Abstract
Background
Cancer-related pain, usually associated with bone metastases, is a frequent and debilitating morbidity in patients with prostate cancer. To date there are only limited data regarding the prognostic role of pain in men with metastatic castration-sensitive prostate cancer (mCSPC). The objective of our analysis was to assess if the presence of pain can be considered an independent prognostic factor in mCSPC patients.
Methods
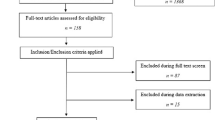
A retrospective analysis was performed on patients with mCSPC referring to six oncology centers in Italy. Clinical and pathological features were recorded. Patients were considered to have pain if this was reported within the patient’s file or in case of a chronic analgesic therapy was found among the concomitant medications. Survivals were estimated by the Kaplan–Meier method, and compared across groups using the log-rank test. Cox proportional hazard models, stratified according to the baseline characteristics, were used to estimate hazard ratios for overall survival (OS). All the variables were significant if p < 0.05.
Results
Data about pain were available for 365 cases and pain was present in 34.8% of patients. Pain was mainly associated with high value of prostate-specific antigen, metastatic bone extension regardless of the site, and lymph node involvement. mCSPC patients with pain had in most of the cases high-volume or Hr disease, and significant shorter OS (27.0 vs. 58.2 months, p < 0.001) and PFS (10.1 vs. 17.4 months, p < 0.001) compared to those without pain. The negative impact of pain on OS remained significant even if adjusted for CHAARTED or LATITUDE classification, and other significant baseline prognostic factors.
Conclusions
This analysis supports the poor prognostic role of cancer-related pain in the setting of mCSPC patients. A prospective validation is required.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7–34.
Weiner AB, Matulewicz RS, Eggener SE, Schaeffer EM. Increasing incidence of metastatic prostate cancer in the United States (2004-2013). Prostate Cancer Prostatic Dis. 2016;19:395–7.
Iacovelli R, Ciccarese C, Mosillo C, Bimbatti D, Fantinel E, Stefani L, et al. Comparison between prognostic classifications in de novo metastatic hormone sensitive prostate cancer. Target Oncol. 2018;13:649–55.
Sweeney CJ, Chen YH, Carducci M, Liu G, Jarrard DF, Eisenberger M, et al. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N. Engl J Med. 2015;373:737–46.
James ND, Sydes MR, Clarke NW, Mason MD, Dearnaley DP, Spears MR, et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet 2016;387:1163–77.
Fizazi K, Tran N, Fein L, Matsubara N, Rodriguez-Antolin A, Alekseev BY, et al. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N. Engl J Med. 2017;377:352–60.
James ND, de Bono JS, Spears MR, Clarke NW, Mason MD, Dearnaley DP, et al. Abiraterone for prostate cancer not previously treated with hormone therapy. N. Engl J Med. 2017;377:338–51.
Davis ID, Martin AJ, Stockler MR, Begbie S, Chi KN, Chowdhury S, et al. Enzalutamide with standard first-line therapy in metastatic prostate cancer. N. Engl J Med. 2019;381:121–31.
Chi KN, Agarwal N, Bjartell A, Chung BH, Pereira de Santana Gomes AJ, Given R, et al. Apalutamide for metastatic, castration-sensitive prostate cancer. N. Engl J Med. 2019;381:13–24.
Armstrong AJ, Szmulewitz RZ, Petrylak DP, Holzbeierlein J, Villers A, Azad A, et al. ARCHES: a randomized, phase iii study of androgen deprivation therapy with enzalutamide or placebo in men with metastatic hormone-sensitive prostate cancer. J Clin Oncol. 2019;37:2974–86.
Halabi S, Vogelzang NJ, Kornblith AB, Ou SS, Kantoff PW, Dawson NA, et al. Pain predicts overall survival in men with metastatic castration-refractory prostate cancer. J Clin Oncol. 2008;26:2544–9.
Gotay CC, Kawamoto CT, Bottomley A, Efficace F. The prognostic significance of patient-reported outcomes in cancer clinical trials. J Clin Oncol. 2008;26:1355–63.
Armstrong AJ, Garrett-Mayer ES, Yang YC, de Wit R, Tannock IF, Eisenberger M. A contemporary prognostic nomogram for men with hormone-refractory metastatic prostate cancer: a TAX327 study analysis. Clin Cancer Res. 2007;13:6396–403.
Miller K, Carles J, Gschwend JE, Van Poppel H, Diels J, Brookman-May SD. The phase 3 COU-AA-302 study of abiraterone acetate plus prednisone in men with chemotherapy-naïve metastatic castration-resistant prostate cancer: stratified analysis based on pain, prostate-specific antigen, and Gleason score. Eur Urol. 2018;74:17–23.
Armstrong AJ, Lin P, Higano CS, Sternberg CN, Sonpavde G, Tombal B, et al. Development and validation of a prognostic model for overall survival in chemotherapy-naïve men with metastatic castration-resistant prostate cancer. Ann Oncol. 2018;29:2200–7.
Halabi S, Lin CY, Small EJ, Armstrong AJ, Kaplan EB, Petrylak D, et al. Prognostic model predicting metastatic castration-resistant prostate cancer survival in men treated with second-line chemotherapy. J Natl Cancer Inst. 2013;105:1729–37.
Cella D, Traina S, Li T, Johnson K, Ho KF, Molina A, et al. Relationship between patient-reported outcomes and clinical outcomes in metastatic castration-resistant prostate cancer: post hoc analysis of COU-AA-301 and COU-AA-302. Ann Oncol. 2018;29:392–7.
Delanoy N, Robbrecht D, Fizazi K, Mercier F, Sartor O, De Wit R, et al. Pain progression at initiation of chemotherapy in metastatic castration-resistant prostate cancer (mCRPC) is associated with a poor prognosis: a post-hoc analysis of FIRSTANA. Ann Oncol. 2019;30:v335–v336.
Glass TR, Tangen CM, Crawford ED, Thompson I. Metastatic carcinoma of the prostate: identifying prognostic groups using recursive partitioning. J Urol. 2003;169:164–9.
Gravis G, Boher JM, Fizazi K, Joly F, Priou F, Marino P, et al. Prognostic factors for survival in noncastrate metastatic prostate cancer: validation of the glass model and development of a novel simplified prognostic model. Eur Urol. 2015;68:196–204.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
R.I.: advisory board member for Pfizer, Janssen, Sanofi, IPSEN, MSD, and Novartis. O.C.: advisory board member/speaker for Pfizer, Janssen, IPSEN, and Astellas. U.D.G.: advisory board or consultant fees from Merck Sharp & Dohme, Bristol Myers Squibb, Janssen, Astellas, Sanofi, Bayer, Pfizer, Ipsen, Novartis, Pharmamar and Institutional research grant from Astrazeneca, Sanofi, and Roche. C.B.: advisory board member for Janssen. G.T.: advisory board member for BMS and Novartis. Chiara Ciccarese (occasional consultant of IPSEN and Merck), M.T., C.M., D.B., F.P., F.M., Chiara Casadei, and M.M. declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Iacovelli, R., Ciccarese, C., Caffo, O. et al. The prognostic value of pain in castration-sensitive prostate cancer. Prostate Cancer Prostatic Dis 23, 654–660 (2020). https://doi.org/10.1038/s41391-020-0255-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-020-0255-x
This article is cited by
-
Association of dynamic change in patient-reported pain with survival in metastatic castrate sensitive prostate cancer—exploratory analysis of LATITUDE study
Prostate Cancer and Prostatic Diseases (2023)