Abstract
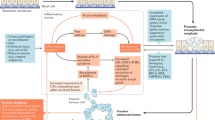
Benign prostatic hyperplasia (BPH) and associated lower urinary tract symptoms are common clinical concerns that affect aging men all over the world. The underlying molecular and cellular mechanisms remain elusive. Over the past few years, a number of animal models of BPH, including spontaneous model, BPH-induction model, xenograft model, metabolic syndrome model, mechanical obstruction model, and transgenic model, have been established that may provide useful tools to fill these critical knowledge gaps. In this review, we therefore outlined the present status quo for animal models of BPH, comparing the pros and cons with respect to their ability to mimic the etiological, histological, and clinical hallmarks of BPH and discussed their applicability for future research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Choi YJ, Kim EK, Fan M, Tang Y, Hwang YJ, Sung SH. Effect of Paecilomyces tenuipes extract on testosterone-induced benign prostatic hyperplasia in Sprague-Dawley rats. Int J Environ Res Public Health. 2019;16:3764–73.
Jin BR, Kim HJ, Seo JH, Kim MS, Lee KH, Yoon IJ, et al. HBX-6, standardized Cornus officinalis and Psoralea corylifolia L. extracts, suppresses benign prostate hyperplasia by attenuating E2F1 activation. Molecules. 2019;24:1719–32.
Popovics P, Schally AV, Salgueiro L, Kovacs K, Rick FG. Antagonists of growth hormone-releasing hormone inhibit proliferation induced by inflammation in prostatic epithelial cells. Proc Natl Acad Sci USA. 2017;114:1359–64.
Zhang J, Ou Z, Zhang X, He W, Wang R, Mo M, et al. Holmium laser enucleation of the prostate versus thulium laser enucleation of the prostate for the treatment of large-volume prostates > 80 ml: 18-month follow-up results. World J Urol. 2019;38:1555–62.
Vignozzi L, Gacci M, Maggi M. Lower urinary tract symptoms, benign prostatic hyperplasia and metabolic syndrome. Nat Rev Urol. 2016;13:108–19.
Madersbacher S, Sampson N, Culig Z. Pathophysiology of benign prostatic hyperplasia and benign prostatic enlargement: a mini-review. Gerontology. 2019;65:458–64.
Hata J, Machida T, Matsuoka K, Hoshi S, Akaihata H, Hiraki H, et al. Complement activation by autoantigen recognition in the growth process of benign prostatic hyperplasia. Sci Rep. 2019;9:599-612.
Nicholson TM, Ricke WA. Androgens and estrogens in benign prostatic hyperplasia: past, present and future. Differentiation. 2011;82:184–99.
Choi BR, Kim HK, Soni KK, Karna KK, Lee SW, So I, et al. Additive effect of oral LDD175 to tamsulosin and finasteride in a benign prostate hyperplasia rat model. Drug Des Dev Ther. 2018;12:1855–63.
Brennen WN, Isaacs JT. Mesenchymal stem cells and the embryonic reawakening theory of BPH. Nat Rev Urol. 2018;15:703–15.
Berry SJ, Coffey DS, Walsh PC, Ewing LL. The development of human benign prostatic hyperplasia with age. J Urol. 1984;132:474–9.
Peters CA, Walsh PC. The effect of nafarelin acetate, a luteinizing-hormone-releasing hormone agonist, on benign prostatic hyperplasia. N Engl J Med. 1987;317:599–604.
Morales A. Androgen replacement therapy and prostate safety. Eur Urol 2002;41:113–20.
Ho CK, Habib FK. Estrogen and androgen signaling in the pathogenesis of BPH. Nat Rev Urol. 2011;8:29–41.
Wang L, Yang JR, Yang LY, Liu ZT. Chronic inflammation in benign prostatic hyperplasia: implications for therapy. Med Hypotheses. 2008;70:1021–3.
Isaacs JT, Coffey DS. Etiology and disease process of benign prostatic hyperplasia. Prostate Suppl. 1989;2:33–50.
Lin VK, Wang SY, Vazquez DV, C CX, Zhang S, Tang L. Prostatic stromal cells derived from benign prostatic hyperplasia specimens possess stem cell like property. Prostate. 2007;67:1265–76.
Huang X, Lee C. Regulation of stromal proliferation, growth arrest, differentiation and apoptosis in benign prostatic hyperplasia by TGF-beta. Front Biosci. 2003;8:s740–9.
He Y, Ou Z, Chen X, Zu X, Liu L, Li Y, et al. LPS/TLR4 signaling enhances TGF-beta response through downregulating BAMBI during prostatic hyperplasia. Sci Rep. 2016;6:27051.
Wang R, Zhang M, Ou Z, He W, Chen L, Zhang J, et al. Long noncoding RNA DNM3OS promotes prostate stromal cells transformation via the miR-29a/29b/COL3A1 and miR-361/TGFbeta1 axes. 2019;11:9442–60.
Lerman LO, Kurtz TW, Touyz RM, Ellison DH, Chade AR, Crowley SD, et al. Animal models of hypertension: a scientific statement from the American Heart Association. Hypertension 2019;73:e87–120.
Burrows DJ, McGown A, Jain SA, De Felice M, Ramesh TM, Sharrack B, et al. Animal models of multiple sclerosis: from rodents to zebrafish. Mult Scler. 2019;25:306–24.
Steiner MS, Couch RC, Raghow S, Stauffer D. The chimpanzee as a model of human benign prostatic hyperplasia. J Urol. 1999;162:1454–61.
Mubiru JN, Hubbard GB, Dick EJ Jr., Furman J, Troyer DA, Rogers J. Nonhuman primates as models for studies of prostate specific antigen and prostatic diseases. Prostate. 2008;68:1546–54.
Walsh PC, Wilson JD. The induction of prostatic hypertrophy in the dog with androstanediol. J Clin Investig. 1976;57:1093–7.
DeKlerk DP, Coffey DS, Ewing LL, McDermott IR, Reiner WG, Robinson CH, et al. Comparison of spontaneous and experimentally induced canine prostatic hyperplasia. J Clin Investig. 1979;64:842–9.
Berry SJ, Strandberg JD, Saunders WJ, Coffey DS. Development of canine benign prostatic hyperplasia with age. Prostate. 1986;9:363–73.
Lowseth LA, Gerlach RF, Gillett NA, Muggenburg BA. Age-related changes in the prostate and testes of the beagle dog. Vet Pathol. 1990;27:347–53.
Murakoshi M, Ikeda R, Fukui N. The effects of chlormadinone acetate (CMA), antiandrogen, on the pituitary, testis, prostate and adrenal gland of the dog with spontaneous benign prostatic hyperplasia. J Toxicol Sci. 2001;26:119–27.
McNeal J. Pathology of benign prostatic hyperplasia. Insight into etiology. Urol Clin North Am. 1990;17:477–86.
Zhou Y, Xiao XQ, Chen LF, Yang R, Shi JD, Du XL, et al. Proliferation and phenotypic changes of stromal cells in response to varying estrogen/androgen levels in castrated rats. Asian J Androl. 2009;11:451–9.
Prins GS, Huang L, Birch L, Pu Y. The role of estrogens in normal and abnormal development of the prostate gland. Ann N Y Acad Sci. 2006;1089:1–13.
Al-Trad B, Aljabali A, Al Zoubi M, Shehab M, Omari S. Effect of gold nanoparticles treatment on the testosterone-induced benign prostatic hyperplasia in rats. Int J Nanomed. 2019;14:3145–54.
Sun F, Baez-Diaz C, Sanchez-Margallo FM. Canine prostate models in preclinical studies of minimally invasive interventions: part II, benign prostatic hyperplasia models. Transl Androl Urol. 2017;6:547–55.
Dai GC, Hu B, Zhang WF, Peng F, Wang R, Liu ZY, et al. Chemical characterization, anti-benign prostatic hyperplasia effect and subchronic toxicity study of total flavonoid extract of Pteris multifida. Food Chem Toxicol. 2017;108:524–31.
McCormick DL, Li J, Tian Y, Guo S, Gu H, Yuan Q, et al. Testosterone-induced benign prostatic hyperplasia rat and dog as facile models to assess drugs targeting lower urinary tract symptoms. PloS ONE. 2018;13:e0191469.
Zou Y, Aboshora W, Li J, Xiao T, Zhang L. Protective effects of Lepidium meyenii (Maca) aqueous extract and lycopene on testosterone propionate-induced prostatic hyperplasia in mice. Phytother Res. 2017;31:1192–8.
Li J, Tian Y, Guo S, Gu H, Yuan Q, Xie X. Testosterone-induced benign prostatic hyperplasia rat and dog as facile models to assess drugs targeting lower urinary tract symptoms. PLoS ONE. 2018;13:e0191469.
Li Z, Xiao H, Wang K, Zheng Y, Chen P, Wang X, et al. Upregulation of oxytocin receptor in the hyperplastic prostate. Front Endocrinol. 2018;9:403.
Nicholson TM, Ricke EA, Marker PC, Miano JM, Mayer RD, Timms BG, et al. Testosterone and 17beta-estradiol induce glandular prostatic growth, bladder outlet obstruction, and voiding dysfunction in male mice. Endocrinology. 2012;153:5556–65.
Yokota T, Honda K, Tsuruya Y, Nomiya M, Yamaguchi O, Gotanda K, et al. Functional and anatomical effects of hormonally induced experimental prostate growth: a urodynamic model of benign prostatic hyperplasia (BPH) in the beagle. Prostate. 2004;58:156–63.
Funahashi Y, O’Malley KJ, Kawamorita N, Tyagi P, DeFranco DB, Takahashi R, et al. Upregulation of androgen-responsive genes and transforming growth factor-beta1 cascade genes in a rat model of non-bacterial prostatic inflammation. Prostate. 2014;74:337–45.
Zhang M, Luo C, Cui K, Xiong T, Chen Z. Chronic inflammation promotes proliferation in the prostatic stroma in rats with experimental autoimmune prostatitis: study for a novel method of inducing benign prostatic hyperplasia in a rat model. World J Urol. 2020. https://doi.org/10.1007/s00345-020-03090-6.
Kramer G, Mitteregger D, Marberger M. Is benign prostatic hyperplasia (BPH) an immune inflammatory disease? Eur Urol. 2007;51:1202–16.
Kim HJ, Park JW, Cho YS, Cho CH, Kim JS, Shin HW, et al. Pathogenic role of HIF-1alpha in prostate hyperplasia in the presence of chronic inflammation. Biochim et Biophys Acta. 2013;1832:183–94.
Dos Santos Gomes FO, Oliveira AC, Ribeiro EL, da Silva BS, Dos Santos LAM, de Lima IT, et al. Intraurethral injection with LPS: an effective experimental model of prostatic inflammation. Inflamm Res. 2018;67:43–55.
Miller GJ, Runner MN, Chung LW. Tissue interactions and prostatic growth: II. Morphological and biochemical characterization of adult mouse prostatic hyperplasia induced by fetal urogenital sinus implants. Prostate. 1985;6:241–53.
Mori F, Oda N, Sakuragi M, Sakakibara F, Kiniwa M, Miyoshi K. New histopathological experimental model for benign prostatic hyperplasia: stromal hyperplasia in rats. J Urol. 2009;181:890–8.
Tian S, Miao M, Bai M, Wu Y, Gao J, Guo L. Effect of stachydrine hydrochloride to the prostate hyperplasia model in mice. Saudi J Biol Sci. 2019;26:782–9.
Kojima Y, Sasaki S, Kubota Y, Imura M, Oda N, Kiniwa M, et al. Up-regulation of alpha1a and alpha1d-adrenoceptors in the prostate by administration of subtype selective alpha1-adrenoceptor antagonist tamsulosin in patients with benign prostatic hyperplasia. J Urol. 2011;186:1530–6.
Kojima Y, Sasaki S, Oda N, Koshimizu TA, Hayashi Y, Kiniwa M, et al. Prostate growth inhibition by subtype-selective alpha(1)-adrenoceptor antagonist naftopidil in benign prostatic hyperplasia. Prostate. 2009;69:1521–8.
Arruzazabala ML, Más R, Molina V, Noa M, Carbajal D, Mendoza N. Effect of D-004, a lipid extract from the Cuban royal palm fruit, on atypical prostate hyperplasia induced by phenylephrine in rats. Drugs R D. 2006;7:233–41.
Marinese D, Patel R, Walden PD. Mechanistic investigation of the adrenergic induction of ventral prostate hyperplasia in mice. Prostate. 2003;54:230–7.
Wang L, Xie L, Tintani F, Xie H, Li C, Cui Z, et al. Aberrant transforming growth factor-beta activation recruits mesenchymal stem cells during prostatic hyperplasia. Stem Cells Transl Med. 2017;6:394–404.
Kim J, Yanagihara Y, Kikugawa T, Ji M, Tanji N, Masayoshi Y, et al. A signaling network in phenylephrine-induced benign prostatic hyperplasia. Endocrinology. 2009;150:3576–83.
Golomb E, Kruglikova A, Dvir D, Parnes N, Abramovici A. Induction of atypical prostatic hyperplasia in rats by sympathomimetic stimulation. Prostate. 1998;34:214–21.
Lopez-Barcons LA. Human benign prostatic hyperplasia heterotransplants as an experimental model. Asian J Androl. 2010;12:157–63.
Wang Y, Revelo MP, Sudilovsky D, Cao M, Chen WG, Goetz L, et al. Development and characterization of efficient xenograft models for benign and malignant human prostate tissue. Prostate. 2005;64:149–59.
Blagosklonny MV, Love HD, Booton SE, Boone BE, Breyer JP, Koyama T, et al. Androgen regulated genes in human prostate xenografts in mice: relation to BPH and prostate cancer. PLoS ONE. 2009;4:e8384.
Tsujimura A, Fukuhara S, Soda T, Takezawa K, Kiuchi H, Takao T, et al. Histologic evaluation of human benign prostatic hyperplasia treated by dutasteride: a study by xenograft model with improved severe combined immunodeficient mice. Urology. 2015;85:274 e1–8.
Aaron-Brooks LM, Sasaki T, Vickman RE, Wei L, Franco OE, Ji Y, et al. Hyperglycemia and T cell infiltration are associated with stromal and epithelial prostatic hyperplasia in the nonobese diabetic mouse. Prostate. 2019;79:980–93.
Ozden C, Ozdal OL, Urgancioglu G, Koyuncu H, Gokkaya S, Memis A. The correlation between metabolic syndrome and prostatic growth in patients with benign prostatic hyperplasia. Eur Urol. 2007;51:199–203. Discussion 4-6.
Vikram A, Kushwaha S, Jena GB. Relative influence of testosterone and insulin in the regulation of prostatic cell proliferation and growth. Steroids. 2011;76:416–23.
Ribeiro DL, Caldeira EJ, Cândido EM, Manzato AJ, Taboga SR, Cagnon VH. Prostatic stromal microenvironment and experimental diabetes. Eur J Histochem. 2006;50:51–60.
Jiang M, Strand DW, Franco OE, Clark PE, Hayward SW. PPARgamma: a molecular link between systemic metabolic disease and benign prostate hyperplasia. Differentiation. 2011;82:220–36.
Escobar EL, Gomes-Marcondes MC, Carvalho HF. Dietary fatty acid quality affects AR and PPARgamma levels and prostate growth. Prostate. 2009;69:548–58.
Vikram A, Jena GB, Ramarao P. Increased cell proliferation and contractility of prostate in insulin resistant rats: linking hyperinsulinemia with benign prostate hyperplasia. Prostate. 2010;70:79–89.
Xu C, Xu Y, Shen Z, Zhou H, Xiao J, Huang T. Effects of metformin on prostatic tissue of rats with metabolic syndrome and benign prostatic hyperplasia. Int Urol Nephrol. 2018;50:611–7.
Zhang X, Na Y, Guo Y. Biologic feature of prostatic hyperplasia developed in spontaneously hypertensive rats. Urology. 2004;63:983–8.
Zhang X, Shen F, Dong L, Zhao X, Qu X. Influence and pathophysiological mechanisms of simvastatin on prostatic hyperplasia in spontaneously hypertensive rats. Urologia Internationalis. 2013;91:467–73.
Yamashita M, Zhang X, Shiraishi T, Uetsuki H, Kakehi Y. Determination of percent area density of epithelial and stromal components in development of prostatic hyperplasia in spontaneously hypertensive rats. Urology. 2003;61:484–9.
Matityahou A, Rosenzweig N, Golomb E. Rapid proliferation of prostatic epithelial cells in spontaneously hypertensive rats: a model of spontaneous hypertension and prostate hyperplasia. J Androl. 2003;24:263–9.
Speakman MJ, Brading AF, Gilpin CJ, Dixon JS, Gilpin SA, Gosling JA. Bladder outflow obstruction—a cause of denervation supersensitivity. J Urol. 1987;138:1461–6.
Broderick G, Longhurst P, Juniewicz P, Wein A, Levin R. A novel canine model of partial outlet obstruction secondary to prostatic hypertrophy. World J Urol. 1994;12:245–8.
Malkowicz SB, Wein AJ, Elbadawi A, Arsdalen KV, Ruggieri MR, Levin RM. Acute biochemical and functional alterations in the partially obstructed rabbit urinary bladder. J Urol. 1986;136:1324–9.
Austin JC, Chacko SK, DiSanto M, Canning DA, Zderic SA. A male murine model of partial bladder outlet obstruction reveals changes in detrusor morphology, contractility and myosin isoform expression. J Urol. 2004;172:1524–8.
Levin RM, Hudson AP. The molecular genetic basis of mitochondrial malfunction in bladder tissue following outlet obstruction. J Urol. 2004;172:438–47.
Lai KP, Huang CK, Fang LY, Izumi K, Lo CW, Wood R, et al. Targeting stromal androgen receptor suppresses prolactin-driven benign prostatic hyperplasia (BPH). Mol Endocrinol. 2013;27:1617–31.
Wennbo H, Kindblom J, Isaksson OG, Törnell J. Transgenic mice overexpressing the prolactin gene develop dramatic enlargement of the prostate gland. Endocrinology. 1997;138:4410–5.
Tutrone RF, Ball RA, Ornitz DM, Leder P, Richie JP. Benign prostatic hyperplasia in a transgenic mouse: a new hormonally sensitive investigatory model. J Urol. 1993;149:633–9.
Muller WJ, Lee FS, Dickson C, Peters G, Pattengale P, Leder P. The int-2 gene product acts as an epithelial growth factor in transgenic mice. EMBO J. 1990;9:907–13.
Hata J, Satoh Y, Akaihata H, Hiraki H, Ogawa S, Haga N, et al. Molecular classification of benign prostatic hyperplasia: a gene expression profiling study in a rat model. Int J Urol. 2016;23:599–612.
Schauer IG, Ressler SJ, Rowley DR. Keratinocyte-derived chemokine induces prostate epithelial hyperplasia and reactive stroma in a novel transgenic mouse model. Prostate. 2009;69:373–84.
Wen S, Chang HC, Tian J, Shang Z, Niu Y, Chang C. Stromal androgen receptor roles in the development of normal prostate, benign prostate hyperplasia, and prostate cancer. Am J Pathol. 2015;185:293–301.
Welsh M, Moffat L, McNeilly A, Brownstein D, Saunders PT, Sharpe RM, et al. Smooth muscle cell-specific knockout of androgen receptor: a new model for prostatic disease. Endocrinology. 2011;152:3541–51.
Acknowledgements
We are grateful to Yi He, PhD, from University of Texas Health Science Center at San Antonio and Ting Wu, PhD, from University of Pittsburgh for helpful hints in grammar check and native expression.
Funding
This research was funded by the National Natural Science Foundation of China 81770758 (to LW).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, J., Zhang, M., Tang, J. et al. Animal models of benign prostatic hyperplasia. Prostate Cancer Prostatic Dis 24, 49–57 (2021). https://doi.org/10.1038/s41391-020-00277-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-020-00277-1
This article is cited by
-
Correlation between metabolic syndrome and periurethral prostatic fibrosis: results of a prospective study
BMC Urology (2024)
-
Shikonin alleviates testosterone-induced benign prostatic hyperplasia in rats via the Nrf2-ARE and NF-κB pathway
Molecular & Cellular Toxicology (2024)
-
Shuangshi Tonglin Capsule treats benign prostatic hyperplasia through the ROS/NLRP3 signaling pathway
International Urology and Nephrology (2023)
-
Icariin ameliorates metabolic syndrome-induced benign prostatic hyperplasia in rats
Environmental Science and Pollution Research (2022)
-
Smoothened inhibition leads to decreased cell proliferation and suppressed tissue fibrosis in the development of benign prostatic hyperplasia
Cell Death Discovery (2021)