Abstract
Introduction
To determine associations between prostate cancer (PCa) tumor burden measured on biopsy or multiparametric magnetic resonance imaging (mpMRI) and outcomes in intermediate-risk (IR) International Society of Urological Pathology (ISUP) grade 2 men managed with primary radical prostatectomy (RP).
Methods
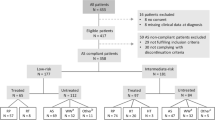
This retrospective, multicenter study was conducted in eight referral centers. The cohort included IR PCa patients who had ISUP 2 at biopsy. We defined biopsy tumor burden as low/high based on the absence/presence of more than 25% positive cores. Tumor burden on imaging was defined as low/high based on maximum lesion diameter, <15 mm and ≥15 mm at mpMRI, respectively. The histological endpoint of the study was adverse features at RP, defined as ≥pT3a stage and/or lymph node invasion and/or ISUP ≥3 at final pathology. The clinical endpoint was biochemical recurrence (BCR) after RP.
Results
A total of 698 IR patients was included, of whom 335 (48%) had adverse features. In multivariate logistic regression analysis, there was no statistical association between tumor burden at biopsy and adverse features (p = 0.7). Tumor size ≥15 mm at mpMRI was significantly associated with adverse pathology (OR 1.65, 95%CI 1.14–2.39; p = 0.01). No significant association was observed between tumor burden at biopsy and BCR (p = 0.4). Tumor size ≥15 mm at mpMRI was significantly associated with BCR (HR 1.96, 95% CI 1.01–3.80; p = 0.04).
Conclusions
Our data support extending the inclusion criteria to ISUP 2 men with >25% positive cores, provided they have a low tumor size at mpMRI (<15 mm). Prospective studies should be performed to validate these findings.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
Data is available on reasonable request from the corresponding author.
References
Tosoian JJ, Mamawala M, Epstein JI, Landis P, Wolf S, Trock BJ, et al. Intermediate and longer-term outcomes from a prospective active-surveillance program for favorable-risk prostate cancer. J Clin Oncol. 2015;33:3379–85.
Tzeng M, Basourakos SP, Davuluri M, Nagar H, Ramaswamy A, Cheng E, et al. Evolving trends in the management of low-risk prostate cancer. Clin Genitourin Cancer. 2022;20:423–30.
Baboudjian M, Breda A, Rajwa P, Gallioli A, Gondran-Tellier B, Sanguedolce F, et al. Active surveillance for intermediate-risk prostate cancer: a systematic review, meta-analysis, and metaregression. Eur Urol Oncol. 2022;5:617–27.
Eastham JA, Auffenberg GB, Barocas DA, Chou R, Crispino T, Davis JW, et al. Clinically localized prostate cancer: AUA/ASTRO guideline, Part II: Principles of active surveillance, principles of surgery, and follow-up. J Urol. 2022;208:19–25.
Willemse PM, Davis NF, Grivas N, Zattoni F, Lardas M, Briers E, et al. Systematic review of active surveillance for clinically localised prostate cancer to develop recommendations regarding inclusion of intermediate-risk disease, biopsy characteristics at inclusion and monitoring, and surveillance repeat biopsy strategy. Eur Urol. 2022;81:337–46.
Bul M, Zhu X, Valdagni R, Pickles T, Kakehi Y, Rannikko A, et al. Active surveillance for low-risk prostate cancer worldwide: the PRIAS study. Eur Urol. 2013;63:597–603.
Kovac E, Vertosick EA, Sjoberg DD, Vickers AJ, Stephenson AJ. Effects of pathological upstaging or upgrading on metastasis and cancer-specific mortality in men with clinical low-risk prostate cancer. BJU Int. 2018;122:1003–9.
Lam TBL, MacLennan S, Willemse PM, Mason MD, Plass K, Shepherd R, et al. EAU-EANM-ESTRO-ESUR-SIOG prostate cancer guideline panel consensus statements for deferred treatment with curative intent for localised prostate cancer from an international collaborative study (DETECTIVE Study). Eur Urol. 2019;76:790–813.
Baboudjian M, Ploussard G. Gleason grade 1 prostate cancer volume at biopsy is associated with upgrading, but not adverse pathology or recurrence after radical prostatectomy: results from a large institutional cohort. Letter. J Urol. 2023;209:72.
Lee DH, Koo KC, Lee SH, Rha KH, Choi YD, Hong SJ, et al. Tumor lesion diameter on diffusion weighted magnetic resonance imaging could help predict insignificant prostate cancer in patients eligible for active surveillance: preliminary analysis. J Urol. 2013;190:1213–7.
de Cobelli O, Terracciano D, Tagliabue E, Raimondi S, Bottero D, Cioffi A, et al. Predicting pathological features at radical prostatectomy in patients with prostate cancer eligible for active surveillance by multiparametric magnetic resonance imaging. PLoS One. 2015;10:e0139696.
Deniffel D, Salinas E, Ientilucci M, Evans AJ, Fleshner N, Ghai S, et al. Does the visibility of grade group 1 prostate cancer on baseline multiparametric magnetic resonance imaging impact clinical outcomes? J Urol. 2020;204:1187–94.
Lantz A, Falagario UG, Ratnani P, Jambor I, Dovey Z, Martini A, et al. Expanding active surveillance inclusion criteria: a novel nomogram including preoperative clinical parameters and magnetic resonance imaging findings. Eur Urol Oncol. 2022;5:187–94.
Barentsz JO, Richenberg J, Clements R, Choyke P, Verma S, Villeirs G, et al. ESUR prostate MR guidelines 2012. Eur Radiol. 2012;22:746–57.
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, et al. PI-RADS prostate imaging - reporting and data system: 2015, Version 2. Eur Urol. 2016;69:16–40.
Morash C, Tey R, Agbassi C, Klotz L, McGowan T, Srigley J, et al. Active surveillance for the management of localized prostate cancer: Guideline recommendations. Can Urol Assoc J. 2015;9:171–8.
Chen RC, Rumble RB, Loblaw DA, Finelli A, Ehdaie B, Cooperberg MR, et al. Active surveillance for the management of localized prostate cancer (cancer care ontario guideline): american society of clinical oncology clinical practice guideline endorsement. J Clin Oncol. 2016;34:2182–90.
EAU Guidelines. Edn. presented at the EAU Annual Congress Amsterdam 2022. ISBN 978-94-92671-16-5.
Gandaglia G, van den Bergh RCN, Tilki D, Fossati N, Ost P, Surcel CI, et al. How can we expand active surveillance criteria in patients with low- and intermediate-risk prostate cancer without increasing the risk of misclassification? Development of a novel risk calculator. BJU Int. 2018;122:823–30.
Diamand R, Albisinni S, Roche JB, Lievore E, Lacetera V, Chiacchio G, et al. Expanding active surveillance criteria for low- and intermediate-risk prostate cancer: can we accurately predict the risk of misclassification for patients diagnosed by multiparametric magnetic resonance imaging-targeted biopsy? Eur Urol Focus. 2022;S2405-4569:00220–6.
Soeterik TFW, van Melick HHE, Dijksman LM, Biesma DH, Witjes JA, van Basten JA. Multiparametric magnetic resonance imaging should be preferred over digital rectal examination for prostate cancer local staging and disease risk classification. Urology. 2021;147:205–12.
Manceau C, Beauval JB, Lesourd M, Almeras C, Aziza R, Gautier JR, et al. MRI characteristics accurately predict biochemical recurrence after radical prostatectomy. J Clin Med. 2020;9:3841.
Baboudjian M, Gondran-Tellier B, Touzani A, Martini A, Diamand R, Roche JB, et al. Magnetic resonance imaging-based t-staging to predict biochemical recurrence after radical prostatectomy: a step towards the iTNM classification. Eur Urol Oncol. 2022;5:617–27.
Triquell M, Regis L, Winkler M, Valdés N, Cuadras M, Celma A, et al. Multiparametric MRI for staging of prostate cancer: a multicentric analysis of predictive factors to improve identification of extracapsular extension before radical prostatectomy. Cancers (Basel). 2022;14:3966.
Stabile A, Mazzone E, Cirulli GO, De Cobelli F, Grummet J, Thoeny HC, et al. Association between multiparametric magnetic resonance imaging of the prostate and oncological outcomes after primary treatment for prostate cancer: a systematic review and meta-analysis. Eur Urol Oncol. 2021;4:519–28.
Sugano D, Sidana A, Jain AL, Calio B, Gaur S, Maruf M, et al. Index tumor volume on MRI as a predictor of clinical and pathologic outcomes following radical prostatectomy. Int Urol Nephrol. 2019;51:1349–55. https://doi.org/10.1007/s11255-019-02168-4.
Tan N, Shen L, Khoshnoodi P, Alcalá HE, Yu W, Hsu W, et al. Pathological and 3 tesla volumetric magnetic resonance imaging predictors of biochemical recurrence after robotic assisted radical prostatectomy: correlation with whole mount histopathology. J Urol. 2018;199:1218–23.
Woo S, Han S, Kim TH, Suh CH, Westphalen AC, Hricak H, et al. Prognostic value of pretreatment MRI in patients with prostate cancer treated with radiation therapy: a systematic review and meta-analysis. AJR Am J Roentgenol. 2020;214:597–604.
Martini A, Gandaglia G, Karnes RJ, Zaffuto E, Bianchi M, Gontero P, et al. Defining the most informative intermediate clinical endpoints for predicting overall survival in patients treated with radical prostatectomy for high-risk prostate cancer. Eur Urol Oncol. 2019;2:456–63.
Author information
Authors and Affiliations
Contributions
Conceived and designed the analysis: MB, AU, GP; performed the analysis: MB, AU, GP; wrote the manuscript: MB; provided critical feedback in shaping the manuscript: all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Baboudjian, M., Uleri, A., Beauval, JB. et al. MRI lesion size is more important than the number of positive biopsy cores in predicting adverse features and recurrence after radical prostatectomy: implications for active surveillance criteria in intermediate-risk patients. Prostate Cancer Prostatic Dis (2023). https://doi.org/10.1038/s41391-023-00693-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41391-023-00693-z
This article is cited by
-
Likelihood of sampling prostate cancer at systematic biopsy as a function of gland volume and number of cores
Prostate Cancer and Prostatic Diseases (2024)
-
Quantified treatment effect at the individual level is more indicative for personalized radical prostatectomy recommendation: implications for prostate cancer treatment using deep learning
Journal of Cancer Research and Clinical Oncology (2024)