotor activity and anxiety-like behaviour (Fig. 1d, e). The above findings indicate that dietary capsaicin can protect against cognitive decline in APP/PS1 mice.
a Escape latency during platform trials in Morris water maze (two-way ANOVA) and representative tracing graphs in platform trials in Morris water maze. b Times of crossing platform and time spent in target quadrant in probe test in Morris water maze (one-way ANOVA followed by Turkey’s test), and representative tracing graphs in probe test in Morris water maze. c Percentage of novel arm entries and time spent in novel arm in Y-maze test. d Distance travelled, number of rearings and the ratio of time spent in central and peripheral areas in open-field test (one-way ANOVA followed by Turkey’s test). e Representative tracing graphs in open-field test. N = 8 per group. Values are presented as the mean ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, two-sided. Tg APP/PS1 transgenic mice, WT wild-type mice, Cap Capsaicin, Ctrl Control.
Dietary capsaicin attenuates Aβ pathologies in APP/PS1 mice
We investigated whether capsaicin treatment before Aβ plaque formation would inhibit Aβ deposition in the brain of APP/PS1 mice. We performed Aβ IHC staining (6E10) for total Aβ plaques and Congo red staining for compact Aβ plaques. Compared with APP/PS1 controls, the mice treated with capsaicin displayed significant reductions in area fraction and plaque density of both total and compact plaques in neocortex and hippocampus (Fig. 2a, b). In addition, Congo red staining showed that Aβ deposition in vessel walls, which reflects the formation of CAA, decreased in mice treated with capsaicin (Fig. 2c, d). Consistent with these data, ELISA assays showed significantly lower levels of Aβ42, Aβ40 and total Aβ in the TBS (extracellular soluble Aβ), SDS (intracellular soluble Aβ) and formic acid (insoluble Aβ) fractions of brain homogenate extracts in mice treated with capsaicin relative to controls (Fig. 2e). Based on the ELISA results, we calculated that dietary capsaicin reduced total Aβ burden by 32.3% in the brain of APP/PS1 mice. Taken together, our findings indicate that dietary capsaicin has preventive potential for AD.
a Representative images of Congo red staining and 6E10 immunohistochemical (IHC) staining in Tg Ctrl and Tg Cap groups. Insets show the representative plaque at higher magnification. Scale bar, 500 μm. b Comparison of 6E10- or Congo red-positive plaques in the neocortex (NC) and hippocampus (HC). c Representative image of cerebral amyloid angiopathy (CAA) visualized using Congo red staining. Insets show the representative morphology of CAAs at higher magnification. Other CAAs are marked with a black star. Scale bar, 200 μm. d Comparison of CAA numbers between Tg Ctrl and Tg Cap mice. e Comparison of Aβ40, Aβ42 and total Aβ levels measured with ELISA in TBS, SDS and formic acid (FA) fractions of brain homogenates. Aβ levels are normalized to the weight of brain tissue. N = 8 per group, Student’s t-test, two-sided. *p < 0.05, **p < 0.01, ***p < 0.001. Bars express mean ± SEM. Tg APP/PS1 transgenic mice, Cap Capsaicin, Ctrl Control.
Capsaicin inhibits Aβ generation via promoting non-amyloidogenic processing of APP
To explore the mechanism underlying Aβ reduction by capsaicin, SH-SY5Y-APP695 cells, which are human neuroblastoma cells overexpressing human wild-type APP695, were treated with or without various concentrations of capsaicin. Aβ is derived from sequential cleavage of APP by β-secretase and γ-secretase within neurons, and then secreted to extracellular space22. First, we measured Aβ40 and Aβ42 levels in cell lysates and culture medium. The results showed that capsaicin treatment decreased levels of both Aβ40 and Aβ42 in SH-SY5Y-APP695 cell lysates in a dose-dependent manner (Fig. 3a, b). Aβ40 levels in culture medium were also dose-dependently reduced by capsaicin (Fig. 3c), whereas Aβ42 in culture medium was undetectable due to the low concentration (data not shown). Next, we examined the levels of full-length APP and products of APP processing in cell lysates. We found that capsaicin did not affect the expression of full-length APP, but dose dependently increased the ratio of CTF-α/CTF-β (Fig. 3d–f). Our in vitro results suggest that capsaicin inhibits Aβ generation via shifting APP processing towards α-cleavage.
a–c Aβ42 and Aβ40 levels in cell lysates and Aβ40 levels in culture medium measured with ELISA in SH-SY5Y-APP695 cells treated with or without various concentrations of capsaicin for 24 h. (N = 3, mean ± SEM; Student’s t test, two-sided. **p < 0.01, ***p < 0.001). d Western blots for full-length APP (APPfl), CTF-α and CTF-β in SH-SY5Y-APP695 cells treated with or without various concentrations of capsaicin for 24 h. e, f Quantitative analysis of APPfl and the ratio of CTF-α/CTF-β in SH-SY5Y-APP695 cell lysates measured by Western blot (N = 3, mean ± SEM; Student’s t test, two-sided. *p < 0.05). g Western blots and quantitative analysis for APP and APP metabolites in brain homogenates of Tg Ctrl and Tg Cap mice (N = 8 per group, mean ± SEM; Student’s t test, two-sided. *p < 0.05, **p < 0.01). h Western blots and quantitative analysis of APP cleavage enzymes and ADAM10 mRNA levels quantified by qRT-PCR in brain homogenates (N = 8 per group, mean ± SEM; Student’s t test, two-sided. **p < 0.01). i Western blots and quantitative analysis of PPARα and TRPV1 in brain homogenates of Tg Ctrl and Tg Cap mice (N = 8 per group, mean ± SEM; Student’s t test, two-sided. *p < 0.05). j Western blots and quantitative analysis of Aβ-degrading and Aβ-transporting receptors in the blood-brain barrier in brain homogenates (N = 8 per group, mean ± SEM; Student’s t test, two-sided.). Tg APP/PS1 transgenic mice, Cap Capsaicin, Ctrl Control.
Next, to verify the effects of capsaicin on APP processing, we measured APP and its processing products in brain homogenates of APP/PS1 mice. Consistent with in vitro results, capsaicin did not affect the expression of full-length APP (Fig. 3g). We found that sAPPα and the ratio of CTF-α/CTF-β significantly increased in the brain of mice treated with capsaicin compared with controls (Fig. 3g). Our in vivo results support that capsaicin inhibits Aβ production via promoting non-amyloidogenic processing of APP.
To further verify above findings, we measured levels of secretases responsible for APP processing. ADAM10 is the major α-secretase that catalyses α-cleavage and promotes non-amyloidogenic processing of APP. ADAM10 is first generated as an inactive proenzyme (proADAM10) and matures into an active protease after the removal of its prodomain23,24. We found that capsaicin treatment significantly increased brain levels of matADAM10 relative to APP/PS1 controls but did not affect proADAM10 protein or mRNA expression (Fig. 3h), suggesting that capsaicin increases the level of ADAM10 by promoting its maturation or activation. However, no significant differences were observed in BACE1 or PS1 responsible for amyloidogenic processing of APP (Fig. 3h).
It has been reported that activation of PPARα, a transcription factor regulating genes involved in fatty acid metabolism, could stimulate ADAM10-mediated proteolysis of APP25. We measured PPARα levels in the brain of APP/PS1 mice and found that PPARα levels increased in the brain of mice treated with capsaicin compared with controls (Fig. 3i), implying that capsaicin may activate ADAM10-mediated APP processing via upregulating PPARα, and thus precluding Aβ generation. We also detected the levels of TRPV1, which is known as capsaicin receptor. Unfortunately, we did not observe significant differences in TRPV1 expression between two groups (Fig. 3i).
Additionally, we tested Aβ clearance-related molecules, including the Aβ-degrading enzymes IDE and NEP, and the Aβ transporters LRP-1 and RAGE, across the blood–brain barrier (BBB). None of these proteins showed significant differences between the two groups (Fig. 3j). Our findings indicate that capsaicin shifts APP processing towards α-cleavage and precludes Aβ generation by promoting the maturation of ADAM10.
Dietary capsaicin attenuates other AD-type pathologies in APP/PS1 mice
We investigated whether capsaicin could affect other AD-type pathologies in the brain of APP/PS1 mice. Capsaicin significantly attenuated hyperphosphorylation of tau in different brain regions and different phosphorylation sites. Phospho-Tau (pS396)-positive neurons in both neocortex and hippocampus were significantly decreased in the brain of capsaicin-treated mice compared to controls (Fig. 4a). The levels of tau phosphorylation at multiple epitopes, including the Ser199 (pS199), pS396 and Thr231, were reduced in the capsaicin group, whereas total tau (tau5) showed no significant differences between the two groups (Fig. 4b). Neuroinflammation was significantly ameliorated in the brain of capsaicin-treated mice compared with APP/PS1 controls, as reflected by decreased levels of activated microglia and astrocytes, and reduced levels of proinflammatory factors, including TNF-α, IFN-γ, and IL-6 (Fig. 4c, d). Capsaicin also attenuated neurodegeneration in APP/PS1 mice. Immunoreactivities of synapse-related proteins, including PSD95, SYN1, SNAP25 and VAMP1, were significantly elevated in the brain of capsaicin-treated APP/PS1 mice (Fig. 4e). Compared with APP/PS1 control mice, the capsaicin-treated mice displayed an increased fluorescence intensity of staining for NeuN and Map-2 and a decreased intensity of activated caspase-3 in the hippocampus (Fig. 4f). Taken together, these findings suggest that capsaicin protects against tau hyperphosphorylation, neuroinflammation and neurodegeneration in the brain of APP/PS1 mice.
a Representative images of PS396 immunohistochemical staining and quantitative analysis of pS396-positive staining in the neocortex and hippocampus. Insets show the representative morphology at higher magnification. Scale bar, 50μm. b Western blots and quantification of phosphorylated tau at multiple sites in brain homogenates. c Immunostaining and quantification of activated microgliosis (CD68) and astrocytosis (GFAP) in neocortex and hippocampus. Insets show the representative morphology at higher magnification. Scale bar, 500μm. d ELISA assays of proinflammatory factors in brain homogenates of Tg Ctrl and Tg Cap mice. e Western blot and quantitative analysis of synapse-related proteins in brain homogenates. f Representative images and quantification of fluorescence intensity (relative integrated density) of neurons and dendrites in the CA1 region of the hippocampus stained with anti-NeuN and anti-MAP-2 immunofluorescence and neuronal apoptosis in the CA3 region stained with activated caspase-3 immunofluorescence. Scale bar, 20μm. N = 8 per group. Data are presented as mean ± SEM. Student’s t test, two-sided. *p < 0.05, **p < 0.01, ***p < 0.001. Tg APP/PS1 transgenic mice, CapCapsaicin, Ctrl Control, IntDen integrated density.
Discussion
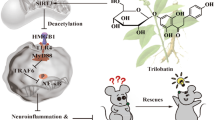
The aetiology of AD is multifactorial, and several potentially modifiable risk factors and protective factors have been identified. Dietary patterns have been linked with the risk of AD26,27,28,29. Healthy diet, as one component of the approach of multidomain lifestyle interventions, has showed beneficial effects on the prevention of cognitive impairment in several trials1,30,31,32. In the present study, we showed that dietary capsaicin, the major pungent ingredient in chili peppers, effectively reduced brain Aβ burden, attenuated neurodegeneration and improved cognition in APP/PS1 mice; our in vivo and in vitro studies indicated that capsaicin inhibited Aβ generation via promoting non-amyloidogenic processing of APP. Overproduction of Aβ plays a key role in the pathogenesis of AD33,34. Aβ is generated by the sequential cleavage of APP via β- and γ-secretases. Alternatively, APP can be cleaved by α-secretase within the Aβ domain, which precludes Aβ generation. Increased α-secretase activity competitively causes decreased β-secretase processing of APP and Aβ production35. ADAM10 is the major α-secretase responsible for ectodomain shedding of APP in the brain36,37. In the present study, we found that capsaicin treatment increased the maturation of ADAM10 and thereby precluded Aβ generation (Fig. 5). Additionally, capsaicin also upregulated the levels of PPARα, which could activate ADAM10-mediated proteolysis of APP25, suggesting that capsaicin might activate ADAM10 via upregulating PPARα.
APP processing includes non-amyloidogenic and amyloidogenic pathways. In the amyloidogenic pathway, Aβ is generated by the sequential cleavage of APP by β- and γ-secretases. In the non-amyloidogenic pathway, APP is cleaved by α-secretase within the Aβ domain, which precludes Aβ generation. Increased α-secretase activity competitively inhibits amyloidogenic processing of APP and decreases Aβ production. ADAM10 is the major α-secretase responsible for ectodomain shedding of APP in the brain. proADAM10 is expressed in endoplasmic reticulum and processed in Golgi where the prodomain of proADAM10 is removed and mature ADAM10 (matADAM10) is generated; then matADAM10 is transported to the plasma membrane and executes catalytic functions. Capsaicin promotes the maturation of ADAM10, thereby shifts APP processing from amyloidogenic pathway to non-amyloidogenic pathway and precludes Aβ generation.
In AD patients, ADAM10 activity has been reduced in both CSF and brain samples38,39. Recently, several rare mutations have been associated with late-onset AD (LOAD) in the prodomain of ADAM10, and two mutations promote amyloid pathology by diminishing α-secretase activity40. This year, a genome-wide association study (GWAS) identified a common variant in ADAM10 which was associated with increased AD risk41, which has been confirmed in Han Chinese population42. These findings suggest that decreased levels and activity of ADAM10 are involved in AD pathogenesis, and ADAM10 is a promising therapeutic target43. The overexpression of ADAM10 can reduce Aβ levels and prevent its deposition in plaques, as well as rescue cognitive defects in animals35. Several drugs intended as indirect α-secretase activators have progressed to the clinical trial stage for AD, such as GABA receptor modulator etazolate, 5-HT4 agonist PRX-03140, and a polyphenolic compound from green tea, epigallocatechin-gallate, but no results have been published to date (NCT00880412, NCT00693004, NCT00951834). At present, no direct α-secretase or ADAM10 activators are used for AD treatment. Our findings suggest that capsaicin is a natural ADAM10 activator and shows potential to attenuate amyloid pathology and protect against AD. However, further mechanism studies, such as inhibition or knockout of ADAM10, are needed to demonstrate ADAM10-mediated inhibition effects of capsaicin on Aβ generation.
TRPV1 is known as capsaicin receptor. Recent studies has reported that genetic upregulation of TRPV1 attenuated Aβ burden in brain of AD model mice and rescued memory decline and Aβ-induced neuronal function and network impairment in vivo and in vitro44,45, although controversial report exists46. In our present study, we did not detected significant differences in TRPV1 expression between mice received capsaicin-rich diet and those received standard diet. TRPV1 is a calcium-permeable non-selective cation channel, it is possible that dietary capsaicin activates TRPV1 via altering its spatial conformation, but doesn’t affect its expression; another possible explanation is that the inhibition effects of capsaicin on Aβ generation is not, at least not completely, dependent on TRPV1 activation.
Our previous cohort study suggests that chili consumption is protective against cognitive impairment in subjects aged 40 years and older14. A very recent paper suggests that higher chili intake is associated with worse memory decline in an open cohort study conducted in China47. However, this study had big biases as it used the objective self-reported memory decline, and the subjects who had higher consumption of chili food were lower educated. Our present findings further support the protective effects of chili consumption on cognition. In previous studies, it has been reported that capsaicin could improve cognitive performance in wild-type mice or rats but with controversy45,48,49, all studies did not find obvious adverse effects of capsaicin administration. Further studies are required to make clear whether capsaicin can enhance memory in wild-type animals or healthy humans. Additionally, capsaicin has been reported to ameliorate tau changes and behaviour impairments in Aβ-independent pathway in stress- or drug-induced nonspecific AD models which only display tau pathologies and cognitive impairment but no Aβ plaques50,51,52. In our present study, APP/PS1 transgenic AD mouse model was used. In this mouse model, tau hyperphosphorylation is secondary to Aβ pathologies. Therefore in our study, the reduction of tau phosphorylation and pathologies should be mainly resulted from the reduction of Aβ production due to capsaicin. Previous and our present studies suggest that capsaicin protects against AD by targeting multiple pathogenesis of disease. Contrast to our results, another study showed that subcutaneous injection of capsaicin enhanced the level of membrane-bound APP in Sprague–Dawley rats53. In this study, capsaicin was given by subcutaneous injection twice in two days which is an acute or short-term treatment, but in our present study and studies mentioned above, capsaicin was given by oral administration or intragastric infusion for a chronic long-term treatment. This may explain the different results among these studies. The dose of capsaicin used in the current study (30 mg/kg) is higher than daily consumption of human. It has been reported that low-dose of capsaicin (1–2 mg/kg) exerted antiepileptic effects54,55, whereas high-dose of capsaicin (10–120 mg/kg) exerted proepileptic effects in animal models56,57. In our study, we did not find the occurrence of obvious seizures or increased mortality in capsaicin-treated mice. The safe and effective doses of capsaicin for AD in both humans and animals need to be determined in future studies.
Capsaicin, as a natural component of spicy food, has potential advantages as an AD intervention strategy. Considering that chili peppers have been a vital part of culinary cultures worldwide and have a long history of application for flavouring, they are feasible to utilized for AD prevention. In addition, capsaicin is a potential therapeutic molecule for various human diseases, such as obesity, cardiovascular diseases, hypertension, and atherosclerosis, which are established risk factors for AD16,17,58. Taken together, the current and previous findings suggest capsaicin may prevent AD by targeting multiple pathways that drive the pathogenesis of AD.
In conclusion, we uncovered an application of capsaicin, the pungent ingredient in chili peppers, as a promising therapy for AD. Capsaicin has moved towards clinical applications and is used currently in topical creams and gels to relieve intractable neuropathic pain, uremic pruritus, and rheumatoid arthritis. Our findings warrant future clinical trials on chili peppers or capsaicin as dietary supplementation for the prevention of AD. Meanwhile, global epidemiological studies are worthy to explore the association between capsaicin-rich diet and AD prevalence.
References
Kivipelto, M., Mangialasche, F. & Ngandu, T. Lifestyle interventions to prevent cognitive impairment, dementia and Alzheimer disease. Nat. Rev. Neurol. 14, 653–666 (2018).
Miller, G. Alzheimer’s research. Stopping Alzheimer’s before it starts. Science 337, 790–792 (2012).
Sperling, R., Mormino, E. & Johnson, K. The evolution of preclinical Alzheimer’s disease: implications for prevention trials. Neuron 84, 608–622 (2014).
Andrieu, S., Coley, N., Lovestone, S., Aisen, P. S. & Vellas, B. Prevention of sporadic Alzheimer’s disease: lessons learned from clinical trials and future directions. Lancet Neurol. 14, 926–944 (2015).
Fan, D. Y. & Wang, Y. J. Early intervention in Alzheimer’s disease: how early is early enough? Neurosci. Bull. 36, 195–197 (2020).
Norton, S., Matthews, F. E., Barnes, D. E., Yaffe, K. & Brayne, C. Potential for primary prevention of Alzheimer’s disease: an analysis of population-based data. Lancet Neurol. 13, 788–794 (2014).
Solomon, A. et al. Advances in the prevention of Alzheimer’s disease and dementia. J. Intern Med 275, 229–250 (2014).
Xu, W. et al. Meta-analysis of modifiable risk factors for Alzheimer’s disease. J. Neurol. Neurosurg. Psychiatry 86, 1299–1306 (2015).
An, Y. et al. Longitudinal and nonlinear relations of dietary and Serum cholesterol in midlife with cognitive decline: results from EMCOA study. Mol. Neurodegener. 14, 51 (2019).
Wakabayashi, T. et al. Differential effects of diet- and genetically-induced brain insulin resistance on amyloid pathology in a mouse model of Alzheimer’s disease. Mol. Neurodegener. 14, 15 (2019).
Yuan, J. et al. Incidence of dementia and subtypes: a cohort study in four regions in China. Alzheimers Dement 12, 262–271 (2016).
Wang, S., Cheng, L., He, S. & Xie, D. Regional pungency degree in China and its correlation with typical climate factors. J. Food Sci. 84, 31–37 (2019).
Sun, D. et al. Spicy food consumption is associated with adiposity measures among half a million Chinese people: the China Kadoorie Biobank study. BMC Public Health 14, 1293 (2014).
Liu, C. H. et al. The associations between a Capsaicin-rich diet and blood amyloid-beta levels and cognitive function. J. Alzheimers Dis. 52, 1081–1088 (2016).
Hooff, G. P., Peters, I., Wood, W. G., Muller, W. E. & Eckert, G. P. Modulation of cholesterol, farnesylpyrophosphate, and geranylgeranylpyrophosphate in neuroblastoma SH-SY5Y-APP695 cells: impact on amyloid beta-protein production. Mol. Neurobiol. 41, 341–350 (2010).
Yang, D. et al. Activation of TRPV1 by dietary capsaicin improves endothelium-dependent vasorelaxation and prevents hypertension. Cell Metab. 12, 130–141 (2010).
Zhang, L. L. et al. Activation of transient receptor potential vanilloid type-1 channel prevents adipogenesis and obesity. Circ. Res. 100, 1063–1070 (2007).
Jiao, S. S. et al. Edaravone alleviates Alzheimer’s disease-type pathologies and cognitive deficits. Proc. Natl Acad. Sci. USA 112, 5225–5230 (2015).
Shen, L.-L. et al. The ProNGF/p75NTR pathway induces tau pathology and is a therapeutic target for FTLD-tau. Mol. Psychiatry 23, 1813–1824 (2018).
Wang, Y. J. et al. Intramuscular delivery of a single chain antibody gene reduces brain Abeta burden in a mouse model of Alzheimer’s disease. Neurobiol. Aging 30, 364–376 (2009).
Wilcock, D. M., Gordon, M. N. & Morgan, D. Quantification of cerebral amyloid angiopathy and parenchymal amyloid plaques with Congo red histochemical stain. Nat. Protoc. 1, 1591–1595 (2006).
Zhang, X. & Song, W. The role of APP and BACE1 trafficking in APP processing and amyloid-beta generation. Alzheimers Res Ther. 5, 46 (2013).
Saftig, P. & Lichtenthaler, S. F. The alpha secretase ADAM10: a metalloprotease with multiple functions in the brain. Prog. Neurobiol. 135, 1–20 (2015).
Vincent, B. Regulation of the alpha-secretase ADAM10 at transcriptional, translational and post-translational levels. Brain Res. Bull. 126, 154–169 (2016).
Corbett, G. T., Gonzalez, F. J. & Pahan, K. Activation of peroxisome proliferator-activated receptor alpha stimulates ADAM10-mediated proteolysis of APP. Proc. Natl Acad. Sci. USA 112, 8445–8450 (2015).
Petersson, S. D. & Philippou, E. Mediterranean diet, cognitive function, and dementia: a systematic review of the evidence. Adv. Nutr. 7, 889–904 (2016).
Martinez-Lapiscina, E. H. et al. Mediterranean diet improves cognition: the PREDIMED-NAVARRA randomised trial. J. Neurol. Neurosurg. Psychiatry 84, 1318–1325 (2013).
Morris, M. C. et al. MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimers Dement. 11, 1007–1014 (2015).
Smith, P. J. et al. Effects of the dietary approaches to stop hypertension diet, exercise, and caloric restriction on neurocognition in overweight adults with high blood pressure. Hypertension 55, 1331–1338 (2010).
Ngandu, T. et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): a randomised controlled trial. Lancet 385, 2255–2263 (2015).
Andrieu, S. et al. Effect of long-term omega 3 polyunsaturated fatty acid supplementation with or without multidomain intervention on cognitive function in elderly adults with memory complaints (MAPT): a randomised, placebo-controlled trial. Lancet Neurol. 16, 377–389 (2017).
Rosenberg, A. et al. Multidomain lifestyle intervention benefits a large elderly population at risk for cognitive decline and dementia regardless of baseline characteristics: the FINGER trial. Alzheimers Dement 14, 263–270 (2018).
Selkoe, D. J. & Hardy, J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol. Med. 8, 595–608 (2016).
Hardy, J. & Selkoe, D. J. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297, 353–356 (2002).
Postina, R. et al. A disintegrin-metalloproteinase prevents amyloid plaque formation and hippocampal defects in an Alzheimer disease mouse model. J. Clin. Invest 113, 1456–1464 (2004).
Kuhn, P. H. et al. ADAM10 is the physiologically relevant, constitutive alpha-secretase of the amyloid precursor protein in primary neurons. EMBO J. 29, 3020–3032 (2010).
Jorissen, E. et al. The disintegrin/metalloproteinase ADAM10 is essential for the establishment of the brain cortex. J. Neurosci. 30, 4833–4844 (2010).
Tyler, S. J., Dawbarn, D., Wilcock, G. K. & Allen, S. J. alpha- and beta-secretase: profound changes in Alzheimer’s disease. Biochem. Biophys. Res. Commun. 299, 373–376 (2002).
Colciaghi, F. et al. [alpha]-Secretase ADAM10 as well as [alpha]APPs is reduced in platelets and CSF of Alzheimer disease patients. Mol. Med. 8, 67–74 (2002).
Suh, J. et al. ADAM10 missense mutations potentiate beta-amyloid accumulation by impairing prodomain chaperone function. Neuron 80, 385–401 (2013).
Jansen, I. E. et al. Genome-wide meta-analysis identifies new loci and functional pathways influencing Alzheimer’s disease risk. Nat. Genet. 51, 404–413 (2019).
Yang, Q. et al. Correlations between single nucleotide polymorphisms, cognitive dysfunction, and postmortem brain pathology in Alzheimer’s disease among Han Chinese. Neurosci. Bull. 35, 193–204 (2019).
Lichtenthaler, S. F. alpha-secretase in Alzheimer’s disease: molecular identity, regulation and therapeutic potential. J. Neurochem. 116, 10–21 (2011).
Balleza-Tapia, H. et al. TrpV1 receptor activation rescues neuronal function and network gamma oscillations from Abeta-induced impairment in mouse hippocampus in vitro. Elife 7, e37703 (2018).
Du Y. et al. TRPV1 activation alleviates cognitive and synaptic plasticity impairments through inhibiting AMPAR endocytosis in APP23/PS45 mouse model of Alzheimer’s disease. Aging Cell 19, e13113 (2020).
Kim, J. et al. Ca2+-permeable TRPV1 pain receptor knockout rescues memory deficits and reduces amyloid-beta and tau in a mouse model of Alzheimer’s disease. Hum. Mol. Genet. 29, 228–237 (2020).
Shi, Z. et al. High chili intake and cognitive function among 4582 adults: an open cohort study over 15 years. Nutrients 11, 1183 (2019).
Shiri, M. et al. Effects of cannabinoid and vanilloid receptor agonists and their interaction on learning and memory in rats. Can. J. Physiol. Pharm. 95, 382–387 (2017).
Li, H. B. et al. Antistress effect of TRPV1 channel on synaptic plasticity and spatial memory. Biol. Psychiatry 64, 286–292 (2008).
Shalaby, M. A., Nounou, H. A. & Deif, M. M. The potential value of capsaicin in modulating cognitive functions in a rat model of streptozotocin-induced Alzheimeras disease. Egyptian J. Neurol. Psychiatry Neurosurgery 55, 48 (2019).
Jiang, X. et al. Capsaicin ameliorates stress-induced Alzheimer’s disease-like pathological and cognitive impairments in rats. J. Alzheimers Dis. 35, 91–105 (2013).
Xu, W. et al. Capsaicin reduces Alzheimer-associated tau changes in the hippocampus of type 2 diabetes rats. PLoS ONE 12, e0172477 (2017).
Pakaski, M. et al. Capsaicin promotes the amyloidogenic route of brain amyloid precursor protein processing. Neurochem. Int. 54, 426–430 (2009).
Lee, T. H. et al. Capsaicin prevents kainic acid-induced epileptogenesis in mice. Neurochem. Int. 58, 634–640 (2011).
Abdel-Salam, O. M. E., Sleem, A. A., Sayed, M., Youness, E. R. & Shaffie, N. Capsaicin exerts anti-convulsant and neuroprotective effects in pentylenetetrazole-induced seizures. Neurochem. Res. 45, 1045–1061 (2020).
Carletti, F., Gambino, G., Rizzo, V., Ferraro, G. & Sardo, P. Neuronal nitric oxide synthase is involved in CB/TRPV1 signalling: focus on control of hippocampal hyperexcitability. Epilepsy Res. 138, 18–25 (2017).
Naziroglu, M. & Ovey, I. S. Involvement of apoptosis and calcium accumulation through TRPV1 channels in neurobiology of epilepsy. Neuroscience 293, 55–66 (2015).
Edwards, J. G. TRPV1 in the central nervous system: synaptic plasticity, function, and pharmacological implications. Prog. Drug Res. 68, 77–104 (2014).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (NSFC) (Grant Nos. 81600949, 81625007, 91749206, 81601112, 31921003 and 81721001).
Author information
Authors and Affiliations
Contributions
Y.J.W. and J.W. conceived and designed the project, J.W., B.L.S., Y.X., D.Y.T., W.W.L., L.L.S., W.S. and Z.M.Z. conducted animal and in vitro exsperiments, J.W., B.L.S., Y.H.L., X.L.B., W.S.J., G.H.Z., W.X., H.D.Z., Z.W. and L.Y.C. performed human sample experiments, J.W., B.L.S., X.W.C, Z.H., Z.M.Z., W.S., J.T.Y., and Y.J.W. analyzed data. J.W., C.Z. and Y.J.W. wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, J., Sun, BL., Xiang, Y. et al. Capsaicin consumption reduces brain amyloid-beta generation and attenuates Alzheimer’s disease-type pathology and cognitive deficits in APP/PS1 mice. Transl Psychiatry 10, 230 (2020). https://doi.org/10.1038/s41398-020-00918-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-020-00918-y
This article is cited by
-
SCG5 and MITF may be novel markers of copper metabolism immunorelevance in Alzheimer’s disease
Scientific Reports (2024)
-
Oral antioxidant edaravone protects against cognitive deficits induced by chronic hypobaric hypoxia at high altitudes
Translational Psychiatry (2024)
-
Beneficial role of capsaicin through modulation of mitochondrial functions in MPTP-injected mice
Neuroscience and Behavioral Physiology (2024)
-
Spicy food intake predicts Alzheimer-related cognitive decline in older adults with low physical activity
Scientific Reports (2023)
-
Capsaicin alleviates neuronal apoptosis and schizophrenia-like behavioral abnormalities induced by early life stress
Schizophrenia (2023)