Abstract
In contrast to traditional perspectives of resilience as a stable, trait-like characteristic, resilience is now recognized as a multidimentional, dynamic capacity influenced by life-long interactions between internal and environmental resources. We review psychosocial and neurobiological factors associated with resilience to late-life depression (LLD). Recent research has identified both psychosocial characteristics associated with elevated LLD risk (e.g., insecure attachment, neuroticism) and psychosocial processes that may be useful intervention targets (e.g., self-efficacy, sense of purpose, coping behaviors, social support). Psychobiological factors include a variety of endocrine, genetic, inflammatory, metabolic, neural, and cardiovascular processes that bidirectionally interact to affect risk for LLD onset and course of illness. Several resilience-enhancing intervention modalities show promise for the prevention and treatment of LLD, including cognitive/psychological or mind–body (positive psychology; psychotherapy; heart rate variability biofeedback; meditation), movement-based (aerobic exercise; yoga; tai chi), and biological approaches (pharmacotherapy, electroconvulsive therapy). Additional research is needed to further elucidate psychosocial and biological factors that affect risk and course of LLD. In addition, research to identify psychobiological factors predicting differential treatment response to various interventions will be essential to the development of more individualized and effective approaches to the prevention and treatment of LLD.
Similar content being viewed by others
Depression vs. resilience in late life
Late-life depression (LLD) is a common and debilitating condition, with less frequent remission and more frequent recurrence following first-line antidepressant treatment compared to depression experienced earlier in life1,2,3,4,5,6,7. Factors contributing to LLD are multifaceted, including biological (e.g., genetic), psychological (e.g., personality), and social influences (e.g., social support). With the world population rapidly aging, it is increasingly important to identify factors that increase resilience to the development and maintenance of LLD.
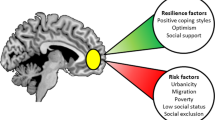
Psychological resilience has been broadly defined as “the capacity to maintain, or regain, psychological well-being in the face of challenge”8. Resilience is a complex construct that can be conceptualized as an attribute (a trait) that is possessed to varying degrees by different individuals, a dynamic process (a state) with bidirectional relations to developmental and environmental factors, and as an outcome in the face of stress and adversity9. Depending on the theoretical perspective, population, and risk factor in question, resilient outcomes may be operationalized as either the presence of a positive outcome (e.g., life satisfaction) or the absence of a negative one (e.g., lack of psychopathology)10. We conceptualize psychological resilience as a multidimensional, dynamic capacity influenced by the interaction of internal factors (e.g., cognitive capacity, personality, physical health) and external resources (e.g., social status, financial stability)11. In the context of major depressive disorder (MDD), psychological resilience refers to the net effects of a variety of psychosocial and biological variables that decrease risk of onset or relapse, decrease illness severity, or increase probability or speed of recovery. The current review describes resilience and vulnerability factors related to LLD. We summarize psychosocial resilience factors that are universal across age groups as well as those unique to aging. We also present results of research investigating the neurobiological, genetic, and immunological biomarkers of resilience.
Psychological resilience factors
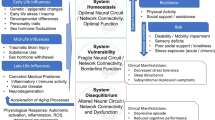
Multiple psychological resilience factors reduce an individual’s risk for depression across the lifespan12. Enduring individual characteristics such as temperament, attachment style, and personality each prospectively predict risk for depression. In addition, multiple psychological processes are proposed to mediate this effect, including beliefs and coping behavior. Psychosocial and biological correlates of LLD are presented in Table 1. A proposed model of how biopsychosocial factors influence risk for LLD and illness course is presented in Fig. 1.
Temperament
Temperament is a basic inherited style, the structure of which has been inferred largely from genetic studies. Meta-analytic data indicate that within the temperament dimensions, harm avoidance (i.e., “behavioral inhibition”) is associated with greater MDD risk and decreased treatment responsivity13. Research on individuals with age-related illness suggests that harm avoidance is similarly predictive of MDD in late life14,15. In contrast, meta-analytic data indicate that positive emotionality (e.g., positive affect, extraversion, and behavioral activation) decreases risk for depression16. Related longitudinal research suggests that the capacity to experience positive emotions such as gratitude, interest, and love is one mechanism by which resilient individuals are buffered against risk for depression following trauma17.
Attachment
Because early life attachment is thought to shape subsequent relationships, attachment theory18,19,20 offers an important framework for understanding the etiology and development of depression across the lifespan21. Insecure attachment is a risk factor for depression onset22, illness severity, and relapse23. Research suggests that increased emotional awareness24 and coping self-efficacy25,26 may be two mechanisms by which secure attachment decreases stress reactivity. Longitudinal studies27 as well as studies of children raised in orphanages28 indicate that disorganized attachment during infancy predicts greater amygdala volume—a hypothesized biomarker of difficulty with emotion regulation28. Although attachment style remains moderately stable throughout life, stressful life events have potential to decrease secure attachment29, while high relationship satisfaction and emotional openness may increase secure attachment29. Research indicates that insecure attachment continues to increase risk for depression in late life30,31.
Personality
Multiple personality factors increase risk for MDD. A recent meta-analysis controlling for baseline depressive symptoms found that low extraversion, high neuroticism, and low conscientiousness predicted depressive symptoms 5 years later32. Research on stroke survivors14, individuals with Parkinson’s Disease33, and nonclinical older adult samples34 has found similar associations, suggesting that these effects are also observed in late life. Another personality characteristic associated with resilience is grit, defined as “perseverance and passion for long-term goals” in the face of setbacks35. A recent study of 337 adults with LLD found that grit was associated with decreased severity of depression, apathy, and anxiety36.
Beliefs and coping behavior
Cognitive behavioral theory posits that an individual’s beliefs about themselves, others, and the environment influence coping behavior and subsequent psychosocial adjustment37,38. For example, two recent meta-analyses found that low self-esteem (i.e., a negative evaluation of one’s self-worth) prospectively increased risk for depression39,40. Similarly, an individual’s self-efficacy for coping with a given stressor impacts the coping strategy they select and how long they persist in their efforts41. Coping can be broadly divided into two domains: active (efforts to directly “solve” the source of stress) and accommodative (efforts to accept or adapt to the source of stress)42. Research indicates that the ability to flexibly apply active vs. accommodative strategies—i.e., using active approaches for controllable stressors and accommodative approaches for uncontrollable stressors—results in more favorable mental health outcomes43,44,45,46,47,48,49. A recent study of 337 adults with LLD found that both active coping self-efficacy and accommodative coping self-efficacy were associated with decreased depressive symptoms, apathy, and anxiety36. In addition, greater baseline accommodative coping self-efficacy predicted antidepressant treatment responsivity50. In general, individuals with high self-efficacy for managing stress through adaptive approaches such as physical exercise, social support, and self-care are more likely to engage in these strategies and less likely to develop prolonged symptoms of depression. In contrast, low coping self-efficacy is associated with passive coping51, avoidance, lower treatment adherence52,53, substance use54, and other maladaptive coping strategies55 that may serve to increase risk or course of depression.
An internal locus of control (i.e., “mastery”)56 is the general belief in one’s ability to influence outcomes57. Because this belief increases self-efficacy for coping with a range of stressors, mastery can be conceptualized as another resilience factor. Similarly, evidence suggests that a “growth mindset”—the belief that one’s abilities can be enhanced through effort—increases resilience by increasing grit58. Finally, a strong sense of meaning/purpose in life increases resilience to depression across the lifespan59. A study of 1475 older Australian adults found that higher sense of purpose was associated with less disability, higher neurocognitive performance, and decreased depressive symptoms, and predicted increased survival in late life59. By contrast, depressed older adults with symptoms of apathy generally have poorer clinical outcomes60, quality of life (QOL)61,62, treatment response63,64, cognitive impairment61, and disability62, possibly resulting from decreased engagement in social- and health-related behaviors62. Other studies have investigated the effect of meaning/purpose on risk for depression among those at high risk due to exposure to acute or chronic stress. A longitudinal study of bereaved adults showed that two construals of meaning—making sense of the loss and finding benefit in the experience—both independently predicted decreased depressive symptoms65. Other studies conducted with cancer survivors66 and individuals with terminal illness67 also report associations between meaning and decreased depressive symptoms. The results of one study suggest that increases in perceived meaning and benefit finding may be one way by which cognitive behavioral therapy helps prevent depression among cancer patients68.
A final category of beliefs that increases risk for poor outcomes in depression is mental illness stigma69. A recent meta-analysis found strong associations between internalized mental illness stigma and poorer psychological resources (hope, self-esteem, empowerment), lower treatment adherence, and greater mental illness symptom severity70. In a study of adults with LLD, higher baseline depression-related stigma predicted worse treatment response, after controlling for baseline depression severity70,71. However, results of a longitudinal study of adults with LLD suggest that mastery may moderate this effect72. In that sample, anticipated stigma only predicted increased depressive symptoms among those with low mastery72. Mastery may counteract the negative impact of anticipated stigma on mental health by increasing older adults’ confidence in their ability to cope with stressors such as interpersonal rejection.
Religion and spirituality
Religion and spirituality have been shown to prospectively reduce risk for depression73. Whether such effects are attributable to religious beliefs, behavior, or social support remains a matter of debate. One investigation of this question involved a study of over 1000 adult Detroit residents74. In that study, religious attendance (e.g., church, temple, synagogue) was associated with greater psychological well-being and less emotional distress; frequency of prayer was associated with lower well-being and more distress; and belief in eternal life was associated with greater well-being but unrelated to distress. Religious attendance was associated with lower distress even after controlling for sociodemographic variables (e.g., age, sex, education), stressors (e.g., health problems, financial problems), social resources (family contact, support, and negative interaction), and psychological resources (self-esteem and personal mastery). By contrast, a study of almost 3000 older Taiwanese adults found that religious attendance no longer predicted decreased depressive symptoms after controlling for health behaviors, social networks, and supports75. Consistent with the results of the Detroit study, religious beliefs and depressive symptoms were unrelated. Finally, a study of almost 8000 US older adults found that frequent attendance of religious services predicted decreased depression onset and frequent private prayer predicted increased depression remission 2 years later. Results of these studies indicate that religious coping behaviors may be more strongly protective against depression than religious beliefs.
Psychological factors specific to late life
The types of stressors encountered by older adults are qualitatively different than those faced by younger age groups. Late-life stressors include voluntary or forced retirement, chronic illness, cognitive decline, caregiving, financial stress, loss of independence, and bereavement. If these types of stressors are less controllable than those encountered by younger age groups, this could make accommodative coping especially essential in geriatric populations76. Indeed, older adults appear to engage in more accommodative coping77 and less instrumental action coping76 compared to younger adults. Wrosch and colleagues propose a developmental theory in which age-adapted selection of coping strategies relates to optimal well-being49. Consistent with this theory, active coping (i.e., “persistence”) was significantly associated with well-being in young adulthood and midlife, but not late life49. Among older adults, accommodative coping was more strongly associated with psychological well-being than was persistence. Another study of LLD found that accommodative coping self-efficacy was uniquely predictive of subsequent remission50.
Attitudes and stereotypes
One category of beliefs especially relevant to resilience in late life includes attitudes about aging itself. Studies in which negative stereotypes about aging are experimentally activated have found that both implicit/subconscious and explicit/conscious stereotypes negatively impact performance in older people attempting physical and cognitive tasks78,79. Other studies employing a cross-cultural approach have found larger age differences in cognitive performance in cultures with more negative stereotypes80,81.
Social role and identity changes
Traditional perspectives have assumed that major life changes inherently stressful82. However, research suggests that role transitions such as retirement exert a wide range of possible mental health effects83,84,85, including increasing well-being when the change represents an escape from a chronically stressful role situation86. A large Canadian survey found that retiring from a low-stress job increased depressive symptoms compared to not retiring, whereas retiring from a high-stress job resulted in an effect twice as large in the opposite direction86.
Several studies have found that a greater number of “absences” in major social role-identities (marital, parental, and employment) is associated with poor late-life psychological adjustment87,88,89. Cumulating evidence indicates that formal volunteering may buffer against this effect by increasing social engagement, life satisfaction, self-worth, personal growth, and sense of purpose/meaning87,90,91,92,93. For example, a US study of almost 400 older adults found that a greater number of major role-identity absences was associated with more negative affect, less positive affect, and less purpose in life87. Formal volunteering was associated with positive affect, and volunteering positively moderated the relationship between role-identity absences and purpose in life87. Other studies have found that older adults typically experience greater increases in life satisfaction with volunteering compared to younger adults91 and that adoption of a volunteer role may offset the negative impact of spousal bereavement on subsequent depressive symptoms94.
Social resilience factors
Systematic reviews suggest that both perceived social support and objective social network size protect against depression in the general population95 as well as in older adults specifically96. Research investigating the mechanisms by which social networks enhance psychological resilience indicates that both emotional support and tangible (“instrumental”) support are important contributors82. Research has also begun to distinguish between the effects of objective vs. perceived social isolation (i.e., loneliness)97. One study of over 1300 older Irish adults concluded that these constructs were distinct and that each independently predicted depressive symptoms98. An even larger US study of 20,000 adults found that loneliness was correlated with a host of other risk factors—less physical exercise, lower sleep quality, lower social engagement, and poorer physical health99. Each of these factors likely interact to predict susceptibility to MDD. A cross-cultural review of additional social resilience factors identified being married or cohabiting, male, and having a higher family income each as associated with reduced risk of depression in the US and Japan100. However, a 3-year longitudinal study of American adults aged 50–67 found that loneliness uniquely predicted depressive symptoms after controlling for demographic and psychosocial covariates such as marital status, perceived stress, and social support101.
Social factors specific to late life
Adults tend to maintain fewer peripheral social partners as they age102. It is hypothesized that an increasing awareness of time as limited influences older adults to prefer smaller and more emotionally satisfying social networks103. Despite changes in social network structure over time, the average degree of loneliness appears fairly constant from middle- to late-life104. In a sample of over 1600 older adults participating in the Health and Retirement Study, 43% reported feeling lonely105. Similarly to findings with younger and middle-ages adults, loneliness increases risk for depression in late life. For example, a study of elderly Finish adults found that loneliness predicted long-term trajectories of depression106. Additional social factors known to increase risk for LLD include bereavement, sleep disturbance, disability, prior depression, and female gender107. A meta-analysis of gender differences in LLD suggests that some of these effects are attenuated once sex differences in prevalence of widowhood, health, and socioeconomic status are accounted for108. Depression is also more common in older adults living in institutions compared to those living at home109,110.
Cognitive factors affecting resilience in LLD
LLD is associated with risk of cognitive decline111,112, mild cognitive impairment (MCI)113,114, and dementia111,115,116. One possible explanation for this association is that LLD and cognitive decline are manifestations of the same underlying neuropathology. Indeed, both LLD and dementia are associated with reduced brain volume117, increased hippocampal atrophy118, increased white matter microstructural changes119, and altered structural and functional connectivity63. Research suggests that chronic stress-associated stimulation of the hypothalamic pituitary adrenal (HPA) axis and associated over-secretion of the stress hormone cortisol contribute to neurodegeneration120,121 that may increase risk for both LLD and cognitive decline. In addition, depressive symptoms may contribute to cognitive decline. Evidence supporting this hypothesis comes from a longitudinal study of 1764 older adults without cognitive impairment at baseline122. In that study, depressive symptoms predicted cognitive decline independent of the neuropathologic hallmarks of dementia. Other research indicates that psychological resilience may be neuroprotective. A recent study found significant associations between self-reported resilience and language performance among 288 adults with LLD123. In addition, the resilience factor grit was associated with greater structural integrity of the genu of the corpus callosum and cingulum, pathways implicated in cognitive and emotion regulation (N = 70)124.
The term cognitive reserve has been used to explain differences in susceptibility to cognitive decline resulting from brain aging, pathology, or insult125. Individual differences in cognitive reserve are determined by such factors as early-life general cognitive ability/intelligence, education level, occupation complexity, physical exercise, social engagement, and ongoing cognitive engagement126. These environmental and social factors are believed to enhance neural networks that promote neuroplasticity. Cognitive reserve may also serve as a psychological resilience factor. A recent systematic review representing data from over 37,000 older adults found that cognitive reserve decreased the association between cognitive impairment and depressed mood127. Of course, factors associated with cognitive reserve such as education are also associated with engagement in health-promoting behaviors that may further protect the individual against both depression and cognitive decline128.
Other research has attempted to identify cognitive factors that predict treatment response in LLD. One recent study found that impairment in response inhibition (a fundamental executive function) predicted poor antidepressant response in LLD129. Other studies have similarly found that baseline impairments in episodic working memory, processing speed, executive function, as well as severity of baseline white matter hyperintensities (WMH) predict decreased LLD improvement with antidepressant treatment130.
Psychobiological resilience factors
The stress response and LLD
Recent research has begun to investigate the biological mechanisms by which chronic stress increases risk for depression131,132,133,134,135. In a psychobiological framework, resilience can be defined as the adaptive maintenance of homeostasis in the face of stress or adversity136. Building psychobiological resilience begins with prenatal and early-life development134. Experimental studies in animals136 as well as observational studies in humans137,138 point to an inverted U-shape between early life challenges and adult stress reactivity, with moderate challenges in early life predicting optimal mental health in adulthood. Animal studies indicate that this so-called “early-life stress inoculation” decreases subsequent cortisol secretion and increases subsequent exploration of novel situations, cognitive control, and ventromedial prefrontal cortical volumes138. It is hypothesized that prefrontal myelination and cortical expansion induced by successful early-life coping lead to enduring adaptive cognitive and emotional changes138. By contrast, high levels of early-life adversity adversely impact attachment, personality, core beliefs, and coping tendencies, ultimately leading to enduring changes in endocrine, autonomic, and immunological processes139, and increasing vulnerability to depression132,140. Genetic factors also contribute. One study found that a functional polymorphism in the promoter region of the serotonin transporter (5-HTT) gene moderated the influence of both childhood stress and later stressful life events on risk for depression141.
One prominent hypothesis for how chronic stress increases risk for depression is through sustained activation and ultimate dysregulation of the HPA axis142. Of the stress hormones, cortisol has received the most research attention due to its widespread regulatory influence143. Research suggests that although uncontrollable stress initially amplifies cortisol secretion, sustained elevated levels of cortisol eventually suppress output of corticotropin-releasing hormone (CRH) and adrenocorticotropin hormone143, resulting in below-normal cortisol levels in chronically stressed populations144. This ultimate blunting of HPA axis responsivity is proposed to underlie the withdrawal and disengagement behaviors that often accompany chronic uncontrollable stress144,145,146. Disrupted HPA axis activity as evidenced by failure to suppress cortisol in the dexamethasone test predicts increased suicide risk in both MDD147 and LLD148, and recent research indicates that low mineralocorticoid receptor availability also increases risk for depression149.
In addition to altering cortisol secretion, sustained activation of the HPA axis results in deficient monoamine transmission, disruption of neurotrophic processes (e.g., the neuroprotective brain-derived neurotrophic factor (BDNF)), oxidative stress, widespread inflammatory processes, and neurodegeneration136,142. Dysfunction in serotonergic and dopaminergic transmission contribute to the common mood and cognitive symptoms observed in depression142,150. Once in motion, this stress-related biological cascade can be exacerbated by environmental factors (e.g., social isolation) or maladaptive coping behaviors (sedentary lifestyle, substance abuse)134.
Neuropeptides
Multiple neuropeptides are known to modulate emotional processing. Neuropeptide Y (NPY) has been proposed as an endogenous mediator of resilience to stress-related psychiatric illness, including depression151. NPY plays a key role in the maintenance of homeostasis and has been implicated in diverse motivational, perceptual, and affective processes including circadian rhythm, anxiety, appetite, alcohol consumption, and pain perception152. NPY receptors are densely expressed in brain regions relevant to mood disorders including the cortex, hippocampus, and amygdala. Low NPY levels have been reported in MDD compared to healthy controls153, and genetic variation associated with low NPY expression increases risk for MDD153,154. Results of one study suggest that this effect may be mediated by increased neuronal response to affective stimuli in the medial prefrontal and anterior cingulate cortices among individuals with low-expression NPY genotypes154.
Endocrine changes in aging and depression
One potential vulnerability for depression specific to older adults is age-associated decline in reproductive hormones. Perimenopause increases risk for both recurrent and new-onset depression155. Loss of normal estradiol (the primary circulating estrogen) cycling is proposed to account for this increased vulnerability via effects on neurotransmitter and mood regulatory systems155. Estrogen receptor polymorphisms have been associated with heightened depression risk in older women156,157, and maintenance of normal estrogen levels is important for several brain regions vulnerable to age-related changes158 (e.g., the prefrontal cortex (PFC) and hippocampus)134,159,160. Similarly, age-related reduction in dehydroepiandrosterone (DHEA) has been linked to depression, cognitive decline, reduced immune function, and decreased physical health161,162. Individuals with LLD demonstrate lower DHEA levels compared to non-depressed older adults, and DHEA levels increase with remission163.
Cardiovascular markers
There is strong evidence for a bi-directional association between depression and cardiovascular disease. Prospective studies indicate that individuals with depression are at nearly twice the risk of developing cardiovascular disease and have nearly a three times higher mortality rate following a cardiac event164,165. Conversely, cardiovascular disease prospectively predicts depression166,167. Although unhealthy behaviors (e.g., unhealthy diet, lack of physical activity) undoubtedly contribute to this effect, lifestyle factors do not fully explain the relation between heart health and depression168.
Autonomic nervous system (ANS) dysregulation is one biological mechanism that may explain the link between cardiovascular risk and depression169. Heart rate variability (HRV) is a surrogate index of resting cardiac vagal outflow that represents the ability of the ANS to adapt to a changing psychological, social and physical environment170. Higher HRV is thought to reflect greater self-regulatory capacity (i.e., regulation of behavioral, cognitive, and emotional processes), and meta-analytic data suggest that this effect is larger for older compared to younger adults171. Recent research suggests that high HRV may serve as a biomarker of resilience to the development of stress-related disorders172 including depression168,173. However, age-related differences have been reported in the frequency of the HRV most predictive of depression. Whereas low high-frequency (HF)-HRV (reflecting parasympathetic activity174) is associated with depression among children173, adolescents173, and young adults175, only low low-frequency (LF)-HRV (reflecting both sympathetic and parasympathetic activity) appears associated with depression among older adults168. Decreased parasympathetic activity with age appears to result in decreased HRV in the general (primarily non-depressed) older adult population176,177, which may partially account for this finding.
Inflammation in aging and depression
Cumulating evidence indicates immune and metabolic dysregulation among individuals with depression178. Immunometabolic dysregulation is associated with more severe and chronic depressive symptoms179,180,181 as well as decreased response to antidepressant treatment182,183,184, and may explain the increased prevalence of cardiovascular disease and diabetes in MDD185. While studies of younger adults typically show upregulation of metabolic processes in depression, studies of LLD report both upregulation and downregulation of these processes186,187,188,189. Both younger190 and older191 adults with depression show increased levels of inflammatory cytokines, secreted proteins that interact with virtually every depression-relevant neurophysiological domain (e.g., neurotransmitter metabolism, neuroendocrine function, and neural plasticity). Additional evidence for the role of inflammation in depression comes from studies indicating that (1) pro-inflammatory factors precede depressive symptom onset191, (2) antidepressant treatments reduce pro-inflammatory factors192, and (3) anti-cytokine therapy decreases depressive symptoms in placebo-controlled trials193. Experimental data indicate that acute psychosocial stress (e.g., public speaking, mental arithmetic) stimulates inflammatory signaling molecules194, and these responses are exaggerated in patients with depression190. Both childhood maltreatment195 and chronic stress in adulthood196,197,198 are associated with increased inflammation. Thus, inflammation may be one pathway by which these psychosocial factors increase risk for depression.
Genetic factors in aging and depression
At least three genes have been associated with increased risk for LLD: the methionine (Met) allele of the neurotrophic factor BDNF199, APOE-e4 (involved in myelin repair and Aβ metabolism), and SLC6A4 (the short allele of the serotonin transporter 5-HTTLPR)200. BDNF is a protein that stimulates neurogenesis and is important for long-term memory. The Met variation of the BDNF gene is associated with decreased BDNF secretion201, poorer memory performance202, and increased risk for a range of neuropsychiatric disorders203. Interestingly, a meta-analysis found that the Met allele predicted MDD among men but not women204. Another meta-analysis found that the Met allele significantly moderated the effect of stressful life events on MDD risk, suggesting that Met carriers are more genetically sensitive to adverse life experiences205. A recent longitudinal study conducted with over 1000 older adults found that epigenetic regulation of the BDNF gene was associated with depression206. Another study of individuals with LLD found that the Met allele predicted poorer response to paroxetine, and that this effect was moderated by the cyclic AMP responsive element binding protein 1 (CREB1)207.
Neuroimaging biomarkers of aging and depression
Emerging research evidence suggests that MDD is associated with reduced structural and functional plasticity208,209. Brain structures important for learning and applying adaptive coping strategies (e.g., the hippocampus and PFC) show atrophy in MDD210,211,212,213,214, possibly resulting from depression-related hypercortisolemia215,216. Similar abnormalities (i.e., decreased limbic structure volumes and reduced PFC activity) have been found in LLD215,217,218,219. However, research suggests that at least with regard to decreased hippocampal volumes220, these effects may be more pronounced for individuals with earlier depression onset. Narayan and colleagues propose that prior depressive episodes, aging, stress, hypercortisolemia, and reduced BDNF levels cause focal atrophy and may decrease the threshold for mood disorders in late life221.
Neuroimaging biomarkers of emotion regulation, coping, and grit
Substantial neuroimaging research has investigated the neural networks implicated in emotion regulation and coping with stress. Despite the neural structures hypothesized to underlie emotion regulation being vulnerable to age-related decline222, behavioral evidence suggests that older adults have better emotion regulation capacity compared to younger adults223. Functional magnetic neuroimaging (fMRI) studies have documented increased activation of the PFC and amygdala224,225 in older vs. younger adults during tasks requiring emotion regulation tasks, possibly reflecting compensation for less efficient cognitive processing224.
Other studies have attempted to identify the neural correlates of adaptive coping with stress. For example, a study of 102 heathy adults found that the functional connectivity of regions associated with the default mode and anterior salience networks was associated with propensity to adopt various coping strategies (e.g., problem-focused, avoidant, social support seeking)226. Others have investigated individual differences in brain structure that relate to personality characteristics associated with resilience to depression. As reported above, grit has been associated with greater structural integrity of the genu of the corpus callosum and cingulum in LLD—pathways implicated in cognitive and emotion regulation36,124. Finally, several studies have documented neuroimaging correlates of self-reported resilience. Among healthy adults, self-reported resilience was correlated with decreased electroencephalogram (EEG) responsivity to adverse images227. In LLD, correlates included low amygdala blood perfusion at rest and greater functional connectivity between the amygdala and the ventral default mode network228.
Structural brain changes and cerebrovascular disease in aging and depression
WMH are another related biological mechanism that may explain the link between cardio- and cerebrovascular disease and depression in late life. LLD is consistently associated with greater WMH severity229,230, and individuals with late onset exhibit greater WMH severity220,231,232,233,234 and greater cognitive impairment234,235,236 compared to those with first onset earlier in life. Depressed older adults who present with WMH are said to have “vascular depression”237, a subtype of depression characterized by cognitive deficits, psychomotor retardation, lack of insight, and disability disproportional to depression severity238. Such vascular abnormalities are linked to greater depressive symptom severity and poorer treatment response239,240. WMH are also strongly and independently associated with symptoms of apathy241,242,243,244,245.
Psychobiological factors associated with early- vs. late-onset LLD
Several studies have investigated the clinical correlates of early- vs. late-onset LLD. One study investigated illness severity and symptoms, cognitive function, antecedent life events, physical health, genetic factors, and vascular health as a function of age of onset in 57 adults with LLD246. They found that early onset was associated with increased symptoms of anxiety and greater heritability. Several studies of LLD have found that early-onset recurrent illness predicts slower treatment responses and greater relapse compared to late onset247. This may be due to a greater number of depressive episodes, which is associated with the depletion of neural248, interpersonal249,250 and psychosocial resources249,250. These results are seemingly in contrast to the results of studies identifying characteristics associated with late onset (i.e., increased subcortical hyperintensities234, decreased cognitive performance234), which have also been associated with inadequate treatment response239,240.
Resilience-enhancing interventions
Resilience-enhancing interventions can be implemented either preventatively to reduce susceptibility to MDD or as a treatment following MDD onset. Prevention strategies promote well-being even in the absence of current psychopathology, and can be applied both to healthy individuals or to those at high risk (i.e., those with chronic stress, trauma, or history of prior depressive episodes). The field of positive psychology251 defines well-being as not the absence of a mental disorder, but rather the presence of well-being, and advocates for the widespread application of such techniques regardless of the presence or absence of psychopathology.
Positive psychology interventions
Positive psychology interventions (PPIs) are “treatment methods or intentional activities that aim to cultivate positive feelings, behaviors, or cognitions”252(p. 468). PPIs typically target hedonic well-being (e.g., positive affect, life satisfaction, happiness)253, eudaimonic well-being (e.g., self-acceptance, positive relations, autonomy, purpose in life)254,255, or both, and are typically amenable to self-administration. Such approaches vary widely in intensity, ranging from a several minute-long daily gratitude journal to more in-depth journal exercises, meditations, and intentional social behaviors. Research suggests that PPIs not only increase well-being, but also improve the individual’s capacity to “bounce back” from adversity. A recent meta-analysis of PPIs including data from over four thousand adults indicated a highly significant, moderate effect of PPIs on both well-being (r = 0.29) and depressive symptoms (r = 0.31)252. PPIs included expressing gratitude256,257, reflecting upon one’s ideal future self257,258,259,260, identifying one’s strengths256, practicing mindfulness261, and practicing compassion/ acts of kindness262,263,264,265. Interventions delivered individually were most effective, followed by those administered in a group, then by self-administered PPIs. The efficacy of PPIs increased linearly with participant age, and depressed individuals benefitted more than non-depressed individuals252. Another PPI shown to improve mood in individuals at risk for depression due to trauma exposure is “benefit finding”, or the intentional identification of positive ways in which their lives have changed as a result of a traumatic event266,267. Bower and colleagues268 propose an integrative conceptual model in which benefit finding promotes the development of interpersonal resources (e.g., adaptive coping strategies including cognitive re-appraisal; increased self-efficacy for coping with stress; more salient sense of one’s values/purpose) and intrapersonal resources (e.g., enhancement of social relationships) that facilitate more adaptive responses to future stressors. More broadly, PPIs that increase one’s feeling of connection to others (e.g., expressive gratitude, practicing compassion may not only directly improve mood but also exert beneficial neuroendocrine, cardiovascular, and immune systems changes269. PPIs aimed at enhancing meaning/purpose (e.g., reflecting upon one’s ideal future self; identifying strengths) are also thought to improve markers of immune functioning270. In addition, it is possible that the positive psychological changes elicited through PPIs have their own mechanistic pathways distinct from those associated with distress—e.g., parasympathetic nervous system activity, growth factors, and other neuroendocrine factors such as oxytocin271. Although PPIs are not recommended as a stand-alone treatment for moderate-to-severe MDD, research suggests that PPIs may be useful for the prevention or adjunct treatment of depression252.
Meditation
Meditation refers to a category of mind–body techniques most commonly involving the directed focus of attention. One form of Buddhist meditation techniques that has been adapted to a variety of secular settings in the West is mindfulness—intentional, non-judgmental, present-focused awareness272. A recent meta-analysis indicated that mindfulness-based interventions are significantly more effective than psychoeducation, supportive psychotherapy, relaxation training, and guided imagery for improving a range of mental and physical health symptoms, with the largest effects demonstrated for mental health symptoms including depression273. A recent review of randomized controlled trials (RCTs) of mindfulness-based interventions for older adults concluded that Mindfulness-Based Stress Reduction (MBSR) is effective for improving symptoms of depression, anxiety, positive affect, insomnia, chronic pain, memory, and executive functioning in late life274. Other research conducted with adults with MDD has found that MBSR is effective for preventing relapse in those with a history of three or more depressive episodes275. A recent systematic review evaluating the biopsychological mechanisms by which mindfulness practice affects clinical outcomes concluded that decreases in cognitive reactivity, emotional reactivity, rumination, and worry may mediate the effect of mindfulness interventions on mental health276. Mindfulness practice appears to alter both brain structure and function, most notably in areas related to attentional control, self-awareness, and emotion regulation277,278,279. In addition, mindfulness interventions may protect against age-related decreases in gray matter volume280, attention performance281, and cellular aging282.
Other forms of yogic-style meditation include those involving repetition of sounds (mantras) or hand positions (mudras). A recent study found that Kirtan Kriya (which combines mantras and mudras) was more effective for improving mental health and cognitive functioning in dementia caregivers compared to passive listening to relaxing music283. Further, Kirtan Kriya reversed the pattern of increased pro-inflammatory cytokine and decreased innate antiviral response gene transcription observed in chronically stressed individuals. Results of this study indicate inflammatory and antiviral transcription pathways as one mechanism by which meditation may increase resilience in older adults284. Another randomized study of mantra meditation for elderly women with hypertension found that chanting significantly reduced depression, anxiety, stress, blood pressure, and cognitive impairment285. Studies investigating the neurohemodynamic correlates of mantra meditation suggest that deactivation of the limbic system may account for observed decreases in physiological arousal and improvements in well-being286.
Psychotherapy
Meta-analytic data suggest that psychotherapy is similarly effective as pharmacological approaches in the treatment of LLD287,288, with a number needed to treat of 3 (ref. 288). A meta-analysis of RCTs comparing psychotherapy for LLD to various control conditions found the largest effects for cognitive behavior therapy (CBT; g = 0.45), problem-solving therapy (g = 0.46), and life review therapy (g = 0.59)287. A more recent review focused on mindfulness-based CBT found such interventions to be similarly effective for improving late-life depressive symptoms (g = 0.55)289. Results of another meta-analysis suggest that music therapy significantly augments the efficacy of standard treatments for LLD290. Because alterations of the HPA axis and the ANS appear to be involved in the development and maintenance of depression, it is conceivable that these dysregulations may interact with psychotherapeutic treatment to influence outcomes. A meta-analysis that attempted to investigate the effect of psychotherapy on HPA and ANS regulation in adults with mixed mental health disorders concluded that this effect could not be estimated due to the large degree of heterogeneity of methodologies across studies291.
Movement-based interventions
Physical exercise is effective for the treatment of MDD, with effect sizes similar to those observed with pharmacological and psychological therapies292. A recent systematic review confirmed that physical activity interventions are also effective in LLD293. In addition to the documented effects on mood, physical activity is associated with improved balance, greater strength, and decreased disability. One mechanism by which exercise interventions affect both physical and mental health may be via reduction in inflammatory cytokine responses resulting from hemodynamic hormonal changes during physical activity294. Movement-based interventions that also incorporate mindfulness are referred to as mind–body therapies (MBTs). A recent review of the efficacy of such interventions for late-life mood and cognitive disorders concluded that MBTs such as yoga and tai chi may outperform conventional physical exercise with regard to effects on mood, QOL, and cognitive functioning295. A review of the neural mechanisms of movement-based vs. stationary meditation found that these two approaches affect multiple common brain regions including those involved in attention, memory, awareness, and emotional processing296. Yoga appears to reduce HPA axis activity in younger healthy adults297,298 as well as in sedentary community-dwelling older adults without depression299. It is possible that restoration of HPA axis dysregulation may be one mechanism by which MBTs improve mood in older adults with depressive symptoms283,300,301,302.
Pharmacotherapy
Second-generation antidepressant medications, including selective serotonin reuptake inhibitors (SSRIs), bupropion, mirtazapine, venlafaxine, and duloxetine are the most commonly prescribed pharmacological treatments for LLD. A recent meta-analysis of second-generation antidepressants vs. placebo for treatment of LLD found that response rates were modest (44% for antidepressant vs. 35% for placebo)3. Response rates were higher for 10–12-week trials (55%) compared to 6–8-week trials (38%)3. However, results of another meta-analysis comparing placebo-controlled vs. comparator trials in LLD suggest that these effects may underestimate those in clinical settings, when patient expectations of improvement may be higher303. Discontinuation rates due to adverse events associated with second-generation antidepressant medication range from 8–27%, compared to 1–11% for placebo3. The precise neurophysiological mechanisms of antidepressant medications remain unknown. However, meta-analytic data indicate normalization of neural responses to positive and negative stimuli in limbic regions as well as increased self-regulatory potential via changes in the dorsolateral PFC304.
Electroconvulsive therapy
Electroconvulsive therapy (ECT) is widely used for the management of severe and refractory MDD305. As the most effective biological treatment for major depression, ECT is associated with remission rates of 51% (ref. 306) and standardized effect sizes of 0.91 compared to sham ECT307. Reviews indicate that ECT is especially effective in older adult patients, with remission rates reaching 73–90% (refs. 308,309,310,311,312). In addition, psychotic and melancholic features predict greater response312. Use of ECT for LLD is safe, well-tolerated, and effective for improving cognition and psychomotor symptoms313. Maintenance ECT, with treatments spaced over weekly to monthly intervals, is often used for relapse prevention314. Recommendations for optimal administration of ECT for LLD can be found elsewhere312. The results of one study investigating the neurological effects of ECT suggest that increases in frontal white matter fractional anisotropy (FA) (typically reduced in LLD compared to age-matched controls315) may account for ECT’s antidepressant effect315. Another study found gray matter volume increases in the right caudate nucleus with ECT treatment for LLD, a change which correlated with improved psychomotor function313.
HRV biofeedback
Small uncontrolled studies have shown some promise for HRV biofeedback in the treatment of MDD316,317, and one RCT found that HRV biofeedback significantly improved depressive symptoms in first-time cardiac surgery patients compared to usual care318. It is thought that strengthened homeostasis or effects on the vagal afferent pathway to frontal cortical areas may account for this effect319. However, no RCTs to date have examined the efficacy of HRV biofeedback in LLD.
Directions for future research and public policy
During this critical time of accelerated global aging, understanding factors that promote resilience vs. risk for the development of LLD and related comorbidities is more important than ever. Continued research in this area is essential for informing practice and public policy to promote successful aging, reduce disability, and mitigate rising healthcare costs. One fruitful area for future research is the investigation of individual characteristics that moderate risk factors for LLD. For example, research suggests that risk factors for depression may vary according to generation110, gender, and cultural origin. Variables such as self-esteem and self-efficacy are highly culturally and contextually dependent, such that the efficacy of interventions targeting these processes will likely vary across cultures. Indeed, PPIs as a whole appear more effective among members of individualist cultures320, perhaps because the rationale for such approaches resonates more strongly with individuals from cultures endorsing the pursuit of individual happiness. It is possible that individuals from collectivist cultures may derive greater benefit from prosocial and other-focused PPIs (e.g., performing acts of kindness, writing a letter of gratitude), compared to self-focused PPIs (e.g., reflecting on personal strengths)252.
Similarly, continued investigation of the biological factors that increase or mitigate risk for depression is of paramount importance. Results of a recent meta-analysis suggest that depressed individuals with higher baseline cortisol levels are less responsive to psychological therapy321. Because elevated cortisol has been linked to concentration and memory difficulties322, the authors propose that difficulty engaging in learning processes may reduce the efficacy of psychotherapy in this subset of MDD patients. Additional research is needed to identify other biomarkers of depression “subtypes” as well as determine as well as the interventions most effective for each. Continued work in this area is essential for the development of more effective approaches to the treatment of LLD.
Resilience-enhancing interventions can be applied not only at the individual level, but also at the level of the family, organization, or community. A recent systematic review of family-oriented interventions found that dyadic interventions such as ecosystems therapy, psychoeducation, family counseling, behavioral therapy, and CBT are feasible and effective for the treatment of LLD323. In addition, the results of two pilot studies suggest that mindfulness training delivered jointly to older adult patients and their caregivers is effective for reducing depressive symptoms324,325,326.
Other research has identified potentially useful directions for organizations that wish to promote resilience in their older adult volunteers. A recent study of almost 400 older adult volunteers identified adequate training, ongoing support, and greater volunteer choice as predictors of larger volunteer mental health benefits327. This effect was mediated by the volunteer’s belief that their work had meaningfully contributed to the well-being of others327.
Factors influencing risk for LLD can also be identified at the community and public policy levels. Because loneliness affects nearly half of the Americans99 and independently predicts severity and course of LLD328, loneliness is a promising target for intervention. Of note, a recent review of seven RCTs found that social robot interventions may be effective for helping to alleviate depressive symptoms in older adults when used during group activities329. Programs to promote greater integration of older adults into their communities (e.g., by facilitating the sharing of meals, joint physical activity, support groups, or interactive volunteer work) are recommended. Of course, factors such as a balanced diet, physical activity, and sufficient sleep are also of paramount importance. As such, public policies that provide food stamps and low-cost healthy food options, safe opportunities for physical exercise, preventative medical care, and financial assistance to those older adults in need will reduce the economic, social, and individual QOL burden of LLD.
Conclusion
With the global population rapidly aging and depression as the leading cause of disability worldwide, identification of factors that increase resilience to LLD is of paramount public health importance. Risk for LLD begins as early as embryonic development and is determined by complex interactions between biological and psychological factors. Research has elucidated both trait-like psychological factors that identify those at greatest risk and psychosocial processes that may be fruitful targets for intervention. Psychobiological factors include endocrine, genetic, inflammatory, neural, and cardiovascular processes that bidirectionally interact to affect LLD risk. Intervention research suggests that MBTs (including yoga299,330,331, MBSR332, tai chi333,334, qigong302, and meditation335,336) have potential for improving HPA axis regulation299 and depressive symptoms in older adults with depression283,300,301,302 as well as in non-depressed older adults330,331,332,334,335,336.
Additional research into the biophysiological mechanisms by which psychosocial processes affect risk for LLD will identify novel targets for intervention. In addition, continued research testing the efficacy of interventions designed to enhance resilience to LLD is critically important. Further work in this area has potential to greatly increase QOL, reduce morbidity, and decrease healthcare costs for aging adults.
References
Alexopoulos, G. S., Young, R. C., Abrams, R. C., Meyers, B. & Shamoian, C. A. Chronicity and relapse in geriatric depression. Biol. Psychiatry 26, 551–564 (1989).
Reynolds, C. F. 3rd et al. Maintenance treatment of major depression in old age. N. Engl. J. Med. 354, 1130–1138 (2006).
Nelson, J. C., Delucchi, K. & Schneider, L. S. Efficacy of second generation antidepressants in late-life depression: a meta-analysis of the evidence. Am. J. Geriatr. Psychiatry 16, 558–567 (2008).
Nelson, J. C., Delucchi, K. L. & Schneider, L. S. Moderators of outcome in late-life depression: a patient-level meta-analysis. Am. J. Psychiatry 170, 651–659 (2013).
Rutherford, B. R. & Roose, S. P. A model of placebo response in antidepressant clinical trials. Am. J. Psychiatry 170, 723–733 (2013).
Sackeim, H. A., Roose, S. P. & Burt, T. Optimal length of antidepressant trials in late-life depression. J. Clin. Psychopharmacol. 25, S34–S37 (2005).
Entsuah, A. R., Huang, H. & Thase, M. E. Response and remission rates in different subpopulations with major depressive disorder administered venlafaxine, selective serotonin reuptake inhibitors, or placebo. J. Clin. Psychiatry 62, 869–877 (2001).
Ryff, C. D., Friedman, E. M., Morozink, J. A. & Tsenkova, V. Psychological resilience in adulthood and later life: implications for health. Annu. Rev. Gerontol. Geriatr. 32, 73–92 (2012).
Smith, G. C. & Hayslip, B. Jr Resilience in adulthood and later life: what does it mean and where are we heading? Annu. Rev. Gerontol. Geriatr. 32, 3 (2012).
Vanderbilt-Adriance, E. & Shaw, D. S. Conceptualizing and re-evaluating resilience across levels of risk, time, and domains of competence. Clin. Child Fam. Psychol. Rev. 11, 30 (2008).
Windle, G. What is resilience? A review and concept analysis. Rev. Clin. Gerontol. 21, 152–169 (2011).
Lavretsky. & Irwin, M. R. Resilience and aging. Aging Health 3, 309–323 (2007).
Kampman, O. & Poutanen, O. Can onset and recovery in depression be predicted by temperament? A systematic review and meta-analysis. J. Affect. Disord. 135, 20–27 (2011).
Afanasiev, S., Aharon-Peretz, J. & Granot, M. Personality type as a predictor for depressive symptoms and reduction in quality of life among stroke survivals. Am. J. Geriatr. Psychiatry 21, 832–839 (2013).
Buchman, A. S. et al. Harm avoidance is associated with progression of parkinsonism in community-dwelling older adults: a prospective cohort study. BMC Geriatr. 14, 54 (2014).
Khazanov, G. K. & Ruscio, A. M. Is low positive emotionality a specific risk factor for depression? A meta-analysis of longitudinal studies. Psychol. Bull. 142, 991 (2016).
Fredrickson, B. L., Tugade, M. M., Waugh, C. E. & Larkin, G. R. What good are positive emotions in crisis? A prospective study of resilience and emotions following the terrorist attacks on the United States on September 11th, 2001. J. Pers. Soc. Psychol. 84, 365 (2003).
Bowlby, J. Attachment and Loss: Attachment. (Basic Books, New York, NY, 1969).
Bowlby, J. Attachment and Loss: Separation. (Basic Books, New York, NY, 1973).
Bowlby, J. Attachment and Loss: Loss, Sadness, and Depression. (Basic Books, New York, NY, 1980).
Dagan, O., Facompré, C. R. & Bernard, K. Adult attachment representations and depressive symptoms: a meta-analysis. J. Affect. Disord. 236, 274–290 (2018).
Kidd, T. et al. Attachment anxiety predicts depression and anxiety symptoms following coronary artery bypass graft surgery. Br. J. Health Psychol. 21, 796–811 (2016).
Conradi, H. J., Kamphuis, J. H. & de Jonge, P. Adult attachment predicts the seven-year course of recurrent depression in primary care. J. Affect Disord. 225, 160–166 (2018).
Monti, J. D. & Rudolph, K. D. Emotional awareness as a pathway linking adult attachment to subsequent depression. J. Couns. Psychol. 61, 374 (2014).
Laird, K. T., Preacher, K. J. & Walker, L. S. Attachment and adjustment in adolescents and young adults with a history of pediatric functional abdominal pain. Clin. J. Pain 31, 152 (2015).
Porter, L. S., Davis, D. & Keefe, F. J. Attachment and pain: recent findings and future directions. Pain 128, 195–198 (2007).
Lyons-Ruth, K., Pechtel, P., Yoon, S. A., Anderson, C. M. & Teicher, M. H. Disorganized attachment in infancy predicts greater amygdala volume in adulthood. Behav. Brain Res. 308, 83–93 (2016).
Tottenham, N. et al. Prolonged institutional rearing is associated with atypically large amygdala volume and difficulties in emotion regulation. Dev. Sci. 13, 46–61 (2010).
McConnell, M. & Moss, E. Attachment across the life span: factors that contribute to stability and change. Aust. J. Educ. Dev. Psychol. 11, 60–77 (2011).
Pietrzak, R. H. & Cook, J. M. Psychological resilience in older US veterans: results from the national health and resilience in veterans study. Depress. Anxiety 30, 432–443 (2013).
Paradiso, S., Naridze, R. & Holm‐Brown, E. Lifetime romantic attachment style and social adaptation in late‐onset depression. Int. J. Geriatr. Psychiatry 27, 1008–1016 (2012).
Hakulinen, C. et al. Personality and depressive symptoms: Individual participant meta‐analysis of 10 cohort studies. Depress. Anxiety 32, 461–470 (2015).
Damholdt, M. F., Callesen, M. B. & Møller, A. Personality characteristics of depressed and non-depressed patients with Parkinson’s disease. J. Neuropsychiatry Clin. Neurosci. 26, 329–334 (2014).
Henriques-Calado, J. et al. Predicting relatedness and self-definition depressive experiences in aging women based on personality traits: a preliminary study. Bull. Menninger Clin. 77, 269–288 (2013).
Duckworth, A. L., Peterson, C., Matthews, M. D. & Kelly, D. R. Grit: perseverance and passion for long-term goals. J. Pers. Soc. Psychol. 92, 1087 (2007).
Laird, K. T. et al. Clinical correlates of resilience factors in geriatric depression. Int. Psychogeriatr. 2018, 1–10 (2018).
McGinn, L. K. Cognitive behavioral therapy of depression: theory, treatment, and empirical status. Am. J. Psychother. 54, 257–262 (2000).
Abramowitz, J. S. The practice of exposure therapy: relevance of cognitive-behavioral theory and extinction theory. Behav. Ther. 44, 548–558 (2013).
Sowislo, J. F. & Orth, U. Does low self-esteem predict depression and anxiety? A meta-analysis of longitudinal studies. Psychol. Bull. 139, 213 (2013).
Orth, U., Robins, R. W., Meier, L. L. & Conger, R. D. Refining the vulnerability model of low self-esteem and depression: Disentangling the effects of genuine self-esteem and narcissism. J. Pers. Soc. Psychol. 110, 133 (2016).
Lazarus, R. S. & Folkman, S. Stress, Appraisal, and Coping. (Springer Publishing Company, New York, 1984).
Thomsen, T. & Greve, W. Accommodative coping in early adolescence: an investigation of possible developmental components. J. Adolesc. 36, 971–981 (2013).
Forsythe, C. J. & Compas, B. E. Interaction of cognitive appraisals of stressful events and coping: testing the goodness of fit hypothesis. Cogn. Ther. Res. 11, 473–485 (1987).
Zakowski, S. G., Hall, M. H., Klein, L. C. & Baum, A. Appraised control, coping, and stress in a community sample: a test of the goodness-of-fit hypothesis. Ann. Behav. Med. 23, 158–165 (2001).
Cheng, C., Lau, H. P. B. & Chan, M. P. S. Coping flexibility and psychological adjustment to stressful life changes: A meta-analytic review. Psychological bulletin 140, 1582–1607 (2014).
Seltzer, M. M., Greenberg, J. S., Floyd, F. J. & Hong, J. Accommodative coping and well-being of midlife parents of children with mental health problems or developmental disabilities. Am. J. Orthopsychiatry 74, 187 (2004).
Boerner, K. Adaptation to disability among middle-aged and older adults: the role of assimilative and accommodative coping. J. Gerontol. B Psychol. Sci. Soc. Sci. 59, P35–P42 (2004).
Isaacowitz, D. M. & Seligman, M. E. P. Cognitive style predictors of affect change in older adults. Int. J. Aging Human. Dev. 54, 233–253 (2002).
Wrosch, C., Dunne, E., Scheier, M. F. & Schulz, R. Self-regulation of common age-related challenges: benefits for older adults’ psychological and physical health. J. Behav. Med. 29, 299–306 (2006).
Laird, K. T., Lavretsky, H., St Cyr, N. & Siddarth, P. Resilience predicts remission in antidepressant treatment of geriatric depression. Int. J. Geriatr. Psychiatry 33, 1596–1603 (2018).
Walker, L. S., Smith, C. A., Garber, J. & Claar, R. L. Testing a model of pain appraisal and coping in children with chronic abdominal pain. Health Psychol. 24, 364–374 (2005).
Anderson, J. R. et al. A dyadic multiple mediation model of patient and spouse stressors predicting patient dietary and exercise adherence via depression symptoms and diabetes self-efficacy. J. Behav. Med. 39, 1020–1032 (2016).
Clarke, J. et al. Effects of mental health self-efficacy on outcomes of a mobile phone and web intervention for mild-to-moderate depression, anxiety and stress: secondary analysis of a randomised controlled trial. BMC Psychiatry 14, 272 (2014).
Newcomb, M. D. & Harlow, L. L. Life events and substance use among adolescents: mediating effects of perceived loss of control and meaninglessness in life. J. Pers. Soc. Psychol. 51, 564 (1986).
Rippetoe, P. A. & Rogers, R. W. Effects of components of protection-motivation theory on adaptive and maladaptive coping with a health threat. J. Pers. Soc. Psychol. 52, 596 (1987).
Wang, Q., Bowling, N. A. & Eschleman, K. J. A meta-analytic examination of work and general locus of control. J. Appl. Psychol. 95, 761 (2010).
Rotter, J. B. Generalized expectancies for internal versus external control of reinforcement. Psychol. Monogr. 80, 1 (1966).
Wang, S. et al. Neuroanatomical correlates of grit: growth mindset mediates the association between gray matter structure and trait grit in late adolescence. Hum. Brain Mapp. 39, 1688–1699 (2018).
Windsor, T. D., Curtis, R. G. & Luszcz, M. A. Sense of purpose as a psychological resource for aging well. Dev. Psychol. 51, 975 (2015).
Yuen, G. S. et al. Apathy in late-life depression: common, persistent, and disabling. Am. J. Geriatr. Psychiatry 23, 488–494 (2015).
Groeneweg-Koolhoven, I., de Waal, M. W. M., van der Weele, G. M., Gussekloo, J. & van der Mast, R. C. Quality of life in community-dwelling older persons with apathy. Am. J. Geriatr. Psychiatry 22, 186–194 (2014).
Tierney, S. M., Woods, S. P., Weinborn, M. & Bucks, R. S. Real-world implications of apathy among older adults: Independent associations with activities of daily living and quality of life. J. Clin. Exp. Neuropsychol 40, 895–903 (2018).
Yuen, G. S. et al. Neuroanatomical correlates of apathy in late-life depression and antidepressant treatment response. J. Affect. Disord. 166, 179–186 (2014).
Lavretsky, H. et al. Citalopram, methylphenidate, or their combination in geriatric depression: a randomized, double-blind, placebo-controlled trial. Am. J. Psychiatry 172, 561–569 (2015).
Davis, C. G., Nolen-Hoeksema, S. & Larson, J. Making sense of loss and benefiting from the experience: two construals of meaning. J. Pers. Soc. Psychol. 75, 561 (1998).
Simonelli, L. E., Fowler, J., Maxwell, G. L. & Andersen, B. L. Physical sequelae and depressive symptoms in gynecologic cancer survivors: meaning in life as a mediator. Ann. Behav. Med. 35, 275–284 (2008).
Nelson, C. J., Rosenfeld, B., Breitbart, W. & Galietta, M. Spirituality, religion, and depression in the terminally ill. Psychosomatics 43, 213–220 (2002).
Antoni, M. H. et al. Cognitive-behavioral stress management intervention decreases the prevalence of depression and enhances benefit finding among women under treatment for early-stage breast cancer. Health Psychol. 20, 20 (2001).
Drapalski, A. L. et al. A model of internalized stigma and its effects on people with mental illness. Psychiatr. Serv. 64, 264–269 (2014).
Livingston, J. D. & Boyd, J. E. Correlates and consequences of internalized stigma for people living with mental illness: a systematic review and meta-analysis. Soc. Sci. Med. 71, 2150–2161 (2010).
Lysaker, P. H., Davis, L. W., Warman, D. M., Strasburger, A. & Beattie, N. Stigma, social function and symptoms in schizophrenia and schizoaffective disorder: associations across 6 months. Psychiatry Res. 149, 89–95 (2007).
Raeifar, E., Halkett, A., Lohman, M. C. & Sirey, J. A. The relation between mastery, anticipated stigma and depression among older adults in a primary care setting. J. Nerv. Ment. Dis. 205, 801–804 (2017).
Miller, L. et al. Religiosity and major depression in adults at high risk: a ten-year prospective study. Am. J. Psychiatry 169, 89–94 (2012).
Ellison, C. G., Boardman, J. D., Williams, D. R. & Jackson, J. S. Religious involvement, stress, and mental health: findings from the 1995 Detroit Area Study. Soc. Forces 80, 215–249 (2001).
Yeager, D. M. et al. Religious involvement and health outcomes among older persons in Taiwan. Soc. Sci. Med. 63, 2228–2241 (2006).
Aldwin, C. M., Sutton, K. J., Chiara, G. & Spiro, A. Age differences in stress, coping, and appraisal: findings from the Normative Aging Study. J. Gerontol. B Psychol. Sci. Soc. Sci. 51, P179–P188 (1996).
Brandtstädter, J. & Renner, G. Tenacious goal pursuit and flexible goal adjustment: explication and age-related analysis of assimilative and accommodative strategies of coping. Psychol. Aging 5, 58 (1990).
Horton, S., Baker, J., Pearce, G. W. & Deakin, J. M. On the malleability of performance: Implications for seniors. J. Appl. Gerontol. 27, 446–465 (2008).
Meisner, B. A. A meta-analysis of positive and negative age stereotype priming effects on behavior among older adults. J. Gerontol. B Psychol. Sci. Soc. Sci. 67, 13–17 (2011).
Levy, B. & Langer, E. Aging free from negative stereotypes: successful memory in China among the American deaf. J. Pers. Soc. Psychol. 66, 989 (1994).
Yoon, C., Hasher, L., Feinberg, F., Rahhal, T. A. & Winocur, G. Cross-cultural differences in memory: the role of culture-based stereotypes about aging. Psychol. Aging 15, 694 (2000).
Thoits, P. A. Conceptual, methodological, and theoretical problems in studying social support as a buffer against life stress. J. Health Soc. Behav. 52, 145–159 (1982).
Midanik, L. T., Soghikian, K., Ransom, L. J. & Tekawa, I. S. The effect of retirement on mental health and health behaviors: the Kaiser Permanente Retirement Study. J. Gerontol. B Psychol. Sci. Soc. Sci. 50, S59–S61 (1995).
Reitzes, D. C., Mutran, E. J. & Fernandez, M. E. Does retirement hurt weil-being? Factors influencing self-esteem and depression among retires and workers. Gerontologist 36, 649–656 (1996).
Lee, J. & Smith, J. P. Work, retirement, and depression. J. Popul. Ageing 2, 57–71 (2009).
Coleman, L. M., Antonucci, T. C. & Adelmann, P. K. Role involvement, gender, and well-being. In F. J. Crosby (Ed.), Spouse, parent, worker: On gender and multiple roles (pp. 138–153). New Haven, CT, US: Yale University Press (1987).
Greenfield, E. A. & Marks, N. F. Formal volunteering as a protective factor for older adults' psychological well-being. J. Gerontol. B Psychol. Sci. Soc. Sci. 59, S258–S264 (2004).
Coleman, L. M., Antonucci, T. C. & Adelmann P. K. Role involvement, gender, and well-being (1987).
Hong, J. & Seltzer, M. M. The psychological consequences of multiple roles: the nonnormative case. J. Health Soc. Behav. 36, 386–398 (1995).
Morrow-Howell, N., Kinnevy, S. & Mann, M. The perceived benefits of participating in volunteer and educational activities. J. Gerontol. Soc. Work 3, 65–80 (1999).
Van Willigen, M. Differential benefits of volunteering across the life course. J. Gerontol. B Psychol. Sci. Soc. Sci. 55, S308–S318 (2000).
Hendricks, J. & Cutler, S. J. Volunteerism and socioemotional selectivity in later life. J. Gerontol. B Psychol. Sci. Soc. Sci. 59, S251–S257 (2004).
Li, Y. & Ferraro, K. F. Volunteering and depression in later life: social benefit or selection processes? J. Health Soc. Behav. 46, 68–84 (2005).
Li, Y. Recovering from spousal bereavement in later life: does volunteer participation play a role? J. Gerontol. B Psychol. Sci. Soc. Sci. 62, S257–S266 (2007).
Santini, Z. I., Koyanagi, A., Tyrovolas, S., Mason, C. & Haro, J. M. The association between social relationships and depression: a systematic review. J. Affect. Disord. 175, 53–65 (2015).
Schwarzbach, M., Luppa, M., Forstmeier, S., König, H. H. & Riedel‐Heller, S. G. Social relations and depression in late life—a systematic review. Int. J. Geriatr. Psychiatry 29, 1–21 (2014).
Hakulinen, C. et al. Social isolation and loneliness as risk factors for myocardial infarction, stroke and mortality: UK Biobank cohort study of 479 054 men and women. Heart 104, 1536–1542 (2018).
Golden, J. et al. Loneliness, social support networks, mood and wellbeing in community‐dwelling elderly. Int. J. Geriatr. Psychiatry 24, 694–700 (2009).
Cigna. Cigna U.S. Loneliness Index https://www.multivu.com/players/English/8294451-cigna-us-loneliness-survey/docs/IndexReport_1524069371598-173525450.pdf (2018). Accessed 25 August 2018.
Inaba, A. et al. Depression in the United States and Japan: gender, marital status, and SES patterns. Soc. Sci. Med. 61, 2280–2292 (2005).
Cacioppo, J. T., Hughes, M. E., Waite, L. J., Hawkley, L. C. & Thisted, R. A. Loneliness as a specific risk factor for depressive symptoms: cross-sectional and longitudinal analyses. Psychol. Aging 21, 140 (2006).
Fung, H. H., Carstensen, L. L. & Lang, F. R. Age-related patterns in social networks among European Americans and African Americans: implications for socioemotional selectivity across the life span. Int. J. Aging Human. Dev. 52, 185–206 (2001).
Carstensen, L. L. Evidence for a life-span theory of socioemotional selectivity. Curr. Dir. Psychol. Sci. 4, 151–156 (1995).
Böger, A. & Huxhold, O. Age-related changes in emotional qualities of the social network from middle adulthood into old age: how do they relate to the experience of loneliness? Psychol. Aging 33, 482–496 (2018).
Perissinotto, C. M., Cenzer, I. S. & Covinsky, K. E. Loneliness in older persons: a predictor of functional decline and death. Arch. Intern. Med. 172, 1078–1084 (2012).
Heikkinen, R.-L. & Kauppinen, M. Depressive symptoms in late life: a 10-year follow-up. Arch. Gerontol. Geriatr. 38, 239–250 (2004).
Cole, M. G. & Dendukuri, N. Risk factors for depression among elderly community subjects: a systematic review and meta-analysis. Am. J. Psychiatry 160, 1147–1156 (2003).
Pinquart, M. & Sörensen, S. Gender differences in self-concept and psychological well-being in old age: a meta-analysis. J. Gerontol. B Psychol. Sci. Soc. Sci. 56, P195–P213 (2001).
McDougall, F. A., Matthews, F. E., Kvaal, K., Dewey, M. E. & Brayne, C. Prevalence and symptomatology of depression in older people living in institutions in England and Wales. Age Ageing 36, 562–568 (2007).
Margrett, J. et al. Depression among centenarians and the oldest old: contributions of cognition and personality. Gerontology 56, 93–99 (2010).
Wilson, R. S. et al. Depressive symptoms, cognitive decline, and risk of AD in older persons. Neurology 59, 364–370 (2002).
Köhler, S. et al. Depressive symptoms and cognitive decline in community‐dwelling older adults. J. Am. Geriatr. Soc. 58, 873–879 (2010).
Barnes, D. E., Alexopoulos, G. S., Lopez, O. L., Williamson, J. D. & Yaffe, K. Depressive symptoms, vascular disease, and mild cognitive impairment: findings from the Cardiovascular Health Study. Arch. Gen. Psychiatry 63, 273–279 (2006).
Geda, Y. E. et al. Depression, apolipoprotein E genotype, and the incidence of mild cognitive impairment: a prospective cohort study. Arch. Neurol. 63, 435–440 (2006).
Saczynski, J. S. et al. Depressive symptoms and risk of dementia The Framingham Heart Study. Neurology 75, 35–41 (2010).
Barnes, D. E. et al. Midlife vs late-life depressive symptoms and risk of dementia: differential effects for Alzheimer disease and vascular dementia. Arch. Gen. Psychiatry 69, 493–498 (2012).
Ballmaier, M. et al. Hippocampal morphology and distinguishing late-onset from early-onset elderly depression. Am. J. Psychiatry 165, 229–237 (2008).
Elbejjani, M. et al. Depression, depressive symptoms, and rate of hippocampal atrophy in a longitudinal cohort of older men and women. Psychol. Med. 45, 1931–1944 (2015).
Hollocks, M. J. et al. Differential relationships between apathy and depression with white matter microstructural changes and functional outcomes. Brain 138, 3803–3815 (2015).
Smith, S. M. & Vale, W. W. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 8, 383 (2006).
Salvat-Pujol, N. et al. Hypothalamic-pituitary-adrenal axis activity and cognition in major depression: the role of remission status. Psychoneuroendocrinology 76, 38–48 (2017).
Wilson, R. S. et al. Clinical-pathologic study of depressive symptoms and cognitive decline in old age. Neurology 83, 702–709 (2014).
Laird, K. T., Lavretsky, H., Wu, P., Krause, B. & Siddarth, P. Neurocognitive correlates of resilience in late-life depression. Am. J. Geriatr. Psychiatry 27, 12–17 (2018).
Vlasova, R. M. et al. Resilience and white matter integrity in geriatric depression. Am. J. Geriatr. Psychiatry 26, 874–883 (2018).
Stern, Y. et al. Whitepaper: defining and investigating cognitive reserve, brain reserve, and brain maintenance. Alzheimers Dement. 1–7 (2018).
Stern, Y. Cognitive reserve. Neuropsychologia 47, 2015–2028 (2009).
Opdebeeck, C., Quinn, C., Nelis, S. M. & Clare, L. Does cognitive reserve moderate the association between mood and cognition? A systematic review. Rev. Clin. Gerontol. 25, 181–193 (2015).
Lachman, M. E. & Agrigoroaei, S. Promoting functional health in midlife and old age: long-term protective effects of control beliefs, social support, and physical exercise. PLoS ONE 5, e13297 (2010).
Sneed, J. R. et al. Response inhibition predicts poor antidepressant treatment response in very old depressed patients. Am. J. Geriatr. Psychiatry 15, 553–563 (2007).
Sheline, Y. I. et al. Support for the vascular depression hypothesis in late-life depression: results of a 2-site, prospective, antidepressant treatment trial. Arch. Gen. Psychiatry 67, 277–285 (2010).
Hammen, C. Stress and depression. Annu. Rev. Clin. Psychol. 1, 293–319 (2005).
Conway, C. C., Raposa, E. B., Hammen, C. & Brennan, P. A. Transdiagnostic pathways from early social stress to psychopathology: a 20‐year prospective study. J. Child Psychol. Psychiatry 59, 855–862 (2018).
Charney, D. S. Psychobiological mechanisms of resilience and vulnerability: implications for successful adaptation to extreme stress. Am. J. Psychiatry 161, 195–216 (2004).
McEwen, B. S. Sex, stress and the hippocampus: allostasis, allostatic load and the aging process. Neurobiol. Aging 23, 921–939 (2002).
Lee, R. S. & Sawa, A. Environmental stressors and epigenetic control of the hypothalamic-pituitary-adrenal axis. Neuroendocrinology 100, 278–287 (2014).
Pfau, M. L. & Russo, S. J. Peripheral and central mechanisms of stress resilience. Neurobiol. Stress 1, 66–79 (2015).
Seery, M. D., Leo, R. J., Lupien, S. P., Kondrak, C. L. & Almonte, J. L. An upside to adversity? Moderate cumulative lifetime adversity is associated with resilient responses in the face of controlled stressors. Psychol. Sci. 24, 1181–1189 (2013).
Lyons, D. M., Parker, K. J., Katz, M. & Schatzberg, A. F. Developmental cascades linking stress inoculation, arousal regulation, and resilience. Front. Behav. Neurosci. 3, 32 (2009).
Pakulak, E., Stevens, C. & Neville, H. Neuro-, cardio-, and immunoplasticity: effects of early adversity. Annu. Rev. Psychol. 69, 131–156 (2018).
Booth, T. et al. Association of allostatic load with brain structure and cognitive ability in later life. Neurobiol. Aging 36, 1390–1399 (2015).
Caspi, A. et al. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 301, 386–389 (2003).
Chopra, K., Kumar, B. & Kuhad, A. Pathobiological targets of depression. Expert Opin. Ther. Targets 15, 379–400 (2011).
Miller, G. E., Chen, E. & Zhou, E. S. If it goes up, must it come down? Chronic stress and the hypothalamic-pituitary-adrenocortical axis in humans. Psychol. Bull. 133, 25 (2007).
Heim, C., Ehlert, U. & Hellhammer, D. H. The potential role of hypocortisolism in the pathophysiology of stress-related bodily disorders. Psychoneuroendocrinology 25, 1–35 (2000).
Gold, P. W. & Chrousos, G. P. Organization of the stress system and its dysregulation in melancholic and atypical depression: high vs low CRH/NE states. Mol. Psychiatry 7, 254 (2002).
Mason, J. W. et al. Psychogenic lowering of urinary cortisol levels linked to increased emotional numbing and a shame-depressive syndrome in combat-related posttraumatic stress disorder. Psychosom. Med. 63, 387–401 (2001).
Mann, J. J. et al. Can biological tests assist prediction of suicide in mood disorders? Int. J. Neuropsychopharmacol. 9, 465–474 (2006).
Jokinen, J. & Nordstrom, P. HPA axis hyperactivity as suicide predictor in elderly mood disorder inpatients. Psychoneuroendocrinology 33, 1387–1393 (2008).
ter Heegde, F., De Rijk, R. H. & Vinkers, C. H. The brain mineralocorticoid receptor and stress resilience. Psychoneuroendocrinology 52, 92–110 (2015).
Leonard, B. E. Inflammation and depression: a causal or coincidental link to the pathophysiology? Acta Neuropsychiatr. 30, 1–16 (2018).
Enman, N. M., Sabban, E. L., McGonigle, P. & Van Bockstaele, E. J. Targeting the neuropeptide Y system in stress-related psychiatric disorders. Neurobiol. Stress 1, 33–43 (2015).
Tatemoto, K. Neuropeptide Y: history and overview. In Neuropeptide Y and related peptides (pp. 1–21). Springer, Berlin, Heidelberg (2004).
Heilig, M. et al. Decreased cerebrospinal fluid neuropeptide Y (NPY) in patients with treatment refractory unipolar major depression: preliminary evidence for association with preproNPY gene polymorphism. J. Psychiatr. Res. 38, 113–121 (2004).
Mickey, B. J. et al. Emotion processing, major depression, and functional genetic variation of neuropeptide Y. Arch. Gen. Psychiatry 68, 158–166 (2011).
Newhouse, P. & Albert, K. Estrogen, stress, and depression: a neurocognitive model. JAMA Psychiatry 72, 727–729 (2015).
Ryan, J., Carriere, I., Scali, J., Ritchie, K. & Ancelin, M. L. Lifetime hormonal factors may predict late-life depression in women. Int. Psychogeriatr. 20, 1203–1218 (2008).
Ryan, J. et al. Oestrogen receptor polymorphisms and late-life depression. Br. J. Psychiatry 199, 126–131 (2011).
Peters, R. Ageing and the brain. Postgrad. Med. J. 82, 84–88 (2006).
Hara, Y., Waters, E. M., McEwen, B. S. & Morrison, J. H. Estrogen effects on cognitive and synaptic health over the lifecourse. Physiol. Rev. 95, 785–807 (2015).
McEwen, B. S. & Morrison, J. H. The brain on stress: vulnerability and plasticity of the prefrontal cortex over the life course. Neuron 79, 16–29 (2013).
Rutkowski, K., Sowa, P., Rutkowska-Talipska, J., Kuryliszyn-Moskal, A. & Rutkowski, R. Dehydroepiandrosterone (DHEA): hypes and hopes. Drugs 74, 1195–1207 (2014).
Samaras, N., Samaras, D., Frangos, E., Forster, A. & Philippe, J. A review of age-related dehydroepiandrosterone decline and its association with well-known geriatric syndromes: is treatment beneficial? Rejuvenation Res. 16, 285–294 (2013).
Fabian, T. J. et al. Endogenous concentrations of DHEA and DHEA-S decrease with remission of depression in older adults. Biol. Psychiatry 50, 767–774 (2001).
Carney, R. M., Freedland, K. E., Sheline, Y. I. & Weiss, E. S. Depression and coronary heart disease: a review for cardiologists. Clin. Cardiol. 20, 196–200 (1997).
Nicholson, A., Kuper, H. & Hemingway, H. Depression as an aetiologic and prognostic factor in coronary heart disease: a meta-analysis of 6362 events among 146 538 participants in 54 observational studies. Eur. Heart J. 27, 2763–2774 (2006).
Hare, D. L., Toukhsati, S. R., Johansson, P. & Jaarsma, T. Depression and cardiovascular disease: a clinical review. Eur. Heart J. 35, 1365–1372 (2013).
Valkanova, V. & Ebmeier, K. P. Vascular risk factors and depression in later life: a systematic review and meta-analysis. Biol. Psychiatry 73, 406–413 (2013).
Brown, L. et al. Heart rate variability alterations in late life depression: a meta-analysis. J. Affect. Disord. 235, 456–466 (2018).
Carney, R. M. & Freedland, K. E. Depression and coronary heart disease. Nat. Rev. Cardiol. 14, 145 (2017).
Appelhans, B. M. & Luecken, L. J. Heart rate variability as an index of regulated emotional responding. Rev. Gen. Psychol. 10, 229 (2006).
Holzman, J. B. & Bridgett, D. J. Heart rate variability indices as bio-markers of top-down self-regulatory mechanisms: a meta-analytic review. Neurosci. Biobehav. Rev. 74, 233–255 (2017).
Carnevali, L., Koenig, J., Sgoifo, A. & Ottaviani, C. Autonomic and brain morphological predictors of stress resilience. Front. Neurosci. 12, 228 (2018).
Koenig, J., Kemp, A. H., Beauchaine, T. P., Thayer, J. F. & Kaess, M. Depression and resting state heart rate variability in children and adolescents—a systematic review and meta-analysis. Clin. Psychol. Rev. 46, 136–150 (2016).
Shaffer, F., McCraty, R. & Zerr, C. L. A healthy heart is not a metronome: an integrative review of the heart's anatomy and heart rate variability. Front. Psychol. 5, 1040 (2014).
Kemp, A. H. et al. Impact of depression and antidepressant treatment on heart rate variability: a review and meta-analysis. Biol. Psychiatry 67, 1067–1074 (2010).
Yoo, H. J. et al. Brain structural concomitants of resting state heart rate variability in the young and old: evidence from two independent samples. Brain Struct. Funct. 223, 727–737 (2018).
Umetani, K., Singer, D. H., McCraty, R. & Atkinson, M. Twenty-four hour time domain heart rate variability and heart rate: relations to age and gender over nine decades. J. Am. Coll. Cardiol. 31, 593–601 (1998).
Vogelzangs, N., Comijs, H. C., Oude Voshaar, R. C., Stek, M. L. & Penninx, B. W. Late-life depression symptom profiles are differentially associated with immunometabolic functioning. Brain Behav. Immun. 41, 109–115 (2014).
Duivis, H. E. et al. Depressive symptoms, health behaviors, and subsequent inflammation in patients with coronary heart disease: prospective findings from the heart and soul study. Am. J. Psychiatry 168, 913–920 (2011).
Kahl, K. G. et al. Prevalence of the metabolic syndrome in unipolar major depression. Eur. Arch. Psychiatry Clin. Neurosci. 262, 313–320 (2012).
Viinamäki, H. et al. Association of depressive symptoms and metabolic syndrome in men. Acta Psychiatr. Scand. 120, 23–29 (2009).
Eller, T., Vasar, V., Shlik, J. & Maron, E. Pro-inflammatory cytokines and treatment response to escitaloprsam in major depressive disorder. Prog. NeuroPsychopharmacol. Biol. Psychiatry 32, 445–450 (2008).
Lanquillon, S., Krieg, J. C., Bening-Abu-Shach, U. & Vedder, H. Cytokine production and treatment response in major depressive disorder. Neuropsychopharmacology 22, 370–379 (2000).
Kloiber, S. et al. Overweight and obesity affect treatment response in major depression. Biol. Psychiatry 62, 321–326 (2007).
Penninx, B. W., Milaneschi, Y., Lamers, F. & Vogelzangs, N.Understanding the somatic consequences of depression: biological mechanisms and the role of depression symptom profile. BMC Med. 11, 129 (2013).
Siennicki-Lantz, A., André-Petersson, L. & Elmståhl, S. Decreasing blood pressure over time is the strongest predictor of depressive symptoms in octogenarian men. Am. J. Geriatr. Psychiatry 21, 863–871 (2013).
Giltay, E. J. et al. Serum cholesterol, apolipoprotein E genotype and depressive symptoms in elderly European men: the FINE study. J. Affect. Disord. 115, 471–477 (2009).
Kuo, S. Y., Lin, K. M., Chen, C. Y., Chuang, Y. L. & Chen, W. J. Depression trajectories and obesity among the elderly in Taiwan. Psychol. Med. 41, 1665–1676 (2011).
Marijnissen, R. M., Naudé, P. J. W., Comijs, H. C., Schoevers, R. A. & Voshaar, R. C. O. Waist circumference and neutrophil gelatinase-associated lipocalin in late-life depression. Brain Behav. Immun. 37, 231–239 (2014).
Miller, A. H., Maletic, V. & Raison, C. L. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol. Psychiatry 65, 732–741 (2009).
Martinez-Cengotitabengoa, M. et al. Peripheral inflammatory parameters in late-life depression: a systematic review. Int. J. Mol. Sci. 17, 8–15 (2016).
Wiedlocha, M. et al. Effect of antidepressant treatment on peripheral inflammation markers - a meta-analysis. Prog. Neuropsychopharmacol. Biol. Psychiatry 80(Pt C), 217–226 (2018).
Kappelmann, N., Lewis, G., Dantzer, R., Jones, P. B. & Khandaker, G. M. Antidepressant activity of anti-cytokine treatment: a systematic review and meta-analysis of clinical trials of chronic inflammatory conditions. Mol. Psychiatry 23, 335–343 (2018).
Bierhaus, A. et al. A mechanism converting psychosocial stress into mononuclear cell activation. Proc. Natl Acad. Sci. USA 100, 1920–1925 (2003).
Danese, A., Pariante, C. M., Caspi, A., Taylor, A. & Poulton, R. Childhood maltreatment predicts adult inflammation in a life-course study. Proc. Natl Acad. Sci. USA 104, 1319–1324 (2007).
McDade, T. W., Hawkley, L. C. & Cacioppo, J. T. Psychosocial and behavioral predictors of inflammation in middle-aged and older adults: the Chicago health, aging, and social relations study. Psychosom. Med. 68, 376–381 (2006).
Kiecolt-Glaser, J. K. et al. Hostile marital interactions, proinflammatory cytokine production, and wound healing. Arch. Gen. Psychiatry 62, 1377–1384 (2005).
Miller, G. E. et al. A functional genomic fingerprint of chronic stress in humans: blunted glucocorticoid and increased NF-κB signaling. Biol. Psychiatry 64, 266–272 (2008).
Pei, Y. et al. The brain-derived neurotrophic-factor (BDNF) val66met polymorphism is associated with geriatric depression: a meta-analysis. Am. J. Med. Genet. B Neuropsychiatr. Genet. 159b, 560–566 (2012).
Tsang, R. S., Mather, K. A., Sachdev, P. S. & Reppermund, S. Systematic review and meta-analysis of genetic studies of late-life depression. Neurosci. Biobehav. Rev. 75, 129–139 (2017).
Egan, M. F. et al. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell 112, 257–269 (2003).
Hariri, A. R. et al. Brain-derived neurotrophic factor val66met polymorphism affects human memory-related hippocampal activity and predicts memory performance. J. Neurosci. 23, 6690–6694 (2003).
Gratacòs, M. et al. Brain-derived neurotrophic factor Val66Met and psychiatric disorders: meta-analysis of case-control studies confirm association to substance-related disorders, eating disorders, and schizophrenia. Biol. Psychiatry 61, 911–922 (2007).
Verhagen, M. et al. Meta-analysis of the BDNF Val66Met polymorphism in major depressive disorder: effects of gender and ethnicity. Mol. Psychiatry 15, 260 (2010).
Hosang, G. M., Shiles, C., Tansey, K. E., McGuffin, P. & Uher, R. Interaction between stress and the BDNF Val66Met polymorphism in depression: a systematic review and meta-analysis. BMC Med. 12, 7 (2014).
Januar, V., Ancelin, M. L., Ritchie, K., Saffery, R. & Ryan, J. BDNF promoter methylation and genetic variation in late-life depression. Transl. Psychiatry 5, e619 (2015).
Murphy, G. M. et al. BDNF and CREB1 genetic variants interact to affect antidepressant treatment outcomes in geriatric depression. Pharmacogenet. Genomics 23, 301–313 (2013).
Garcia, R. Stress, synaptic plasticity, and psychopathology. Rev. Neurosci. 13, 195–208 (2002).
Duman, R. S., Malberg, J., Nakagawa, S. & D’Sa, C. Neuronal plasticity and survival in mood disorders. Biol. Psychiatry 48, 732–739 (2000).
Öngür, D., Drevets, W. C. & Price, J. L. Glial reduction in the subgenual prefrontal cortex in mood disorders. Proc. Natl Acad. Sci. USA 95, 13290–13295 (1998).
Rosoklija, G. et al. Structural abnormalities of subicular dendrites in subjects with schizophrenia and mood disorders: preliminary findings. Arch. Gen. Psychiatry 57, 349–356 (2000).
Cotter, D. et al. Reduced neuronal size and glial cell density in area 9 of the dorsolateral prefrontal cortex in subjects with major depressive disorder. Cereb. Cortex 12, 386–394 (2002).
Miguel-Hidalgo, J. J. & Rajkowska, G. Morphological brain changes in depression. CNS Drugs 16, 361–372 (2002).
Rajkowska, G. Cell pathology in mood disorders. Semin. Clin. Neuropsychiatry 7, 281–292 (2002).
Geerlings, M. I. & Gerritsen, L. Late-life depression, hippocampal volumes, and hypothalamic-pituitary-adrenal axis regulation: a systematic review and meta-analysis. Biol. Psychiatry 82, 339–350 (2017).
Dwivedi, Y. Involvement of brain-derived neurotrophic factor in late-life depression. Am. J. Geriatr. Psychiatry 21, 433–449 (2013).
Aizenstein, H. J. et al. Altered functioning of the executive control circuit in late-life depression: episodic and persistent phenomena. Am. J. Geriatr. Psychiatry 17, 30–42 (2009).
Crocco, E. A., Castro, K. & Loewenstein, D. A. How late-life depression affects cognition: neural mechanisms. Curr. Psychiatry Rep. 12, 34–38 (2010).
Khundakar, A. A. & Thomas, A. J. Morphometric changes in early-and late-life major depressive disorder: evidence from postmortem studies. Int. Psychogeriatr. 21, 844–854 (2009).
Janssen, J. et al. Hippocampal volume and subcortical white matter lesions in late-life depression: comparison of early-and late-onset depression. J. Neurol. Neurosurg. Psychiatry 78, 638–640 (2007).
Narayan, M., Bremner, J. D. & Kumar, A. Neuroanatomic substrates of late-life mental disorders. J. Geriatr. Psychiatry Neurol. 12, 95–106 (1999).
Ebner, N. C. & Fischer, H. Emotion and aging: evidence from brain and behavior. Front. Psychol. 5, 996 (2014).
Urry, H. L. & Gross, J. J. Emotion regulation in older age. Curr. Dir. Psychol. Sci. 19, 352–357 (2010).
Allard, E. S. & Kensinger, E. A. Age-related differences in neural recruitment during the use of cognitive reappraisal and selective attention as emotion regulation strategies. Front. Psychol. 5, 296 (2014).
Dolcos, S., Katsumi, Y. & Dixon, R. A. The role of arousal in the spontaneous regulation of emotions in healthy aging: a fMRI investigation. Front. Psychol. 5, 681 (2014).
Santarnecchi, E. et al. Brain functional connectivity correlates of coping styles. Cogn. Affect. Behav. Neurosci. 18, 495–508 (2018).
Chen, D. et al. Negative association between resilience and event-related potentials evoked by negative emotion. Sci. Rep. 8, 7149 (2018).
Leaver, A. M. et al. Resilience and amygdala function in older healthy and depressed adults. J. Affect. Disord. 237, 27–34 (2018).
Coffey, C. E., Figiel, G. S., Djang, W. T., Saunders, W. B. & Weiner, R. D. White matter hyperintensity on magnetic resonance imaging: clinical and neuroanatomic correlates in the depressed elderly. J. Neuropsychiatry Clin. Neurosci 1, 135–144 (1989).
Dolan, R. J., Poynton, A. M., Bridges, P. K. & Trimble, M. R. Altered magnetic resonance white-matter T 1 values in patients with affective disorder. Br. J. Psychiatry 157, 107–110 (1990).
Lavretsky, H., Lesser, I. M., Wohl, M. & Miller, B. L. Relationship of age, age at onset, and sex to depression in older adults. Am. J. Geriatr. Psychiatry 6, 248–256 (1998).
Herrmann, L. L., LeMasurier, M. & Ebmeie, K. P. White matter hyperintensities in late life depression: a systematic review. J. Neurol. Neurosurg. Psychiatry 79, 619–624 (2007).
de Groot, J. C. et al. Cerebral white matter lesions and depressive symptoms in elderly adults. Arch. Gen. Psychiatry 57, 1071–1076 (2000).
Salloway, S. et al. MRI and neuropsychological differences in early-and late-life-onset geriatric depression. Neurology 46, 1567–1574 (1996).
Dillon, C. et al. Late-versus early-onset geriatric depression in a memory research center. Neuropsychiatr. Dis. Treat. 5, 517 (2009).
Rapp, M. A. et al. Neuropsychological differences between late-onset and recurrent geriatric major depression. Am. J. Psychiatry 162, 691–698 (2005).
Krishnan, K. R., Hays, J. C. & Blazer, D. G. MRI-defined vascular depression. Am. J. Psychiatry 154, 497–501 (1997).
Alexopoulos, G. S., Meyers, B. S., Young, R. C. & Kakuma, T. Clinically defined vascular depression. Am. J. Psychiatry 154, 562 (1997).
Taylor, W. D., Aizenstein, H. J. & Alexopoulos, G. S. The vascular depression hypothesis: mechanisms linking vascular disease with depression. Mol. Psychiatry 18, 963–974 (2013).
Taylor, W. D., Steffens, D. C. & Krishnan, K. R. Psychiatric disease in the twenty-first century: The case for subcortical ischemic depression. Biol. Psychiatry 60, 1299–1303 (2006).
Kamat, R. et al. Apathy is associated with white matter abnormalities in anterior, medial brain regions in persons with HIV infection. J. Clin. Exp. Neuropsychol. 36, 854–866 (2014).
Alves, G. S. et al. Clinical characteristics in subcortical ischemic white matter disease. Arq. Neuropsiquiatr. 67(2A), 173–178 (2009).
Grool, A. M. et al. Structural MRI correlates of apathy symptoms in older persons without dementia AGES-Reykjavik Study. Neurology 82, 1628–1635 (2014).
Bruce, D. G. et al. Apathy in older patients with type 2 diabetes. Am. J. Geriatr. Psychiatry 23, 615–621 (2015).
Ligthart, S. A. et al. Association of vascular factors with apathy in community-dwelling elderly individuals. Arch. Gen. Psychiatry 69, 636–642 (2012).
Baldwin, R. C. & Tomenson, B. Depression in later life: a comparison of symptoms and risk factors in early and late onset cases. Br. J. Psychiatry 167, 649–652 (1995).
Driscoll, H. C. et al. Late‐onset major depression: clinical and treatment‐response variability. Int. J. Geriatr. Psychiatry 20, 661–667 (2005).
Whyte, E. M. et al. Geriatric depression treatment in nonresponders to selective serotonin reuptake inhibitors. J. Clin. Psychiatry 65, 1634–1641 (2004).
Karp, J. F. et al. Time to remission in late‐life depression: analysis of effects of demographic, treatment, and life‐events measures. Depression 1, 250–256 (1993).
Reynolds, C. Fr et al. Effects of age at onset of first lifetime episode of recurrent major depression on treatment response and illness course in elderly patients. Am. J. Psychiatry 155, 795–799 (1998).
Seligman, M. E. & Csikszentmihalyi, M. Positive Psychology: An Introduction (Vol. 55, p. 5–14), American Psychological Association (2000).
Sin, N. L. & Lyubomirsky, S. Enhancing well‐being and alleviating depressive symptoms with positive psychology interventions: a practice‐friendly meta‐analysis. J. Clin. Psychol. 65, 467–487 (2009).
Diener, E. Subjective well-being. Psychol. Bull. 95, 542 (1984).
Ryan, R. M. & Deci, E. L. On happiness and human potentials: a review of research on hedonic and eudaimonic well-being. Annu. Rev. Psychol. 52, 141–166 (2001).
Ryff, C. D. Happiness is everything, or is it? Explorations on the meaning of psychological well-being. J. Pers. Soc. Psychol. 57, 1069 (1989).
Seligman, M. E. P., Steen, T. A., Park, N. & Peterson, C. Positive psychology progress: empirical validation of interventions. Am. Psychol. 60, 410 (2005).
Sheldon, K. M. & Lyubomirsky, S. How to increase and sustain positive emotion: the effects of expressing gratitude and visualizing best possible selves. J. Posit. Psychol. 1, 73–82 (2006).
Peters, M. L., Flink, I. K., Boersma, K. & Linton, S. J. Manipulating optimism: can imagining a best possible self be used to increase positive future expectancies? J. Posit. Psychol. 5, 204–211 (2010).
Peters, M. L., Meevissen, Y. M. C. & Hanssen, M. M. Specificity of the Best Possible Self intervention for increasing optimism: comparison with a gratitude intervention. Ter. Psicol. 31, 93–100 (2013).
Meevissen, Y. M. C., Peters, M. L. & Alberts, H. J. E. M. Become more optimistic by imagining a best possible self: effects of a two week intervention. J. Behav. Ther. Exp. Psychiatry 42, 371–378 (2011).
Zautra, A. J. et al. Comparison of cognitive behavioral and mindfulness meditation interventions on adaptation to rheumatoid arthritis for patients with and without history of recurrent depression. J. Consult. Clin. Psychol. 76, 408 (2008).
Lyubomirsky, S., Sheldon, K. M. & Schkade, D. Pursuing happiness: the architecture of sustainable change. Rev. Gen. Psychol. 9, 111 (2005).
Anik, L., Aknin, L. B., Norton, M. I. & Dunn, E. W. Feeling Good About Giving: the Benefits (And Costs) of Self-interested Charitable Behavior. Harvard Business School Marketing Unit Working Paper (2009).
Lyubomirsky, S., Tkach, C. & Sheldon K. M. Pursuing sustained happiness through random acts of kindness and counting one’s blessings: tests of two six-week interventions. Unpublished raw data (2004).
Buchanan, K. E. & Bardi, A. Acts of kindness and acts of novelty affect life satisfaction. J. Soc. Psychol. 150, 235–237 (2010).
Helgeson, V. S., Reynolds, K. A. & Tomich, P. L. A meta-analytic review of benefit finding and growth. J. Consult. Clin. Psychol. 74, 797 (2006).
Algoe, S. B. & Stanton, A. L. Is benefit finding good for individuals with chronic disease? In C. L. Park, S. C. Lechner, M. H. Antoni, & A. L. Stanton (Eds.), Medical illness and positive life change: Can crisis lead to personal transformation? (pp. 173–193). Washington, DC, US: American Psychological Association (2009).
Bower, J. E., Moskowitz, J. T. & Epel, E. Is benefit finding good for your health? Pathways linking positive life changes after stress and physical health outcomes. Curr. Dir. Psychol. Sci. 18, 337–341 (2009).
Uchino, B. N., Cacioppo, J. T. & Kiecolt-Glaser, J. K. The relationship between social support and physiological processes: a review with emphasis on underlying mechanisms and implications for health. Psychol. Bull. 119, 488 (1996).
Bower, J. E., Kemeny, M. E., Taylor, S. E. & Fahey, J. L. Finding positive meaning and its association with natural killer cell cytotoxicity among participants in a bereavement-related disclosure intervention. Ann. Behav. Med. 25, 146–155 (2003).
Bower, J. E. & Segerstrom, S. C. Stress management, finding benefit, and immune function: positive mechanisms for intervention effects on physiology. J. Psychosom. Res. 56, 9–11 (2004).
Kabat-Zinn, J. Mindfulness‐based interventions in context: past, present, and future. Clin. Psychol. Sci. Pract. 10, 144–156 (2003).
Khoury, B. et al. Mindfulness-based therapy: a comprehensive meta-analysis. Clin. Psychol. Rev. 33, 763–771 (2013).
Hazlett-Stevens, H., Singer, J. & Chong, A. Mindfulness-based stress reduction and mindfulness-based cognitive therapy with older adults: a qualitative review of randomized controlled outcome research. Clin. Gerontol. 1–12 (2018).
Piet, J. & Hougaard, E. The effect of mindfulness-based cognitive therapy for prevention of relapse in recurrent major depressive disorder: a systematic review and meta-analysis. Clin. Psychol. Rev. 31, 1032–1040 (2011).
Gu, J., Strauss, C., Bond, R. & Cavanagh, K. How do mindfulness-based cognitive therapy and mindfulness-based stress reduction improve mental health and wellbeing? A systematic review and meta-analysis of mediation studies. Clin. Psychol. Rev. 37, 1–12 (2015).
Luders, E. Exploring age‐related brain degeneration in meditation practitioners. Ann. N. Y. Acad. Sci. 1307, 82–88 (2014).
Boccia, M., Piccardi, L. & Guariglia, P. The meditative mind: a comprehensive meta-analysis of MRI studies. BioMed Res. Int. 2015, 1–11 (2015).
Tang, Y.-Y., Hölzel, B. K. & Posner, M. I. The neuroscience of mindfulness meditation. Nat. Rev. Neurosci. 16, 213 (2015).
Chiesa, A. & Serretti, A. A systematic review of neurobiological and clinical features of mindfulness meditations. Psychol. Med. 40, 1239–1252 (2010).
Kurth, F., Cherbuin, N. & Luders, E. Aging mindfully to minimize cognitive decline. J. Cogn. Enhanc. 1, 108–114 (2017).
Epel, E., Daubenmier, J., Moskowitz, J. T., Folkman, S. & Blackburn, E. Can meditation slow rate of cellular aging? cognitive stress, mindfulness, and telomeres. Ann. N. Y. Acad. Sci. 1172, 34–53 (2009).
Lavretsky, H. et al. A pilot study of yogic meditation for family dementia caregivers with depressive symptoms: effects on mental health, cognition, and telomerase activity. Int. J. Geriatr. Psychiatry 28, 57–65 (2013).
Black, D. S. et al. Yogic meditation reverses NF-kappaB and IRF-related transcriptome dynamics in leukocytes of family dementia caregivers in a randomized controlled trial. Psychoneuroendocrinology 38, 348–355 (2013).
Amin, A. et al. Beneficial effects of OM chanting on depression, anxiety, stress and cognition in elderly women with hypertension. Indian J. Clin. Anat. Physiol. 3, 253–255 (2016).
Kalyani, B. G. et al. Neurohemodynamic correlates of ‘OM’chanting: a pilot functional magnetic resonance imaging study. Int. J. Yoga 4, 3 (2011).
Cuijpers, P., Karyotaki, E., Pot, A. M., Park, M. & Reynolds Iii, C. F. Managing depression in older age: psychological interventions. Maturitas 79, 160–169 (2014).
Pinquart, M., Duberstein, P. R. & Lyness, J. M. Treatments for later-life depressive conditions: a meta-analytic comparison of pharmacotherapy and psychotherapy. Am. J. Psychiatry 163, 1493–1501 (2006).
Kishita, N., Takei, Y. & Stewart, I. A meta‐analysis of third wave mindfulness‐based cognitive behavioral therapies for older people. Int. J. Geriatr. Psychiatry 32, 1352–1361 (2017).
Zhao, K., Bai, Z. G., Bo, A. & Chi, I. A systematic review and meta‐analysis of music therapy for the older adults with depression. Int. J. Geriatr. Psychiatry 31, 1188–1198 (2016).
Laufer, S., Engel, S., Knaevelsrud, C. & Schumacher, S. Cortisol and alpha-amylase assessment in psychotherapeutic intervention studies: a systematic review. Neurosci. Biobehav. Rev. 95, 235–262 (2018).
Wegner, M. et al. Effects of exercise on anxiety and depression disorders: review of meta-analyses and neurobiological mechanisms. CNS Neurol. Disord. Drug Targets 13, 1002–1014 (2014).
Blake, H., Mo, P., Malik, S. & Thomas, S. How effective are physical activity interventions for alleviating depressive symptoms in older people? A systematic review. Clin. Rehabil. 23, 873–887 (2009).
Hoffman-Goetz, L. & Pedersen, B. K. Exercise and the immune system: a model of the stress response? Immunol. Today 15, 382–387 (1994).
Laird, K. T., Paholpak, P., Roman, M., Rahi, B. & Lavretsky, H. Mind-body therapies for late-life mental and cognitive health. Curr. Psychiatry Rep. 20, 2 (2018).
Acevedo, B. P., Pospos, S. & Lavretsky, H. The neural mechanisms of meditative practices: novel approaches for healthy aging. Curr. Behav. Neurosci. Rep. 3, 328–339 (2016).
Pascoe, M. C. & Bauer, I. E. A systematic review of randomised control trials on the effects of yoga on stress measures and mood. J. Psychiatr. Res. 68, 270–282 (2015).
Pascoe, M. C., Thompson, D. R. & Ski, C. F. Yoga, mindfulness-based stress reduction and stress-related physiological measures: a meta-analysis. Psychoneuroendocrinology 86, 152–168 (2017).
Gothe, N. P., Keswani, R. K. & McAuley, E. Yoga practice improves executive function by attenuating stress levels. Biol. Psychol. 121(Pt A), 109–116 (2016).
Prakhinkit, S., Suppapitiporn, S., Tanaka, H. & Suksom, D. Effects of Buddhism walking meditation on depression, functional fitness, and endothelium-dependent vasodilation in depressed elderly. J. Altern. Complement. Med. 20, 411–416 (2014).
Shahidi, M. et al. Laughter yoga versus group exercise program in elderly depressed women: a randomized controlled trial. Int. J. Geriatr. Psychiatry 26, 322–327 (2011).
Tsang, H. W. et al. Psycho-physical and neurophysiological effects of qigong on depressed elders with chronic illness. Aging Ment. Health 17, 336–348 (2013).
Sneed, J. R. et al. Design makes a difference: a meta-analysis of antidepressant response rates in placebo-controlled versus comparator trials in late-life depression. Am. J. Geriatr. Psychiatry 16, 65–73 (2008).
Ma, Y. Neuropsychological mechanism underlying antidepressant effect: a systematic meta-analysis. Mol. Psychiatry 20, 311 (2015).
Davidson, J. R. Major depressive disorder treatment guidelines in America and Europe. J. Clin. Psychiatry 71, e04 (2010).
Dierckx, B., Heijnen, W. T., van den Broek, W. W. & Birkenhäger, T. K. Efficacy of electroconvulsive therapy in bipolar versus unipolar major depression: a meta‐analysis. Bipolar Disord. 14, 146–150 (2012).
Carney, S. et al. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet 361, 799–808 (2003).
O'Connor, M. K. et al. The influence of age on the response of major depression to electroconvulsive therapy: a CORE Report. Am. J. Geriatr. Psychiatry 9, 382–390 (2001).
Hickie, I., Mason, C., Parker, G. & Brodaty, H. Prediction of ECT response: validation of a refined sign-based (CORE) system for defining melancholia. Br. J. Psychiatry 169, 68–74 (1996).
Tew, J. D. Jr et al. Acute efficacy of ECT in the treatment of major depression in the old-old. Am. J. Psychiatry 156, 1865–1870 (1999).
Van der Wurff, F. B., Stek, M. L., Hoogendijk, W. J. G. & Beekman, A. T. F. The efficacy and safety of ECT in depressed older adults: a literature review. Int. J. Geriatr. Psychiatry 18, 894–904 (2003).
Flint, A. J. & Gagnon, N. Effective use of electroconvulsive therapy in late-life depression. Can. J. Psychiatry 47, 734–741 (2002).
Bouckaert, F. et al. Grey matter volume increase following electroconvulsive therapy in patients with late life depression: a longitudinal MRI study. J. Psychiatry Neurosci. 41, 105 (2016).
Sackeim, H. A. Electroconvulsive therapy in late-life depression. Clin. Geriatr. Psychopharmacol. 4, 385–422 (2004).
Nobuhara, K. et al. Effects of electroconvulsive therapy on frontal white matter in late-life depression: a diffusion tensor imaging study. Neuropsychobiology 50, 48–53 (2004).
Karavidas, M. K. et al. Preliminary results of an open label study of heart rate variability biofeedback for the treatment of major depression. Appl. Psychophysiol. Biofeedback 32, 19–30 (2007).
Siepmann, M., Aykac, V., Unterdörfer, J., Petrowski, K. & Mueck-Weymann, M. A pilot study on the effects of heart rate variability biofeedback in patients with depression and in healthy subjects. Appl. Psychophysiol. Biofeedback 33, 195–201 (2008).
Patron, E. et al. Biofeedback assisted control of respiratory sinus arrhythmia as a biobehavioral intervention for depressive symptoms in patients after cardiac surgery: a preliminary study. Appl. Psychophysiol. Biofeedback 38, 1–9 (2013).
Lehrer, P. M. & Gevirtz, R. Heart rate variability biofeedback: how and why does it work? Front. Psychol. 5, 756 (2014).
Lyubomirsky, S., Dickerhoof, R., Boehm, J. K. & Sheldon, K. M. Becoming happier takes both a will and a proper way: an experimental longitudinal intervention to boost well-being. Emotion 11, 391 (2011).
Fischer, S., Strawbridge, R., Vives, A. H. & Cleare, A. J. Cortisol as a predictor of psychological therapy response in depressive disorders: systematic review and meta-analysis. Br. J. Psychiatry 210, 105–109 (2017).
Schlosser, N., Wolf, O. T. & Wingenfeld, K. Cognitive correlates of hypothalamic–pituitary–adrenal axis in major depression. Expert Rev. Endocrinol. Metab. 6, 109–126 (2011).
Stahl, S. T. et al. Systematic review of dyadic and family‐oriented interventions for late‐life depression. Int. J. Geriatr. Psychiatry 31, 963–973 (2016).
Paller, K. A. et al. Benefits of mindfulness training for patients with progressive cognitive decline and their caregivers. Am. J. Alzheimer's Dis. Other Demen. 30, 257–267 (2015).
Birnie, K., Garland, S. N. & Carlson, L. E. Psychological benefits for cancer patients and their partners participating in mindfulness‐based stress reduction (MBSR). Psychooncology 19, 1004–1009 (2010).
Berk, L., Warmenhoven, F., van Os, J. & van Boxtel, M. Mindfulness training for people with dementia and their caregivers: rationale, current research, and future directions. Front. Psychol. 9, 982 (2018).
Tang, F., Choi, E. & Morrow-Howell, N. Organizational support and volunteering benefits for older adults. Gerontologist 50, 603–612 (2010).
Holvast, F. et al. Loneliness is associated with poor prognosis in late-life depression: Longitudinal analysis of the Netherlands study of depression in older persons. J. Affect. Disord. 185, 1–7 (2015).
Chen, S. C., Jones, C. & Moyle, W. Social robots for depression in older adults: a systematic review. J. Nurs. Scholarsh 50, 612 (2018).
Bonura, K. B. & Tenenbaum, G. Effects of yoga on psychological health in older adults. J. Phys. Act. Health 11, 1334–1341 (2014).
Eyre, H. A. et al. A randomized controlled trial of Kundalini yoga in mild cognitive impairment. Int. Psychogeriatr. 29, 557–567 (2017).
Zhang, J. X. et al. Mindfulness-based stress reduction for chronic insomnia in adults older than 75 years: a randomized, controlled, single-blind clinical trial. Explore (NY) 11, 180–185 (2015).
Dechamps, A. et al. Effects of exercise programs to prevent decline in health-related quality of life in highly deconditioned institutionalized elderly persons: a randomized controlled trial. Arch. Intern. Med. 170, 162–169 (2010).
Lam, L. C. W. et al. A 1-year randomized controlled trial comparing mind body exercise (Tai Chi) with stretching and toning exercise on cognitive function in older Chinese adults at risk of cognitive decline. J. Am. Med. Dir. Assoc. 13, 568.e15–568.e20 (2012).
Vasudev, A. et al. A training programme involving automatic self-transcending meditation in late-life depression: preliminary analysis of an ongoing randomised controlled trial. BJPsych. Open 2, 195–198 (2016).
Waelde, L. C., Meyer, H., Thompson, J. M., Thompson, L. & Gallagher-Thompson, D. Randomized controlled trial of inner resources meditation for family dementia caregivers. J. Clin. Psychol. 73, 1629–1641 (2017).
Acknowledgements
This work was supported by NIH grants AT009198, MH097892, AT008383, and MH114981, as well as by Alzheimer’s Research and Prevention Foundation grants to Dr. Lavretsky.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H.L. received research support from Allergan/Forest Laboratories. The remaining authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Laird, K.T., Krause, B., Funes, C. et al. Psychobiological factors of resilience and depression in late life. Transl Psychiatry 9, 88 (2019). https://doi.org/10.1038/s41398-019-0424-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-019-0424-7
This article is cited by
-
Communication Preferences and Factors Predicting Smartphone Addiction Among Four Generations of Australians: Gender and Generational Differences
Journal of Technology in Behavioral Science (2024)
-
The impact of mind–body therapies on the mental health of women victims of violence: A meta-analysis
Archives of Women's Mental Health (2024)
-
Depression, anxiety, stress, and PTSD symptoms during the first and second COVID-19 waves: a comparison of elderly, middle-aged, and young people in Iran
BMC Psychiatry (2023)
-
Multidimensional computational study to understand non-coding RNA interactions in breast cancer metastasis
Scientific Reports (2023)
-
Biological factors influencing depression in later life: role of aging processes and treatment implications
Translational Psychiatry (2023)