Abstract
Nutriments have been deemed to impact all physiopathologic processes. Recent evidences in molecular medicine and clinical trials have demonstrated that adequate nutrition treatments are the golden criterion for extending healthspan and delaying ageing in various species such as yeast, drosophila, rodent, primate and human. It emerges to develop the precision-nutrition therapeutics to slow age-related biological processes and treat diverse diseases. However, the nutritive advantages frequently diversify among individuals as well as organs and tissues, which brings challenges in this field. In this review, we summarize the different forms of dietary interventions extensively prescribed for healthspan improvement and disease treatment in pre-clinical or clinical. We discuss the nutrient-mediated mechanisms including metabolic regulators, nutritive metabolism pathways, epigenetic mechanisms and circadian clocks. Comparably, we describe diet-responsive effectors by which dietary interventions influence the endocrinic, immunological, microbial and neural states responsible for improving health and preventing multiple diseases in humans. Furthermore, we expatiate diverse patterns of dietotheroapies, including different fasting, calorie-restricted diet, ketogenic diet, high-fibre diet, plants-based diet, protein restriction diet or diet with specific reduction in amino acids or microelements, potentially affecting the health and morbid states. Altogether, we emphasize the profound nutritional therapy, and highlight the crosstalk among explored mechanisms and critical factors to develop individualized therapeutic approaches and predictors.
Similar content being viewed by others
Introduction
What we eat and how this may influence our health has aroused interest for millennia. In ancient times, wild animals and foraged plants exclusively were acquired as foods, and diet fluctuated with the seasons.1 Currently, we can obtain food continually due to advanced technology in agriculture and animal husbandry. As a consequence, the impact of nutrition on humans has intrinsically diverged in human evolution. In many cases, diets are inundated with excessive amounts of calories, highly processed foods and a mass of salt, trans fat and sugar.2 Hence, dramatic changes in the quantity, quality, and frequency of foods are considered to result in human maladaptation.
Nutrition influences all physiological processes. Calorie restriction (CR) without malnutrition was put forward long time ago, and was demonstrated to prolong lifespan in rats in 1935 (Fig. 1). However, the mechanisms by which CR improves healthspan and longevity have remained elusive until a few decades ago. First, mutations in a single gene involved in nutrient-sensing signalling pathways are found to substantially lengthen the lifespans of nematode worms.3 Consequently, nutrition-mediated mechanisms increasing healthspan and lifespan are discovered.4 In the clinical interpretation of these findings, conscious variation of nutrition has been advocated as a promising method for enhancing healthspan and safeguarding against a multitude of diseases such as cancer, obesity and Alzheimer disease (AD). The concept that diet could affect the risks of developing certain diseases and impact therapeutic effects has prevailed.5 It is unfortunate that the effectiveness of most of these diets has not been rigorously evaluated, and viewpoints in this realm are not always based on solid mechanistic insight.
Research history of dietary regulation. The advances in dietary regulation can be roughly divided into three stages. Stage I, dietary regulation discovery and initial characterizations of their effects on body function, including dietary interventions based on traditional eating habits, such as fasting,367 caloric restriction,368 high-fibre diet,369 fermented/probiotic diet,370 Mediterranean diet371 and protein restriction,372 some new dietary interventions, such as ketogenic diet,373 glucose and carbohydrate restriction,374 amino acid intervention375 and micronutrient intervention.376,377 Stage II, key molecular mechanisms of dietary interventions were identified, including mTORC1,378 AMPK,379 FOXOs380 and Sirtuins and NAD.381 To date, whether and how metabolic interventions interfere with these signalling pathways to function has been extensively studied. Stage III, dietary interventions have been being explored for their impact on the overall metabolic network of the body to develop precision-nutrition therapeutics
The quantity, quality and composition of foods as well as meal timing directly impact our healthspan by regulating nutrient availability (Table 1). Despite varying dietary strategies, many sets of dietary guidelines have already been reported to be capable of extending lifetime in at least one model organism and improving health conditions in humans. Considering the dissimilarity across populations and individuals, variation in responses to diverse regimens is practical to anticipate. Precision nutrition, which means establishing personalized dietary plans to promote health, has lately been suggested as a novel strategy.6 Thus, many companies have attempted to implement precision nutrition to maximize lifespan while failing to maximize individual benefits by incorporating omics-based signatures.
In this review, we detail the nutritional molecular mechanisms that result in improved health. We also propose to decipher factors associated with sex, genetics and age that affect responses to dietary intervention. Altogether, we develop an approach to precision-nutrition medicine to elucidate the health-promoting effects of diet-based therapeutics.
Nutrient-associated molecular mechanisms
The mechanisms that are universally attributed to the effects of diet on disease and health are mainly classified into two types (Table 2; Fig. 2): (1) nutrient-mediated mechanisms, covering metabolic regulators, nutritive metabolism pathways, epigenetic mechanisms and circadian clocks; and (2) diet-responsive effectors, including the diet-endocrine axis, the diet-immune axis, the diet-gut axis, the diet-senescence axis and the diet-nerve axis.
Molecular and effector mechanisms of dietary intervention. Dietary intervention engages by alterable consumption of numerous nutrients, including carbohydrates, lipids, amino acids, micronutrients, and metabolites. Nutrient signals under diverse dietary interventions lead to activation of multiple biochemical pathways. As a result, these pathways involve in downstream effectors like cytokines secretion, immunoregulation, gut microbiome homoeostasis, elimination of senescent cells, and neuromodulation
Nutrient-mediated mechanisms
Metabolic regulators
mTORC1
One of the principal nutrient-related molecular mechanisms is the mammalian target of rapamycin (mTOR) kinase. mTORC1 and mTORC2, which are both composed of diverse protein subunits phosphorylating distinct substrates, constitute two separate complexes of mTOR kinases.7 In short, mTORC1 controls an extensive range of external stimuli, including the availability of oxygen, insulin/insulin-like growth factor 1 (IGF-1), glucose, amino acids and cholesterol, while mTORC2 acts predominantly as an effector of phosphoinositide 3-kinase (PI3K) signalling.8 mTORC1 integrates numerous extracellular and intracellular nutrient signals, which involve autophagy, ribosomal biogenesis and protein translation as well as biogenesis of proteins, lipids and nucleotides. A previous report discussed the regulation of mTORC1 in detail.7 Inhibition of mTOR activity via genetic modification and rapamycin treatment extends lifespan in species such as yeast, worms, Drosophila and mice.7,9 Subsequently, diet restriction has been found to impair mTOR activity and favourably impact many age-related disorders in humans. For example, calorie-restricted diets enhance longevity, improve cognitive function, reinforce cardiac function, ameliorate metabolic pathogenesis and reduce the incidence of cancer.10,11,12,13 Likewise, mTOR inhibitors such as rapamycin and perhexiline maleate have been found to mimic the health-promoting effect of CR.13,14 However, the wide-scale application of rapamycin has been impeded owing to a number of side effects.15 In mice, the ‘off-target’ restraint of mTORC2, which could be interrupted via chronic therapy with rapamycin in vivo, probably mediates these side effects.16,17 These findings have provoked great interest in distinguishing feasible dosing regimens for rapamycin and any other drugs specifically targeting mTORC1 that could maximize the beneficial effects and minimize the side effects simultaneously.
AMPK
AMP-activated protein kinase (AMPK), as a sensor of mitochondrial stress and nutrient status, can be activated under elevated AMP:ATP ratio conditions, embodying a detector of cellular energy status.18 AMPK switches on several catabolic pathways, such as glycolysis and fatty acid oxidation, to supplement ATP levels as a timely response to low levels of cellular energy.19 In addition, AMPK regulates many cellular signalling pathways, including mTORC1, by phosphorylating raptor and tuberous sclerosis complex 2 (TSC2).18 Both intermittent fasting (IF) and time-constrained feeding have been reported to activate AMPK to improve health and function in multiple species.20,21 In C. elegans, glucose restriction (GR) stimulates mitohormesis, which is defined as the upregulation of reactive oxygen species (ROS) levels along with the activation of the oxidative stress resistance response. This GR-induced effect depends on AMPK and is essential for its prolonged longevity function.22,23 In addition, AMPK activation contributes to longevity in C. elegans by sustaining mitochondrial network homoeostasis and functional coordination with peroxisomes to promote fatty acid oxidation.24 Importantly, metformin, an agonist of AMPK, has been found to decrease the level of blood glucose, retard tumour progression and reverse cognitive impairment.13,25 In summary, the data suggest that AMPK stimulators as dietary adjuvants may favour healthspan.
FOXO
Forkhead box O transcription factors (FOXOs) are downstream effectors of the insulin pathway involved in the regulation of stress responses, metabolic homoeostasis, cellular proliferation, and development.26 The FOXO family includes FOXO1, FOXO3, FOXO4 and FOXO6, which share a forkhead domain that is an exceedingly conserved DNA-binding domain (DBD) consisting of 100 amino acids.26 FOXO activity in response to diverse environmental stimuli is a pivotal regulator in cellular homoeostasis and is chiefly modified via phosphorylation, acetylation, ubiquitylation and methylation.26 Crucially, FOXO activity is mainly inhibited by PI3K-AKT signalling but is activated by various cell stresses.26 Furthermore, FOXO is regulated by AMPK, sirtuins, and mTOR status.26 For example, in mammalian cell cultures, AMPK activation also directly participates in FOXO3 phosphorylation and FOXO3 transcriptional regulation without affecting its nuclear localization.26 The role of FOXO is observed in nutritional intervention. CR on peptone dilutions and solid-media growth plates stimulates daf-16/FOXO activity to extend lifespan in C. elegans.27 In addition, the activation of dFOXO in a fat body of an adult Drosophila alters the response to dietary restriction but is not thought to extend lifespan.28 In mice, FOXO1 is increased in liver and skeletal muscles, while FOXO4 is elevated in skeletal muscles and adipose tissue, after CR.29,30 FOXO3+/− and FOXO3−/− mice do not live longer after experiencing CR.31 In addition, CCAAT/enhancer-binding protein β (C/EBPβ), which is an Aβ and inflammatory cytokine-activated transcription factor, could result in abnormal neural excitation and cognitive dysfunction by selectively triggering inhibitory GABAergic neuronal degeneration through blocking FOXOs. C. elegans neurons overexpressing CEBP-2 or LGMN-1 (asparagine endopeptidase) can shorten lifespan and diminish daf-16/FOXO-induced longevity.32 Consequently, FOXO has been discovered to support the beneficial effects of CR. Although it is commendably acknowledged that the regulation of CR-mediated lifespan extension critically requires FOXO, we still have no idea how FOXO regulates the gene expression programme that is significant for superior survival during CR. Moreover, identifying factors such as protein partners and posttranslational modifications, which play vital roles in regulating the pattern by which FOXO signalling integrates signals derived from miscellaneous external stimuli in vivo, is crucial for comprehending its function during CR. Further context-specific research concerning the function of FOXO is urgently needed.
Sirtuins and NAD
Sirtuins perform multiple catalytic functions, including roles as deacylases, demalonylase, desuccinylase, depalmitoylase, demyristoylase, and mono-ADP-ribosyltransferase.33 Levels of the cellular coenzyme nicotinamide adenine dinucleotide (NAD+) play a vital role in the deacylase function of sirtuins.34 A sirtuin consists of seven subunits (SIRT1-7) with distinct intracellular localizations. SIRT1, SIRT6 and SIRT7 are distributed in cell nuclei; SIRT2 is mostly in the cytoplasm but is transported into the nucleus; and SIRT3, SIRT4 and SIRT5 reside in mitochondria. They have important effects in regulating cellular metabolism, especially in glucose and lipid metabolism.33 In yeast, Sir2 (a homologue of mammalian SIRT1) is essential for CR-modulated lifespan.35,36 Similarly, Sir2 homologues also participate in the regulation of lifespan in nematodes and flies and significantly influence their responses to dietary limitation.37 An accumulating body of research has revealed that CR increases the expression of SIRT1 in several types of tissue in rats and human cell cultures,38,39 and humans with CR exhibit upregulated SIRT1 levels in skeletal muscle.40 Furthermore, SIRT3 and SIRT5 cause similar alterations in response to CR in mice.41,42 Mice overexpressing SIRT1 by genetic modification display a lean and metabolically active phenotype with decreased circulating levels of cholesterol and insulin and improved glucose tolerance.43 Nevertheless, mice systemically overexpressing SIRT1 fail to show prolonged lifespan.44 The overexpression of SIRT2 extends the lifetime of progeroid mice, which lack BubR1.45 Deletion of SIRT3 shortens lifespan,46 while elevated SIRT3 levels can enhance haematopoietic stem cell regenerative capacity.47 A large amount of evidence shows that obliteration of SIRT6 and SIRT7 induces infirmity and shortens lifespan.48,49 However, overexpression of SIRT6 in transgenic male mice can extend healthspan and lifespan;50 in addition, lifespan is lengthened in mice with Hutchinson–Gilford progeria syndrome that overexpress SIRT7.51 Mechanistically, the transcriptional activity of FOXO is directly regulated by SIRT1 via deacetylation, which could activate FOXO-dependent transcription of genes responsive to stress and prolong lifespan.52,53 In mice, the activity of the transcription factor coactivator PGC-1α, considered a key regulator of mitochondrial biogenesis and function, is also deacetylated and regulated by SIRT1.54 When AMPK phosphorylates PGC-1α at Thr177 and Ser538, PGC-1α is prone to deacetylation and activation by SIRT1.55 SIRT3 plays an essential role in enhancing the mitochondrial glutathione antioxidant defence system in cochlear cells as well as in the neocortex and liver to promote CR-mediated reduction of oxidative damage.56 This finding is consistent with a deficiency oxidative metabolism since misregulation of protein acetylation in SIRT3 knockout mice during CR.57 SIRT6 enhances DNA stability and represses senescence by facilitating DNA double-strand break repair through poly (ADP-ribose) polymerase 1 (PARP1) activation.58 The suppression of long interspersed element class 1 (LINE1) elements via SIRT6, which leads to DNA damage and inflammation, is probably another pivotal mechanism.59
Sirtuins are connected with diet as well as with metabolism, owing to their demand for NAD+. NAD+ functions as not only a master coenzyme in redox reactions but also a cofactor for non-redox NAD+-dependent enzymes, including sirtuins and PARPs.34 NPT1 (a NAD+ synthesis enzyme) and PNC1 (encoding a vital enzyme in the NAD+ salvage pathway) are essential for CR-mediated lifespan extension in both yeast and worms.36,60,61 NAD+ levels decrease with age and age-associated diseases.35 Specifically, senescent cells progressively accumulate in liver and visceral white adipose tissue (WAT) with ageing; moreover, they secrete inflammatory cytokines to induce tissue-resident macrophages to proliferate and express the NAD+-consuming enzyme CD38. These macrophages reinforce the activity of CD38-dependent NADase, thereby decreasing tissue NAD+ levels.62 Furthermore, adipose-specific overexpression of nicotinamide phosphoribosyltransferase (NAMPT, an NAD+ synthetase), which is the rate-limiting enzyme in a principal NAD+ synthesis pathway, was recently reported to elevate NAD+ levels in several types of tissue; ameliorate numerous measures of physical performance, metabolic health and cognition; and extend the lifespans of female mice.63 Hence, interventions to increase levels of NAD+ have prospective therapeutic capacity in ageing and age-related disease.35 With regard to this point, dietary intervention is extremely beneficial since it can boost NAD+ levels.64 Nutritional supplementation with NAD+ or NAD+ precursors is under active investigation as an approach to promote healthy ageing and intervene in diseases.35 For example, in women with prediabetes who are overweight or obese, nicotinamide mononucleotide, an NAD+ precursor, has been shown to ameliorate muscle insulin sensitivity, insulin signalling and remodelling.65 Similarly, nicotinamide riboside (NR), an NAD+ precursor, strikingly reshapes metabolic and stress-response pathways that decline with ageing by inhibiting the clock repressor PER2. NR supplementation in old mice can replenish NAD+ to youthful levels that enable improved health.66 In APP/PS1 mutant transgenic mice, dietary NR increases brain NAD+ levels, reduces the expression of proinflammatory cytokines, and improves cognitive and synaptic functions.67
In summary, accumulating studies to date indicate that the sirtuin family probably has a significant function during CR. Current investigations are aimed at defining the targets and enzymatic functions of sirtuins, as well as the character of each sirtuin in the regulation of healthspan, longevity and metabolism.
Nutritive metabolism pathways
Mineral metabolism
Numerous minerals are universally acknowledged to exert bioactivity through element chelation, giving rise to modulation of antioxidant capacity or microbiome metabolism among other physiological processes. Dietary minerals mainly include Na, K, Cl, P, Fe, Zn, Mg, Se, Cu, I and Ca. The dietary functions of some essential elements in health and disease deserve to be thoroughly clarified.
First, potassium, as the most abundant cation intracellularly, has a close mutual association with sodium. The potassium-sodium gradient between intracellular and extracellular compartments prompts a series of cellular processes that maintain homoeostasis of other metabolites. The potassium balance in mitochondria is a dominant mechanism that regulates mitochondrial redox capacity and charge. Many studies have reported that changes in mitochondrial potassium currents may play substantial roles in the development of neurodegenerative and cardiovascular diseases.68
Calcium is essential for living organisms along with normal body function. Calcium is linked with an array of biological events, including immune responses, cell death, cell differentiation, transmission of nerve impulses, muscle contraction, and enzyme activation. Calcium status disorders are conducive to the pathogenesis of bone diseases as well as increased risks of metabolic diseases and epithelial cancer.69 Calcium can only be provided with food to satisfy its requirement. Therefore, it is highly necessary to obtain the proper quantity of calcium from the diet for normal body function. Since a low cytosolic calcium level is crucial for cellular function, calcium oscillations are able to act as secondary messengers for diverse stimuli that range from proliferation to apoptosis. Calcium has an extensive array of roles within the mitochondria. Calcium is critically essential for the activities of a few enzymes within the electron-transport chain and tricarboxylic acid (TCA) cycle, including the rate-limiting isocitrate dehydrogenase. It also promotes the conveyance of adenylate and synthesis of ATP within mitochondria, with a parallel elevation in mitochondrial membrane potential.68
In addition, magnesium is not only the most abundant divalent intracellular cation in humans but also the second most concentrated intracellular ion after potassium. It has been traditionally considered that magnesium is the cofactor of approximately 300 regulatory enzymes; however, current databases have enumerated over 600 enzymes for which magnesium acts as a cofactor. Magnesium is related to several primary cellular processes, including ATP-dependent biochemical processes, where magnesium is a component of the activated Mg-ATP complex, glucose metabolism, DNA synthesis, RNA expression, blood pressure control, and neural and muscular cell signalling. Magnesium deficiency has also been reported to be linked with oxidative stress, low-grade inflammation, insulin resistance, and metabolic syndrome.70
Next, Fe, as an indispensable mineral for sustaining homoeostasis in humans, is critically essential for quite a few cellular reactions, encompassing DNA synthesis, cell division and growth, immune responses, protein metabolism, oxygen transportation through haemoglobin, production of various neurotransmitters, thyroid hormone regulation, oxidation–reduction reactions and erythropoietic functioning within connective tissue. In addition, Fe is also a vital element for numerous enzymes related to metabolic reactions, such as peroxidase and catalase, together with cytochrome. Although one of the main functions of Fe is boosting oxygen diffusion into mitochondria, it might also be adverse owing to its oxidative role in somatic cells. In addition, Fe is absorbed or stored in the form of ferritin in oxidative enzymes. This component is largely found in myoglobin and haemoglobin, and the systemic level is controlled by the balance of its intake, utilization and storage.71
Cu, as a trace element pivotal for enzyme function, has a valuable dual function as both a pro-oxidant and an antioxidant. It functions as a catalytic cofactor for an array of enzymes, including Cu/Zn superoxide dismutase (SOD), lysyl oxidase, and ceruloplasmin (CPO), which plays a significant role in the integrity and strength of the heart and blood vessels. Cu is also a crucial requisite for Fe absorption and mitochondrial respiration. Excess copper directly binding to lipoylated constituents in the TCA cycle could further lead to aggregation of lipoylated protein as well as subsequent iron-sulfur cluster protein loss, which would force proteotoxic stress and eventually cell death.72 Increased Cu levels are able to enhance ROS production and consequent oxidative stress, causing the oxidation of DNA, proteins, lipids, homocysteine and other molecules. Cu deficiency, on the other hand, can induce peroxidative damage. Moreover, not only Cu deficiency but also Cu overload has crucial effects in atherogenesis.71
Zn is the second most common transition metal in humans after Fe. Considering its prevalence in the structure of diverse proteins and enzymes, Zn plays a predominant role in normal cell structure and catalytic function, particularly in the central nervous and immune systems. It is also pivotal to cell growth and division as well as repair, haemostasis, energy-producing functions, wound healing, carbohydrate catabolism, thrombosis, encompassing fibrinolysis, NO synthesis, coagulation and anticoagulation. In addition, intracellular Zn plays a vital role in redox signalling pathways and contributes to antiapoptotic, antioxidant and anti-inflammatory activities. However, Zn deficiency can result in the oxidation and degradation of essential proteins such as protein kinase C (PKC), the production of C-reactive protein (CRP) and inflammatory cytokines, and ingestion of particles by monocytes and macrophages. Moreover, Zn deficiency can affect the development of diverse organs, including the brain, heart, lung, kidney and skeleton.71
Ultimately, selenium (Se), as a micronutrient indispensable for the human body, performs its roles as a component of the amino acid selenocysteine (Sec) which is considered the 21st amino acid in selenoproteins. Se exhibits various significant activities, including antioxidant, immunomodulatory, thyroid metabolism and human fertility effects. Inserted into mammalian selenoproteins, Se is different from other minerals interacting with proteins as cofactors. Throughout the mammalian body, selenoprotein genes have already been validated; however, functions have only been described for half of them. Via a mechanism involving the recoding of the stop codon UGA during translation, Sec is generally located at the active site of an enzyme. The 3′ UTR of the Sec incorporation sequence (SECIS) region is where Sec is incorporated into a protein. The majority of selenoproteins are associated with multiple biological reactions concerning the regulation of the redox state and antioxidant function. One of the well-characterized functions of selenoproteins is redox activity, which is mostly due to three isoforms of thioredoxin reductases and deiodinases as well as five members of the glutathione peroxidase (GPX) family. Se is also crucial for modulating not only inflammatory responses, since it can attenuate the activation of the nuclear factor (NF)-κB pathway, but also thyroid function, on account of the action of deiodinases in the conversion of T4 into its active form T3. Furthermore, Se is critically vital for neurological function, since it can protect the brain from oxidative damage, along with male fertility and reproduction.73
In summary, conflicting evidence remains regarding mineral interventions for improving health. Clinical studies of mineral patterns, quantity, and bioactivity in diet must be performed to fully understand the role of mineral metabolism in health and disease.
Lipid metabolism
Dietary lipids play a conspicuous role in mental and behavioural health. Dietary fatty acids, as structural building barriers of diverse membranes, are connected with pro- and anti-inflammation mediators, making them infinitely important for human health, growth, development and preservation. The quantity and quality of dietary fat has undergone a vast alteration over the past 10,000 years. A switch from a diet abundant in omega-3 polyunsaturated fatty acids (ω−3 PUFAs) towards a Western diet almost deficient in ω−3 PUFAs but increasing levels of saturated fatty acids (SFAs), trans fatty acids (TFAs) and ω−6 PUFAs results from this alteration accompanying the industrial revolution.74 Accordingly, balanced quantities and proportions of dietary lipids may be critical to maintaining health. First, accumulating research on nutrient restriction in mice and flies by tracers reveals an upregulation in both the synthesis and catabolism of fat, showing a shift towards superior lipid utilization.75,76 Inhibition of fatty acid synthesis by blocking acetyl-CoA carboxylase (ACC) or mitochondrial β-oxidation leads to a failure of dietary restriction-mediated lifespan extension.75 Studies in SIRT1+/− cell culture and mice suggest that SIRT1 contributes to fat mobilization in white adipocytes by blocking the transcriptional effects of the fat regulator peroxisome proliferator-activated receptor-γ (PPARγ).77 AMPK is also reported to regulate lipid metabolism by phosphorylating ACC.78 In worms, it is critically essential for dietary restriction-mediated longevity and amelioration of proteotoxic effects in polyQ Huntington models to suppress genes controlling fatty acid synthesis, oxidation, and desaturation, such as the nuclear hormone receptors NHR-49/PPARα and NHR-80/HNF4.79 Importantly, alternate-day fasting (ADF) is reported to reduce fat mass and cholesterol levels but increase the levels of diverse fatty acids and their catabolite β-hydroxybutyrate in a clinical trial.80 Furthermore, a ketogenic diet (KD) has been found to be safe and has the potential to extend healthspan or improve health. The KD boosts fatty acid β-oxidation in the liver to generate ketone bodies, including acetoacetate, acetone, and β-hydroxybutyrate. Ketones are transferred into the bloodstream to various tissues, where they are transformed to acetyl-CoA to fuel the TCA cycle.81 In terms of types of fatty acids, the elevated abundance of the mono- and poly-unsaturated fatty acids in response to CR is a key component of cell membrane structures and has been revealed to upregulate pro-survival mechanisms such as cellular detoxification.82 Taken together, these results indicate that reinforced rates of both fatty acid synthesis and breakdown are vital controllers of CR-mediated longevity. In addition, special attention should be given when assessing the influence of CR on fatty acid metabolism.
Proteostasis
Many proteins and amino acids play a crucial role in sustaining health.83 Some specific proteins are detrimental to healthspan, since inhibiting these proteins has been found to extend lifespan in C. elegans.83 Protein interventions hold the capacity to prevent age-associated insoluble proteins from intracellular accumulation, but an increasing number of studies have shown that CR enables the balance of protein synthesis and degradation. First, CR is reported to stimulate endoplasmic reticulum stress and expedite proteostasis, which contributes to prolonging longevity in C. elegans.84 Proteostasis inevitably suppresses unfolded protein synthesis and opportunistically degrades these toxic proteins, which requires responses to unfolded proteins, including autophagy and ER stress responses.85,86 Key autophagic genes have been found to be indispensable for CR-mediated benefits in healthspan improvement from yeast to humans.87,88 For example, intermittent time-restricted feeding (iTRF) mediates lifespan extension in Drosophila, which depends on circadian regulation and autophagy. Autophagic activation at night has been found to be sufficient and essential for the benefit of CR for longevity.87 Similarly, mammalian MON2 (a Golgi protein) is upregulated in long-lived C. elegans, and MON2 is essential for stimulating autophagic flux by activating the Atg8 orthologue GABARAP/LGG-1 in C. elegans to extend longevity.89 Starvation, lipid restriction, proteins or special amino acid deletions activate autophagy.90,91,92 Overactive autophagy in response to fasting is partially regulated by activation of AMPK and SIRT1 activity and inhibition of mTOR activity.13 The autophagic response is also implemented to mobilize stored lipids via lipophagy to coordinate the metabolic response to food availability and CR.91 Moreover, total protein restriction or lack of specific amino acids, such as branched-chain amino acids (BCAAs), methionine, cystine and glutamine, are thought to stimulate the autophagic/lysosomal response, partially depending on mTOR suppression and general control nonderepressible 2 (GCN2) activation.93,94,95,96 For example, fasting drives lysosomal export of cystine in the fat bodies of Drosophila; subsequently, cystine is transformed to cysteine and metabolized to acetyl-CoA by promoting CoA metabolism. This process limits TORC1 reactivation to maintain autophagy.92 As an evolutionarily conserved serine/threonine kinase, GCN2 is able to sense changes in amino acid levels to modulate diverse nutrient-response pathways. For example, GCN2 coordinates inflammation and integrated stress responses to control malignant growth and immune homoeostasis. Ribosome stalling and elevated levels of uncharged tRNAs can activate GCN2, and activated GCN2 induces the phosphorylation of eukaryotic translation initiation factor 2 (eIF2), subsequently selectively stimulating ATF4-mediated translation but blocking translation of most mRNAs.97 Fibroblast growth factor 21 (FGF21) secretion in response to acute protein restriction partially depends on the GCN2-ATF4 pathways, whereas chronic protein restriction is capable of directly stimulating ATF4-induced FGF21 release in a GCN2-independent manner.98 The sophisticated effects of diet interventions on proteostasis deserve to be elucidated in detail in the context of different strategies and in different tissues.
Mitochondrial function
The centre of many metabolic processes is mitochondrial function. Mitochondria are metabolic centres that constructively respond to diet through mitochondrial quality control and alteration of mitochondrial function.99,100 Likewise, mitochondrial dysfunction underlies various age-associated diseases.99 However, the effects of CR on the improvement of mitochondrial homoeostasis and mitochondrial function are inconsistent. One potential mechanism is that CR reduces mitochondria-generated ROS and ROS-induced damage.101 Compared to hepatocellular mitochondria from rats fed a normal diet, those isolated from rats fed a 40% CR diet were found to exhibit reductions in membrane potential, oxygen consumption and ROS production while maintaining ATP generation, which demonstrated that CR was able to augment mitochondrial efficiency.102 In the skeletal muscle of humans, short-term CR has no effect on the function of mitochondrial enzymes but facilitates mitochondrial biogenesis to decrease oxidative stress, suggesting that CR is capable of inducing the formation of highly efficient mitochondria.40 However, chronic CR fails to boost mitochondrial richness but optimizes oxidative damage to DNA and protein through augmentation of antioxidant scavengers and reduction of oxidant emission.103 Similarly, deficiency in mitochondrial fitness, proteotoxicity and mitonuclear protein imbalance can trigger mitochondrial stress, which results in mitophagy and the mitochondrial unfolded protein response (mtUPR) to maintain mitochondrial homoeostasis.104,105 In worms and long-lived Snell dwarf mice, the mtUPR can sustain mitochondrial protein stoichiometry and alter electron-transport chain components to prolong lifespan.106,107 Incremental studies have also indicated that CR prevents the impairment of mitochondrial DNA (mtDNA).108 In addition, fasting or CR induces mitophagy, suggesting enhanced turnover of damaged mitochondria.109 Interestingly, the TCA cycle, a main metabolic pathway in the mitochondria, is a centre of cellular energy and metabolism through the compact connection of glucose, amino acids and lipid oxygenolysis. α-Ketoglutarate (αKG) is an intermediate in the TCA cycle, and αKG supplementation has the potential to inhibit the ageing process and prolong healthspan (reviewed in detail in ref. 110) Taken together, it is evident that mitochondrial functionality is indispensable for CR-modulated health. However, this evidence is necessary to decipher how dietary interventions affect mitochondrial homoeostasis across species and within various tissues under the circumstances of disease and health.
Epigenetic mechanisms
Epigenetics is defined as regulation of gene expression through frequent alteration of the expression of genetic material and DNA configuration in the absence of sequence mutations. Epigenetic alterations are moderate and stepwise but could possibly be rejuvenated. Epigenetics deals with three main types of modulations: (1) DNA methylation, (2) histone modification and (3) ncRNA-mediated gene expression. Indeed, nutritional interventions impact the methylation of DNA, histone modification and ncRNA expression.111,112
First, DNA methylation constitutes covalent modifications in CPG dinucleotides, which show a pair of cytosines at the 5-carbon position of CG dinucleotides. DNA methylation enzymes include DNA methyltransferase 1 (DNMT1), DNMT2, DNMT3a and DNMT3b. Folate metabolism preserves balanced quantities of deoxyribonucleic acid and is thus crucial to DNA replication. As a cofactor for enzymes, folate is essential for synthesizing nucleotides and thymidylate.113 Similarly, one-carbon units are mainly derived from serine during the homocysteine-methionine conversion process; furthermore, the conversion of methionine to S-adenosyl methionine (SAM) requires the consumption of abundant ATP that can be produced within the de novo synthesis of serine. Liver kinase B1 (LKB1) loss augments DNA methylation through activation of de novo serine biosynthesis.114 Serine hydroxymethyltransferase 2 (SHMT2), a key enzyme of serine biosynthesis in mitochondria, has been observed to promote histone and DNA methylation by increasing SAM synthesis.115 In addition, an enhancement of αKG levels has been found to lead to the differentiation of tumour cells in a 5-hydroxymethylcytosine (5hmC)-dependent manner.116 Methyl donor SAM replenishment is derived from dietetic methionine, serine, vitamins B2, B6 and B12, and folate. A lack of these can weaken DNA methylation, potentially extending lifespan and improving health.115,117,118
Histone proteins include eight proteins containing histones H2A, H2B, H3 and H4 in complex octameric structures. As a histone linker, histone H1 connects DNA couplers with nucleosomes. Histone modifications, including methylation, acetylation, phosphorylation, succinylation, hydroxybutyrylation and lactylation, may undergo changes during metabolic events.119 For example, CR restricts SAM availability to suppress histone H3K4 methyltransferase SET-2 activity, which leads to activate TFEB/FOXA-mediated autophagy to prolong lifespan.120 Meanwhile, high levels of αKG are thought to sustain trimethyl H3K27 and H3K9, and αKG supplementation ameliorates age-related osteoporosis.121 Moreover, the histone acetyltransferase p300 enhances the expression of senescence genes. Mechanistically, p300 contributes to the formation of activatory enhancer elements within noncoding regions and dynamic hyperacetylation of chromatin.122 Conversely, CR-activated sirtuins and p300 inhibitors reduce the overall level of histone acetylation to improve health.123,124,125 Finally, succinyl-CoA derived from the TCA cycle is the direct donor for succinylation. Lysine acetyl transferase 2A (KAT2A) was first identified as a succinyltransferase and catalyses the succinylation of histone H3 at lysine 79 by binding to succinyl-CoA, which profoundly promotes gene expression.126 KAT2A performs protein succinylation, while SIRT7 is endowed with the capacity to act as a histone desuccinylase when DNA is damaged.127
Noncoding RNAs (ncRNAs) include long noncoding RNAs (lncRNAs), microRNAs (miRNAs), circular RNAs (circRNAs) and packaging RNAs (pRNAs), and account for a large proportion of the human genome. The majority of ncRNAs fail to encode proteins but exert pivotal effects on translation, mRNA stability and transcription.128 As well-established ncRNAs, miRNAs are thought to mediate nutrient-sensing pathways.129 For example, some miRNAs have been discovered to regulate metabolic signalling, such as mTOR, AMPK, sirtuins and insulin/IGF-1, by targeting their mRNAs.130,131 Likewise, some gene signatures based on diet-mediated miRNA expression can predict health improvement.132,133 In addition, miRNAs are capable of being released into body fluids and plasma either in a vesicle-free form or packaged in extracellular vesicles, highlighting that these marked miRNAs could be utilized as non-invasive diagnostic tools and predictors of health and diet-associated diseases.134,135 Ultimately, dietary miRNAs originating from plants and milk biologically affect the health of the host by directly targeting host genes and indirectly affecting the function of the microbiome, leading to the development of novel methods to treat or prevent diseases.136
Polyphenols are abundant in vegetables and fruits, and possess polyhydroxy phenols as a fundamental structure. They comprise ten subgroups, including acetophenones, lignins, xanthones, benzoquinones, flavonoids and phenolic compounds, based on their chemical structures and properties.13 Dietary polyphenols are implemented to prevent certain diseases through diverse mechanisms, such as silencing genes and epigenetic modifications.13,137,138,139,140,141 For example, polyphenols function to induce histone modifications such as deacetylation and methylation and inhibit DNA methyltransferase for health improvement. Polyphenols exhibit properties of fundamental alteration of neoplastic or systemic epigenomes and are of great interest for disease prevention and health improvement.
Circadian clock modulators
As an inherent punctual system, the circadian clock has the capacity to sustain an all-day rhythmic cycle of physiology, behaviour and metabolism.142 Clock genes have rhythmic activity to regulate the transcription of numerous genes in a cyclic way, which in turn synchronizes diverse physiological processes to external stimuli, such as photostimulation. Under this condition, dysfunction of circadian rhythms plays an important role in health.143 For example, Reg3γ, a C-type lectin antimicrobial peptide in host, connects intestinal circadian clock to ileal microbes. Fat-enriched diet results in a continuous expression of Reg3γ to drive microbial oscillators.144 Apart from light, clock genes can also respond to cues from diet. In mouse and fly models, CR, especially at night, has been found to enhance the amplitude of clock gene mRNA expression to promote longevity.145,146 Without the primary clock genes period (Per) and timeless (Tim), flies or mice fail to extend their healthspan even upon TRF.145,146 In clinic, it is widely accepted that TRF ameliorates metabolic health. Compared with mid-day TRF (mTRF, food intake restricted to the middle of the day), early TRF (eTRF, food intake restricted to the early part of the day) is reported to be more conducive to enhancing insulin sensitivity, increasing gut microbial diversity, ameliorating inflammation, and reducing body weight, adiposity and fasting glucose.147 Mechanistically, circadian rhythms have been found to mediate brain and muscle Arnt-like protein 1 (BMAL1) in the brain and muscle to drive daily fluctuations in NAD+ levels and shape the light cycle.148 Furthermore, SIRT1 and AMPK interact with these circadian factors and modulate their activity.142,148,149 In addition, both autophagy and circadian regulation are necessary when iTRF extends fly longevity and decreases the profile of age-associated biomarkers in the gut and muscle. On an AL diet, autophagic induction only at night is sufficient to prolong lifespan.87 Conversely, night-time eating results in misalignment between central and peripheral (glucose) endogenous circadian rhythms and impairs glucose tolerance to increase diabetes risk.150 Moreover, deficiency of the specific circadian clock regulators Tim and Per has been reported to lead to changes in cellular respiration through an increase in the uncoupling protein UCP4C in the gut and eventually contributes to prolonging the lifespan of male Drosophila.151 How nutrient signals affect circadian clock genes is worth investigating. To identify the metabolic functions of the circadian clock, the integration of multi-omics, including metabolomic and sequential circadian transcriptomic analyses, in many human tissue types is indispensable. Seeking the circadian rhythm-modulated pathways and the potential mechanisms by which they affect health is entirely relevant for individualized translational applications.
Diet-responsive effectors
The diet-endocrine axis
Nutritional complementarity is primary to an organism’s growth and development via regulation of multiple cytokines (leptin, adiponectin, asprosin, acyl-CoA-binding protein and so on) and growth factors and hormones (insulin/IGF-1, FGF21, thyroid-stimulating hormone, prolactin, corticosterone, etc.).80,152,153,154,155,156 Insulin and IGF-1 were first discovered to be connected to diet and health. The orthologue of the mammalian insulin/IGF-1 receptor in C. elegans is encoded by the daf-2 gene, and its mutation has been found to dramatically prolong longevity.3 As in C. elegans, relative mutations in genes in the insulin/IGF-1 pathway, such as chico (the insulin receptor substrate-like signalling protein gene),157 and mutations in the insulin-like receptor gene (InR),158 are observed to prolong lifespan in flies. In mice, knockout of insulin receptor substrate 1 (IRS1) or IRS2 heterozygosity throughout the body and selective deletion of the insulin receptor in fat tissue are capable of extending lifespan.93 Similarly, CR or caloric restriction mimetics (CRMs) have been reported to decrease the circulating levels of insulin and IGF-1, which has been regarded as a pivotal longevity mechanism of CR.13,90,159 Mediators of CR, such as mTOR, FOXO and SIRTs, are involved in insulin/IGF-1 signalling.160 Generally speaking, it is likely that the favourable effects of CR partially depend on the inhibition of the insulin/IGF-1 pathway, and these dietary strategies or drugs for suppressing this signalling may have the potential to improve healthspan in translational research. Nevertheless, IGF-1 is also secreted by Lepr+ mesenchymal cells surrounding intestinal crypts, and stimulates local intestinal stem cells (ISCs) functionality and proliferation via binding to epithelial IGF1R.161 Moreover, FGF21 is a stress-inducible hormone involved in multiple pivotal metabolic signalling pathways.162 Both total protein restriction and particular amino acid restriction stimulate FGF21 secretion in the liver and adipose tissue to augment energy expenditure and improve insulin sensitivity.163,164 In responses to protein restriction, FGF21 plays an important role in diverse species, including humans. Long-term protein restriction stimulates the release of FGF21 in hepatocytes and elevates circulatory levels in male C57BL/6J mice and Sprague Dawley rats.163,165,166 In the clinic, protein restriction (4–6 weeks) can elevate circulating FGF21 levels.163,165 Time-restricted fasting also potently promotes FGF21 release, and transgenic FGF21 expression and recombinant FGF21 treatment have the capacity to improve insulin sensitivity and glucose tolerance in mice.162 Interestingly, transgenic overproduction of FGF21 has no effect on food intake or the mTORC1 pathway but independently prolongs healthspan in mice.167 Altogether, continuing studies are required to further decipher the specific mechanism by which CR and protein restriction stimulate FGF21 secretion, and these dietary treatments and FGF21 mimics are implemented to sustain health and treat metabolic diseases.
The diet-immune axis
Diet has been discovered to impact the immune system (has been reviewed in ref. 168) The immune system uniquely contains various cell subpopulations and multiple layers with particular and complementary effectors. In response to external stimuli, including cancer and infection, the interdependent immune network endows the host with the capacity to protect against these various stimuli.169,170 Confronted with these challenges, every organ exhibits tissue-specific immune responses, which are mainly reflected in resident immune cells that boost tissue repair and rapid local protection. These immune cells obviously adapt to the regional environment and show metabolic characteristics analogous to those of resident tissue.171,172 In parallel, several immune cell subsets and multitudinous immunological factors dynamically exist in circulation and migrate throughout the body to conduct immunosurveillance. When at rest, these immune cells commonly need few nutrients.173 However, immune cells targeting dangerous stimuli definitely require heavy consumption of nutrients and energy for functional activation and enormous expansion.174 Therefore, the immune system exhibits extreme adaptation and high resilience to thrive under fluctuating nutrient conditions.
The quantity and composition of the diet directly affects the immunological response by modulating nutrient availability. Fluctuations in nutrients and/or reduced dietary intake hold the potential to favour optimal immune responses. Recently, some studies have demonstrated that CR or IF can moderate the harmful off-target effects of allergies and autoimmunity and protect against cancer and pathogens through optimization of immune responses.175,176,177,178,179 In C. elegans, CR or reduced insulin/IGF-1 signalling has been found to downregulate p38-ATF7 signalling to a basal level in the innate immunity pathway, which contributes to the extension of lifespan.178 In mice and humans, CR ameliorates ageing-associated alterations in transcriptional regulatory networks, expression of key genes and the composition of cell types. The proinflammatory type of immune cells has been conjectured to contribute to ageing, while CR beneficially disrupts age-induced eccentric interactions within immune cells, similar to the excessiveness of proinflammatory ligand–receptor combinations.179 In healthy humans, a 2-year CR inhibits platelet-activating factor acetyl hydrolase (PAF-AH) in the adipose tissue surrounding the thymus, inactivates the NLRP3 inflammasome, prevents age-associated inflammation, and reduces thymic lipoatrophy to improve metabolic health.100 In terms of dietary constituents, ions are crucial in immune system homoeostasis. An excessive sodium chloride diet not only motivates maturation of DCs and activates their antigen-presenting capacity but also stimulates the generation of proinflammatory factors and autoantibodies, which leads to lymphadenectasis and splenomegaly, and exacerbates renal diseases.180 Likewise, the potassium channel K2P18.1 mediates the development and function of Tregs by facilitating FOXP3 expression. Mechanistically, K2P18.1 forcibly drives continuous Ca2+ influx to increase FOXP3 transcription in an NFAT- and NF-κB-dependent manner. The K2P18.1 activator nitroxoline is detrimental for urinary tract infections via a rapid and reversible increases in Tregs.181 Moreover, Se supplementation has been found to enhance GPX4 expression in T cells, increase the numbers of follicular helper T cells and promote antibody responses in mice and humans after influenza vaccination.182 Similarly, zinc as a dietary supplement stimulates the secretion of bone morphogenetic protein 4 (BMP4) in thymic endothelial cells by binding to its receptor GPR39 to promote T-cell development.183 In lipid-associated metabolism, ketogenesis-derived β-hydroxybutyrate enables epigenetic modification of Lys 9 of histone H3 (H3K9) and hydroxybutyrylates Ppargc1a (which encodes PGC-1α) and Foxo1, upregulating Pck1 expression to enhance the pentose phosphate and glycogen pathways in CD8+ memory T cells.184 In addition, an ω−3 PUFA-rich diet has been found to decrease the quantity of effector memory CD4+ T cells in fat tissues and alter lipid profiles in plasma, adipose tissues and lymphoid organs. Docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) treatments interfere with cytoskeletal rearrangements to decrease the motility of CD4+ T cells and recede their polarity.185 Other small-molecule metabolites in the diet also influence the differentiation and function of immune cells. Supplementation with spermidine, an endogenous polyamine metabolite, reinstates memory B-cell responses. Specifically, spermidine stimulates the hypusination of eukaryotic initiation factor 5A (eIF5A), further activating TFEB-dependent autophagy.186
Nutrients mediate the function and composition of the microbiota to indirectly coordinate host immune responses. First, fat-enriched diet is reported to perturb the diversity of gut microbiome and repress the expression of major histocompatibility complex class II (MHC class II) in ISCs, which impairs immunosurveillance for tumorigenesis.187 Similar to high-fat diet, high salt intake is found to particularly deplete Lactobacillus murinus in mice, enrich Th17 cells and increase blood pressure.188 However, a high-salt diet augments gut permeability and increases the gut and intratumor richness of Bifidobacterium, which results in tumour regression by activating the antitumor function of NK cells.189 In addition, dietary fibres can be decomposed into short-chain fatty acids (SCFAs). These SCFAs optimize the proliferation of innate lymphoid cells (ILCs) in the intestines. For example, SCFAs stimulate expansion of ILC2 cells by binding to their receptors, such as free fatty acid receptor 2 (FFAR2) and FFAR3. However, SCFAs have the capacity to inhibit the proliferation of ILC2s in an FFAR-independent manner.190 Taken together, alimentative treatments, including CR and compositional alteration, optimize and educate the immune response in an evolutionarily conserved manner.
The diet-gut axis
Recently, diet-modulated gut microbiomes and their metabolites have exerted prominent roles in shaping health.191 Considering that diet-induced changes significantly affect the gut microbiome, CR has the potential to exert an immense effect.192 CR markedly alters the composition and function of gut microbiota to stimulate beige fat and improve metabolic state. Mechanistically, CR has been found to reduce lipid A biosynthesis through diminution of bacterial enzymes, further recruit alternative activated macrophages and eosinophils in adipose tissue to ameliorate diet-induced fatty liver and promote the development of beige fat.193 Meanwhile, CR enables to reprogramme the microbial bile acid metabolism to stimulate brown adipose.194 However, a very-low-calorie diet severely impairs bacterial abundance and restructures the gut microbiome. Specifically, this dietary strategy has been found to result in an enrichment in Clostridioides difficile that was related to weight loss and metabolic improvement in a toxin-dependent manner.192 In addition, Lactobacillus gasseri located in upper small intestine is found to upregulate the expression of long-chain acyl-CoA synthetase 3 (ACSL3), which serves as a pre-absorptive mediator to regulate systemic glucose homoeostasis.195 Clinically, fasting influences the generation of SCFAs by altering bacterial taxa and related genes in the gut. The profiles of immune cells, such as Treg, Th17 cells and CD8+ effector T cells, and microbial taxa, including Akkermansia, Ruminococcaceae, Desulfovibrionaceae and Hydrogenoanaerobacterium, have been speculated to control systolic blood pressure in response to fasting.196 Homogeneously, CR re-establishes a favourable gut microbiome, contributed to higher overall alpha diversity and augments the proportions of naïve B and T cells to delay immunosenescence.197 Recent evidence in clinical indicates that the low-energy diet (LED) significantly increases the richness of Akkermansia and Christensenellaceae R-7 associated with metabolic improvements in overweight and prediabetic individuals.198 Inconsistently, other clinical results have indicated that the gut microbiome remains highly individual-specific but stable in response to CR.11 In addition, dietary contents impact microbial community stability.199 For example, ketone bodies reduces the levels of proinflammatory Th17 cells in the gut by electively suppressing bifidobacterial growth.200 In contrast, a high-fibre diet has no effect on microbial community diversity but increased microbiome-encoded glycan-degrading carbohydrate active enzymes (CAZymes), whereas a diet enriched in fermented food steadily decreases inflammatory markers and increases microbiota diversity.201 Dietary fibres are universally considered to be beneficial for our health via the gut microbiome.202,203,204 For example, a recent study with multiomic signatures shows that arabinoxylan consumption is capable to reduce cholesterol level mainly depending on a reduction in low-density lipoprotein (LDL) and an increase in bile acids. By contrast, long-chain inulin results in liver inflammation and upregulation of alanine aminotransferase.203 One of potential mechanisms is that the different structures of dietary fibres contribute to the variants of gut microbiome and their metabolic functionality.204 However, dietary fibre deficiency has been found to expedite the utilization of glycoproteins by mucus-eroding microbiota, such as Citrobacter rodentium, eventually leading to intestinal barrier dysfunction and lethal colitis.205 Dietary thymidine and serine alter microbial composition via different mechanisms. The microbial conversion of 5-fluoro 2'deoxyuridine (FUdR) into toxic 5-fluorouridine-5'-monophosphate (FUMP) in C. elegans can be accelerated by thymidine supplementation. In contrast, serine alters one-carbon metabolism to exacerbate DNA toxicity in E. coli.206 Ultimately, the gut microbiota plays a crucial role in the interaction between host circadian rhythms and dietary timing (which has been reviewed in refs. 207,208) For instance, dietary ingredients and rhythmicity modulate the small intestine microbiome. Changes in feeding contents or time have been found to disrupt the circadian clock, resulting in barrier disruption with extensive import of microbial products to drive Crohn-like enteritis.209
The diet-senescence axis
Cellular senescence has been identified as a permanent arrest of cell proliferation in response to several stressful stimuli, such as DNA damage, oxidative stress, telomere dysfunction, oncogene activation and metabolic dysfunction.210 Accumulation of senescent cells in tissues contributes to organismal ageing and age-associated diseases and impairs tissue regeneration via diverse secretory factors and metabolites.211 Various nutritive therapies have been found to eliminate these senescent cells. CR holds the capacity to decrease the markers of senescent cells and relieve age-related inflammation in both mouse and human tissues.212 These mechanisms reinforce DNA repair, inactivation of oncogenes, reduced ROS production and clearance of damaged mitochondria by mitophagy (which has been reviewed previously210) Other dietary interventions, such as KD213 and methionine or serine restriction,115,214 can similarly eliminate senescent cells. β-hydroxybutyrate generated by KD decreased the level of senescence biomarkers in endothelial cells and vascular smooth muscle.215 In comparison, methionine restriction reduced senescent biomarkers by enhancing methylation and promoting one-carbon metabolism.216 Studies have demonstrated that appropriate nutritional treatments can deplete senescent cells to shape a supporting metabolic state.
The diet-nerve axis
The contribution of the nervous system to food intake and preferences is pivotal. Several landmark reports have shown that the central nervous system (CNS) maintains systemic metabolic homoeostasis in response to nutrition. For example, Capa, a homologue of mammalian neuromedin U, is released from a complex of six neurosecretory cells in the CNS of Drosophila in response to circulating levels of nutrients. Capa is capable of controlling energy homoeostasis via combination with its receptor (CapaR) in peripheral tissues to sustain energy homoeostasis.217 Similar to the CNS, the peripheral nervous system (PNS) also possesses nutritional sensors. For instance, neuropod cells in the intestine are indispensable for the preference for nutritive sugars over non-caloric sweeteners via glutamatergic signalling transduction.218 Enteroendocrine cells, as sensors of alimental amino acids in the gut, secrete diuretic hormone 31 (DH31). DH31 binds to the DH31 receptor (DH31R) in brain neurons and excites courtship behaviour.219 Importantly, nutritional intervention profoundly impacts the functionality and homoeostasis of the nervous system. Age-associated cognitive decline is reported to be delayed by CR in complex mammals, including humans.220 One potential possibility is that CR can promote neuron survival and enhance neurogenesis.221 Less cell proliferation and fewer progenitor cells and neuroblasts observed in sites of neurogenesis in the subgranular layer (SGL) and dentate gyrus subventricular zone (SVZ) are viewed as classical structural alterations induced by ageing.222 Interestingly, alternate-day CR increases cell survival and induces neurogenesis in the dentate gyrus in 8-week-old mice.223 Moreover, CR is discovered to elevate the levels of neurotrophi ω−3 and brain-derived neurotrophic factor.223 Similarly, an increased proliferation of progenitor cells and neural stem cells in the hippocampus is only observed in young mice undergoing 40% CR.224 It is non-negligible that mice with both CR and calorie dilution (CD) exhibit a prominent elevation of starvation-related genes in hypothalamus.225 In addition, composition-specific diet influences neurologic function. A diet with reduced dietary protein intake or rapamycin treatment reduces food intake in mice, but these mice do not appear to be hyperphagic when returned to a protein-rich diet. A very low-protein diet caused weight decline and improved glucose tolerance partially through hypothalamic mTOR signalling.226 Then, KD is found to improve cognition in mild cognitive impairment,227 while methionine restriction increases FGF21 expression in serum, liver, and brain to alleviate age-associated cognitive decline.228 Interestingly, the supplement with non-essential amino acids results in appetitive suppression through direct activation of hypothalamic orexin/hypocretin neurons.229
Although the regulation of nutrient-sensitive mechanisms and their modulators by nutrimental intervention is well established, subsequent studies can concentrate on identifying and validating the polymorphic functions, which will require translational studies to develop individualized therapeutic regimens for disease treatment and healthspan improvement.
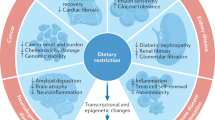
Dietary intervention in tissue health and disease
Considering that diet-mediated signalling exists throughout the entire body, diet-derived effects on health and disease have been assumed to occur in all tissues and organs. Consequently, nutrients influence tissue to varying degrees and in diverse ways (Fig. 3). Furthermore, multiple nutritional interventions have been demonstrated to broadly relieve the disease process and immensely strengthen the therapeutic effect (Table 3). In addition, we also summarize the current clinical trials related to dietary interventions (Table 4). Thus, we elaborate the mechanisms by which diet impacts health and diseases, which can aid in the development of precision-nutrition therapeutics.
Functionality of dietary interventions on human tissues under health and diverse disease states. Dietary interventions exert beneficial effects across varying aspects, including nervous system, cardiovascular system, respiratory system, metabolic system, renal system, endocrine system, and digestive system as well as cancer
Metabolic syndrome
Overnutrition contributes to accelerate disease progression, notably with obesity and type 2 diabetes. A myriad of diseases, including non-alcoholic fatty liver disease (NAFLD), diabetes, cardiovascular disease (CVD), and cancer, are positively correlated with obesity.230 A fat-rich diet and excessive intake of BCAAs, tryptophan and its metabolites, and methionine result in weight gain and obesity-associated disease (reviewed in ref. 231) For example, a mixed-protein diet mimicking a Western diet exacerbates insulin resistance and obesity. Mechanistically, the mixed protein source can give rise to elevation of acylcarnitines and microbially generated BCAAs in plasma and liver, further activating mTORC1/S6 kinase 1 (S6K1) to increase glucose synthesis in hepatocytes.232 As a novel diet-dependent obesogen, microbial δ-valerobetaine (VB) is found to exacerbate visceral fat mass and phenotypic obesity by inhibiting mitochondrial fatty acid oxidation.233
Obesity comprehensively leads to comorbidities. Thus, dietary intervention is a promising method to combat these disorders. Specifically, CR has the capability to polarize M2 macrophages and recruit eosinophils in fat to promote beige fat within the visceral and subcutaneous fat tissue, consequently contributing to metabolic improvements and lipopenia in mice.234 Likewise, CR induced a high release of adiponectin to impact adiposity by activating PGC-1α, SIRT1, and NAMPT in adipose tissues.235 In clinical, CR is observed to reduce the levels of alkaline phosphatase (ALP) and gamma-glutamyl transferase (GGT) in health adults to improve liver function.236 Moreover, diet patterns are pivotal in obesity-related metabolic dysfunction. In WAT, isocaloric IF is capable of polarizing macrophage alternative activation via induction of vascular endothelial growth factor (VEGF) expression, then improving metabolic dysfunction primarily through adipose thermogenesis.237 Alternating prolonged fasting (PF) has been found to reduce high-risk factors for cancer and ageing-associated diseases in the absence of obvious detriments in a pilot clinical trial.238,239 Compared to controls, TRF leads to a considerable decrease in oxidative stress, insulin resistance and body weight240 and improves cardiometabolic health for patients with metabolic syndrome.241 Similarly, an every-other-day fasting (EODF) regimen prominently mitigates insulin resistance, NAFLD and obesity. EODF not only increases mitochondrial protein content and fatty acid synthesis enzymes in WAT242 but also accelerates the intake of microbially fermented acetate and lactate in beige cells by electively upregulating their transporters.243 However, the benefits of TRF on loss of body fat and body weight are not superior to that of daily CR in obese patients.244 Ultimately, alteration of nutritive components is a feasible way to mitigate metabolic syndrome. The Mediterranean diet (MD) and plant-rich diets are associated with a lower risk of type 2 diabetes in healthy adults.245,246 BCAA restriction has the capacity to improve health in obese and non-obese animals as well as in humans.165 Likewise, branched-chain keto acids (BCKAs) have been found to suppress inguinal WAT browning and thermogenesis to expedite high-fat diet-induced obesity, and telmisartan significantly reduced BCKA levels to result in enhanced WAT browning and reduced adiposity.247 In addition, daily high-fibre supplementation combined with faecal microbiota transplantation (FMT) or yogurt consumption alleviates insulin resistance in cases with metabolic syndrome or severe obesity.248,249 In similar, daily high-inulin supplementation combined with excise are able to reduce cholesterol level in plasma and liver, improve glucose tolerance and lead to weight loss through augmentation of inulin-degrading bacteria in gut.250 In contrast, a CNOT6L inhibitor elevates hepatokine FGF21 and growth differentiation factor 15 (GDF15) levels by stabilizing their mRNAs and resulted in a remarkable improvement of overnutrition-induced metabolic syndrome.251
Although there is clearly a relationship between nutritional intervention and metabolic disorders, the type and pattern of diet may be vital. A case-by-case analysis is required to demonstrate how nutrition interventions are regarded as adjuvant treatments for metabolic syndromes.
Cardiovascular diseases
Diet is closely linked to the onset of cardiovascular health and disease, and cardiovascular health has frequently focalized dietary interventions.252 High-fat/salt diets and diets extremely high in BCAAs are high-risk factors for CVD.253 For example, dietary BCAA intake significantly facilitates platelet activation, further increasing the risks of arterial thrombosis and cardiovascular disease.254 Recently, CR is found to reduce adverse factors for CVD and improve the parameters of cardiac function, such as energy expenditure, heart rate and blood pressure.255 One possible mechanism is that CR reduces free leptin in plasma, which reverses myocardial hypertrophy and reduces lipid accumulation.256 CR also reduces oxidative stress, fibrosis, cardiac inflammation and myocyte hypertrophy in mice.257 Studies have further demonstrated the advantages of CR, such as lessened triglyceride, cholesterol and LDL levels.258,259 A 5-day fast reduces blood pressure by altering the gut microbiome and systemic immune status.196 In obese models induced by a fat-rich diet, a 5-day fasting-mimicking diet (FMD) is able to promote visceral and subcutaneous fat loss and protect against obesity-impaired cardiac vascularity and function.260 Similar to CR, IF retards hypertension pathogenesis in rats through alteration of the metabatic profile in the caecum and plasma and the composition of the gut microbiota.261 Considering food composition, high fibre consumption is discovered to elevate the richness of SCFA-generated microbial communities to include Bacteroides acidifaciens. SCFA supplementation as well as a high-fibre diet stabilize circadian rhythm and downregulate Egr1, a master cardiovascular regulator, in the heart and kidney and are conducive to decreasing cardiac hypertrophy and fibrosis as well as blood pressure.262 A low-carbohydrate, high-fat KD elevated the levels of ketone bodies to stimulate the proliferation of cardiac endothelial cells and prevent heart hypertrophy.263 Particularly, MD alters the microbial compositions and function to benefit cardiometabolic homoeostasis,264,265 and reduce diverse cardiovascular events like atherosclerosis to prevent cardiovascular disease.266,267 Altogether, dietary interventions partially mediate these cardiovascular complications.
Intestinal malfunction
Diet has been discovered to profoundly affect intestinal functions. Digestive disorders such as colitis and Crohn disease are regulated by nutritional therapies. Some diet plans are adverse for intestinal function. For instance, long-term red meat consumption has been related to a high risk of digestive disease. The N-glycolylneuraminic acid (Neu5Gc) enriched in red meat can only be synthesized by gut microbiota. The red-meat diet induces an increase in Bacteroidales and Clostridiales to produce Neu5Gc, which is a potential factor contributing to the inflammation-mediated promotion of diseases and carcinomas.268 In active human Crohn disease, ω−3 and ω−6 PUFAs induce chemokine expression in epithelial cells by activating toll-like receptor 2 (TLR2)/inositol-requiring enzyme 1α (IRE1α) in epithelial cells.269 Some nutrition strategies are enabled to sustain intestinal homoeostasis. First, dietary restriction is demonstrated to improve intestinal barrier function and maintain intestinal homoeostasis via upregulation of Myc270 and activation of IRE1/X-box binding protein 1 (XBP1) endoplasmic reticulum (ER) stress signalling,271 excitation of autophagy in worms272 and alteration of the intestinal microbiome.11 Specific dietetic techniques for dietary restriction of fermentable carbohydrates and CR have been discovered to improve functional gastrointestinal symptoms and combat the worsening of digestive disorders.273 In contrast, the effects of a high-fibre, low-fat diet reduces inflammatory markers and reverses intestinal dysbiosis to benefit patients with ulcerative colitis.274 In addition, KD has been found to be able to protect the function of the intestinal barrier and alleviate colitis. Mechanistically, KD contributes to a reproducible increase in Akkermansia but declines in Escherichia/Shigella, subsequently eliminating inflammatory factors (Ccl4, IL-17α, IL-18, IL-22) and restraining the differentiation of group 3 innate lymphoid cells (ILC3s) and related inflammatory cytokines (IL-17α, IL-18, IL-22, Ccl4).275 Moreover, L-serine restriction markedly reduces the abundance and availability of E. coli LF82, which catabolizes L-serine and blunts the inflammatory response.276 Overall, suitable dietary patterns appear beneficial to properly sustain metabolic and barrier functionality in the gut, but the detailed mechanisms should be further deciphered.
Renal diseases
Renal functionality is broadly dependent on the state of nourishment. Notoriously, overnutrition impairs renal function. For example, dietary protein aggravates progression to chronic kidney disease (CKD) in mice. A high-protein diet elevates levels of uraemic toxins such as microbial indoxyl sulfate, indole and hydrogen sulfide (H2S).277 Likewise, excessive salt intake has been suspected to exacerbate the symptoms of renal diseases via multiple mechanisms (reviewed in ref. 278) Consequently, appropriate alimentative methods alleviate renal dysfunction. Recent studies in murine models have indicated that dietary protein restriction decreases renal ammonia excretion by altering ammonia metabolism.279 In peritoneal dialysis, the restriction of dietary advanced glycation end products (AGEs) remodels gut-microbiome composition, suggesting that a reduction in AGEs potentially alleviates CKD progression.280 Streptozotocin is most often used to elicit diabetic kidney disease (DKD) in murine models. In these mouse models, the replenishment of β-hydroxybutyrate inhibits glycogen synthase kinase 3β (GSK3β) but activates Nrf2 to improve diabetic albuminuria and glomerulopathy and substantially mitigate podocyte injury and senescence.281 Similarly, acetate generated in a high-fibre diet markedly reduced renal fibrosis.262 Some studies have uncovered the possible mechanisms by which nourishment may impact renal ischaemia-reperfusion, showing that nutritive intervention has therapeutic potential.282 In clinical, coronary heart disease patients with T2DM undergo a MD for 5 years, and they exhibit a remarkably decline of creatinine-based glomerular filtration rate, suggesting that MD could preserve kidney function.283 Taken together, these studies confirm that optimized diets serve as a dominant factor for relieving renal dysfunction.
Nervous system and neurodegenerative diseases
Due to potential neuroprotection, optimal diet has received serious attention. First, declines in neurological function are able to be delayed by dietary restriction in mice284 and macaques,285 which may be attributed to SIRT1 activation. CR and KD improve late-life memory in mice.286,287 In obese rats, CR is capable of accelerating proteasome-dependent degradation and ameliorating neuronal cell death to protect overall neuronal function.288 Furthermore, CR stimulates the secretion of neurotrophic factors and relieves diverse stress responses as well as DNA maintenance in mice.289,290 Added to the diet cycle and components, FMD contributes to improved cognitive performance in aged mice and in humans through elevation of NeuroD1 but reduced IGF-1 levels and PKA activity in hippocampal neurogenesis.238 In the Drosophila brain, dietary spermidine induces the levels of eIF5A hypusination, which reverses age-associated memory and locomotion loss and rescues the homoeostasis of senescent mitochondria.291 Fortuitously, the behaviour and gut microbiomes of offspring can be disrupted by maternal obesity. SCFA supplementation or a high-fibre diet in either offspring or mothers alleviates microglial maturation defects and synaptic impairments.292 In summary, favourable diet strategies have the capacity to align the function of the nervous system for health improvement.
Consequently, an increasing number of studies have focused on the beneficial effects of diet in treating neurological diseases.293 Initially, CR suppresses inflammation and oxidative stress, remodels gut microbiota, and protects the substantia nigra and dopaminergic neurons, which is conducive to relieving symptoms of Parkinson disease (PD).294 In addition, oral NR to replenish NAD+ elevates the level of cerebral NAD+, which reduces inflammatory factor levels in both cerebrospinal fluid and plasma. Thus, NR supplementation is a safe and effective treatment for PD.295 Nutritional intervention also benefits the symptoms of AD. The evidence shows that dietary intervention during neurodegenerative treatments enables relief of the AD process.296 CR in early-life rats preserves the blood–brain barrier to delay AD pathologies.297 Comparably, MD as well as the supplementation with medium-chain triglycerides (MCT) improves gait and cognitive functions in healthy older adults.298,299 As a neurodegenerative disease, Huntington disease (HD) is distinguished by protein aggregates in the brain, and CR advantageously functions in HD mice via histone acetylation modifications.300 Similarly, TRF enables perfect motor function and circadian rhythms in HD mice.301 Finally, multiple sclerosis (MS) is a common CNS autoimmunity. IF results in increased richness of gut bacterial, enhancement of antioxidative microbial metabolic pathways, and alteration of T-cell subtype proportions in the gut to ameliorate the clinical course and pathology of MS.154 It is evident that alimentative strategies profoundly affect neurological health and disease, and future studies should utilize diet as a promising therapy for neurological disease.
Muscle and skeletal tissue
Muscle is regarded as one of the main metabolic organs, and muscle homoeostasis is dramatically mediated by food intake. It is evident that CR preserves muscle structure in late life302 and improve the muscle integrity in aged mouse.303 From a mechanistic perspective, CR can improve mitochondrial function and autophagy to provide muscular benefits.304 CR prevents muscular apoptosis by upregulating IL-15.305 Additional targets for CR are found to reduce oxidative stress for persistent advantages in muscle tissue.306 CR also optimizes proteasome-dependent degradation to sustain muscle health in mice.307 Considering CR-induced superiority, diet intervention is frequently applied to determine its efficacy in muscle-associated disease. In the early stages of senescence-related sarcopenia, CR sustains muscle mass in mice308 as well as in monkeys.309 Regrettably, CR has no effect on muscle health in patients with sarcopenia.310 What is more, the influences of fasting on muscle repair are uncovered. During fasting or KD, muscle stem cells (MuSCs) are converted to highly resilient deep quiescence where they could obtain ameliorative survival under diverse cellular stress.311 However, patients ingesting dietary proteins mainly at breakfast were found to have higher muscle function than those ingesting dietary proteins at dinner. In the early active phase, protein intake, especially BCAA supplementation, promotes skeletal muscle hypertrophy in a muscle-clock-reliable manner.312 Although the results thus far have been unpromising in clinical trials, a multitude of investigations into how the rhythm and pattern of diets influence human health are needed.
Bone health highly depends on a balanced diet, particularly with regard to dietary mineral intake. Unexpectedly, CR has been demonstrated to increase the risk of bone weakening and osteoporosis313 and reduce bone-healing ability in catagmatic or osteoporotic rats.314 A potential reason may be a decrease in mineral intake. Conversely, CR paired with exercise or mild CR is able to improve bone health.315,316 The administration of αKG ameliorates osteoporosis in aged mice. In detail, αKG promotes the proliferation, colony formation, migration, and osteogenesis of bone marrow mesenchymal stem cells (MSCs) by demethylating H3K9me3 and H3K27me3 to increase Nanog expression and BMP signalling. Activated MSCs attenuate age-related bone loss by promoting osteogenesis and accelerating bone regeneration.121 In general, meticulous dietary adjustments should be closely explored to strengthen bone health.
Endocrine system
The endocrine system regulates the effectors of nutritional intervention, but dietary intake influences hormonal signalling. Therefore, the interactions between endocrine modulation and diets are quite complicated. Endocrine-associated metabolic pathways and CR are intricately connected and mainly involve ghrelin, leptin, adiponectin, FGF21, GDF15, ACBP and asprosin. For example, ghrelin, as a hunger hormone, is acylated to stimulate glycogenolysis in response to CR.317 Following long-term CR, growth-associated hormonic pathways are observably repressed in rats.318 The hypothalamic/pituitary axis is heavily influenced by nutrients.319 Diet-mediated alterations in hormones and cytokines occur in multiple neuroendocrine axes. In the adrenal glands, CR amplifies the neuroprotective effects of glucocorticoids.320 CR has been found to inhibit hormone secretion in mammary glands and ovulation in females to perturb signal transduction between the pituitary gland and gonads.321 Likewise, testosterone levels are elevated in men by CR.322 Fasting induces profound changes in the hypothalamic-pituitary-thyroid (HPT) axis. In a clinical trial, long-term ADF leads to reduced secretion of circulating fT3 and high release of parathyroid hormone (PTH) but sustains normal function of the thyroid gland.80 Similarly, fasting leads to phosphorylation of thyroid hormone receptor β 2 isoform (THRB2) at the S101 site via AMPK/cyclin-dependent kinase 2 (CDK2) activation, and repress the functions of thyroid-stimulating hormone and thyrotropin-releasing hormone through negative feedback.323 Overall, the disparity and the interplay in the endocrine system exhibit a systemic function of diet and demonstrate that nutritional strategies extensively affect endocrine regulation and that these hormones are pivotal for determining applicable diet-based therapies for endocrine disorders.
Respiratory system
Diet profoundly impacts health and disease in the respiratory system. Originally, CR is found to prevent pulmonary mycobacterium tuberculosis (MTB) infection. Mechanistically, CR impairs both fatty acid β-oxidation and mTOR activity but induces glycolysis and autophagy in immune cells. As a result, CR reduces the bacterial load and consolidated the MTB reservoir in foam cells to restrain lung damage and endow lung epithelial cells with an intact barrier capable of confining spreading.175 After infection of Enterovirus D68 (EV-D68), a respiratory virus, fasting is observed to attenuate virus replication through stimulating of autophagic flux.324 Furthermore, dietary tryptophan affects sensitivity to anti-MTB therapy. Tryptophan catabolites bind and activate aryl hydrocarbon receptor (AhR), which combines with rifampicin (RIF) and rifabutin (RFB), anti-MTB drugs. Subsequently, they function to increase drug metabolism and reinforce the host antibacterial response to consolidate the efficacy of anti-MTB drugs.324 In obese patients with chronic obstructive pulmonary disease (COPD), CR paired with exercise is capable of improving dyspnoea and obstruction symptoms.325 Regarding food composition, a low-carbohydrate, high-fat KD is discovered to potentially preserve respiratory function through a decrease in the storage of carbon dioxide in the lungs.326 Meanwhile, KD remodels the metabolic process of γδ T cells and contributes to γδ T-cells expansion, which improves lung barrier functions to resist influenza virus infection.327 In addition, dietary fibre has a vital effect on lung functions. A fibre-rich diet augments the proportion of fibre-fermenting bacteria in both the lung and gut to elevate circulating levels of SCFAs, subsequently leading to a decline in allergic inflammation in the lung. SCFAs reinforce the production of dendritic cells (DCs) and macrophage precursors in bone marrow haematopoiesis, and DCs with high phagocytic capacity migrate to the lungs to promote the effector function of Th2 cells. Likewise, SCFAs bind to G protein-coupled receptor 41 (GPR41) to alleviate allergic inflammation.328 In addition, a high-fibre diet prolongs the survival of influenza-infected mice. The main mechanism is that SCFAs facilitate the functionality of CD8+ effector T cells, and also induce the polarity of macrophages by enhancing precursor generation in bone marrow haematopoiesis. These macrophages restrain the secretion of the chemokine CXCL1, which recruits neutrophils in the airways.329 Initial studies highlights that the manner of nutritional therapy is key to improving the respiratory microecosystem and function for lung health.
Cancer
Dietotheroapy is a prime adjunctive therapy for cancer treatment involving multiple intrinsic and extrinsic pathways. In intrinsic mechanisms, nutrient interventions are capable of suppressing neoplastic growth and metastasis. In a mouse model, TRF is found to delay tumour initiation, retard obesity-promoted malignant growth and decrease pulmonary metastatic foci, partially dependent on insulin levels and metabolic state.330 In addition, CR and FMD have been found to reduce IGF-1 levels to delay cancer risk.90,138,331 CR is capable of upregulating p53, a tumour suppressor, in mice.332 Similarly, GR deacetylates and degrades mutant p53 in an autophagy-dependent way. CR highly stimulates SIRT1 in mice with p53 deficiency, suggesting that deacetylation of mutant p53 results in a reduced level.333 αKG has the capacity to repress tumour growth depending on p53 status, while succinate has been found to competitively inhibit the effect of αKG to abrogate p53-mediated tumour suppression. The accumulation of αKG is able to increase 5-hydroxymethylcytosine (5hmC) modification in chromatin, accompanied by tumour cell differentiation and decreasing tumour cell fitness.116 mTOR and FOXO3 are additional mechanisms found to be associated with CR and tumour suppression.93 Ketone bodies derived from stromal cells can be absorbed by tumour cells to exhibit metabolic adaptation and an aggressive phenotype.334 Similarly, β-hydroxybutyrate is an alternative cell-intrinsic or systemic fuel that can promote the progression of pancreatic ductal adenocarcinoma.335 A fat-enriched KD elevates the circulatory levels of acetoacetate, which expedites malignant growth in mice inoculated with BRAFV600E-mutant melanoma cells;336 whereas KD combined with IF leads to an increase of uric acid but reduction of leptin, insulin and glucose in patients with recurrent brain tumours.337 Conversely, ω−3 and ω−6 PUFAs selectively induces ferroptosis in malignant cells, and an ω−3 PUFA-enriched diet dramatically decelerates murine tumour growth.338 Consistently, CR as well as KD with altering fat composition slows malignant growth via an imbalanced ratio of saturated to unsaturated fatty acids cooperating with decreasing tumorous stearoyl-CoA desaturase activity.339 Thus, the specific species of fatty acid determine whether KD impairs tumour growth. Dietary restriction of protein and special amino acids, such as serine, methionine and BCAAs, can retard tumour growth (reviewed in ref. 93) For example, an improved survival during concomitant dietary restriction of serine and glycine is found in mouse models of multiple tumour types.340 Moreover, a series of drugs mimicking amino acid restriction have been put forward. Systemic cysteinase enables to deplete cysteine and inhibit tumour growth through elevating ROS and inducing tumour-selective ferroptosis.341,342 Glutamine antagonism in tumour-bearing mice exhibits forceful antitumor effects by down-regulating oxidative and glycolytic metabolism of malignant cells but dramatically promoting oxidative metabolism of effector T cells.343 In mineral metabolism, Se replenishment has antioxidant and DNA repair effects to exert anticancer properties. As a cofactor in DNA repair genes, zinc is shown to perform a DNA repair function through estimation of oxidized guanine. Similarly, magnesium exerts coenzyme functions for diverse DNA polymerases.69 In the extrinsic pathways, short-term fasting induces the depletion of regulatory T cells (Tregs) in mice, thereby improving anticancer immunosurveillance.90,331 In a clinical trial, FMD contributes to better clinical outcomes in patients with cancer. Mechanistically, FMD decreases the ratio of Tregs to immunosuppressive myeloid cells in peripheral areas and strengthens intratumoral cytotoxic effectors to profoundly remodel antitumor immunity.344 Considering the nutrient proportions, a low-protein diet reduces tumour growth through expansion of antigen-presenting cells (APCs) and CD8+ T cells.345 Unexpectedly, abnormal accumulation of potassium ions (K+) in the tumour interstitial fluid has been found to contribute to impaired T-cell effector function through suppression of AKT-mTOR signalling.346
Dietary regulation also influences responsiveness to cancer treatments (which has been reviewed in ref. 347) A short-term fasting or FMD synergizes with chemotherapy to inhibit tumour growth by enhancing CD8+-dependent tumour cytotoxicity or chemotherapy-induced DNA damage.90,348,349 KD potentiates cytotoxic chemotherapy in the treatment of tumours in murine pancreatic cancer;350 by contrast, dietetic cystine restriction retard tumour growth and enhance the efficiency of chemotherapy in colon cancer model.351 In a murine hormone-receptor-positive breast cancer model, PF or FMD reinforces the efficiency of endocrine therapeutic drugs such as fulvestrant and tamoxifen in mice and humans. Furthermore, a FMD combining fulvestrant and CDK4/6 inhibitor causes extensive tumour regression and reversed drug resistance.159 In targeted therapy, fasting synergizes with the efficacy of sorafenib in hepatocellular carcinoma accompanied by acquired resistance to sorafenib.352 Immunotherapy is particularly strengthened with diverse eating strategies in mouse models. Short-dated fasting decreases the serum levels of IGF-1 and IGF-1 receptor expression in neoplastic cells and potentiates the therapeutic effect of PD-1 inhibitors to suppress malignant progression.331 Similarly, picropodophyllin, as an inhibitor of IGF-1R, stimulates the infiltration of intratumoral cytotoxic T lymphocytes (CTLs) but reduces Tregs to enhance the efficacy of chemoimmunotherapy with a combination of immunogenic chemotherapeutics and PD-1 antagonists.138 KD is capable to suppress colorectal tumorigenesis,353 and KD or oral 3-hydroxybutyrate supplementation re-establishes therapeutic responses to immune checkpoint inhibitors.354 Mechanistically, ketone bodies profoundly impact human T-cell responses by markedly enhancing the capacity of CD4+ T cells, CD8+ T cells, and Tregs and augmenting the formation of memory T cells.355 In addition, dietary consumption enriched in fibre and yogurt is associated with a low risk of lung cancer.356 A fibre-rich diet as well as prebiotics supplementation prolongs progression-free survival and synergizes anti-PD-1-based therapy in mice.357,358 In syngeneic murine cancer models, SCFA supplementation heightens the anticancer capacity of ROR1-targeting CAR-T cells and antigen-specific CTLs.359 However, high SCFA levels limit the accumulation of memory T cells and tumour-specific T cells as well as the expression of ICOS on T cells and CD80/CD86 on DCs to resist CTLA-4 blockade.360 For dietary microelements, replenishing NAD+ boosts T-cell-based immunotherapy by facilitating the cell-killing function of CTLs.361 In multiple types of tumours, the biogenesis of NAD+ upregulates PD-L1 expression within IFNγ stimulation and prompts tumour immune escape in a CTL-dependent manner. Therefore, anti-PD-L1 treatment is more effective in tumours with high NAMPT, and NAD+ supplementation augments the efficacy of PD-L1 blockade in tumours resistant to immunotherapy.362 Another study also indicates that a high concentration of K+ in the tumour microenvironment (TME) is able to suppress the tumour-killing function of CTLs but had no effect on T-cell proliferation by restraining CTL nutrient absorption.363 However, a high K+ concentration improves the anticancer activity, pluripotency and durability of adoptively transferred CD8+ T cells by reprogramming their metabolic processes.363 Thus, potassium in the TME reshapes T-cell metabolism to enhance anticancer immunological reactions. Taken together, proper adjustment of the manner and composition of diet not only decelerates tumour progression but also potentiates the therapeutic effect of diversified antineoplastic strategies.
Conclusion
Notably, nutritive intervention is viewed as an efficient therapy for counteracting morbidities and maintaining health. However, there are several unsolved questions about dietary functionality and its clinical application. First, future studies should elaborate the period and circadian rhythm of dietary treatment, such as curative endurance, cycles or meal times. Moreover, future studies should focus on the mechanisms by which strict nutrient constituents such as lipids, carbohydrates, amino acids, micronutrients and metabolites modulate morbid progression and health state. It is crucial to understand how to combine diet with sleep, exercise and other therapeutic interventions to acquire cooperative effects that improve healthspan at the tissue and organismal levels. Considering that gene variants like Pkn involved in nutrition-mediated response,364 the genetic disparity should be defined in personalized nutrition intervention. The nutritive functions in diverse genotypes also require further exploration. Due to the heterogeneity within individuals in response to dietary interventions, biomarker panels need to be developed, and diet-based therapeutic strategies need to be optimized. Similarly, it has been reported that diet-mediated functions exhibit sexual disparity in diverse organisms including humans.365,366 Therefore, gender necessitates consideration in nutrient-based therapies. Because demographic factors such as age and sex and genotypes are currently not taken into consideration in diet-based trials, successive studies need to elucidate their pivotal roles based on multi-omics data analysis and systems biology.
Considering that nutrient restriction is unattainable in humans, replacement therapies that in part imitate the advantages of dietary intervention at the physiological and molecular levels are prospective. This approach requires seeking behavioural interventions or compounds that serve as mimetics and inducers of alimentation intervention, such as CRMs.90,137,138,140 It is hopeful that individualized diets could potentially delay disease progression and maximize health maintenance.
References
Pontzer, H., Wood, B. M. & Raichlen, D. A. Hunter-gatherers as models in public health. Obes. Rev. 19, 24–35 (2018).
Sonnenburg, J. L. & Sonnenburg, E. D. Vulnerability of the industrialized microbiota. Science 366, eaaw9255 (2019).
Kenyon, C. J. The genetics of ageing. Nature 464, 504–512 (2010).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. The hallmarks of aging. Cell 153, 1194–1217 (2013).
Wilson, K. A. et al. Evaluating the beneficial effects of dietary restrictions: a framework for precision nutrigeroscience. Cell Metab. 33, 2142–2173 (2021).
Rodgers, G. P. & Collins, F. S. Precision nutrition-the answer to “what to eat to stay healthy”. JAMA 324, 735–736 (2020).
Liu, G. Y. & Sabatini, D. M. mTOR at the nexus of nutrition, growth, ageing and disease. Nat. Rev. Mol. Cell Biol. 21, 183–203 (2020).
Szwed, A., Kim, E. & Jacinto, E. Regulation and metabolic functions of mTORC1 and mTORC2. Physiol. Rev. 101, 1371–1426 (2021).
Harrison, D. E. et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 460, 392–395 (2009).
Dorling, J. L., Martin, C. K. & Redman, L. M. Calorie restriction for enhanced longevity: the role of novel dietary strategies in the present obesogenic environment. Ageing Res. Rev. 64, 101038 (2020).
Sowah, S. A. et al. Calorie restriction improves metabolic state independently of gut microbiome composition: a randomized dietary intervention trial. Genome Med. 14, 30 (2022).
O'Flanagan, C. H., Smith, L. A., McDonell, S. B. & Hursting, S. D. When less may be more: calorie restriction and response to cancer therapy. BMC Med. 15, 106 (2017).
Madeo, F., Carmona-Gutierrez, D., Hofer, S. J. & Kroemer, G. Caloric restriction mimetics against age-associated disease: targets, mechanisms, and therapeutic potential. Cell Metab. 29, 592–610 (2019).
Chong, C. R., Sallustio, B. & Horowitz, J. D. Drugs that affect cardiac metabolism: focus on perhexiline. Cardiovasc Drugs Ther. 30, 399–405 (2016).
Schreiber, K. H. et al. A novel rapamycin analog is highly selective for mTORC1 in vivo. Nat. Commun. 10, 3194 (2019).
Lamming, D. W. et al. Rapamycin-induced insulin resistance is mediated by mTORC2 loss and uncoupled from longevity. Science 335, 1638–1643 (2012).
Sarbassov, D. D. et al. Prolonged rapamycin treatment inhibits mTORC2 assembly and Akt/PKB. Mol. Cell 22, 159–168 (2006).
González, A., Hall, M. N., Lin, S. C. & Hardie, D. G. AMPK and TOR: the yin and yang of cellular nutrient sensing and growth control. Cell Metab. 31, 472–492 (2020).
Lin, S. C. & Hardie, D. G. AMPK: sensing glucose as well as cellular energy status. Cell Metab. 27, 299–313 (2018).
Fontana, L. & Partridge, L. Promoting health and longevity through diet: from model organisms to humans. Cell 161, 106–118 (2015).
Kapahi, P., Kaeberlein, M. & Hansen, M. Dietary restriction and lifespan: lessons from invertebrate models. Ageing Res. Rev. 39, 3–14 (2017).
Schulz, T. J. et al. Glucose restriction extends Caenorhabditis elegans life span by inducing mitochondrial respiration and increasing oxidative stress. Cell Metab. 6, 280–293 (2007).
Ristow, M. & Zarse, K. How increased oxidative stress promotes longevity and metabolic health: the concept of mitochondrial hormesis (mitohormesis). Exp. Gerontol. 45, 410–418 (2010).
Weir, H. J. et al. Dietary restriction and AMPK increase lifespan via mitochondrial network and peroxisome remodeling. Cell Metab. 26, 884–896.e5 (2017).
Boccardi, V., Murasecco, I. & Mecocci, P. Diabetes drugs in the fight against Alzheimer's disease. Ageing Res. Rev. 54, 100936 (2019).
Calissi, G., Lam, E. W. & Link, W. Therapeutic strategies targeting FOXO transcription factors. Nat. Rev. Drug Disco. 20, 21–38 (2021).
Greer, E. L. & Brunet, A. Different dietary restriction regimens extend lifespan by both independent and overlapping genetic pathways in C. elegans. Aging Cell 8, 113–127 (2009).
Giannakou, M. E., Goss, M. & Partridge, L. Role of dFOXO in lifespan extension by dietary restriction in Drosophila melanogaster: not required, but its activity modulates the response. Aging Cell 7, 187–198 (2008).
Miyauchi, T. et al. Up-regulation of FOXO1 and reduced inflammation by β-hydroxybutyric acid are essential diet restriction benefits against liver injury. Proc. Natl Acad. Sci. USA 116, 13533–13542 (2019).
Furuyama, T. et al. Effects of aging and caloric restriction on the gene expression of Foxo1, 3, and 4 (FKHR, FKHRL1, and AFX) in the rat skeletal muscles. Microsc. Res. Tech. 59, 331–334 (2002).
Shimokawa, I. et al. The life-extending effect of dietary restriction requires Foxo3 in mice. Aging Cell 14, 707–709 (2015).
Xia, Y. et al. Neuronal C/EBPβ/AEP pathway shortens life span via selective GABAnergic neuronal degeneration by FOXO repression. Sci. Adv. 8, eabj8658 (2022).
Zhu, S. et al. The roles of sirtuins family in cell metabolism during tumor development. Semin. Cancer Biol. 57, 59–71 (2019).
Xie, N. et al. NAD(+) metabolism: pathophysiologic mechanisms and therapeutic potential. Signal Transduct. Target Ther. 5, 227 (2020).
Covarrubias, A. J., Perrone, R., Grozio, A. & Verdin, E. NAD(+) metabolism and its roles in cellular processes during ageing. Nat. Rev. Mol. Cell Biol. 22, 119–141 (2021).
Lin, S. J., Defossez, P. A. & Guarente, L. Requirement of NAD and SIR2 for life-span extension by calorie restriction in Saccharomyces cerevisiae. Science 289, 2126–2128 (2000).
Houtkooper, R. H., Pirinen, E. & Auwerx, J. Sirtuins as regulators of metabolism and healthspan. Nat. Rev. Mol. Cell Biol. 13, 225–238 (2012).
Cohen, H. Y. et al. Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science 305, 390–392 (2004).
Han, X. et al. Targeting Sirtuin1 to treat aging-related tissue fibrosis: From prevention to therapy. Pharm. Ther. 229, 107983 (2022).
Civitarese, A. E. et al. Calorie restriction increases muscle mitochondrial biogenesis in healthy humans. PLoS Med. 4, e76 (2007).
Gomes, P. et al. The yin and yang faces of the mitochondrial deacetylase sirtuin 3 in age-related disorders. Ageing Res. Rev. 57, 100983 (2020).
Nakagawa, T., Lomb, D. J., Haigis, M. C. & Guarente, L. SIRT5 Deacetylates carbamoyl phosphate synthetase 1 and regulates the urea cycle. Cell 137, 560–570 (2009).
Bordone, L. et al. SIRT1 transgenic mice show phenotypes resembling calorie restriction. Aging Cell 6, 759–767 (2007).
Herranz, D. et al. Sirt1 improves healthy ageing and protects from metabolic syndrome-associated cancer. Nat. Commun. 1, 3 (2010).
North, B. J. et al. SIRT2 induces the checkpoint kinase BubR1 to increase lifespan. EMBO J. 33, 1438–1453 (2014).
Benigni, A. et al. Sirt3 deficiency shortens life span and impairs cardiac mitochondrial function rescued by Opa1 gene transfer. Antioxid. Redox Signal 31, 1255–1271 (2019).
Brown, K. et al. SIRT3 reverses aging-associated degeneration. Cell Rep. 3, 319–327 (2013).
Kawahara, T. L. et al. SIRT6 links histone H3 lysine 9 deacetylation to NF-kappaB-dependent gene expression and organismal life span. Cell 136, 62–74 (2009).
Vakhrusheva, O. et al. Sirt7 increases stress resistance of cardiomyocytes and prevents apoptosis and inflammatory cardiomyopathy in mice. Circ. Res. 102, 703–710 (2008).
Korotkov, A., Seluanov, A. & Gorbunova, V. Sirtuin 6: linking longevity with genome and epigenome stability. Trends Cell Biol. 31, 994–1006 (2021).
Sun, S. et al. Vascular endothelium-targeted Sirt7 gene therapy rejuvenates blood vessels and extends life span in a Hutchinson-Gilford progeria model. Sci. Adv. 6, eaay5556 (2020).
Berdichevsky, A., Viswanathan, M., Horvitz, H. R. & Guarente, L. C. elegans SIR-2.1 interacts with 14-3-3 proteins to activate DAF-16 and extend life span. Cell 125, 1165–1177 (2006).
Brunet, A. et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303, 2011–2015 (2004).
Wang, R. et al. Metabolic stress modulates Alzheimer's β-secretase gene transcription via SIRT1-PPARγ-PGC-1 in neurons. Cell Metab. 17, 685–694 (2013).
Cantó, C. et al. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature 458, 1056–1060 (2009).
Someya, S. et al. Sirt3 mediates reduction of oxidative damage and prevention of age-related hearing loss under caloric restriction. Cell 143, 802–812 (2010).
Hebert, A. S. et al. Calorie restriction and SIRT3 trigger global reprogramming of the mitochondrial protein acetylome. Mol. Cell 49, 186–199 (2013).
Van Meter, M. et al. JNK phosphorylates SIRT6 to stimulate DNA double-strand break repair in response to oxidative stress by recruiting PARP1 to DNA breaks. Cell Rep. 16, 2641–2650 (2016).
Simon, M. et al. LINE1 derepression in aged wild-type and SIRT6-deficient mice drives inflammation. Cell Metab. 29, 871–885.e5 (2019).
Anderson, R. M., Bitterman, K. J., Wood, J. G., Medvedik, O. & Sinclair, D. A. Nicotinamide and PNC1 govern lifespan extension by calorie restriction in Saccharomyces cerevisiae. Nature 423, 181–185 (2003).
Moroz, N. et al. Dietary restriction involves NAD+-dependent mechanisms and a shift toward oxidative metabolism. Aging Cell 13, 1075–1085 (2014).
Covarrubias, A. J. et al. Senescent cells promote tissue NAD(+) decline during ageing via the activation of CD38(+) macrophages. Nat. Metab. 2, 1265–1283 (2020).
Yoshida, M. et al. Extracellular vesicle-contained eNAMPT delays aging and extends lifespan in mice. Cell Metab. 30, 329–342.e5 (2019).
Ramsey, K. M. et al. Circadian clock feedback cycle through NAMPT-mediated NAD+ biosynthesis. Science 324, 651–654 (2009).
Yoshino, M. et al. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science 372, 1224–1229 (2021).
Levine, D. C. et al. NAD(+) controls circadian reprogramming through PER2 nuclear translocation to counter aging. Mol. Cell 78, 835–849.e7 (2020).
Hou, Y. et al. NAD(+) supplementation reduces neuroinflammation and cell senescence in a transgenic mouse model of Alzheimer's disease via cGAS-STING. Proc. Natl Acad. Sci. USA 118, e2011226118 (2021).
Killilea, D. W. & Killilea, A. N. Mineral requirements for mitochondrial function: a connection to redox balance and cellular differentiation. Free Radic. Biol. Med. 182, 182–191 (2022).
Wawrzyniak, N. & Suliburska, J. Nutritional and health factors affecting the bioavailability of calcium: a narrative review. Nutr. Rev. 79, 1307–1320 (2021).
Dominguez, L., Veronese, N. & Barbagallo, M. Magnesium and hypertension in old age. Nutrients 13, 139 (2020).
Mohammadifard, N. et al. Trace minerals intake: risks and benefits for cardiovascular health. Crit. Rev. Food Sci. Nutr. 59, 1334–1346 (2019).
Tsvetkov, P. et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science 375, 1254–1261 (2022).
Donadio, J. L. S., Duarte, G. B. S., Borel, P., Cozzolino, S. M. F. & Rogero, M. M. The influence of nutrigenetics on biomarkers of selenium nutritional status. Nutr. Rev. 79, 1259–1273 (2021).
Custers, E., Kiliaan, E. M. & Amanda, J. Dietary lipids from body to brain. Prog. Lipid Res. 85, 101144 (2022).
Katewa, S. D. et al. Intramyocellular fatty-acid metabolism plays a critical role in mediating responses to dietary restriction in Drosophila melanogaster. Cell Metab. 16, 97–103 (2012).
Bruss, M. D., Khambatta, C. F., Ruby, M. A., Aggarwal, I. & Hellerstein, M. K. Calorie restriction increases fatty acid synthesis and whole body fat oxidation rates. Am. J. Physiol. Endocrinol. Metab. 298, E108–E116 (2010).
Picard, F. et al. Sirt1 promotes fat mobilization in white adipocytes by repressing PPAR-gamma. Nature 429, 771–776 (2004).
Indraccolo, S. et al. Phosphorylated acetyl-CoA carboxylase is associated with clinical benefit with regorafenib in relapsed glioblastoma: REGOMA trial biomarker analysis. Clin. Cancer Res. 26, 4478–4484 (2020).
Marcellino, B. K., Ekasumara, N. & Mobbs, C. V. Dietary restriction and glycolytic inhibition reduce proteotoxicity and extend lifespan via NHR-49. Curr. Neurobiol. 9, 1–7 (2018).
Stekovic, S. et al. Alternate day fasting improves physiological and molecular markers of aging in healthy, non-obese humans. Cell Metab. 30, 462–476.e6 (2019).
Kolb, H. et al. Ketone bodies: from enemy to friend and guardian angel. BMC Med. 19, 313 (2021).
Chamoli, M. et al. Polyunsaturated fatty acids and p38-MAPK link metabolic reprogramming to cytoprotective gene expression during dietary restriction. Nat. Commun. 11, 4865 (2020).
Le Couteur, D. G. et al. Branched chain amino acids, aging and age-related health. Ageing Res. Rev. 64, 101198 (2020).
Matai, L. et al. Dietary restriction improves proteostasis and increases life span through endoplasmic reticulum hormesis. Proc. Natl Acad. Sci. USA 116, 17383–17392 (2019).
Ferro-Novick, S., Reggiori, F. & Brodsky, J. L. ER-phagy, ER homeostasis, and ER quality control: implications for disease. Trends Biochem. Sci. 46, 630–639 (2021).
Levine, B. & Kroemer, G. Biological functions of autophagy genes: a disease perspective. Cell 176, 11–42 (2019).
Ulgherait, M. et al. Circadian autophagy drives iTRF-mediated longevity. Nature 598, 353–358 (2021).
Hansen, M., Rubinsztein, D. C. & Walker, D. W. Autophagy as a promoter of longevity: insights from model organisms. Nat. Rev. Mol. Cell Biol. 19, 579–593 (2018).
Jung, Y. et al. MON-2, a Golgi protein, mediates autophagy-dependent longevity in Caenorhabditis elegans. Sci. Adv. 7, eabj8156 (2021).
Pietrocola, F. et al. Caloric restriction mimetics enhance anticancer immunosurveillance. Cancer Cell 30, 147–160 (2016).
Saito, T. et al. Autophagy regulates lipid metabolism through selective turnover of NCoR1. Nat. Commun. 10, 1567 (2019).
Jouandin, P. et al. Lysosomal cystine mobilization shapes the response of TORC1 and tissue growth to fasting. Science 375, eabc4203 (2022).
Green, C. L., Lamming, D. W. & Fontana, L. Molecular mechanisms of dietary restriction promoting health and longevity. Nat. Rev. Mol. Cell Biol. 23, 56–73 (2022).
Sutter, B. M., Wu, X., Laxman, S. & Tu, B. P. Methionine inhibits autophagy and promotes growth by inducing the SAM-responsive methylation of PP2A. Cell 154, 403–415 (2013).
White, E., Lattime, E. C. & Guo, J. Y. Autophagy regulates stress responses, metabolism, and anticancer immunity. Trends Cancer 7, 778–789 (2021).
Ouyang, Y., Wu, Q., Li, J., Sun, S. & Sun, S. S-adenosylmethionine: a metabolite critical to the regulation of autophagy. Cell Prolif. 53, e12891 (2020).
Falcón, P., Escandón, M., Brito, Á. & Matus, S. Nutrient sensing and redox balance: GCN2 as a new integrator in aging. Oxid. Med. Cell Longev. 2019, 5730532 (2019).
Laeger, T. et al. Metabolic responses to dietary protein restriction require an increase in FGF21 that is delayed by the absence of GCN2. Cell Rep. 16, 707–716 (2016).
Amorim, J. A. et al. Mitochondrial and metabolic dysfunction in ageing and age-related diseases. Nat. Rev. Endocrinol. 18, 243–258 (2022).
Spadaro, O. et al. Caloric restriction in humans reveals immunometabolic regulators of health span. Science 375, 671–677 (2022).
Qiu, X., Brown, K., Hirschey, M. D., Verdin, E. & Chen, D. Calorie restriction reduces oxidative stress by SIRT3-mediated SOD2 activation. Cell Metab. 12, 662–667 (2010).
López-Lluch, G. et al. Calorie restriction induces mitochondrial biogenesis and bioenergetic efficiency. Proc. Natl Acad. Sci. USA 103, 1768–1773 (2006).
Lanza, I. R. et al. Chronic caloric restriction preserves mitochondrial function in senescence without increasing mitochondrial biogenesis. Cell Metab. 16, 777–788 (2012).
Song, J., Herrmann, J. M. & Becker, T. Quality control of the mitochondrial proteome. Nat. Rev. Mol. Cell Biol. 22, 54–70 (2021).
Ruan, L. et al. Mitochondria-associated proteostasis. Annu. Rev. Biophys. 49, 41–67 (2020).
Ozkurede, U. & Miller, R. A. Improved mitochondrial stress response in long-lived Snell dwarf mice. Aging Cell 18, e13030 (2019).
Durieux, J., Wolff, S. & Dillin, A. The cell-non-autonomous nature of electron transport chain-mediated longevity. Cell 144, 79–91 (2011).
Lam, J. & McKeague, M. Dietary modulation of mitochondrial DNA damage: implications in aging and associated diseases. J. Nutr. Biochem 63, 1–10 (2019).
Mehrabani, S., Bagherniya, M., Askari, G., Read, M. I. & Sahebkar, A. The effect of fasting or calorie restriction on mitophagy induction: a literature review. J. Cachexia Sarcopenia Muscle 11, 1447–1458 (2020).
Gyanwali, B. et al. Alpha-Ketoglutarate dietary supplementation to improve health in humans. Trends Endocrinol. Metab. 33, 136–146 (2022).
Campbell, S. L. & Wellen, K. E. Metabolic signaling to the nucleus in cancer. Mol. Cell 71, 398–408 (2018).
Diehl, K. L. & Muir, T. W. Chromatin as a key consumer in the metabolite economy. Nat. Chem. Biol. 16, 620–629 (2020).
Zheng, Y. & Cantley, L. C. Toward a better understanding of folate metabolism in health and disease. J. Exp. Med. 216, 253–266 (2019).
Kottakis, F. et al. LKB1 loss links serine metabolism to DNA methylation and tumorigenesis. Nature 539, 390–395 (2016).
Wu, Q., Chen, X., Li, J. & Sun, S. Serine and metabolism regulation: a novel mechanism in antitumor immunity and senescence. Aging Dis. 11, 1640–1653 (2020).
Morris, J. P. T. et al. α-Ketoglutarate links p53 to cell fate during tumour suppression. Nature 573, 595–599 (2019).
Parkhitko, A. A., Jouandin, P., Mohr, S. E. & Perrimon, N. Methionine metabolism and methyltransferases in the regulation of aging and lifespan extension across species. Aging Cell 18, e13034 (2019).
Mota-Martorell, N. et al. Methionine transsulfuration pathway is upregulated in long-lived humans. Free Radic. Biol. Med. 162, 38–52 (2021).
Sun, L., Zhang, H. & Gao, P. Metabolic reprogramming and epigenetic modifications on the path to cancer. Protein Cell. https://doi.org/10.1007/s13238-021-00846-7 (2021).
Lim, C. Y. et al. SAMS-1 coordinates HLH-30/TFEB and PHA-4/FOXA activities through histone methylation to mediate dietary restriction-induced autophagy and longevity. Autophagy 3, 1–17 (2022).
Wang, Y. et al. Alpha-ketoglutarate ameliorates age-related osteoporosis via regulating histone methylations. Nat. Commun. 11, 5596 (2020).
Sen, P. et al. Histone acetyltransferase p300 induces de novo super-enhancers to drive cellular senescence. Mol. Cell 73, 684–698 e8 (2019).
Morselli, E. et al. Caloric restriction and resveratrol promote longevity through the Sirtuin-1-dependent induction of autophagy. Cell Death Dis. 1, e10 (2010).
Pietrocola, F. et al. Aspirin recapitulates features of caloric restriction. Cell Rep. 22, 2395–2407 (2018).
Madeo, F., Eisenberg, T., Pietrocola, F. & Kroemer, G. Spermidine in health and disease. Science 359, eaan2788 (2018).
Smestad, J., Erber, L., Chen, Y. & Maher, L. J. 3rd Chromatin succinylation correlates with active gene expression and is perturbed by defective TCA cycle metabolism. iScience 2, 63–75 (2018).
Li, L. et al. SIRT7 is a histone desuccinylase that functionally links to chromatin compaction and genome stability. Nat. Commun. 7, 12235 (2016).
Goodall, G. J. & Wickramasinghe, V. O. RNA in cancer. Nat. Rev. Cancer 21, 22–36 (2021).
Victoria, B., Nunez Lopez, Y. O. & Masternak, M. M. MicroRNAs and the metabolic hallmarks of aging. Mol. Cell Endocrinol. 455, 131–147 (2017).
Cardoso, A. L. et al. Towards frailty biomarkers: candidates from genes and pathways regulated in aging and age-related diseases. Ageing Res. Rev. 47, 214–277 (2018).
Caravia, X. M., Roiz-Valle, D., Morán-Álvarez, A. & López-Otín, C. Functional relevance of miRNAs in premature ageing. Mech. Ageing Dev. 168, 10–19 (2017).
Victoria, B. et al. Circulating microRNA signature of genotype-by-age interactions in the long-lived Ames dwarf mouse. Aging Cell 14, 1055–1066 (2015).
Csiszar, A. et al. Caloric restriction confers persistent anti-oxidative, pro-angiogenic, and anti-inflammatory effects and promotes anti-aging miRNA expression profile in cerebromicrovascular endothelial cells of aged rats. Am. J. Physiol. Heart Circ. Physiol. 307, H292–H306 (2014).
Mori, M. A., Ludwig, R. G., Garcia-Martin, R., Brandão, B. B. & Kahn, C. R. Extracellular miRNAs: from biomarkers to mediators of physiology and disease. Cell Metab. 30, 656–673 (2019).
Kumar, S., Vijayan, M., Bhatti, J. S. & Reddy, P. H. MicroRNAs as peripheral biomarkers in aging and age-related diseases. Prog. Mol. Biol. Transl. Sci. 146, 47–94 (2017).
Jia, M. et al. Cross-kingdom regulation by dietary plant miRNAs: an evidence-based review with recent updates. Food Funct. 12, 9549–9562 (2021).
Wu, Q. et al. Isobacachalcone induces autophagy and improves the outcome of immunogenic chemotherapy. Cell Death Dis. 11, 1015 (2020).
Wu, Q. et al. IGF1 receptor inhibition amplifies the effects of cancer drugs by autophagy and immune-dependent mechanisms. J. Immunother. Cancer 9, e002722 (2021).
Ouyang, Y. et al. Chalcone derivatives: role in anticancer therapy. Biomolecules 11, 894 (2021).
Chen, G. et al. 3,4-Dimethoxychalcone induces autophagy through activation of the transcription factors TFE3 and TFEB. EMBO Mol. Med 11, e10469 (2019).
Carmona-Gutierrez, D. et al. The flavonoid 4,4'-dimethoxychalcone promotes autophagy-dependent longevity across species. Nat. Commun. 10, 651 (2019).
Reinke, H. & Asher, G. Crosstalk between metabolism and circadian clocks. Nat. Rev. Mol. Cell Biol. 20, 227–241 (2019).
Rijo-Ferreira, F. & Takahashi, J. S. Genomics of circadian rhythms in health and disease. Genome Med 11, 82 (2019).
Frazier, K. et al. High-fat diet disrupts REG3γ and gut microbial rhythms promoting metabolic dysfunction. Cell Host Microbe 30, 809–823.e6 (2022).
Acosta-Rodríguez, V. et al. Circadian alignment of early onset caloric restriction promotes longevity in male C57BL/6J mice. Science 376, 1192–1202 (2022).
Katewa, S. D. et al. Peripheral circadian clocks mediate dietary restriction-dependent changes in lifespan and fat metabolism in Drosophila. Cell Metab. 23, 143–154 (2016).
Xie, Z. et al. Randomized controlled trial for time-restricted eating in healthy volunteers without obesity. Nat. Commun. 13, 1003 (2022).
Levine, D. C. et al. NADH inhibition of SIRT1 links energy state to transcription during time-restricted feeding. Nat. Metab. 3, 1621–1632 (2021).
Patke, A., Young, M. W. & Axelrod, S. Molecular mechanisms and physiological importance of circadian rhythms. Nat. Rev. Mol. Cell Biol. 21, 67–84 (2020).
Chellappa, S. L. et al. Daytime eating prevents internal circadian misalignment and glucose intolerance in night work. Sci. Adv. 7, eabg9910 (2021).
Ulgherait, M. et al. Circadian regulation of mitochondrial uncoupling and lifespan. Nat. Commun. 11, 1927 (2020).
Tacad, D. K. M. et al. Satiety associated with calorie restriction and time-restricted feeding: peripheral hormones. Adv. Nutr. 13, 792–820 (2022).
López-Otín, C. & Kroemer, G. Hallmarks of Health. Cell 184, 33–63 (2021).
Cignarella, F. et al. Intermittent fasting confers protection in CNS autoimmunity by altering the gut microbiota. Cell Metab. 27, 1222–1235 e6 (2018).
Bravo-San Pedro, J. M. et al. Acyl-CoA-binding protein is a lipogenic factor that triggers food intake and obesity. Cell Metab. 30, 754–767.e9 (2019).
Mishra, I. et al. Protein tyrosine phosphatase receptor δ serves as the orexigenic asprosin receptor. Cell Metab. 34, 549–563.e8 (2022).
Clancy, D. J. et al. Extension of life-span by loss of CHICO, a Drosophila insulin receptor substrate protein. Science 292, 104–106 (2001).
Tatar, M. et al. A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science 292, 107–110 (2001).
Caffa, I. et al. Fasting-mimicking diet and hormone therapy induce breast cancer regression. Nature 583, 620–624 (2020).
Pollak, M. The insulin and insulin-like growth factor receptor family in neoplasia: an update. Nat. Rev. Cancer 12, 159–169 (2012).
Deng, M. et al. Lepr(+) mesenchymal cells sense diet to modulate intestinal stem/progenitor cells via Leptin-Igf1 axis. Cell Res. https://doi.org/10.1038/s41422-022-00643-9 (2022).
Geng, L., Lam, K. S. L. & Xu, A. The therapeutic potential of FGF21 in metabolic diseases: from bench to clinic. Nat. Rev. Endocrinol. 16, 654–667 (2020).
Yu, D. et al. The adverse metabolic effects of branched-chain amino acids are mediated by isoleucine and valine. Cell Metab. 33, 905–922.e6 (2021).
Wei, S. et al. Intermittent protein restriction protects islet β cells and improves glucose homeostasis in diabetic mice. Sci. Bull. 67, 733–747 (2022).
Fontana, L. et al. Decreased consumption of branched-chain amino acids improves metabolic health. Cell Rep. 16, 520–530 (2016).
Hill, C. M. et al. FGF21 is required for protein restriction to extend lifespan and improve metabolic health in male mice. Nat. Commun. 13, 1897 (2022).
Zhang, Y. et al. The starvation hormone, fibroblast growth factor-21, extends lifespan in mice. Elife 1, e00065 (2012).
Collins, N. & Belkaid, Y. Control of immunity via nutritional interventions. Immunity 55, 210–223 (2022).
Ansaldo, E., Farley, T. K. & Belkaid, Y. Control of immunity by the microbiota. Annu Rev. Immunol. 39, 449–479 (2021).
Wu, Q. et al. YAP/TAZ-mediated activation of serine metabolism and methylation regulation is critical for LKB1-deficient breast cancer progression. Biosci. Rep. 37, BSR20171072 (2017).
Weisberg, S. P., Ural, B. B. & Farber, D. L. Tissue-specific immunity for a changing world. Cell 184, 1517–1529 (2021).
Wu, Q., Yu, X., Li, J., Sun, S. & Tu, Y. Metabolic regulation in the immune response to cancer. Cancer Commun. (Lond.) 41, 661–694 (2021).
Geltink, R. I. K., Kyle, R. L. & Pearce, E. L. Unraveling the complex interplay between T cell metabolism and function. Annu Rev. Immunol. 36, 461–488 (2018).
Ma, E. H. et al. Metabolic profiling using stable isotope tracing reveals distinct patterns of glucose utilization by physiologically activated CD8(+) T cells. Immunity 51, 856–870.e5 (2019).
Palma, C. et al. Caloric restriction promotes immunometabolic reprogramming leading to protection from tuberculosis. Cell Metab. 33, 300–318.e12 (2021).
Collins, N. et al. The bone marrow protects and optimizes immunological memory during dietary restriction. Cell 178, 1088–1101.e15 (2019).
Jordan, S. et al. Dietary intake regulates the circulating inflammatory monocyte pool. Cell 178, 1102–1114.e17 (2019).
Wu, Z. et al. Dietary restriction extends lifespan through metabolic regulation of innate immunity. Cell Metab. 29, 1192–1205.e8 (2019).
Ma, S. et al. Caloric restriction reprograms the single-cell transcriptional landscape of Rattus norvegicus aging. Cell 180, 984–1001.e22 (2020).
Xiao, Z. X. et al. High salt diet accelerates the progression of murine lupus through dendritic cells via the p38 MAPK and STAT1 signaling pathways. Signal Transduct. Target Ther. 5, 34 (2020).
Ruck, T. et al. K(2P)18.1 translates T cell receptor signals into thymic regulatory T cell development. Cell Res. 32, 72–88 (2022).
Yao, Y. et al. Selenium-GPX4 axis protects follicular helper T cells from ferroptosis. Nat. Immunol. 22, 1127–1139 (2021).
Iovino, L. et al. Activation of the Zinc-sensing receptor GPR39 promotes T cell reconstitution after hematopoietic cell transplant in mice. Blood. https://doi.org/10.1182/blood.2021013950 (2022).
Zhang, H. et al. Ketogenesis-generated β-hydroxybutyrate is an epigenetic regulator of CD8(+) T-cell memory development. Nat. Cell Biol. 22, 18–25 (2020).
Cucchi, D. et al. Omega-3 polyunsaturated fatty acids impinge on CD4+ T cell motility and adipose tissue distribution via direct and lipid mediator-dependent effects. Cardiovasc. Res. 116, 1006–1020 (2020).
Zhang, H. et al. Polyamines control eIF5A hypusination, TFEB translation, and autophagy to reverse B cell senescence. Mol. Cell 76, 110–125 e9 (2019).
Beyaz, S. et al. Dietary suppression of MHC class II expression in intestinal epithelial cells enhances intestinal tumorigenesis. Cell Stem. Cell 28, 1922–1935.e5 (2021).
Wilck, N. et al. Salt-responsive gut commensal modulates T(H)17 axis and disease. Nature 551, 585–589 (2017).
Rizvi, Z. A. et al. High-salt diet mediates interplay between NK cells and gut microbiota to induce potent tumor immunity. Sci. Adv. 7, eabg5016 (2021).
Sepahi, A., Liu, Q., Friesen, L. & Kim, C. H. Dietary fiber metabolites regulate innate lymphoid cell responses. Mucosal. Immunol. 14, 317–330 (2021).
Alexander, M. & Turnbaugh, P. J. Deconstructing mechanisms of diet-microbiome-immune interactions. Immunity 53, 264–276 (2020).
von Schwartzenberg, R. J. et al. Caloric restriction disrupts the microbiota and colonization resistance. Nature 595, 272–277 (2021).
Fabbiano, S. et al. Functional gut microbiota remodeling contributes to the caloric restriction-induced metabolic improvements. Cell Metab. 28, 907–921.e7 (2018).
Li, M. et al. Gut microbiota-bile acid crosstalk contributes to the rebound weight gain after calorie restriction in mice. Nat. Commun. 13, 2060 (2022).
Bauer, P. V. et al. Lactobacillus gasseri in the upper small intestine impacts an ACSL3-dependent fatty acid-sensing pathway regulating whole-body glucose homeostasis. Cell Metab. 27, 572–587.e6 (2018).
Maifeld, A. et al. Fasting alters the gut microbiome reducing blood pressure and body weight in metabolic syndrome patients. Nat. Commun. 12, 1970 (2021).
Sbierski-Kind, J. et al. Effects of caloric restriction on the gut microbiome are linked with immune senescence. Microbiome 10, 57 (2022).
Jian, C. et al. Gut microbiota predicts body fat change following a low-energy diet: a PREVIEW intervention study. Genome Med 14, 54 (2022).
Johnson, A. J. et al. Daily sampling reveals personalized diet-microbiome associations in humans. Cell Host Microbe 25, 789–802.e5 (2019).
Ang, Q. Y. et al. Ketogenic diets alter the gut microbiome resulting in decreased intestinal Th17. Cells Cell 181, 1263–1275.e16 (2020).
Wastyk, H. C. et al. Gut-microbiota-targeted diets modulate human immune status. Cell 184, 4137–4153.e14 (2021).
Liu, H. et al. Ecological dynamics of the gut microbiome in response to dietary fiber. ISME J. https://doi.org/10.1038/s41396-022-01253-4 (2022).
Lancaster, S. M. et al. Global, distinctive, and personal changes in molecular and microbial profiles by specific fibers in humans. Cell Host Microbe 30, 848–862.e7 (2022).
Deehan, E. C. et al. Precision microbiome modulation with discrete dietary fiber structures directs short-chain fatty acid production. Cell Host Microbe 27, 389–404.e6 (2020).
Desai, M. S. et al. A dietary fiber-deprived gut microbiota degrades the colonic mucus barrier and enhances pathogen susceptibility. Cell 167, 1339–1353.e21 (2016).
Ke, W. et al. Dietary serine-microbiota interaction enhances chemotherapeutic toxicity without altering drug conversion. Nat. Commun. 11, 2587 (2020).
Zheng, D., Ratiner, K. & Elinav, E. Circadian influences of diet on the microbiome and immunity. Trends Immunol. 41, 512–530 (2020).
Choi, H., Rao, M. C. & Chang, E. B. Gut microbiota as a transducer of dietary cues to regulate host circadian rhythms and metabolism. Nat. Rev. Gastroenterol. Hepatol. 18, 679–689 (2021).
Tuganbaev, T. et al. Diet diurnally regulates small intestinal microbiome-epithelial-immune homeostasis and enteritis. Cell 182, 1441–1459.e21 (2020).
Wiley, C. D. & Campisi, J. The metabolic roots of senescence: mechanisms and opportunities for intervention. Nat. Metab. 3, 1290–1301 (2021).
Di Micco, R., Krizhanovsky, V., Baker, D. & d'Adda di Fagagna, F. Cellular senescence in ageing: from mechanisms to therapeutic opportunities. Nat. Rev. Mol. Cell Biol. 22, 75–95 (2021).
Fontana, L. et al. The effects of graded caloric restriction: XII. Comparison of mouse to human impact on cellular senescence in the colon. Aging Cell 17, e12746 (2018).
Roberts, M. N. et al. A ketogenic diet extends longevity and healthspan in adult mice. Cell Metab. 26, 539–546.e5 (2017).
Parkhitko, A. A. et al. A genetic model of methionine restriction extends Drosophila health- and lifespan. Proc. Natl Acad. Sci. USA 118, e2110387118 (2021).
Han, Y. M. et al. β-Hydroxybutyrate prevents vascular senescence through hnRNP A1-mediated upregulation of Oct4. Mol. Cell 71, 1064–1078.e5 (2018).
Gao, X. et al. Dietary methionine influences therapy in mouse cancer models and alters human metabolism. Nature 572, 397–401 (2019).
Koyama, T. et al. A nutrient-responsive hormonal circuit mediates an inter-tissue program regulating metabolic homeostasis in adult Drosophila. Nat. Commun. 12, 5178 (2021).
Buchanan, K. L. et al. The preference for sugar over sweetener depends on a gut sensor cell. Nat. Neurosci. 25, 191–200 (2022).
Lin, H. H. et al. A nutrient-specific gut hormone arbitrates between courtship and feeding. Nature 602, 632–638 (2022).
Witte, A. V., Fobker, M., Gellner, R., Knecht, S. & Flöel, A. Caloric restriction improves memory in elderly humans. Proc. Natl Acad. Sci. USA 106, 1255–1260 (2009).
Bondolfi, L., Ermini, F., Long, J. M., Ingram, D. K. & Jucker, M. Impact of age and caloric restriction on neurogenesis in the dentate gyrus of C57BL/6 mice. Neurobiol. Aging 25, 333–340 (2004).
Luo, J., Daniels, S. B., Lennington, J. B., Notti, R. Q. & Conover, J. C. The aging neurogenic subventricular zone. Aging Cell 5, 139–152 (2006).
Lee, J., Seroogy, K. B. & Mattson, M. P. Dietary restriction enhances neurotrophin expression and neurogenesis in the hippocampus of adult mice. J. Neurochem 80, 539–547 (2002).
Encinas, J. M. et al. Division-coupled astrocytic differentiation and age-related depletion of neural stem cells in the adult hippocampus. Cell Stem Cell 8, 566–579 (2011).
Liu, X. et al. Calorie restriction and calorie dilution have different impacts on body fat, metabolism, behavior, and hypothalamic gene expression. Cell Rep. 39, 110835 (2022).
Wu, Y. et al. Very-low-protein diets lead to reduced food intake and weight loss, linked to inhibition of hypothalamic mTOR signaling, in mice. Cell Metab. 33, 888–904.e6 (2021).
Fortier, M. et al. A ketogenic drink improves cognition in mild cognitive impairment: Results of a 6-month RCT. Alzheimers Dement 17, 543–552 (2021).
Ren, B. et al. Methionine restriction alleviates age-associated cognitive decline via fibroblast growth factor 21. Redox Biol. 41, 101940 (2021).
Viskaitis, P. et al. Ingested non-essential amino acids recruit brain orexin cells to suppress eating in mice. Curr. Biol. 32, 1812–1821.e4 (2022).
Larabee, C. M., Neely, O. C. & Domingos, A. I. Obesity: a neuroimmunometabolic perspective. Nat. Rev. Endocrinol. 16, 30–43 (2020).
Simonson, M., Boirie, Y. & Guillet, C. Protein, amino acids and obesity treatment. Rev. Endocr. Metab. Disord. 21, 341–353 (2020).
Choi, B. S. et al. Feeding diversified protein sources exacerbates hepatic insulin resistance via increased gut microbial branched-chain fatty acids and mTORC1 signaling in obese mice. Nat. Commun. 12, 3377 (2021).
Liu, K. H. et al. Microbial metabolite delta-valerobetaine is a diet-dependent obesogen. Nat. Metab. 3, 1694–1705 (2021).
Fabbiano, S. et al. Caloric restriction leads to browning of white adipose tissue through type 2 immune signaling. Cell Metab. 24, 434–446 (2016).
Miller, K. N. et al. Aging and caloric restriction impact adipose tissue, adiponectin, and circulating lipids. Aging Cell 16, 497–507 (2017).
Dorling, J. L. et al. Effect of 2 years of calorie restriction on liver biomarkers: results from the CALERIE phase 2 randomized controlled trial. Eur. J. Nutr. 60, 1633–1643 (2021).
Kim, K. H. et al. Intermittent fasting promotes adipose thermogenesis and metabolic homeostasis via VEGF-mediated alternative activation of macrophage. Cell Res. 27, 1309–1326 (2017).
Brandhorst, S. et al. A periodic diet that mimics fasting promotes multi-system regeneration, enhanced cognitive performance, and healthspan. Cell Metab. 22, 86–99 (2015).
Pak, H. H. et al. Fasting drives the metabolic, molecular and geroprotective effects of a calorie-restricted diet in mice. Nat. Metab. 3, 1327–1341 (2021).
Cienfuegos, S. et al. Effects of 4- and 6-h time-restricted feeding on weight and cardiometabolic health: a randomized controlled trial in adults with obesity. Cell Metab. 32, 366–378.e3 (2020).
Wilkinson, M. J. et al. Ten-hour time-restricted eating reduces weight, blood pressure, and atherogenic lipids in patients with metabolic syndrome. Cell Metab. 31, 92–104.e5 (2020).
Harney, D. J. et al. Proteomics analysis of adipose depots after intermittent fasting reveals visceral fat preservation mechanisms. Cell Rep. 34, 108804 (2021).
Li, G. et al. Intermittent fasting promotes white adipose browning and decreases obesity by shaping the gut microbiota. Cell Metab. 26, 672–685.e4 (2017).
Liu, D. et al. Calorie restriction with or without time-restricted eating in weight loss. N. Engl. J. Med. 386, 1495–1504 (2022).
Wang, F. et al. Plasma metabolite profiles related to plant-based diets and the risk of type 2 diabetes. Diabetologia 65, 1119–1132 (2022).
Meslier, V. et al. Mediterranean diet intervention in overweight and obese subjects lowers plasma cholesterol and causes changes in the gut microbiome and metabolome independently of energy intake. Gut 69, 1258–1268 (2020).
Ma, Q. X. et al. BCAA-BCKA axis regulates WAT browning through acetylation of PRDM16. Nat. Metab. 4, 106–122 (2022).
Mocanu, V. et al. Fecal microbial transplantation and fiber supplementation in patients with severe obesity and metabolic syndrome: a randomized double-blind, placebo-controlled phase 2 trial. Nat. Med. 27, 1272–1279 (2021).
Daniel, N. et al. Gut microbiota and fermentation-derived branched chain hydroxy acids mediate health benefits of yogurt consumption in obese mice. Nat. Commun. 13, 1343 (2022).
Rodriguez, J. et al. Physical activity enhances the improvement of body mass index and metabolism by inulin: a multicenter randomized placebo-controlled trial performed in obese individuals. BMC Med. 20, 110 (2022).
Katsumura, S. et al. Deadenylase-dependent mRNA decay of GDF15 and FGF21 orchestrates food intake and energy expenditure. Cell Metab. 34, 564–580.e8 (2022).
Sciarretta, S. et al. Caloric restriction mimetics for the treatment of cardiovascular diseases. Cardiovasc Res. 117, 1434–1449 (2021).
Lopaschuk, G. D., Karwi, Q. G., Tian, R., Wende, A. R. & Abel, E. D. Cardiac energy metabolism in heart failure. Circ. Res. 128, 1487–1513 (2021).
Xu, Y. et al. Branched-chain amino acid catabolism promotes thrombosis risk by enhancing tropomodulin-3 propionylation in platelets. Circulation 142, 49–64 (2020).
Redman, L. M. et al. Metabolic slowing and reduced oxidative damage with sustained caloric restriction support the rate of living and oxidative damage theories of aging. Cell Metab. 27, 805–815 e4 (2018).
Sloan, C. et al. Central leptin signaling is required to normalize myocardial fatty acid oxidation rates in caloric-restricted ob/ob mice. Diabetes 60, 1424–1434 (2011).
An, H. S. et al. Caloric restriction reverses left ventricular hypertrophy through the regulation of cardiac iron homeostasis in impaired leptin signaling mice. Sci. Rep. 10, 7176 (2020).
Bhanpuri, N. H. et al. Cardiovascular disease risk factor responses to a type 2 diabetes care model including nutritional ketosis induced by sustained carbohydrate restriction at 1 year: an open label, non-randomized, controlled study. Cardiovasc Diabetol. 17, 56 (2018).
Wei, M. et al. Fasting-mimicking diet and markers/risk factors for aging, diabetes, cancer, and cardiovascular disease. Sci. Transl. Med. 9, eaai8700 (2017).
Mishra, A. et al. Fasting-mimicking diet prevents high-fat diet effect on cardiometabolic risk and lifespan. Nat. Metab. 3, 1342–1356 (2021).
Shi, H. et al. Restructuring the gut microbiota by intermittent fasting lowers blood pressure. Circ. Res. 128, 1240–1254 (2021).
Marques, F. Z. et al. High-fiber diet and acetate supplementation change the gut microbiota and prevent the development of hypertension and heart failure in hypertensive mice. Circulation 135, 964–977 (2017).
Weis, E. M. et al. Ketone body oxidation increases cardiac endothelial cell proliferation. EMBO Mol. Med. 14, e14753 (2022).
Rinott, E. et al. The effects of the Green-Mediterranean diet on cardiometabolic health are linked to gut microbiome modifications: a randomized controlled trial. Genome Med. 14, 29 (2022).
Wang, D. D. et al. The gut microbiome modulates the protective association between a Mediterranean diet and cardiometabolic disease risk. Nat. Med. 27, 333–343 (2021).
Delgado-Lista, J. et al. Long-term secondary prevention of cardiovascular disease with a Mediterranean diet and a low-fat diet (CORDIOPREV): a randomised controlled trial. Lancet 399, 1876–1885 (2022).
Jimenez-Torres, J. et al. Mediterranean diet reduces atherosclerosis progression in coronary heart disease: an analysis of the CORDIOPREV randomized controlled trial. Stroke 52, 3440–3449 (2021).
Zaramela, L. S. et al. Gut bacteria responding to dietary change encode sialidases that exhibit preference for red meat-associated carbohydrates. Nat. Microbiol. 4, 2082–2089 (2019).
Schwärzler, J. et al. PUFA-induced metabolic enteritis as a fuel for Crohn's disease. Gastroenterology 162, 1690–1704 (2022).
Akagi, K. et al. Dietary restriction improves intestinal cellular fitness to enhance gut barrier function and lifespan in D. melanogaster. PLoS Genet. 14, e1007777 (2018).
Luis, N. M. et al. Intestinal IRE1 is required for increased triglyceride metabolism and longer lifespan under dietary restriction. Cell Rep. 17, 1207–1216 (2016).
Gelino, S. et al. Intestinal autophagy improves healthspan and longevity in C. elegans during dietary restriction. PLoS Genet. 12, e1006135 (2016).
Prince, A. C. et al. Fermentable carbohydrate restriction (low FODMAP diet) in clinical practice improves functional gastrointestinal symptoms in patients with inflammatory Bowel disease. Inflamm. Bowel Dis. 22, 1129–1136 (2016).
Fritsch, J. et al. Low-fat, high-fiber diet reduces markers of inflammation and dysbiosis and improves quality of life in patients with ulcerative colitis. Clin. Gastroenterol. Hepatol. 19, 1189–1199.e30 (2021).
Kong, C. et al. Ketogenic diet alleviates colitis by reduction of colonic group 3 innate lymphoid cells through altering gut microbiome. Signal Transduct. Target Ther. 6, 154 (2021).
Kitamoto, S. et al. Dietary L-serine confers a competitive fitness advantage to Enterobacteriaceae in the inflamed gut. Nat. Microbiol. 5, 116–125 (2020).
Lobel, L., Cao, Y. G., Fenn, K., Glickman, J. N. & Garrett, W. S. Diet posttranslationally modifies the mouse gut microbial proteome to modulate renal function. Science 369, 1518–1524 (2020).
Wilck, N., Balogh, A., Markó, L., Bartolomaeus, H. & Müller, D. N. The role of sodium in modulating immune cell function. Nat. Rev. Nephrol. 15, 546–558 (2019).
Lee, H. W. et al. Effect of dietary protein restriction on renal ammonia metabolism. Am. J. Physiol. Ren. Physiol. 308, F1463–F1473 (2015).
Yacoub, R. et al. Advanced glycation end products dietary restriction effects on bacterial gut microbiota in peritoneal dialysis patients; a randomized open label controlled trial. PLoS ONE 12, e0184789 (2017).
Fang, Y. et al. The ketone body β-hydroxybutyrate mitigates the senescence response of glomerular podocytes to diabetic insults. Kidney Int 100, 1037–1053 (2021).
Yamamoto, T. et al. Eicosapentaenoic acid attenuates renal lipotoxicity by restoring autophagic flux. Autophagy 17, 1700–1713 (2021).
Podadera-Herreros, A. et al. Long-term consumption of a mediterranean diet or a low-fat diet on kidney function in coronary heart disease patients: the CORDIOPREV randomized controlled trial. Clin. Nutr. 41, 552–559 (2022).
Gräff, J. et al. A dietary regimen of caloric restriction or pharmacological activation of SIRT1 to delay the onset of neurodegeneration. J. Neurosci. 33, 8951–8960 (2013).
Bendlin, B. B. et al. Effects of aging and calorie restriction on white matter in rhesus macaques. Neurobiol. Aging 32, 2319.e1–11 (2011).
Newman, J. C. et al. Ketogenic diet reduces midlife mortality and improves memory in aging mice. Cell Metab. 26, 547–557.e8 (2017).
Rojic-Becker, D., Portero-Tresserra, M., Martí-Nicolovius, M., Vale-Martínez, A. & Guillazo-Blanch, G. Caloric restriction modulates the monoaminergic and glutamatergic systems in the hippocampus, and attenuates age-dependent spatial memory decline. Neurobiol. Learn Mem. 166, 107107 (2019).
Shruthi, K. et al. Amelioration of neuronal cell death in a spontaneous obese rat model by dietary restriction through modulation of ubiquitin proteasome system. J. Nutr. Biochem. 33, 73–81 (2016).
Vermeij, W. P. et al. Restricted diet delays accelerated ageing and genomic stress in DNA-repair-deficient mice. Nature 537, 427–431 (2016).
Duan, W. et al. Dietary restriction normalizes glucose metabolism and BDNF levels, slows disease progression, and increases survival in huntingtin mutant mice. Proc. Natl Acad. Sci. USA 100, 2911–2916 (2003).
Liang, Y. et al. eIF5A hypusination, boosted by dietary spermidine, protects from premature brain aging and mitochondrial dysfunction. Cell Rep. 35, 108941 (2021).
Liu, X. et al. High-fiber diet mitigates maternal obesity-induced cognitive and social dysfunction in the offspring via gut-brain axis. Cell Metab. 33, 923–938.e6 (2021).
Fontana, L., Ghezzi, L., Cross, A. H. & Piccio, L. Effects of dietary restriction on neuroinflammation in neurodegenerative diseases. J. Exp. Med. 218, e20190086 (2021).
Zhou, Z. L. et al. Neuroprotection of fasting mimicking diet on MPTP-induced Parkinson's disease mice via gut microbiota and metabolites. Neurotherapeutics 16, 741–760 (2019).
Brakedal, B. et al. The NADPARK study: a randomized phase I trial of nicotinamide riboside supplementation in Parkinson's disease. Cell Metab. 34, 396–407.e6 (2022).
Bredesen, D. E. et al. Reversal of cognitive decline in Alzheimer's disease. Aging 8, 1250–1258 (2016).
Tomi, M., Zhao, Y., Thamotharan, S., Shin, B. C. & Devaskar, S. U. Early life nutrient restriction impairs blood-brain metabolic profile and neurobehavior predisposing to Alzheimer's disease with aging. Brain Res. 1495, 61–75 (2013).
Mutoh, T. et al. Impact of medium-chain triglycerides on gait performance and brain metabolic network in healthy older adults: a double-blind, randomized controlled study. Geroscience. https://doi.org/10.1007/s11357-022-00553-z (2022).
Ghosh, T. S. et al. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: the NU-AGE 1-year dietary intervention across five European countries. Gut 69, 1218–1228 (2020).
Moreno, C. L., Ehrlich, M. E. & Mobbs, C. V. Protection by dietary restriction in the YAC128 mouse model of Huntington's disease: Relation to genes regulating histone acetylation and HTT. Neurobiol. Dis. 85, 25–34 (2016).
Wang, H. B. et al. Time-restricted feeding improves circadian dysfunction as well as motor symptoms in the Q175 mouse model of Huntington's disease. eNeuro 5, ENEURO.0431–17.2017 (2018).
Mattison, J. A. et al. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 489, 318–321 (2012).
Ham, D. J. et al. Distinct and additive effects of calorie restriction and rapamycin in aging skeletal muscle. Nat. Commun. 13, 2025 (2022).
Gutiérrez-Casado, E. et al. The impact of aging, calorie restriction and dietary fat on autophagy markers and mitochondrial ultrastructure and dynamics in mouse skeletal muscle. J. Gerontol. A Biol. Sci. Med. Sci. 74, 760–769 (2019).
Marzetti, E. et al. Changes in IL-15 expression and death-receptor apoptotic signaling in rat gastrocnemius muscle with aging and life-long calorie restriction. Mech. Ageing Dev. 130, 272–280 (2009).
Jang, Y. C. et al. Dietary restriction attenuates age-associated muscle atrophy by lowering oxidative stress in mice even in complete absence of CuZnSOD. Aging Cell 11, 770–782 (2012).
Hepple, R. T., Qin, M., Nakamoto, H. & Goto, S. Caloric restriction optimizes the proteasome pathway with aging in rat plantaris muscle: implications for sarcopenia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 295, R1231–R1237 (2008).
van Norren, K. et al. Behavioural changes are a major contributing factor in the reduction of sarcopenia in caloric-restricted ageing mice. J. Cachexia Sarcopenia Muscle 6, 253–268 (2015).
McKiernan, S. H. et al. Caloric restriction delays aging-induced cellular phenotypes in rhesus monkey skeletal muscle. Exp. Gerontol. 46, 23–29 (2011).
Burks, T. N. & Cohn, R. D. One size may not fit all: anti-aging therapies and sarcopenia. Aging 3, 1142–1153 (2011).
Benjamin, D. I. et al. Fasting induces a highly resilient deep quiescent state in muscle stem cells via ketone body signaling. Cell Metab. 34, 902–918.e6 (2022).
Aoyama, S. et al. Distribution of dietary protein intake in daily meals influences skeletal muscle hypertrophy via the muscle clock. Cell Rep. 36, 109336 (2021).
Villareal, D. T. et al. Effect of two-year caloric restriction on bone metabolism and bone mineral density in non-obese younger adults: a randomized clinical trial. J. Bone Min. Res. 31, 40–51 (2016).
Bodnar, M., Skalicky, M., Viidik, A. & Erben, R. G. Interaction between exercise, dietary restriction and age-related bone loss in a rodent model of male senile osteoporosis. Gerontology 58, 139–149 (2012).
Cao, J. J. Caloric restriction combined with exercise is effective in reducing adiposity and mitigating bone structural deterioration in obese rats. Ann. N. Y Acad. Sci. 1433, 41–52 (2018).
Huang, T. H. & Ables, G. P. Dietary restrictions, bone density, and bone quality. Ann. N. Y Acad. Sci. 1363, 26–39 (2016).
Trivedi, A., Babic, S., Heiman, M., Gibson, W. T. & Chanoine, J. P. Ghrelin, ghrelin o-acyltransferase, and carbohydrate metabolism during pregnancy in calorie-restricted mice. Horm. Metab. Res. 49, 64–72 (2017).
Molon-Noblot, S., Laroque, P., Coleman, J. B., Hoe, C. M. & Keenan, K. P. The effects of ad libitum overfeeding and moderate and marked dietary restriction on age-related spontaneous pituitary gland pathology in Sprague-Dawley rats. Toxicol. Pathol. 31, 310–320 (2003).
Bédard, K., Bédard, J., Rocheleau, G., Ferland, G. & Gaudreau, P. Aging and diets regulate the rat anterior pituitary and hypothalamic transcriptome. Neuroendocrinology 97, 146–159 (2013).
Qiu, G. et al. Neuroprotection provided by dietary restriction in rats is further enhanced by reducing glucocortocoids. Neurobiol. Aging 33, 2398–2410 (2012).
Tabrizi, F. P. F., Farhangi, M. A., Vaezi, M. & Hemmati, S. The effects of spinach-derived thylakoid supplementation in combination with calorie restriction on anthropometric parameters and metabolic profiles in obese women with polycystic ovary syndrome: a randomized, double-blind, placebo-controlled clinical trial. Nutr. J. 19, 82 (2020).
Schulte, D. M. et al. Caloric restriction increases serum testosterone concentrations in obese male subjects by two distinct mechanisms. Horm. Metab. Res. 46, 283–286 (2014).
Minakhina, S., De Oliveira, V., Kim, S. Y., Zheng, H. & Wondisford, F. E. Thyroid hormone receptor phosphorylation regulates acute fasting-induced suppression of the hypothalamic-pituitary-thyroid axis. Proc. Natl Acad. Sci. USA 118, e2107943118 (2021).
Puyskens, A. et al. Aryl hydrocarbon receptor modulation by tuberculosis drugs impairs host defense and treatment outcomes. Cell Host Microbe 27, 238–248.e7 (2020).
McDonald, V. M. et al. Should we treat obesity in COPD? The effects of diet and resistance exercise training. Respirology 21, 875–882 (2016).
Rubini, A. et al. Effects of twenty days of the ketogenic diet on metabolic and respiratory parameters in healthy subjects. Lung 193, 939–945 (2015).
Goldberg, E. L. et al. Ketogenic diet activates protective γδ T cell responses against influenza virus infection. Sci. Immunol. 4, eaav2026 (2019).
Trompette, A. et al. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat. Med. 20, 159–166 (2014).
Trompette, A. et al. Dietary fiber confers protection against flu by shaping Ly6c(-) patrolling monocyte hematopoiesis and CD8(+) T cell metabolism. Immunity 48, 992–1005.e8 (2018).
Das, M. et al. Time-restricted feeding normalizes hyperinsulinemia to inhibit breast cancer in obese postmenopausal mouse models. Nat. Commun. 12, 565 (2021).
Ajona, D. et al. Short-term starvation reduces IGF-1 levels to sensitize lung tumors to PD-1 immune checkpoint blockade. Nat. Cancer 1, 75–85 (2020).
Ma, D. et al. Upregulation of the ALDOA/DNA-PK/p53 pathway by dietary restriction suppresses tumor growth. Oncogene 37, 1041–1048 (2018).
de Cabo, R. et al. Serum from calorie-restricted animals delays senescence and extends the lifespan of normal human fibroblasts in vitro. Aging 7, 152–166 (2015).
Wu, Q. et al. Exosomes from the tumour-adipocyte interplay stimulate beige/brown differentiation and reprogram metabolism in stromal adipocytes to promote tumour progression. J. Exp. Clin. Cancer Res. 38, 223 (2019).
Gouirand, V. et al. Ketogenic HMG-CoA lyase and its product β-hydroxybutyrate promote pancreatic cancer progression. EMBO J. 41, e110466 (2022).
Xia, S. et al. Prevention of dietary-fat-fueled ketogenesis attenuates BRAF V600E tumor growth. Cell Metab. 25, 358–373 (2017).
Voss, M. et al. Short-term fasting in glioma patients: analysis of diet diaries and metabolic parameters of the ERGO2 trial. Eur. J. Nutr. 61, 477–487 (2022).
Dierge, E. et al. Peroxidation of n-3 and n-6 polyunsaturated fatty acids in the acidic tumor environment leads to ferroptosis-mediated anticancer effects. Cell Metab. 33, 1701–1715.e5 (2021).
Lien, E. C. et al. Low glycaemic diets alter lipid metabolism to influence tumour growth. Nature 599, 302–307 (2021).
Maddocks, O. D. K. et al. Modulating the therapeutic response of tumours to dietary serine and glycine starvation. Nature 544, 372–376 (2017).
Cramer, S. L. et al. Systemic depletion of L-cyst(e)ine with cyst(e)inase increases reactive oxygen species and suppresses tumor growth. Nat. Med 23, 120–127 (2017).
Badgley, M. A. et al. Cysteine depletion induces pancreatic tumor ferroptosis in mice. Science 368, 85–89 (2020).
Leone, R. D. et al. Glutamine blockade induces divergent metabolic programs to overcome tumor immune evasion. Science 366, 1013–1021 (2019).
Vernieri, C. et al. Fasting-mimicking diet is safe and reshapes metabolism and antitumor immunity in patients with cancer. Cancer Disco. 12, 90–107 (2022).
Rubio-Patino, C. et al. Low-protein diet induces IRE1alpha-dependent anticancer immunosurveillance. Cell Metab. 27, 828–842 e7 (2018).
Eil, R. et al. Ionic immune suppression within the tumour microenvironment limits T cell effector function. Nature 537, 539–543 (2016).
Yilmaz, O. H. Dietary regulation of the origins of cancer. Sci. Transl. Med. 10, eaat8285 (2018).
de Groot, S. et al. Fasting mimicking diet as an adjunct to neoadjuvant chemotherapy for breast cancer in the multicentre randomized phase 2 DIRECT trial. Nat. Commun. 11, 3083 (2020).
Di Biase, S. et al. Fasting-mimicking diet reduces HO-1 to promote T cell-mediated tumor cytotoxicity. Cancer Cell 30, 136–146 (2016).
Yang, L. et al. Ketogenic diet and chemotherapy combine to disrupt pancreatic cancer metabolism and growth. Med 3, 119–136.e8 (2022).
Wu, J. et al. Cyst(e)ine in nutrition formulation promotes colon cancer growth and chemoresistance by activating mTORC1 and scavenging ROS. Signal Transduct. Target Ther. 6, 188 (2021).
Krstic, J. et al. Fasting improves therapeutic response in hepatocellular carcinoma through p53-dependent metabolic synergism. Sci. Adv. 8, eabh2635 (2022).
Dmitrieva-Posocco, O. et al. β-Hydroxybutyrate suppresses colorectal cancer. Nature 605, 160–165 (2022).
Ferrere, G. et al. Ketogenic diet and ketone bodies enhance the anticancer effects of PD-1 blockade. JCI Insight 6, e145207 (2021).
Hirschberger, S. et al. Very-low-carbohydrate diet enhances human T-cell immunity through immunometabolic reprogramming. EMBO Mol. Med 13, e14323 (2021).
Yang, J. J. et al. Association of dietary fiber and yogurt consumption with lung cancer risk: a pooled analysis. JAMA Oncol. 6, e194107 (2020).
Spencer, C. N. et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science 374, 1632–1640 (2021).
Dizman, N. et al. Nivolumab plus ipilimumab with or without live bacterial supplementation in metastatic renal cell carcinoma: a randomized phase 1 trial. Nat. Med. 28, 704–712 (2022).
Luu, M. et al. Microbial short-chain fatty acids modulate CD8(+) T cell responses and improve adoptive immunotherapy for cancer. Nat. Commun. 12, 4077 (2021).
Coutzac, C. et al. Systemic short chain fatty acids limit antitumor effect of CTLA-4 blockade in hosts with cancer. Nat. Commun. 11, 2168 (2020).
Wang, Y. et al. NAD(+) supplement potentiates tumor-killing function by rescuing defective TUB-mediated NAMPT transcription in tumor-infiltrated T cells. Cell Rep. 36, 109516 (2021).
Lv, H. et al. NAD(+) metabolism maintains inducible PD-L1 expression to drive tumor immune evasion. Cell Metab. 33, 110–127.e5 (2021).
Vodnala, S. K. et al. T cell stemness and dysfunction in tumors are triggered by a common mechanism. Science 363, eaau0135 (2019).
Havula, E. et al. Genetic variation of macronutrient tolerance in Drosophila melanogaster. Nat. Commun. 13, 1637 (2022).
Mitchell, S. J. et al. Daily fasting improves health and survival in male mice independent of diet composition and calories. Cell Metab. 29, 221–228.e3 (2019).
Green, C. L. et al. Sex and genetic background define the metabolic, physiologic, and molecular response to protein restriction. Cell Metab. 34, 209–226.e5 (2022).
Granger, B. Some account of the fasting-woman at titbury. Edinb. Med. Surg. J. 5, 319–326 (1809).
Rous, P. The influence of diet on transplanted and spontaneous mouse tumors. J. Exp. Med. 20, 433–451 (1914).
Kiehm, T. G., Anderson, J. W. & Ward, K. Beneficial effects of a high carbohydrate, high fiber diet on hyperglycemic diabetic men. Am. J. Clin. Nutr. 29, 895–899 (1976).
Finzi, M. Fermented milks (yogurt) in the diet therapy of chronic constipation. Minerva Med. 57, varia 979+ (1966).
Ferro-Luzzi, A. et al. Changing the Mediterranean diet: effects on blood lipids. Am. J. Clin. Nutr. 40, 1027–1037 (1984).
Stoltzner, G. Effects of life-long dietary protein restriction on mortality, growth, organ weights, blood counts, liver aldolase and kidney catalase in Balb/C mice. Growth 41, 337–348 (1977).
Wilder, R. J. The effects of ketonemia on the course of epilepsy. Mayo Clinic Proceedings 2, 307–308 (1921).
Rollo, J. Account of two cases of diabetes mellitus, with remarks. Ann. Med. 2, 85–105 (1797).
AMINO acid imbalance and supplementation. J. Am. Med. Assoc. 161, 884–885 (1956).
Steenbock, H. Vitamines and nutrition. Science 47, 119 (1918).
Salmon, W. D. The effect of mineral supplements on growth and reproduction. Science 60, 457 (1924).
Hara, K. et al. Raptor, a binding partner of target of rapamycin (TOR), mediates TOR action. Cell 110, 177–189 (2002).
Sim, A. T. & Hardie, D. G. The low activity of acetyl-CoA carboxylase in basal and glucagon-stimulated hepatocytes is due to phosphorylation by the AMP-activated protein kinase and not cyclic AMP-dependent protein kinase. FEBS Lett. 233, 294–298 (1988).
Costa, R. H., Grayson, D. R. & Darnell, J. E. Jr. Multiple hepatocyte-enriched nuclear factors function in the regulation of transthyretin and alpha 1-antitrypsin genes. Mol. Cell Biol. 9, 1415–1425 (1989).
Imai, S., Armstrong, C. M., Kaeberlein, M. & Guarente, L. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature 403, 795–800 (2000).
Honjoh, S., Yamamoto, T., Uno, M. & Nishida, E. Signalling through RHEB-1 mediates intermittent fasting-induced longevity in C. elegans. Nature 457, 726–730 (2009).
Catterson, J. H. et al. Short-term, intermittent fasting induces long-lasting gut health and TOR-independent lifespan extension. Curr. Biol. 28, 1714–1724.e4 (2018).
Kaeberlein, M. et al. Regulation of yeast replicative life span by TOR and Sch9 in response to nutrients. Science 310, 1193–1196 (2005).
Hansen, M. et al. A role for autophagy in the extension of lifespan by dietary restriction in C. elegans. PLoS Genet. 4, e24 (2008).
Weng, M., Golden, K. L. & Lee, C. Y. dFezf/Earmuff maintains the restricted developmental potential of intermediate neural progenitors in Drosophila. Dev. Cell 18, 126–135 (2010).
Bonawitz, N. D., Chatenay-Lapointe, M., Pan, Y. & Shadel, G. S. Reduced TOR signaling extends chronological life span via increased respiration and upregulation of mitochondrial gene expression. Cell Metab. 5, 265–277 (2007).
Jensen, K., McClure, C., Priest, N. K. & Hunt, J. Sex-specific effects of protein and carbohydrate intake on reproduction but not lifespan in Drosophila melanogaster. Aging Cell 14, 605–615 (2015).
Lee, K. P. et al. Lifespan and reproduction in Drosophila: New insights from nutritional geometry. Proc. Natl Acad. Sci. USA 105, 2498–2503 (2008).
Stefana, M. I. et al. Developmental diet regulates Drosophila lifespan via lipid autotoxins. Nat. Commun. 8, 1384 (2017).
Fanson, B. G., Weldon, C. W., Pérez-Staples, D., Simpson, S. J. & Taylor, P. W. Nutrients, not caloric restriction, extend lifespan in Queensland fruit flies (Bactrocera tryoni). Aging Cell 8, 514–523 (2009).
Solon-Biet, S. M. et al. The ratio of macronutrients, not caloric intake, dictates cardiometabolic health, aging, and longevity in ad libitum-fed mice. Cell Metab. 19, 418–430 (2014).
Powers, R. W. 3rd, Kaeberlein, M., Caldwell, S. D., Kennedy, B. K. & Fields, S. Extension of chronological life span in yeast by decreased TOR pathway signaling. Genes Dev. 20, 174–184 (2006).
Lee, B. C. et al. Methionine restriction extends lifespan of Drosophila melanogaster under conditions of low amino-acid status. Nat. Commun. 5, 3592 (2014).
Ruckenstuhl, C. et al. Lifespan extension by methionine restriction requires autophagy-dependent vacuolar acidification. PLoS Genet. 10, e1004347 (2014).
Lu, J. et al. Sestrin is a key regulator of stem cell function and lifespan in response to dietary amino acids. Nat. Aging 1, 60–72 (2021).
Brown-Borg, H. M. et al. Growth hormone signaling is necessary for lifespan extension by dietary methionine. Aging Cell 13, 1019–1027 (2014).
Miller, R. A. et al. Methionine-deficient diet extends mouse lifespan, slows immune and lens aging, alters glucose, T4, IGF-I and insulin levels, and increases hepatocyte MIF levels and stress resistance. Aging Cell 4, 119–125 (2005).
Zimmerman, J. A., Malloy, V., Krajcik, R. & Orentreich, N. Nutritional control of aging. Exp. Gerontol. 38, 47–52 (2003).
Sanz, A. et al. Methionine restriction decreases mitochondrial oxygen radical generation and leak as well as oxidative damage to mitochondrial DNA and proteins. FASEB J. 20, 1064–1073 (2006).
Sasikumar, A. N., Killilea, D. W., Kennedy, B. K. & Brem, R. B. Potassium restriction boosts vacuolar acidity and extends lifespan in yeast. Exp. Gerontol. 120, 101–106 (2019).
Klang, I. M. et al. Iron promotes protein insolubility and aging in C. elegans. Aging 6, 975–991 (2014).
Kumar, J. et al. Zinc levels modulate lifespan through multiple longevity pathways in Caenorhabditis elegans. PLoS ONE 11, e0153513 (2016).
Cohen, K. et al. Caloric restriction ameliorates cardiomyopathy in animal model of diabetes. Exp. Cell Res. 350, 147–153 (2017).
Kanda, Y. et al. Dietary restriction preserves the mass and function of pancreatic β cells via cell kinetic regulation and suppression of oxidative/ER stress in diabetic mice. J. Nutr. Biochem. 26, 219–226 (2015).
Waldman, M. et al. Regulation of diabetic cardiomyopathy by caloric restriction is mediated by intracellular signaling pathways involving 'SIRT1 and PGC-1α'. Cardiovasc. Diabetol. 17, 111 (2018).
Baumeier, C. et al. Caloric restriction and intermittent fasting alter hepatic lipid droplet proteome and diacylglycerol species and prevent diabetes in NZO mice. Biochim. Biophys. Acta 1851, 566–576 (2015).
Castaño-Martinez, T. et al. Methionine restriction prevents onset of type 2 diabetes in NZO mice. FASEB J. 33, 7092–7102 (2019).
Shibolet, O., Alper, R., Avraham, Y., Berry, E. M. & Ilan, Y. Immunomodulation of experimental colitis via caloric restriction: role of Nk1.1+ T cells. Clin. Immunol. 105, 48–56 (2002).
Liu, G. et al. Methionine restriction on oxidative stress and immune response in dss-induced colitis mice. Oncotarget 8, 44511–44520 (2017).
Robertson, L. T. et al. Protein and calorie restriction contribute additively to protection from renal ischemia reperfusion injury partly via leptin reduction in male mice. J. Nutr. 145, 1717–1727 (2015).
Shushimita, S. et al. Mannan-binding lectin is involved in the protection against renal ischemia/reperfusion injury by dietary restriction. PLoS ONE 10, e0137795 (2015).
Shin, B. K., Kang, S., Kim, D. S. & Park, S. Intermittent fasting protects against the deterioration of cognitive function, energy metabolism and dyslipidemia in Alzheimer's disease-induced estrogen deficient rats. Exp. Biol. Med. 243, 334–343 (2018).
Liu, Y. et al. SIRT3 mediates hippocampal synaptic adaptations to intermittent fasting and ameliorates deficits in APP mutant mice. Nat. Commun. 10, 1886 (2019).
Lazic, D. et al. Every-other-day feeding exacerbates inflammation and neuronal deficits in 5XFAD mouse model of Alzheimer's disease. Neurobiol. Dis. 136, 104745 (2020).
Sharp, Z. D. et al. Minimal effects of dietary restriction on neuroendocrine carcinogenesis in Rb+/- mice. Carcinogenesis 24, 179–183 (2003).
Sundaram, S. & Yan, L. Dietary energy restriction reduces high-fat diet-enhanced metastasis of Lewis lung carcinoma in mice. Oncotarget 7, 65669–65675 (2016).
Simone, B. A. et al. Caloric restriction counteracts chemotherapy-induced inflammation and increases response to therapy in a triple negative breast cancer model. Cell Cycle 17, 1536–1544 (2018).
Fontana, L. et al. Dietary protein restriction inhibits tumor growth in human xenograft models. Oncotarget 4, 2451–2461 (2013).
Orillion, A. et al. Dietary protein restriction reprograms tumor-associated macrophages and enhances immunotherapy. Clin. Cancer Res. 24, 6383–6395 (2018).
Ma, E. H. et al. Serine is an essential metabolite for effector T cell expansion. Cell Metab. 25, 345–357 (2017).
Maddocks, O. D., Labuschagne, C. F., Adams, P. D. & Vousden, K. H. Serine metabolism supports the methionine cycle and DNA/RNA methylation through de novo ATP synthesis in cancer cells. Mol. Cell 61, 210–221 (2016).
Sullivan, M. R. et al. Increased serine synthesis provides an advantage for tumors arising in tissues where serine levels are limiting. Cell Metab. 29, 1410–1421.e4 (2019).
Golbourn, B. J. et al. Loss of MAT2A compromises methionine metabolism and represents a vulnerability in H3K27M mutant glioma by modulating the epigenome. Nat. Cancer 3, 629–648 (2022).
Poillet-Perez, L. et al. Autophagy maintains tumour growth through circulating arginine. Nature 563, 569–573 (2018).
Geiger, R. et al. L-Arginine modulates T cell metabolism and enhances survival and anti-tumor activity. Cell 167, 829–842.e13 (2016).
Li, J. T. et al. BCAT2-mediated BCAA catabolism is critical for development of pancreatic ductal adenocarcinoma. Nat. Cell Biol. 22, 167–174 (2020).
Thandapani, P. et al. Valine tRNA levels and availability regulate complex I assembly in leukaemia. Nature 601, 428–433 (2022).
Gottschalk Højfeldt, S. et al. Relapse risk following truncation of pegylated asparaginase in childhood acute lymphoblastic leukemia. Blood 137, 2373–2382 (2021).
Acknowledgements
This study was supported by the National Key Research and Development Program of China (2020YFA0803201), the Grants from the National Natural Science Foundation of China (31830053, 31920103007) to Prof. Ping Wang.
Author information
Authors and Affiliations
Contributions
P.W. conceived of the paper. Q.W. and Z.J.G. wrote and edited the paper and generated the figures. X.Y. revised the paper. All authors have read and approved the final paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, Q., Gao, ZJ., Yu, X. et al. Dietary regulation in health and disease. Sig Transduct Target Ther 7, 252 (2022). https://doi.org/10.1038/s41392-022-01104-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41392-022-01104-w
This article is cited by
-
Epigenetic Orchestration of Neurodegenerative Disorders: A Possible Target for Curcumin as a Therapeutic
Neurochemical Research (2024)
-
A metabolome atlas of mouse brain on the global metabolic signature dynamics following short-term fasting
Signal Transduction and Targeted Therapy (2023)
-
Effects of raw and hydrolysed Nannochloropsis gaditana biomass included at low level in finishing diets for gilthead seabream (Sparus aurata) on fillet quality and shelf life
Journal of Applied Phycology (2023)