Abstract
Background
To investigate the role of urine spermine and Spermine Risk Score in prediction of high-grade prostate cancer (HGPCa, ISUP grade group ≥2).
Methods
Nine hundred and five consecutive men with elevated PSA were prospectively recruited from two hospitals. Core analyses focused on consecutive men with PSA 4–20 ng/mL (n = 600). Pre-biopsy urine without prior prostatic massage was analyzed for spermine level with ultra-high performance liquid chromatography with triple quadrupole mass spectrometer (UPLC-MS/MS). The proportions of PCa and HGPCa were compared across different spermine ranges. Logistic regressions were used to form different models, and their performances were compared using area under curve (AUC) and decision curve analysis (DCA).
Results
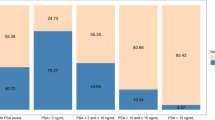
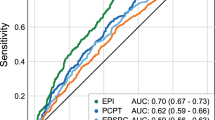
PCa and HGPCa were diagnosed in 30.8% (185/600) and 17.2% (103/600) men, respectively, and were significantly associated with lower urine spermine levels. Between the lowest and highest quartiles of spermine results, a threefold increase in PCa risk (49.3% vs. 16.7%) and 3.5-fold increase in ISUP grade group ≥2 PCa risk (31.3% vs. 8.7%) were observed. Multivariate analysis showed PSA, prostate volume (PV), digital rectal examination (DRE), and spermine, which were independent predictors for PCa and HGPCa, and a Spermine Risk Score with these factors achieved the highest AUC of 0.78 for PCa and 0.82 for HGPCa. At 90% sensitivity for HGPCa, 36.7% biopsies and 24.4% ISUP grade group 1 diagnoses could have been avoided, with a negative predictive value of 95.4%. DCA revealed net clinical benefit of the Spermine Risk Score. Internal validation with bootstrapping showed good discrimination and calibration.
Conclusion
Urine spermine and Spermine Risk Score identified men at higher risk of HGPCa and reduced unnecessary biopsies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Hugosson J, Roobol MJ, Månsson M, Tammela TLJ, Zappa M, Nelen V, et al. A 16-yr follow-up of the European Randomized study of screening for prostate cancer. Eur Urol. 2019;76:43–51.
Chiu PK, Ng CF, Semjonow A, Zhu Y, Vincendeau S, Houlgatte A, et al. A multicentre evaluation of the role of the Prostate Health Index (PHI) in regions with differing prevalence of prostate cancer: adjustment of PHI reference ranges is needed for European and Asian settings. Eur Urol. 2019;75:558–61.
Braun K, Sjoberg DD, Vickers AJ, Lilja H, Bjartell AS. A four-kallikrein panel predicts high-grade cancer on biopsy: independent validation in a community cohort. Eur Urol. 2016;69:505–11.
Rodríguez SVM. Diagnostic accuracy of prostate cancer antigen 3 (PCA3) prior to first prostate biopsy: a systematic review and meta-analysis. Can Urol Assoc J. 2020;14:E214–9.
Van Neste L, Hendriks RJ, Dijkstra S, Trooskens G, Cornel EB, Jannink SA, et al. Detection of high-grade prostate cancer using a urinary molecular biomarker-based risk score. Eur Urol. 2016;70:740–48.
Kasivisvanathan V, Rannikko AS, Borghi M, Panebianco V, Mynderse LA, Vaarala MH, et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med. 2018;378:1767–77.
Schipper RG, Romijn JC, Cuijpers VM, Verhofstad AA. Polyamines and prostatic cancer. Biochem Soc Trans. 2003;31:375–80.
van der Graaf M, Schipper RG, Oosterhof GO, Schalken JA, Verhofstad AA, Heerschap A. Proton MR spectroscopy of prostatic tissue focused on the detection of spermine, a possible biomarker of malignant behavior in prostate cancer. MAGMA. 2000;10:153–9.
Tsoi TH, Chan CF, Chan WL, Chiu KF, Wong WT, Ng CF, et al. Urinary polyamines: a pilot study on their roles as prostate cancer detection biomarkers. PLoS ONE. 2016;11:e0162217.
Russell DH. Increased polyamine concentrations in the urine of human cancer patients. Nat N Biol. 1971;233:144–5.
Fair WR, Wehner N, Brorsson U. Urinary polyamine levels in the diagnosis of carcinoma of the prostate. J Urol. 1975;114:88–92.
Hradec E, Jiránek J, Macek K. Urinary spermidine levels in cases of carcinoma of the prostate. Preliminary report. Scand J Urol Nephrol Suppl. 1980;55:71–3.
Horn Y, Beal SL, Walach N, Lubich WP, Spigel L, Marton LJ. Further evidence for the use of polyamines as biochemical markers for malignant tumors. Cancer Res. 1982;42:3248–51.
Horn Y, Beal SL, Walach N, Lubich WP, Spigel L, Marton LJ. Relationship of urinary polyamines to tumor activity and tumor volume in patients. Cancer Res. 1984;44:4675–8.
Sakai S, Ito Y, Koide T, Tei K, Hara A, Sawada H. Detection of urinary polyamine by a new enzymatic differential assay. (III). Studies on urinary polyamines in patients with malignant genitourinary diseases. Hinyokika Kiyo. 1986;32:343–50.
Sakai S, Koide T, Ito Y, Tei K, Deguchi T, Matsuda S, et al. Urine polyamine in patients with malignant urological diseases using a polyamine-test enzyme kit. Hinyokika Kiyo. 1986;32:661–5.
Chen R, Ren S, Chinese Prostate Cancer Consortium, Yiu MK, Fai NC, Cheng WS, et al. Prostate cancer in Asia: a collaborative report. Asian J Urol. 2014;1:15–29.
Acknowledgements
The authors would like to thank Ms Shaz Cheng for her help in this project.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
K-LW holds a patent for urinary polyamines as prostate cancer detection biomarkers (patent no. US20180172695A1). The other authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Chiu, P.KF., Fung, YH., Teoh, J.YC. et al. Urine spermine and multivariable Spermine Risk Score predict high-grade prostate cancer. Prostate Cancer Prostatic Dis 24, 542–548 (2021). https://doi.org/10.1038/s41391-020-00312-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-020-00312-1