Abstract
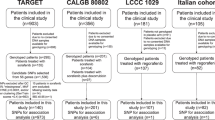
No biomarkers are available to predict patients at risk of developing hypertension induced by VEGF-pathway inhibitors. This study aimed to identify predictive biomarkers of hypertension induced by these drugs using a discovery-replication approach. The discovery set included 140 sorafenib-treated patients (TARGET study) genotyped for 973 SNPs in 56 genes. The most statistically significant SNPs associated with grade ≥2 hypertension were tested for association with grade ≥2 hypertension in the replication set of a GWAS of 1039 bevacizumab-treated patients from four clinical trials (CALGB/Alliance). In the discovery set, rs444904 (G > A) in PIK3R5 was associated with an increased risk of sorafenib-induced hypertension (p = 0.006, OR = 3.88 95% CI 1.54–9.81). In the replication set, rs427554 (G > A) in PIK3R5 (in complete linkage disequilibrium with rs444904) was associated with an increased risk of bevacizumab-induced hypertension (p = 0.008, OR = 1.39, 95% CI 1.09–1.78). This study identified a predictive marker of drug-induced hypertension that should be evaluated for other VEGF-pathway inhibitors.
ClinicalTrials.gov Identifier:NCT00073307 (TARGET).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
21 December 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41397-021-00264-2
References
Wilhelm SM, Carter C, Tang LY, Wilkie D, McNabola A, Rong H, et al. BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res. 2004;64:7099–109.
Nexavar (Sorafenib). 2018. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/021923s020lbl.pdf. Accessed 28 Jul 2020.
Costa LJ, Drabkin HA. Renal cell carcinoma: new developments in molecular biology and potential for targeted therapies. Oncologist. 2007;12:1404–15.
Li Y, Li S, Zhu Y, Liang X, Meng H, Chen J, et al. Incidence and risk of sorafenib-induced hypertension: a systematic review and meta-analysis. J Clin Hypertens. 2014;16:177–85.
Mir O, Coriat R, Cabanes L, Ropert S, Billemont B, Alexandre J, et al. An observational study of bevacizumab-induced hypertension as a clinical biomarker of antitumor activity. Oncologist. 2011;16:1325–32.
Qi WX, Lin F, Sun YJ, Tang LN, He AN, Yao Y, et al. Incidence and risk of hypertension with pazopanib in patients with cancer: a meta-analysis. Cancer Chemother Pharm. 2013;71:431–9.
Zhu X, Stergiopoulos K, Wu S. Risk of hypertension and renal dysfunction with an angiogenesis inhibitor sunitinib: systematic review and meta-analysis. Acta Oncologica. 2009;48:9–17.
Maitland ML, Kasza KE, Karrison T, Moshier K, Sit L, Black HR, et al. Ambulatory monitoring detects sorafenib-induced blood pressure elevations on the first day of treatment. Clin Cancer Res. 2009;15:6250–7.
Karar J, Maity A. PI3K/AKT/mTOR pathway in angiogenesis. Front Mol Neurosci. 2011;4:51.
Horowitz JR, Rivard A, Van Der Zee R, Hariawala M, Sheriff DD, Esakof DD, et al. Vascular endothelial growth factor/vascular permeability factor produces nitric oxide-dependent hypotension: evidence for a maintenance role in quiescent adult endothelium. Arterioscler Thromb Vasc Biol. 1997;17:2793–2800.
Rini BI. Quantifying hypertension in patients with cancer treated with sorafenib. Lancet Oncol. 2008;9:86–87.
Qin C, Cao Q, Li P, Wang S, Wang J, Wang M, et al. The influence of genetic variants of sorafenib on clinical outcomes and toxic effects in patients with advanced renal cell carcinoma. Sci Rep. 2016;6:20089.
Robinson ES, Khankin EV, Karumanchi SA, Humphreys BD. Hypertension induced by vascular endothelial growth factor signaling pathway inhibition: Mechanisms and potential use as a biomarker. Semin Nephrol. 2010;30:591–601.
Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N. Engl J Med. 2007;356:125–34.
Crona DJ, Skol AD, Leppanen VM, Glubb DM, Etheridge AS, Hilliard E, et al. Genetic variants of VEGFA and FLT4 are determinants of survival in renal cell carcinoma patients treated with sorafenib. Cancer Res. 2019;79:231–41.
Quintanilha JCF, Wang J, Sibley AB, Jiang C, Etheridge AS, Shen F et al. Bevacizumab-induced hypertension and proteinuria: a genome-wide analysis of more than 1,000 patients. Br J Cancer 2021. https://doi.org/10.1038/s41416-021-01557-w
Kindler HL, Niedzwiecki D, Hollis D, Sutherland S, Schrag D, Hurwitz H, et al. Gemcitabine plus bevacizumab compared with gemcitabine plus placebo in patients with advanced pancreatic cancer: Phase III trial of the Cancer and Leukemia Group B (CALGB 80303). J Clin Oncol. 2010;28:3617–22.
Dickler MN, Barry WT, Cirrincione CT, Ellis MJ, Moynahan ME, Innocenti F, et al. Phase III trial evaluating letrozole as first-line endocrine therapy with or without bevacizumab for the treatment of postmenopausal women with hormone receptor-positive advanced-stage breast cancer: CALGB 40503 (Alliance). J Clin Oncol. 2016;34:2602–9.
Kelly WK, Halabi S, Carducci M, George D, Mahoney JF, Stadler WM, et al. Randomized, double-blind, placebo-controlled phase III trial comparing docetaxel and prednisone with or without bevacizumab in men with metastatic castration-resistant prostate cancer: CALGB 90401. J Clin Oncol. 2012;30:1534–40.
Rugo HS, Barry WT, Moreno-Aspitia A, Lyss AP, Cirrincione C, Leung E, et al. Randomized phase III trial of paclitaxel once per week compared with nanoparticle albumin-bound nab-paclitaxel once per week or ixabepilone with bevacizumab as first-line chemotherapy for locally recurrent or metastatic breast cancer: CALGB 40502/NCCTG N0. J Clin Oncol. 2015;33:2361–9.
Innocenti F, Owzar K, Cox NL, Evans P, Kubo M, Zembutsu H, et al. A genome-wide association study of overall survival in pancreatic cancer patients treated with gemcitabine in CALGB 80303. Clin Cancer Res. 2012;18:577–84.
Baldwin RM, Owzar K, Zembutsu H, Chhibber A, Kubo M, Jiang C, et al. A genome-wide association study identifies novel loci for paclitaxel-induced sensory peripheral neuropathy in CALGB 40101. Clin Cancer Res. 2012;18:5099–109.
Rashkin SR, Chua KC, Ho C, Mulkey F, Jiang C, Mushiroda T, et al. A pharmacogenetic prediction model of progression-free survival in breast cancer using genome-wide genotyping data from CALGB 40502 (Alliance). Clin Pharm Ther. 2019;105:738–45.
Machiela MJ, Chanock SJ. LDlink: A web-based application for exploring population-specific haplotype structure and linking correlated alleles of possible functional variants. Bioinformatics. 2015;31:3555–7.
Zhang W, Gamazon ER, Zhang X, Konkashbaev A, Liu C, Szilágyi KL, et al. SCAN database: facilitating integrative analyses of cytosine modification and expression QTL. Database. 2015;27:bav025.
Boyle AP, Hong EL, Hariharan M, Cheng Y, Schaub MA, Kasowski M, et al. Annotation of functional variation in personal genomes using RegulomeDB. Genome Res. 2012;22:1790–7.
Dayem Ullah AZ, Oscanoa J, Wang J, Nagano A, Lemoine NR, Chelala C. SNPnexus: assessing the functional relevance of genetic variation to facilitate the promise of precision medicine. Nucleic Acids Res. 2018;46:W109–W113.
Ardlie KG, DeLuca DS, Segrè AV, Sullivan TJ, Young TR, Gelfand ET, et al. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science. 2015;348:648–60.
Zuo C, Shin S, Keleş S. AtSNP: Transcription factor binding affinity testing for regulatory SNP detection. Bioinformatics. 2015;31:3353–5.
Zhong H, Chiles K, Feldser D, Laughner E, Hanrahan C, Georgescu MM, et al. Modulation of hypoxia-inducible factor 1α expression by the epidermal growth factor/phosphatidylinositol 3-kinase/PTEN/AKT/FRAP pathway in human prostate cancer cells: Implications for tumor angiogenesis and therapeutics. Cancer Res. 2000;60:1541–5.
Forsythe JA, Jiang BH, Iyer NV, Agani F, Leung SW, Koos RD, et al. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol Cell Biol. 1996;16:4604–13.
Tassan N AL, Khalil D, Shinwari J, Sharif L AL, Bavi P, Abduljaleel Z, et al. A Missense mutation in PIK3R5 gene in a family with ataxia and oculomotor apraxia. Hum Mutat. 2012;33:351–4.
Li LY, Kim HJ, Park SA, Lee SH, Kim LK, Lee JY, et al. Genetic profiles associated with chemoresistance in patient-derived xenograft models of ovarian cancer. Cancer Res Treat. 2019;51:1117–27.
Buniello A, Macarthur JAL, Cerezo M, Harris LW, Hayhurst J, Malangone C, et al. The NHGRI-EBI GWAS Catalog of published genome-wide association studies, targeted arrays and summary statistics 2019. Nucleic Acids Res. 2019;47:D1005–D1012.
Shymanets A, Prajwal, Bucher K, Beer-Hammer S, Harteneck C, Nürnberg B. P87 and p101 subunits are distinct regulators determining class I B phosphoinositide 3-kinase (PI3K) specificity. J Biol Chem. 2013;288:31059–68.
Serban D, Leng J, Cheresh D. H-ras regulates angiogenesis and vascular permeability by activation of distinct downstream effectors. Circ Res. 2008;102:1350–8.
Blattler A, Farnham PJ. Cross-talk between site-specific transcription factors and DNA methylation states. J Biol Chem. 2013;288:34287–94.
Sherry ST. dbSNP: the NCBI database of genetic variation. Nucleic Acids Res. 2001;29:308–11.
Funding
This work was supported by the National Cancer Institute of the National Institutes of Health under Award Numbers U10CA180821 and U10CA180882 (to the Alliance for Clinical Trials in Oncology), U24CA196171, R21CA139280-01, and K07CA140390-01. JCFQ was supported by the São Paulo Research Foundation (FAPESP 2018/04491-2).
Author information
Authors and Affiliations
Contributions
JCFQ contributed to formal analysis, investigation and writing – original draft, review and editing. AR contribute to formal analysis and investigation. JW contributed to data curation, methodology and formal analysis. ASE contributed to formal analysis, investigation and writing – review and editing. SD contributed to formal analysis and investigation. CEP was responsible for resources. ADS contributed to data curation, formal analysis and methodology. DJC contributed to formal analysis and writing – review and editing. DL contributed to data curation, methodology and supervision. FI was responsible for conceptualization and funding acquisition, and contributed to formal analysis, investigation, methodology, supervision and writing – original draft, review and editing.
Corresponding authors
Ethics declarations
Competing interests
JCFQ, JW, DL, and FI are coinventors of a patent application, serial number 16/932,002. FI is an advisor for Emerald Lake Safety. These relationships have been disclosed to and are under management by UNC-Chapel Hill. CEP was employed by Bayer Health Care Pharmaceuticals at the time this work was conceived.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The Funding Information has been revised.
Supplementary information
Rights and permissions
About this article
Cite this article
Quintanilha, J.C.F., Racioppi, A., Wang, J. et al. PIK3R5 genetic predictors of hypertension induced by VEGF-pathway inhibitors. Pharmacogenomics J 22, 82–88 (2022). https://doi.org/10.1038/s41397-021-00261-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41397-021-00261-5